Abstract
Keywords
Introduction
Acetaminophen (APAP) is one of the most commonly used analgesic and antipyretic drugs in the world. 1 The dosage is safe within a certain range, while exceeding the range can cause severe liver damage. Hundreds of millions of people use APAP every year, and some people will suffer liver damage from taking APAP, which is caused by N-acetyl-p-benzoquinone imine (NAPQI), a reactive intermediate produced by drug metabolism. 2 NAPQI is metabolized by CYP2E1 and CYP3A4 isoenzymes, the Glutathione (GSH) combines with NAPOI and detoxifies to mercapto acid, and is eliminated in the bile and blood. The accumulation of NAPQI will lead to the consumption of GSH in the body. NAPQI will covalently bond with the electronic groups contained in the cell, causing a series of biochemical reactions such as mitochondrial oxidative stress, which eventually lead to apoptosis and severe liver damage.3–5 When APAP-induced liver poisoning occurs, pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) are released. It is reported that the PI3K/AKT signaling pathway may regulate the transcriptional activity of the NF-κB pathway through phosphorylation and promotion of I-κBα degradation in liver damage induced by APAP. 6 Therefore, the PI3K/AKT signaling pathways–mediated inflammation and apoptosis were hypothesized to act as a vital role in liver damage induced by APAP.
Until now, the treatment of APAP poisoning mainly uses N-acetylcysteine (NAC) to detoxify. However, NAC has some shortcomings, such as certain gastrointestinal reactions and long administration time. 7 Thus, exploring the possible molecular mechanism of APAP-induced liver damage and suitable treatment of toxic drugs is meaningful.
Dihydromyricetin (DHM) is a kind of white powder soluble in ethanol, methanol, and water, which is a flavonoid isolated from Ampelopsis grossedentata. It has been shown to have a variety of pharmacological properties, such as anti-inflammatory, antioxidant, anti-tumor, anti-hypertensive, and prevent atherosclerosis effects, and has fewer side effects. 8 Researchers have proved that DHM could obviously inhibit TNF-α expression to suppress inflammation. 9 It has been demonstrated that DHM exerted protective effect against carbon tetrachloride-induced hepatotoxicity in mice. 10 Researchers have demonstrated that DHM improves endothelial dysfunction in mice by inhibiting oxidative stress. Massive studies have proved that DHM could suppress the TNF-α/NF-κB signaling pathway in colorectal cancer cells, 11 breast cancer cells, and ovarian cancer cells. 12 At present, there are some reports on the mechanism of DHM in vitro. However, there are few studies on its anti-inflammatory, anti-oxidant, and anti-tumor effects in vivo, and we are still not clear about its mechanism in vivo. Therefore, it is very necessary to study whether DHM exerts its hepatoprotective effect in vivo.
At this research, the pharmacokinetics and function of DHM as a hepatoprotective drug in suppressing liver damage after APAP treatment were studied in mice. We found that DHM had a hepatoprotective effect on APAP-induced hepatotoxicity, which was shown by suppressing oxidative stress and inflammatory responses, and reducing hepatocyte apoptosis by activating the PI3K/AKT signaling pathway.
Materials and Methods
Reagents and Antibodies
DHM (purity > 97.5%) was purchased from Jilin University (Jilin, China). APAP was acquired from Macklin Industrial Co., Ltd (Beijing, China). All the biochemical kits (including alanine aminotransferase (ALT) and malondialdehyde (MDA), etc) were obtained from Sigma company (USA). TNF-α, IL-1β, TUNEL apoptosis and Hoechst 33 258 kits were obtained from the R&D (USA). Immunofluorescence detection kit was obtained from Boxbio science & Technology Co., Ltd (Beijing China). All primary and secondary antibodies used in this study were purchased from Abcam (USA). No reference hepatoprotective agent like silymarin was used in this study.
Animals
Six- to eight-week-old male ICR mice were obtained from JILIN University, with Certificate No. JLUN (JI) 2019-0005. The mice were acclimated to the environment for about a week before the start of the experiment. All animal experiments were strictly in accordance with the “Regulations on the Management of Laboratory Animals” (license No. ECLA-JASTC-18065) issued by the Experimental Animal Ethics Committee of Jilin Agricultural Science and Technology College. The animals were separated into four groups at random (n = 15): (1) Control, (2) 250 mg/kg APAP, (3) 250 mg/kg APAP + 10 mg/kg DHM, (4) 250 mg/kg APAP + 20 mg/kg DHM.
Animals in group 3 and 4 were pretreated with different concentrations of DHM (10 or 20 mg/kg) for one week. On the seventh day, mice were treated with APAP (250 mg/kg) after 1 h of DHM pretreatment. After 24 h, all mice were sacrificed by cervical dislocation immediately, and the serum of mice were separated at 2000 rpm for 8 min and stored at −80 °C for following experiments. Meanwhile, liver and spleen tissues were collected and weighted to calculate the organ indices, and then part of the liver tissues were fixed in 10% formalin solution (m/v) for making tissue sections, and the rest of the liver samples were preserved in the refrigerator at −80 °C.
Pharmacokinetic Assay
Five mice of groups 3 and 4 were taken to examine the pharmacodynamics of DHM and fasted for 12 h before the experiment; 200 μL blood was collected from the tail vein and centrifuged at 2000 rpm for 8 min at 0.0, 0.08, 0.17, 0.25, 0.33, 0.50, 0.75, 1.0, 2.0, 4.0, 6.0, 8.0, and 12.0 h after administration. The serum was separated from the supernatant and stored in a refrigerator at −80 °C.
To establish an LC-MS/MS method for the determination of DMH in mice plasma, the Phenomenex Lunar chromatographic column was selected to separate the liquid chromatography (LC). The mobile phase includes 0.1% formic acid aqueous solution (A) and acetonitrile (B). The gradients and parameters were set according to the reference. 13 Electrospray ionization (ESI) and multiple reaction selective ion monitoring (MRM) were used for sample detection. The ion source temperature was 150 °C, the dissolution temperature was 200 °C, the capillary voltage was 2.65 kV, and the extractor voltage was 5500 V. The purity of nitrogen was ≥99.99%. Desolvent gas and cone air flow were set to 700 L/h and 50 L/h, respectively, and mass scanning range was 50 ∼ 400 m/z.
Biochemical Marker Assay
According to the kit protocol, the serum was obtained at 2000 rpm for 8 min for subsequent experiments. The levels of aspartate aminotransferase (AST), ALT, glutathione (GSH), and MDA were quantified by the commercial detection kits of Sigma company. The levels of TNF-α and IL-1β were detected by enzyme-linked immunosorbent assay (ELISA) kits of R&D company.
Histological Analysis
In order to explore the changes in liver tissue, fresh liver samples were fixed in 10% formaldehyde for one day. Then the liver sample was embedded in paraffin and made into sections (Leica RM2125 RTS). Afterward, the sections were detected by hematoxylin and eosin (H&E) staining and Hoechst 33 258 staining (10 μg/mL), respectively. Meanwhile, TUNEL apoptosis detection kit was used for histopathological analysis. The Image-Pro plus software (version 6.0, USA) was used to evaluate the degree of liver apoptosis.
Immunofluorescence Assay
According to the operation instructions, Boxbio science & Technology immunofluorescence detection kits (Beijing, China) were used to perform the immunofluorescence assay to assess the expression levels of CYP2E1 and 4-HNE in APAP-induced hepatotoxicity. The ImageJ software (Media Cybernetics, USA) was used to catch the signals.
Western Blot Analysis
Protein samples were prepared to homogenize by RIPA lysate from mice liver tissues which were deposed in −80 °C, and BCA protein concentration determination kit was used to detect the protein concentration. 20 μg/lane proteins were separated using 10% SDS-PAGE and transferred onto a PVDF membrane, and the membranes with primary antibody recognizing p-IKKα/β (1:1000, Abcam), IKKα (1:1000, Abcam), IKKβ (1:1000, Abcam), p-I-κBα (1:1000, Abcam), I-κBα (1:1000, Abcam), p-NF-κB (1:1000, Abcam), NF-κB (1:1000, Abcam), p-PI3K (1:1000, Abcam), PI3K (1:1000, Abcam), p-AKT (1:1000, Abcam), AKT (1:1000, Abcam), Bax (1:1000, Abcam) and Bcl-2(1:1000, Abcam) were incubated at 4 °C overnight, respectively. GAPDH was used as an internal reference, and the membrane with secondary antibody (HRP-conjugated sheep anti-mouse IgG, 1:1000, Abcam) was incubated at room temperature for 2 h, respectively; then, the ECL reagent (Genshare, China) was added to the membrane. Finally, ImageJ software was carried out to catch the signals.
Statistical Analysis
Statistical analysis was carried out by GraphPad 8.0 software. The data were analyzed using one-way analysis of variance (ANOVA) or t-test analysis. The data were presented as mean ± SD. P < .05 was served as the cutoff for the significant difference.
Results
Metabolic Expression of DHM in Mice
The high sensitivity and selectivity of DHM can be obtained in the negative ion acquisition mode of the ESI source. As shown in Figure 1B, the tuned DHM mass transition was m/z 319.0-192.9, which was determined as the best DHM sensitivity and peak range. In order to achieve rapid separation of DHM in mice blood, gradient elution was used. DHM (10 ng mL−1) standard solution was added to plasma as a control, and the changes of DHM in the blood of mice after administration were measured. The results showed that the retention time of DHM was about 4.00 min, and the total running time was 6.4 min (Figure 1C1, 2, 3). As shown in Figure 1D, the concentration of DHM in mice was increased in the veins of mice about at 5 min after gavage and maximum concentration of DHM was reached in mice of DHM-administered (10 and 20 mg/kg) groups about at 1 h, while the DHM concentration in plasma was reached half of the maximum value at 2 h, and almost reduced to one-fourth of the highest concentration at 4 h. The results suggested that data analysis could verify the efficacy of medicinal in about 6-8 h.

Pharmacokinetic analysis of DHM in the serum of mice. (A) Molecular structure and plausible fragmentation pathways of DHM. (B) The mass spectrum of DHM, molecular ion peaks at m/z 319.0 [M-H]+, fragmentation peaks at m/z 300.9, 192.9, and 124.9. (C1) Blank plasma. (C2) Blank plasma spiked with DHM. (C3) Plasma samples after intragastric administration of DHM. The retention time is 4 min. (D) The plasma concentration-time curves of DMY(×± s, n = 5). Abbreviation: DHM, dihydromyricetin.

Effects of DHM on serum transaminases and oxidative stress indices of the liver in liver damage induced by APAP. (A) The expression level of ALT in serum; (B) the expression level of AST in serum; (C) GSH content; (D) MDA content; (E) CYP2E and (F) 4-HNE immunofluorescence at 400× magnification; (G) changes of CYP2E1; and (H) 4-HNE of relative fluorescence intensity. (Mean ± S.D, n = 8. *P < .05, **P < .01 indicated all groups compared with control group; #P < .05, ##P < .01 indicated all groups compared with APAP group.) Abbreviations: 4-HNE, 4-hydroxy-2-nonenal; ALT, alanine aminotransferase; APAP, acetaminophen; AST, aspartate aminotransferase; CYP2E1, Cytochrome P450 2E1; DHM, dihydromyricetin; MDA, malondialdehyde.

DHM reduces hepatocyte necrosis in mice. (A) Hematoxylin-eosin (H&E) staining of liver tissue (400×). The yellow arrows suggest the apoptotic cavitation of hepatocytes; (B) Analysis of the degree of necrosis of peripheral vein cells in liver tissue. 0 = no damage, 1 = 0–10%, 2 = 11%–25%, 3 = 26%–45%, 4 = 46%–75%, 5 = > 75%. Values are expressed as mean ± S.D, n = 8. *P < .05, **P < .01 indicated all groups compared with control group; #P < .05, ##P < .01 indicated all groups compared with APAP group. Abbreviations: APAP, acetaminophen; DHM, dihydromyricetin.
Effects of DHM on Liver Indices and Serum Transaminases in APAP-Induced Mice
The weight of mice after APAP 250 mg/kg administration was lower than that of the control and DHM groups (P < .05), and the liver and spleen indices were obviously higher than those of the control and DHM groups (P < .05 or P < .01) (Table 1), indicating that DHM has a positive activity on the liver of mice. DHM-pretreatment can regulate APAP-induced liver damage and improve the body's recovery. ALT and AST levels in serum were used to evaluate the degree of liver injury of the mice in APAP-induced and DHM-pretreatment groups (Figure 2A and B). The levels of ALT and AST in the serum of mice after APAP stimulation were significantly increased compared with the control group (P < .01), and the results showed that the mice model of liver injury APAP-exposure (250 mg/kg) was successfully established. However, the levels of ALT and AST in the serum of mice treated with two different concentrations of DHM were obviously reduced (P < .01), and a better effect was examined in the high-dose DHM-pretreatment group (20 mg/kg).
Effects of DHM on Body Weights and Organ Indices in Mice.

Note. n = 8, Mean ± S.D; *P < .05, **P < .01 versus control group; #P < .05, ##P < .01 versus APAP group.
Abbreviations: APAP, acetaminophen; DHM, dihydromyricetin.
Effects of DHM on Oxidative Stress Indices of Liver
The GSH level in the liver of the APAP-exposure group was significantly reduced compared with the control group (P < .05), while they were significantly increased after DHM treatment (P < .05) (Figure 2C). The MDA level in the liver of the APAP-exposure group was obviously increased compared with the control group (P < .05), which suggested that APAP could cause the damage of OS. Similarly, the DHM-administered group was able to significantly alleviate the APAP-induced increasing of MDA levels (P < .05) (Figure 2D). These results demonstrated that DHM might relieve OS damage of liver induced by APAP. To deeper investigate whether OS is related to the liver damage caused by APAP, immunofluorescence technique was used to detect the OS indicators levels of CYP2E1 and 4-HNE in liver tissue. As shown in Figure 2E and F, stronger CYP2E1 and 4-HNE fluorescence intensities were observed in liver tissue after one day of APAP induction. However, after DHM-pretreatment (10 and 20 mg/kg), the increasing trends were significantly suppressed, especially in the high-dose group (Figure 2G and H) (P < .01). We can conclude that DHM can at least partially alleviate the liver damage caused by APAP by inhibiting OS.
DHM Reduces Necrosis of Hepatocytes Caused by APAP-Induced Liver Injury
Full hepatocytes and clear hepatic lobes in the liver tissue can be observed without showing histopathological changes in the normal liver tissue group. In the APAP-induced liver injury group, hepatocyte cavitation was extremely prominent throughout the entire visual field of the microscope. Meanwhile, dense condensation of hepatocyte nuclei could be clearly observed. The microstructure of the DHM pretreatment (20 mg/kg) high-dose group was similar to that of the normal group, with occasional hepatocyte degeneration and necrosis (Figure 3A). In the DHM pretreatment (10 mg/kg) low-dose group, hepatocyte cords were not clear enough, and hepatocytes were slightly cavitated, which was basically normal. The necrotic score of histopathological changes was shown in Figure 3B, which indicated that DHM pretreatment might reduce the liver pathological changes induced by APAP (P < .01).
DHM Inhibits the Apoptosis of Hepatocyte
To explore the relationship between DHM pretreatment and APAP-induced hepatocyte apoptosis, we used TUNEL staining and Hoechst 33 258 staining to investigate the degree of apoptosis of hepatocyte nuclei in liver tissue. It was observed that almost all liver cells in the APAP group had significant hepatocyte nuclear division and coagulation. However, in DHM (10 mg/kg) pretreatment of APAP group, a small number of hepatocytes developed nuclear fragmentation and condensation. DHM (20 mg/kg) pretreatment group exhibited normal contours and the regular homogeneous fluorescence intensity (P < .01) (Figure 4A and B). In addition, the results shown in Figure 4A and C were as expected, with almost no positive cell expression in the normal control group. In contrast, massive TUNEL-positive cell expression occurred in the APAP group, while the pretreatment of DHM (10 mg/kg and 20 mg/kg) could reverse this result and exerted a certain dose dependence (P < .01).
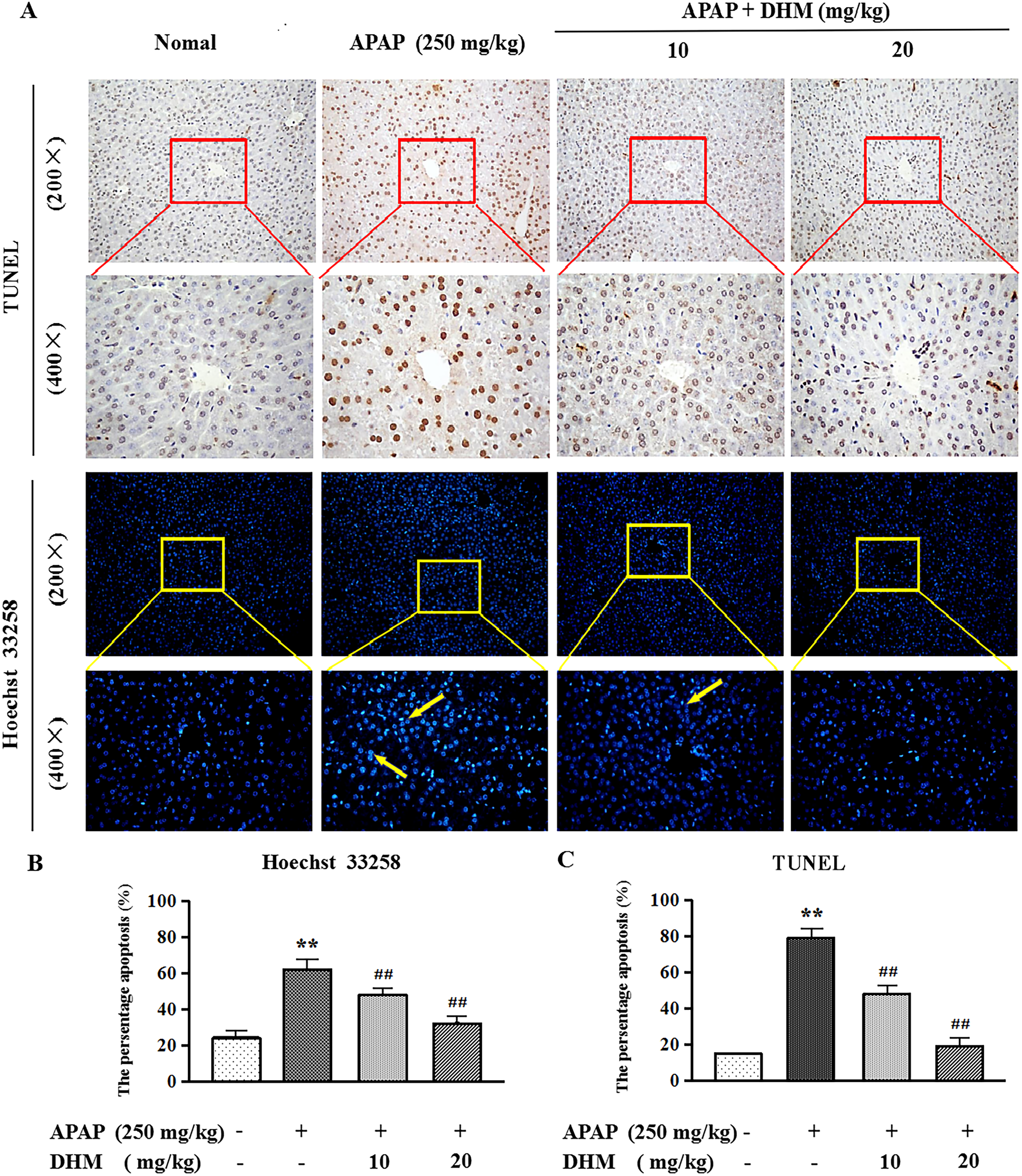
DHM inhibits the apoptosis of hepatocyte. (A) Liver tissue TUNEL (400×) and Hoechst 33 258 (400×) staining. Yellow arrows suggest apoptotic cells. Percentage of (B) Hoechst 33 258 and (C) TUNEL positive cells. Values represent mean ± SE (n = 8). *P < .05, **P < .01 indicated all groups compared with control group; #P < .05, ##P < .01 indicated all groups compared with APAP group. Abbreviations: APAP, acetaminophen; DHM, dihydromyricetin.
To further explore the effect of DHM pretreatment on hepatocyte apoptosis, the expression levels of PI3K/AKT signaling pathway-related proteins and downstream apoptosis-related factors (such as Bax and Bcl-2) in all groups were determined. The western blot results showed that the expression levels of p-PI3K and AKT were significantly reduced after APAP treatment (P < .05 or P < .01), while the DHM pretreatment group (10 and 20 mg/kg) could partially reverse these expression (Figure 5C and D). In addition, the results showed that treatment with APAP could obviously decrease the expression of Bcl-2 and increase the expression of Bax in liver tissue. Similarly, DHM pretreatment groups (10 and 20 mg/kg) can partially reverse this expression (P < .05 or P < .01).

Effects of DHM on serum TNF-α and IL-1β levels and liver tissue–related protein expression in mice in liver damage induced by APAP. (A) TNF-α levels. (B) IL-1β levels. (C) Western blot analysis of p-PI3K, PI3K, p-AKT, AKT, Bax, and Bcl-2. (D) Quantification of relative protein expression of p-PI3K, PI3K, p-AKT, AKT, Bax, and Bcl-2. (E) Western blot analysis of phosphorylated and total IKKα, IKKβ, IκBα, and NF-κB. (F) Quantification of relative protein expression of IKKα, IKKβ, IκBα, and NF-κB. Values represent mean ± SD (n = 8). *P < .05, **P < .01 indicated all groups compared with control group; #P < .05, ##P < .01 indicated all groups compared with APAP group. Abbreviations: APAP, acetaminophen; DHM, dihydromyricetin.

Molecular mechanism of DHM inhibiting PI3K/AKT pathway. Abbreviation: DHM, dihydromyricetin.
DHM Inhibits Hepatocyte Inflammation
To verify the correlation between APAP-induced liver toxicity and inflammatory response, the contents of the pro-inflammatory cytokines TNF-α and IL-1β in serum were determined. We found that the contents of TNF-α and IL-1β in serum of APAP-induced mice were increased than those in the control group (P < .01) (Figure 5A and B). However, the contents of TNF-α and IL-1β were significantly reduced in DHM pretreatment groups (10 and 20 mg/kg) (P < .05 or P < .01).
To evaluate liver protection-related signaling molecules in liver samples, western blot analysis was performed to detect the expression levels of phosphorylated and non-phosphorylated forms of NF-κB signaling pathway–related proteins. The expression levels of p-IKKα/β, p-NF-κB, and p-IκBα in liver samples were generally increased due to APAP induction, but all protein data showed a downward trend after treatment with DHM (Figure 5E and F). Therefore, it was shown that DHM might suppress the activation and expression of the NF-κB signaling pathway and significantly down-regulate the expression of these proteins (P < .05 or P < .01).
Discussion
APAP poisoning has always been one of the main causes of drug-induced liver disease. Due to limited treatment options, injuries and related adverse events remain a global problem. 14 Massive studies indicate that APAP overdose is the most universal cause of severe hepatotoxicity. Its main characteristics are extensive necrosis of liver cells, elevated ALT, AST, TNF-α, and IL-1β, GSH depletion, the increasing of MDA, CYP2E1, and 4-HNE.1, 4, 15 Liver damage induced by APAP in mouse model is often carried out to investigate the potential molecular mechanisms of hepatotoxicity induced by APAP.
In recent years, natural herbal medicines have become a research hotspot in the development of new disease treatment drugs. Aeginetia indica and celastrol have been shown to be potential treatments for hepatotoxicity induced by APAP.16, 17 DHM is a flavonoid isolated from ampelopsis. Previous studies have proved that DHM has a certain protective effect on chemical and alcoholic liver damage. DHM can regulate the expression of target genes induced by TNF-α by suppressing NF-κB activation. 18 Thus, we explored whether DHM could suppress the inflammation and oxidative stress in the liver induced by APAP and the corresponding molecular mechanism.
At this research, we first investigated the metabolic expression of DHM in mice, and we found that DHM could be detected in mice serum about five minutes after gavage and reached its highest level in about 1 h. To explore the alleviating effect of DHM on liver damage induced by APAP, ALT and AST levels in serum were used to evaluate the degree of liver injury of the mice in APAP-induced and DHM-pretreatment groups, and the results demonstrated that DHM could alleviate the increasing of ALT and AST levels in mouse serum caused by APAP induction.
In this report, the oxidative stress response of liver tissue caused by APAP exposure was reflected in the depletion of GSH, the increase in MDA, 4-HNE, and CYP2E1 levels, which are in line with the previous studies.19, 20 The results showed that pretreatment with different doses of DHM reversed the exhaustion of liver GSH and the increase in MDA, 4-HNE, and CYP2E1 levels induced by APAP. Long-term or over-injection of APAP can cause hepatic sinusoidal macrophages to be activated and release a variety of pro-inflammatory cytokines, such as IL-1β and TNF-α. 21 The ELISA results demonstrated that DHM could decrease the content of IL-1β and TNF-α induced by APAP. To further verify the above experimental results, H&E, Tunel, and Hoechst 33 258 staining analysis were used to clearly and intuitively observe the liver damage caused by APAP and the protective effect of DHM pretreatment on the liver, indicating that DHM could protect liver cells from liver damage induced by APAP. In summary, the experimental results showed that the protective mechanism of DHM on APAP-induced liver toxicity may depend on inhibiting oxidative stress and inflammation. The antioxidant activity of DHM is closely associated with its chemical structure, including the quantity and positions of the hydroxy groups. 22 The ortho-trihydroxyl group (hydroxyl in the C-3’, 4’, and 5’ position) in B-ring of DHM has been demonstrated to have strong effect against free radicals and provide an antioxidant defense.23, 24
The PI3K/AKT signaling pathway is associated with the process of cell apoptosis, and the process was regulated by Bcl-2 and Bax. Bcl-2 and Bax were the downstream target proteins of the PI3K/AKT signaling pathway. 25 To further explore the relationship between apoptosis and liver damage induced by APAP, western blot analysis was performed to assess the expression levels of Bcl-2 and Bax. We found that the PI3K/AKT pathway APAP-induced liver injury was successfully inhibited after 7 days of DHM pretreatment, and the protein levels of Bax and Bcl-2 were close to that of the control group. DHM treatment successfully inhibited the expression of Bax and increased the expression of Bcl-2.
In this study, the expression of PI3K/AKT-mediated NF-κB pathway–related proteins was detected to further explore the mechanism of DHM inhibiting liver damage induced by APAP. The transcriptional activity of the NF-κB pathway is regulated by PI3K/AKT through phosphorylation and promotion of I-κB degradation. Numerous studies have shown that the NF-κB pathway has become one of the most typical signaling pathways in various disease (including liver and inflammatory diseases) mechanisms.14, 26 Western blot analysis suggested that DHM can reduce the expression of IKKα, IKKβ, and I-κBα and inhibit the activation of NF-κB. Therefore, the potential mechanism of DHM protection against liver damage induced by APAP may be related to blocking the activation of the NF-κB pathway by inhibiting the conduction of the PI3K/AKT signaling pathway.
Conclusions
In summary, DHM could alleviate APAP-induced liver damage in mice by suppressing the oxidative stress in liver tissue, improving the inflammatory response and decreasing hepatocyte apoptosis. Its mechanism of action was mediated by the PI3K/AKT signaling pathway.
Footnotes
Author Contribution
Jingli Gong and Juntao Gao contributed to the study conception and design. Jingli Gong and Xiaomei Zhang performed the data collection. Xiaomei Zhang analyzed the data. Huan Yu and Huifeng Zhang performed the statistical analysis. Jingli Gong and Huan Yu drafted the manuscript. Juntao Gao revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jilin Medical University Doctoral Foundation Project (grant number JYBS2021020LK).
