Abstract
Cardiovascular disease (CVD), a leading cause of morbidity and mortality, is among the most prevalent health problems worldwide and effective strategies for its prevention and treatment are urgently required. In this regard, increasing research has demonstrated that natural drugs offer antihypertensive, antiatherosclerotic, and cardioprotective activities, and many are applied widely for the treatment of CVD and its manifestations such as myocardial infarction, peripheral vascular diseases, and coronary heart disease. Natural drugs have significant advantages in the treatment of CVD due to their efficacy and safety profiles. Saponins are an important class of active components of plant natural products and play an important role in the treatment of CVD. This review covers the most up-to-date information on saponins concerning their cardioprotective effects and mechanisms of action.
According to World Health Organization statistics, cardiovascular disease (CVD) is responsible for an estimated 17.9 million deaths each year. 1 CVD is a group of diseases that includes heart failure (HF), myocardial infarction (MI), cardiac ischemia (CI), and cardiac hypertrophy (CH). Due to the rising prevalence of consequent mortality and disability, more effective treatments for CVD are urgently required. Recently, herbal medicine therapy has been increasingly regarded as an alternative and promising strategy for the treatment of CVD. Saponins are an important class of active compounds and are widely distributed in nature, particularly in fungi, ferns, plants, animals, and marine life. Numerous studies have shown that saponins extracted from various Chinese herbal medicines have cardioprotective effects, offering great potential for the development of new drugs for CVD therapy. This review will focus on the latest research into the cardioprotective effects of saponins and their underlying molecular mechanisms.
Saponins, glycosides of triterpenes and steroids, are widely distributed in higher plants and medicinal plants such as notoginseng (Panax notoginseng (Burkill) F.H.Chen), ginseng (Panax ginseng C.A.Meyer), and milkvetch (Astragalus propinquuos Schischkin) are rich sources of these compounds. In the last 2 decades, extensive research into saponins has uncovered a wide range of physiological activities including antitumor, glucose-lowering, immunoregulatory, antiviral, and cardioprotective effects. 2
Panax notoginseng Saponin Cardioprotective Effects
Panax notoginseng saponins (PNS) are the main active class of components of this species. Among these, ginsenosides Rb1, Rg1, Rd, and notoginsenoside R1 are the major constituents. 2 The cardioprotective effects of PNS have demonstrated potential in the treatment of MI, CH, and HF.
Panax notoginseng Saponins and Cardiac Hypertrophy
Notoginsenoside R1 has been found to be effective in inhibiting isoproterenol (ISO)-induced CH, manifesting increased cardiomyocyte viability, decreased ANP, β-MHC, TNF-α, IL-6, and IL-1β mRNA levels, increased I-κBα protein levels, inhibition of p65 phosphorylation, and decreased Caspase-3 activity. Its underlying mechanism may be associated with attenuation of the inflammation response via inhibition of the NF-κB signaling pathway, thereby preventing ISO-induced CH. 3 Energy metabolism disturbance is found in various CVD, such as coronary heart disease, diabetic cardiomyopathy, and HF. Luo Kai et al have provided convincing evidence that PNS could improve cardiac function, regulating energy metabolism by enhancing fatty acid β-oxidation and glucose oxidation in ISO-induced CH. 4 In addition, notoginsenoside R1 is closely correlated with the inactivation of CC chemokine receptor 2 to blind the proinflammatory monocyte subset and interfere with the subsequent activities of various inflammatory factors to protect against CH. 5 In summary, PNS are promising compounds for the treatment of cardiac hypertrophy through multiple signaling pathways.
Panax notoginseng Saponins and Myocardial Ischemia Reperfusion Injury
Recent studies have shown the potential role of PNS in myocardial ischemia-reperfusion injury (MIRI). Gao et al have reported that PNS can reduce ISO-induced acute MIRI in rats as evidenced by a reduction in the release of damaged myocardial enzymes, an increase in SOD activity in serum and myocardial tissue, and a decrease in MDA. 6 Among the cardioprotective pathways involved, activation of the phosphatidylinositol 3-kinase (PI3K)/AKT signaling pathway has been implicated, with the consequent reduction in oxidative stress and inflammatory response, alleviation of myocardial apoptosis, and improvement in cardiac function in I/R rats. 7 In addition, Wang et al showed that pretreatment with PNS significantly reduced the expression of LC3-II and Beclin-1 while elevating the expression of p62, suggesting that PNS ameliorates MIRI by regulation of autophagy.8,9 Furthermore, gene expression levels obtained by RNA-Seq reveal that PNS could reactivate PI3K/AKT and AMPK signaling pathways and protect cardiomyocytes from oxygen-glucose depletion-induced apoptosis. Moreover, RNA-seq demonstrated that the expression levels of many noncoding RNAs, such as miRNAs and lncRNAs, were significantly affected after PNS treatment, suggesting that PNS could protect cardiomyocytes through regulating noncoding RNAs. 10
Panax notoginseng Saponins and Heart Failure
The protective effects of PNS against the development of HF were investigated in a Rattus model, indicating cardiomyocyte apoptosis inhibition, downregulation of p-ERK, p-JNK, and p-p38 protein expression and inhibition of the secretion of serum inflammatory factors. 11 Myocardial infarction remains the most common cause of HF worldwide. Wang et al demonstrated that PNS exerted a cardioprotective effect in acute myocardial infarction. RNA-seq (transcriptome profiling) revealed that PNS and its main constituent ginsenoside Rg1 upregulated angiogenesis and autophagy-related genes. 12 Moreover, PNS enhanced glucose deprivation-induced autophagy through phosphorylation of AMPK Thr172 and CaMKII Thr287 in cardiomyocytes. 12 In research by Chen et al, PNS upregulated the expression of PPARα, RXRα, and PGC1α to protect against HF in rats. Interestingly, the addition of the PPARα inhibitor GW6471 canceled the beneficial effects of PNS indicating PPARα as a possible target of PNS for HF treatment. The combined evidence reveals the cardioprotective effects of PNS against HF. 13
In summary, the activity of Panax notoginseng saponins against CVD through multiple pathways presents a significant opportunity for the development of cardioprotective and antihypertrophic drugs (Figure 1).

Effects and mechanisms of Panax notoginseng Saponins on heart diseases.
Panax ginseng Ginsenoside Cardioprotective Effects
As an ancient Chinese herbal medicine, Panax ginseng has been used both as food and medicine for nutrient supplements and disease treatment. It contains many active components including ginsenosides Rd, Rg1, and Rg3, collectively known as ginsenosides, of which more than 30 have been identified to date. Recent studies have shown that ginsenosides are effective in preventing and treating a variety of CVD including MI, CH, and HF.
Panax ginseng Ginsenosides and Cardiac Hypertrophy
Various studies have revealed that ginsenosides exert an antihypertrophic effect. One study showed that ginsenoside Rd contributes to the attenuation of CH in vivo and in vitro, significantly improving pressure overload-induced contractile dysfunction, fibrosis, CH, inflammation, and oxidative stress in mice. 14 Ginsenoside Rd also inhibited the phenylephrine (PE)-induced a hypertrophic response in cultured cardiomyocytes. Similar to ginsenoside Rd, ginsenoside Rg1 also protects against CH induced by Ang II in neonatal rat cardiomyocytes. Ginsenoside Rg1 can improve the energy metabolism of hypertrophic cardiomyocytes by regulating the SIRT1/PGC-1α pathway. 15 In addition, ginsenoside Rg3 has a prominent role in anti-inflammation. A recent study found that ginsenoside Rg3 prevents Ang II-induced myocardial hypertrophy, inactivating NLRP3 inflammasome and oxidative stress by modulating the SIRT1/NF-κB pathway. 16 Thus, ginsenosides have demonstrated antihypertrophic effects.
Panax ginseng Ginsenosides and Myocardial Ischemia-Reperfusion Injury
Ginsenoside Rb1 and Rb2 have cardioprotective effects against MIRI. Ginsenoside Rb1 improves impaired autophagic flux, 17 activates the phosphorylation of mTOR, 18 and decreases levels of ROS by inhibiting mitochondrial complex I. 19 Ginsenoside Rb1 also increases ATP content and decreases ROS production, thereby attenuating hypoxia/reoxygenation-induced apoptosis. Ginsenoside Rb2 alleviates MIRI in rats by activating SIRT1 expression, reducing myocardium inflammation, and alleviating oxidative stress. 20 Additionally, other ginsenosides, such as Re and Rg1, have also been found to play a protective role against MIRI. Ginsenoside Re pretreatment has a positive protective effect on the myocardium in rats with acute myocardial ischemia, possibly due to the activation of the JAK2/STAT3 signaling pathway. 21 Ginsenoside Rg1 ameliorates MIRI with a proposed mechanism involving inhibition of myocardial apoptosis and modulation of energy metabolism through binding to RhoA. 22 Other studies have shown that ginsenoside Rg1 robustly inhibits the release of Cytc to regulate myocardial apoptosis, the regulatory process being driven by up-regulation of eNOS protein. 23 Ginsenoside Rg1 has also been found to improve myocardial arrhythmia after ischemia/reperfusion by inhibiting PPAR-γ-mediated inflammation. 24 These findings provide new insights for the application of ginsenosides against MIRI.
Panax ginseng Ginsenosides and Heart Failure
The role of ginsenosides in the treatment of HF has also been reported. Evidence has emerged that disturbance of mitochondrial energy metabolism is implicated in the development of HF. Ginsenoside Rb1 can improve mitochondrial energy metabolism in HF rats. 25 In further research, ginsenoside Rb1 demonstrated anti-HF activity by inhibiting cardiomyocyte autophagy of rats through the regulation of Rho/ROCK and PI3K/mTOR pathways. 26 There is also evidence that ginsenoside Rg3, Rg1, and Re exert anti-CHF effects. Both ginsenoside Rg3 and Rg1 enhance the activity of SERCA2a to improve damaged Ca2+ homeostasis in HF.27,28 Ginsenoside Re can ameliorate isoproterenol-induced myocardial fibrosis and HF by regulation of the TGF-β1/Smad3 pathway. 29 These studies support further investigation into ginsenosides as potential therapeutic agents against HF.
These findings indicate that several ginsenosides have important cardioprotective effects and potential against myocardial ischemia-reperfusion injury (Figure 2).
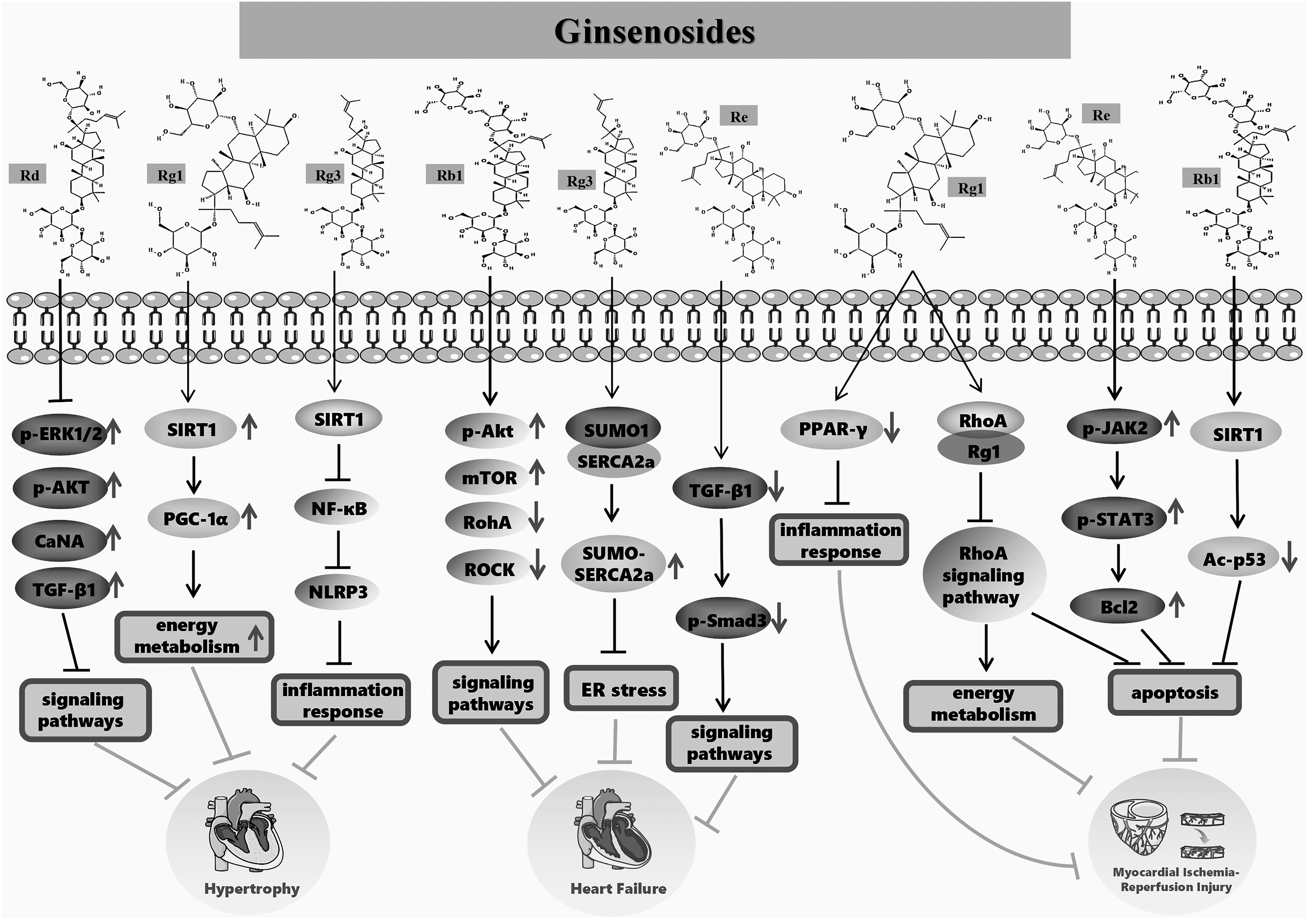
Effects and mechanisms of ginsenosides on heart diseases.
Astragalus Propinquuos Astragaloside IV Cardioprotective Effects
Astragaloside IV is an important active astragaloside component of Astragalus propinquuos and is prominent in studies into CVD. Recent reports indicate that astragaloside IV can protect against MIRI, inhibit myocardial hypertrophy and myocardial fibrosis, enhance myocardial contractility, and improve diastolic dysfunction.30–32
Astragalus Propinquuos Astragaloside IV and Cardiac Hypertrophy
The protective effect of astragaloside IV against CH in vivo or in vitro has been widely reported. A recent study has found that astragaloside IV prevents myocardial hypertrophy induced by mechanical stress via autophagy activation and inflammation reduction. 33 In addition, astragaloside IV inhibits oxidative stress to prevent cardiac remodeling, possibly mediated by activation of Sirt3 and NRF2/HO-1 pathways resulting in attenuation of cardiomyocyte hypertrophy. 13 Astragaloside IV could also enhance the suppressor of IKKε (SIKE), a negative regulator of the interferon pathway, and consequently ameliorate pathological CH in mice via inactivation of the TANK-binding kinase 1 (TBK1)/PI3K/AKT signaling pathway. 34 The above studies show that astragaloside IV can exert antihypertrophic effects through multiple pathways.
Astragalus Propinquuos Astragaloside IV and Myocardial Ischemia-Reperfusion Injury
Studies have uncovered various mechanisms for the protective effects of astragaloside IV against MIRI. Firstly, inhibition of autophagy has been increasingly recognized as an underlying mechanism, with studies revealing multiple signaling pathways. Astragaloside IV upregulated GATA-4 expression which enhanced the interaction between Bcl-2 and Beclin-1, subsequently increasing the expression of Bcl-2 and p62, thereby playing a protective role toward cardiomyocytes. 35 Li et al also reported that astragaloside IV regulates PINK1-Parkin-mediated autophagy and prevents MIRI in rats. Secondly, 36 astragaloside IV treatment protected against MIRI by inhibiting cardiomyocyte apoptosis. 37 Studies have shown that astragaloside IV alleviates MIRI in rats by regulating PI3K/AKT/GSK-3β signaling pathways. 38 It was also found that astragaloside IV not only regulates miR-21/PTEN to reduce the release of Caspase-3, Bcl-2, and Cyt-C 39 but also mediates NRF2/HO-1 signaling pathways to attenuate MIRI.40,41 In addition, astragaloside IV inhibits endoplasmic reticulum (ER) stress to exert protective effects against MIRI. It has been reported that astragaloside IV protects cardiomyocytes from ER stress by reducing 2-DG-induced expression of GPR 78, GRP 94, and IRE1 and preventing mPTP opening. 30 In further research, astragaloside IV inhibits MIRI-induced ER stress of H9c2 cardiomyocytes through a mechanism involving the PERK pathway. 42 Moreover, astragaloside IV ameliorated MIRI by modulating succinate and lysophospholipid metabolism and scavenging ROS via the Nrf2 signal pathway. 43 Therefore, astragaloside IV could be a valuable candidate drug for treating MIRI.
Astragalus Propinquuos Astragaloside IV and Heart Failure
Accumulating evidence suggests that astragaloside IV can protect against HF with many mechanisms potentially involved. Recent studies have highlighted the function of astragaloside IV in promoting the translocation of nuclear factor-erythroid 2-related factor 2 (Nrf2), a key transcription factor responsible for regulating antioxidant defense systems, from the cytoplasm to the nucleus, thereby inducing the transcription of downstream antioxidant genes. 44 Therefore, it can be further speculated that astragaloside IV prevents HF by counteracting oxidative stress through the Nrf2/HO-1 pathway. In agreement, Feng et al also discovered that astragaloside IV attenuated Dox-induced cardiomyocyte apoptosis and mitochondrial dysfunction partially by activation of Nrf-2/HO-1 signaling. 45 Other research conducted by Liu et al disclosed that astragaloside IV decreased oxidative stress and mitochondrial damage, inhibited ventricular remodeling, and ultimately improved cardiac function by suppressing HF-induced Senp1-upregulation. 46 Additionally, astragaloside IV significantly improves HF in rats, possibly by activating the AMPK/PPARα signaling pathway to improve myocardial energy metabolism. 47 On the other hand, astragaloside IV may inhibit endothelial to mesenchymal transition mediated by the AKT/GSK3-β/SNAIL signaling pathway, thereby inhibiting myocardial fibrosis in HF.48,49 Therefore, astragaloside IV has been shown to display cardioprotection against HF. Taken together, these results provide evidence that astragaloside IV has an important pharmacological role in cardioprotection (Figure 3).

Effects and mechanisms of astragaloside IV on heart diseases.
Conclusions and Perspective
This review summarizes the role of 3 well-studied and representative plant saponins in the treatment of CVD, with their effects and mechanisms (Table 1). In the context of continuing research into the mechanisms underlying the treatment of CVD, the promising results summarized herein warrant further prospective research into saponins and their potential medical indications and clinical applications.
Effects and Mechanisms of Saponins on CVD.

Footnotes
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (82200550;81960662;82160064), the Applied Basic Research Programs of Yunnan Provincial Science and Technology Department (202101AT070154), the Open Research Foundation of Yunnan Key Laboratory of Bioactive Peptides in Yunnan Province (HXDT-2022-1), the program Innovative Research Team in Science and Technology in Kunming Medical University (CXTD202003), and the Yunnan Provincial Science and Technology Department (No 202105AF150015). We would like to thank MogoEdit (![]() ) for its English editing during the preparation of this manuscript.
) for its English editing during the preparation of this manuscript.
Authors’ Note
PNS, ginsenoside, and astragaloside IV structures were obtained from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). Parts of the figure were drawn using images from Servier Medical Art. Servier Medical Art by Servier is licensed under a Creative Commons Attribution 3.0 Unported License (![]() ).
).
Authors' Contributions
Data collection: GU Xuerong.Statistical analysis: JIA Yingnong, YANG Weimin. Analysis and interpretation of the data: SHEN Baochun, DU Guan-hua. Drafting of the manuscript: MA Xin, LU Jun. Critical revision of the manuscript: ZHENG Chang-Bo.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Yunnan Provincial Science and Technology Department, program Innovative Research Team in Science and Technology in Kunming Medical University, National Natural Science Foundation of China, Applied Basic Research Programs of Yunnan Provincial Science and Technology Department, Open Research Foundation of Yunnan Key Laboratory of Bioactive Peptides in Yunnan Province (grant number No 202105AF150015, CXTD202003, 82200550;81960662;82160064, 202101AT070154, HXDT-2022-1).
