Abstract
Background
Pyroptosis, a form of inflammatory programmed cell death, has recently emerged as a pivotal factor in the pathogenesis of myocardial ischemia/reperfusion (MI/R) injury. Despite its significance, effective therapeutic strategies targeting MI/R-induced pyroptosis remain elusive in current clinical practice. Previous studies have demonstrated the promising anti-inflammatory effects of Angelica sinensis polysaccharide (ASP) in the context of certain inflammatory disorders.
Objectives
We aimed to investigate the effects of ASP on pyroptosis in MI/R injury and elucidate the potential molecular mechanisms by combining transcriptomic analysis with complementary in vivo and in vitro experiments.
Materials and methods
H9c2 cells were used to establish a hypoxia/reoxygenation (H/R) model, and MI/R injury was induced in rats by ligating and releasing the left anterior coronary artery. Myocardial tissue samples were harvested for transcriptomic sequencing and bioinformatic analyses. Cardioprotective effects were evaluated through electrocardiography, echocardiography, and histological examination. Enzyme-linked immunosorbent assay (ELISA) was employed to quantify levels of inflammatory mediators. Biochemical assays were conducted to assess myocardial injury biomarkers and Caspase-1 activity. Western blotting was performed to analyze the protein expression levels of Fibronectin 1 (FN1), Nuclear factor kappa B (NF-κB) p65, phosphorylated NF-κB p65 (p-NF-κB p65), Nod-like receptor protein 3 (NLRP3), and Gasdermin-D (GSDMD).
Results
Pretreatment with ASP conferred potent cardioprotective effects in a rat model of MI/R injury, as evidenced by significant attenuation of infarct size, myocardial enzyme levels, and ST-segment elevation on electrocardiography, alongside notable improvements in cardiac function. Transcriptomic profiling unveiled that differentially expressed genes modulated by ASP treatment were predominantly implicated in the pyroptosis-elicited inflammatory response. Concordantly, both in vivo and in vitro experiments substantiated that ASP treatment effectively attenuated MI/R-induced pyroptosis, as manifested by diminished levels of pyroptosis-related indicators, encompassing the proportion of TUNEL-positive cells, Caspase-1 activation, GSDMD cleavage, and the liberation of pro-inflammatory cytokines. Further mechanistic investigations revealed ASP inhibition of the FN1/NF-κB/NLRP3 signaling pathway.
Conclusions
Our investigation, leveraging system-level transcriptomic profiling, demonstrates that ASP confers cardioprotective effects against MI/R injury through the suppression of cardiomyocyte pyroptosis, culminating from downregulation of the FN1/NF-κB/NLRP3 pathway.
Introduction
Globally, acute myocardial infarction (AMI) is a significant contributor to mortality. 1 The condition typically arises from the abrupt obstruction of blood flow to the heart muscle due to the rupture or erosion of atherosclerotic plaques within the coronary arteries. While the prompt reestablishment of blood flow via percutaneous coronary intervention or thrombolysis is deemed the most effective strategy for salvaging the ischemic myocardium, reperfusion therapy may paradoxically culminate in MI/R injury. 2 This phenomenon contributes to adverse clinical outcomes, including infarct size extension, cardiac dysfunction, heightened risk of arrhythmias, and impaired coronary microvascular function.3,4 Due to the unclear pathophysiological mechanisms underlying MI/R injury, effective interventions for this condition are lacking in clinical practice. 3 Therefore, there is an urgent need to elucidate the molecular mechanisms underlying MI/R injury and develop effective therapeutic strategies.
Myocardial inflammatory response is pivotal in the development of MI/R injury, underscoring the importance of targeting the response as a fundamental approach in both preventing and treating MI/R injury.2,5 In recent studies, particularly those published in 2022 6 and 2023, 7 pyroptosis has been increasingly implicated as a crucial contributor to the amplification of inflammation and exacerbation of cardiac injury during MI/R. The initiation of pyroptosis in the context of MI/R injury is influenced significantly by the activation of inflammasomes, with the Nod-like receptor protein 3 (NLRP3) inflammasome being notably critical in the process. 8 The NLRP3 inflammasome consists of NLRP3, an apoptosis-associated speck-like protein-containing CARD (ASC), and pro-Caspase-1. 8 In the heart, the traditional activation sequence of the NLRP3 inflammasome unfolds through two distinct phases: initial priming followed by activation. 8 The priming stage is driven primarily by Toll-like receptor 4 (TLR4) recognizing pathogen-associated molecular patterns or damage-associated molecular patterns, which activates nuclear factor kappa B (NF-κB) signaling, ultimately leading to the increased expression of NLRP3 and other inflammasome components. The activation phase of the NLRP3 inflammasome complex is initiated through external and/or internal triggers, including fluctuations in intracellular potassium levels and the destabilization of lysosomes. The activated NLRP3 inflammasome mediates Caspase-1 activation, leading to the production of pro-inflammatory cytokines and cleavage of Gasdermin-D (GSDMD). 9 The cleavage of GSDMD (specifically, the N-terminal fragment of GSDMD, N-GSDMD) establishes pores in the cell membrane, facilitating subsequent expulsion of pro-inflammatory cytokines and ultimately triggering inflammatory cell death. 10 Therefore, targeting NLRP3-dependent pyroptosis is a promising strategy for mitigating MI/R injury.
Traditional Chinese medicine (TCM) has been demonstrated as an efficacious therapy against MI/R injury, with Angelica sinensis (Oliv.) Diels being one of the widely employed medicinal herbs. 11 Renowned as the “female ginseng”, the highly esteemed herb in TCM has been utilized extensively for its hematopoietic, analgesic, and blood circulation-promoting properties. 12 Beyond its traditional applications, A. sinensis has garnered significant interest in contemporary pharmacological research owing to its diverse bioactive compounds and broad therapeutic potential.13,14 A network pharmacology approach was used to identify the active compounds, therapeutic targets, and pharmacological mechanisms of A. sinensis in the treatment of AMI. 13 The investigation utilized herbal databases and bioinformatics tools to identify Angelica sinensis polysaccharide (ASP) as a pivotal active component of A. sinensis, whereby ASP confers the cardioprotective properties through antioxidant, anti-inflammatory, and anti-apoptotic mechanisms. 13 In vitro and in vivo experiments have validated the antioxidant and anti-apoptotic effects of ASP in mitigating MI/R injury.15,16 Extensive investigations have been conducted to elucidate the anti-inflammatory effects of ASP in some inflammatory disorders.17–19 For instance, studies have demonstrated the ability of ASP to effectively suppress the production of pro-inflammatory cytokines, such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α), in murine models of colitis. 17 Furthermore, ASP could mitigate joint inflammation associated with rheumatoid arthritis by modulating gut microbiome composition. 18 Notably, recent advances in 2023 within the field of biomaterials have demonstrated the potential of pH- and redox-responsive ASP-loaded nanoparticles to attenuate the release of pro-inflammatory cytokines and promote gut microbial homeostasis at sites of colonic inflammation. 19 While the anti-inflammatory properties of ASP have been previously reported, 20 its role in modulating inflammatory responses following MI/R injury and the potential involvement of NLRP3 inflammasome-mediated pyroptosis in its mechanism of action remain largely unexplored.
The present study explored the cardioprotective properties of ASP in the context of MI/R injury, conducting both in vivo experiments with MI/R-afflicted rats and in vitro studies on hypoxia/reoxygenation (H/R)-damaged H9c2 cells. Additionally, ASP's mechanisms of action were explored through comprehensive transcriptomic analysis. Subsequent experiments were carried out to confirm the critical mechanisms identified within ASP. In-depth investigations into the regulatory effects of ASP on inflammatory responses and its role in ameliorating MI/R injury can expand our knowledge of its therapeutic applications and uncover novel strategies for managing MI/R injury.
Materials and Methods
Preparation of ASP
ASP was obtained by hot-water extraction and ethanol precipitation as described previously.21,22 Briefly, the dried roots of Angelica sinensis from Minxian County (Gansu, China) were used as the raw material. The roots were first defatted with ethanol, followed by two 2-h extractions in boiling water (100 °C). The extracted solution was then concentrated, and a calcium hydroxide suspension was slowly added until flocs emerged. The resulting mixture was centrifuged to collect the supernatant. After adjusting the pH to 5–6, the supernatant was concentrated and precipitated with an equal volume of ethanol for 24 h. The precipitate was collected by centrifugation and dried in a constant-temperature oven to obtain the crude polysaccharide. To remove proteins and pigments, the crude polysaccharide was subjected to the freeze-thaw method. The decolorized polysaccharide solution was then dialyzed against distilled water and further purified using a Sephadex G-50 column. Finally, the purified polysaccharide solution was lyophilized to obtain ASP. The total sugar content of ASP, determined using the phenol-sulfuric acid method, was found to be greater than 90%.
Animals, Grouping, and Treatment
Sprague-Dawley male rats (240 ± 20 g) were obtained from the Experimental Animal Center of Lanzhou University (Lanzhou, Gansu, China). The animals were housed under controlled environmental conditions, with a temperature of 22 ± 2 °C, a 12-h light/dark cycle, and ad libitum access to water and standard rodent chow. The present study adhered to the Guide for the Care and Use of Laboratory Animals of the First Hospital of Lanzhou University. The study protocol was approved by the Animal Care Committee of the First Hospital of Lanzhou University. A flowchart of the experimental design is illustrated in Figure 1.

Experimental design flowchart. Sprague-Dawley rats were assigned randomly to the Sham, Myocardial ischemia/reperfusion (MI/R), or MI/R + Angelica sinensis polysaccharide (ASP) groups. Cardiac function, infarct size, and histological analyses were performed to evaluate the cardioprotective effects of ASP pretreatment against MI/R injury. Subsequent transcriptomic profiling was conducted on cardiac tissue samples, and bioinformatics analyses were performed on the transcriptomic data to identify potential therapeutic targets and signaling pathways modulated by ASP. In parallel, complementary in vitro experiments using H9c2 cardiomyocytes subjected to hypoxia/reoxygenation (H/R) injury and in vivo studies were conducted to further validate the mechanisms underlying the cardioprotective actions of ASP.
The rats were randomly divided into three groups: Sham, MI/R, and MI/R + ASP. The MI/R model was established following the procedure outlined in our previous publication, 23 involving the ligation and subsequent release of the left anterior descending coronary artery. The effectiveness of MI/R induction was confirmed by observing the ST-segment deviation on an electrocardiogram (Chengdu Technology Market, Chengdu, China). In the Sham group, rats were subjected a surgical protocol identical to that of the MI/R group, with the exception of coronary artery ligation. Based on our previous laboratory study, 13 rats in the MI/R + ASP group received a daily dose of 200 mg/kg ASP administered via a stomach tube for four weeks prior to MI/R modeling. Simultaneously, rats in both the Sham and MI/R groups were given equivalent volumes of distilled water through intragastric administration.
Echocardiographic Analysis
Cardiac function and structural parameters were assessed using the Vevo 2100 high-resolution ultrasound imaging system (VisualSonics, Toronto, Canada).
24
All measurements were performed by a blinded operator, and data were averaged from three consecutive cardiac cycles. Prior to the imaging procedure, animals were anesthetized with 2% isoflurane and positioned on a heated platform to maintain body temperature. Transthoracic echocardiography was performed in M-mode and B-mode using a linear array transducer. Left ventricular (LV) dimensions, including end-diastolic diameter (LVEDD) and end-systolic diameter (LVESD), as well as LV end-diastolic volume (LVEDV) and end-systolic volume (LVESV), were obtained from M-mode tracings. Fractional shortening (FS) and ejection fraction (EF) were calculated as indicators of systolic function, according to the following equations:
Triphenyltetrazolium Chloride Staining
Triphenyltetrazolium chloride (TTC) staining (Solarbio, Beijing, China) was used to assess myocardial infarct size. Following euthanasia, rat hearts were removed and subjected to a 30-min freezing process at −20 °C. Subsequently, the hearts were cut into 2-mm sections and immersed in a 2% TTC solution at 37 °C for 30 min. The sections were then preserved in 4% paraformaldehyde and captured digitally for analysis. An investigator, who was unaware of the treatment groups, measured the infarct sizes using ImageJ 1.53k software (National Institutes of Health, Bethesda, MA, USA).
Hematoxylin-Eosin Staining
Hematoxylin-Eosin (H&E) staining technique was used for assessing the histological structure of the myocardium. Initially, the cardiac tissues were fixed using 4% paraformaldehyde, followed by a dehydration process in a series of ethanol solutions of increasing concentration, cleared in xylene, and finally, embedded in paraffin wax. Subsequently, 5-μm-thick sections were cut from areas 2 mm below the point of ligation, stained using a commercially available H&E staining kit (Solarbio), and examined under a light microscope (Olympus IX73, Tokyo, Japan).
TUNEL Analysis
The terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) technique was applied to evaluate cell death, employing a kit obtained from Beyotime (Shanghai, China) and according to the guidelines of the manufacturer. 24 In brief, the prepared cell culture and tissue samples underwent TUNEL reaction mixture treatment and were incubated at 37 °C for 1 h. Endogenous peroxidase activity was quenched, after which the sections were incubated with streptavidin-horseradish peroxidase (HRP) conjugate, followed by exposure to the 3,3′-Diaminobenzidine (DAB) substrate solution for chromogenic detection of TUNEL-positive cells. Counterstaining with hematoxylin was performed to visualize the nuclei. The TUNEL-positive cardiomyocytes were quantified in multiple fields of view under the Olympus IX73 microscope.
Immunohistochemistry
The cardiac tissue slices underwent a process of deparaffinization and subsequent rehydration through varying concentrations of ethanol. This was followed by an antigen retrieval process. To inhibit the activity of inherent peroxidases, the slices were treated with a 3% hydrogen peroxide solution for 10 min and then blocked using 5% bovine serum albumin (BSA) provided by Sangon Biotech (Shanghai, China). After washing in phosphate-buffered saline, they were left to incubate overnight at 4 °C with a primary antibody specific for Fibronectin 1 (FN1; Abcam, Cambridge, USA). This was followed by a 1-h incubation with an IgG secondary antibody. Visualization was achieved using a diaminobenzidine substrate, and counterstaining was performed with hematoxylin. Imaging was conducted using the Olympus IX73 microscope, and the average optical density in the positively stained regions for each group was quantified using ImageJ 1.53k.
Transcriptomic Profiling
RNA was extracted from cardiac tissues with TRIzol reagent (Invitrogen, Carlsbad, USA), according to the manufacturer's protocol. The integrity and concentration of the RNA were evaluated using a Fragment Analyzer (Agilent, Santa Clara, USA). A cDNA library was generated following mRNA poly(A) enrichment through PCR. BGI Genomics (Wuhan, China) conducted the transcriptome sequencing on a DNBSEQ technology platform (MGI, Shenzhen, China). Initial sequencing outputs were processed using SOAPnuke (https://github.com/BGI-flexlab/SOAPnuke) to remove impurities, resulting in clean data in FASTQ format. The processed sequences were then aligned to the rat genome version 6.0 using HISAT2 (http://daehwankimlab.github.io/hisat2/), and transcriptome mapping was executed with Bowtie2. Quantification of gene expression was performed using the RSEM tool.
Analysis of differential gene expression was executed using the DESeq2 toolkit within R/Bioconductor v3.6.2 (R Foundation for Statistical Computing, Vienna, Austria). Genes exhibiting a fold change (FC) > 2.0 and a p value < 0.05 were categorized as differentially expressed genes (DEGs). To identify potential targets of ASP for mitigating MI/R injury, the intersecting DEGs (common DEGs, cDEGs) among ASP-influenced genes (comparison between the MI/R + ASP and MI/R groups) and genes associated with MI/R injury (comparison between the MI/R and Sham groups) were determined using a Venn diagram. Additionally, to elucidate the phenotypic alterations, analyses encompassing Gene Ontology (GO) enrichment for biological processes (BP), molecular functions (MF), and cellular components (CC), along with Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway assessment, were conducted in the Metascape platform. 25
Bioinformatic Analysis
To further pinpoint hub genes, protein-protein interaction (PPI) network analysis for the cDEGs was carried out using the STRING database (https://string-db.org/) and visualized using Cytoscape v3.7.2 (Cytoscape Consortium, San Diego, USA). The degree centrality of each node in the network was calculated to evaluate its topological importance. Next, module analysis was performed using Molecular Complex Detection (MCODE) plugin to identify densely connected regions in the PPI network. Genes with high degree connectivity and module scores were defined as candidate hub genes. 26 Furthermore, one rat dataset (GSE122020) and two human datasets (GSE97320 and GSE59867) obtained from the Gene Expression Omnibus (GEO) database were used for external validation to identify the key hub genes.
In an effort to uncover the underlying molecular mechanisms associated with the key hub genes, Gene Set Enrichment Analysis (GSEA) was employed in GSEA 4.1.0 software (https://mybiosoftware.com/gsea-gene-set-enrichment-analysis.html), focusing on the canonical pathways outlined in the C2 data sets. 27 GSEA yielded two critical values: the Normalized Enrichment Score (NES) and the False Discovery Rate (FDR). The NES represents the enrichment strength of a pathway in relation to the expression patterns of key hub genes. The NES operates over a spectrum from −1 to 1, with positive scores signifying upregulation and negative scores denoting downregulation. The FDR is a statistical measure that corrects for multiple hypothesis testing and estimates the probability of false positives among the significant enrichments. An FDR < 0.05 signifies significant enrichment of the expression of the key hub genes within specific gene sets associated with pathways. 28
Cell Culture and Treatment
The H9c2 rat embryonic ventricular myocardial cell line was obtained from the China Infrastructure of Cell Line Resource located in Beijing, China. The H9c2 cells were cultivated in Dulbecco's Modified Eagle Medium (DMEM), enriched with 10% fetal bovine serum from Gibco (Grand Island, USA).
For the in vitro simulation of MI/R, an H/R model was used, based on methodologies we’ve detailed in prior publications.23,29 Briefly, H9c2 cells were exposed to a low-oxygen environment (1% O2, 94% N2, and 5% CO2) within a hypoxic chamber for 3 h and subsequently returned to normal oxygen levels under standard culture conditions for 6 h. As a baseline comparison, control cells were maintained in DMEM under normoxic conditions. To explore impact of ASP on H9c2 cells, a 50 μg/mL ASP pre-treatment was administered for 4 h prior to the H/R exposure (H/R + ASP). 13 Additionally, H9c2 cells were treated with a canonical inflammasome activator, nigericin (Nig; 5 μM; Selleckchem, Houston, USA), to validate the role of pyroptosis.
Cell Viability Assay
The viability of cells was evaluated using a Cell Counting Kit-8 (CCK-8) as per guidelines provided by the manufacturer (Biosharp, Anhui, China). 29 Initially, H9c2 cells were distributed into 96-well plates, achieving a density close to 1 × 104 cells per well, and then incubated for 24 h. Following the treatment phase, each well received 10 μL CCK-8 solution. Subsequently, the plates were incubated further for 2 h. Absorbance readings for each well were obtained at 450 nm using the Infinite M200 PRO microplate reader (Tecan, Mannedorf, Switzerland).
Myocardial Injury Biomarker Assay
The levels of lactate dehydrogenase (LDH), a cytosolic enzyme released upon cellular damage, were quantified to evaluate myocardial injury. LDH activity was assessed using a commercially available colorimetric assay kit (Solarbio) according to the manufacturer's instructions. 29 Briefly, samples were diluted with the provided assay buffer to ensure that measurements fell within the linear range of the standard curve. The reaction was initiated by adding the substrate mixture, and the absorbance was measured at 450 nm using the Infinite M200 PRO microplate reader. LDH activity was calculated based on the absorbance values and the standard curve.
The levels of cardiac troponin T (cTnT), a sensitive and specific biomarker for myocardial injury, were quantified using an enzyme-linked immunosorbent assay (ELISA) kit (Meibiao Biology, Jiangsu, China). The assay was performed according to the manufacturer's instructions. Briefly, serum samples were diluted with the provided sample diluent to ensure that the measurements fell within the linear range of the standard curve. Diluted samples and standards were added to the pre-coated 96-well microplate and incubated with a biotinylated anti-cTnT antibody. After washing to remove unbound antibodies, a streptavidin-HRP conjugate, which binds to the biotinylated antibody, was added. Following incubation and subsequent washing steps, a chromogenic substrate solution, 3,3′,5,5′-tetramethylbenzidine (TMB), was added, and the enzymatic reaction was allowed to proceed. The reaction was terminated by adding a stop solution, and the absorbance was measured at 450 nm using the Infinite M200 PRO microplate reader. The cTnT concentrations in the samples were calculated based on the absorbance values and the standard curve generated from the known concentrations of cTnT standards.
Quantification of Inflammatory Cytokines by ELISA
The concentrations of inflammatory cytokines, including IL-1β, interleukin-18 (IL-18), and TNF-α, were quantified using ELISA kits obtained from Beyotime. The assays were performed according to the manufacturer's instructions, with strict adherence. Briefly, cell or serum samples were diluted with the provided sample diluents to ensure that the measurements fell within the linear range of the respective standard curves. Diluted samples and standards were added to the pre-coated 96-well microplates and incubated with biotinylated detection antibodies specific for each cytokine. After washing to remove unbound antibodies, streptavidin-HRP conjugates were added, which bind to the biotinylated antibodies. Following incubation and subsequent washing steps, TMB solutions were added, and the enzymatic reactions were allowed to proceed. The reactions were terminated by adding stop solutions, and the absorbance was measured at 450 nm using the Infinite M200 PRO microplate reader. The concentrations of IL-1β, IL-18, and TNF-α in the samples were calculated based on the respective absorbance values and standard curves generated from known concentrations of the cytokine standards.
Caspase-1 Activity Assay
The activity of Caspase-1, a key enzyme involved in the inflammatory response, was measured using a commercially available assay kit (Beyotime). The assay was performed according to the manufacturer's instructions, employing a spectrophotometric method based on the cleavage of a synthetic substrate by Caspase-1. Briefly, tissue or cell lysates were prepared and incubated with the provided reaction buffer and the caspase-specific substrate Ac-YVAD-pNA (acetyl-Tyr-Val-Ala-Asp p-nitroanilide). Upon cleavage by Caspase-1, the substrate releases the chromophore p-nitroaniline (pNA), which exhibits a distinct yellow color that can be quantified by measuring the absorbance at 405 nm. The absorbance readings were obtained using the Infinite M200 PRO microplate reader. Caspase-1 activity in the samples was calculated based on the absorbance values and a standard curve generated from known concentrations of the pNA standard. Caspase-1 activity was expressed as the percentage change relative to the control group.
Western Blot
Proteins were isolated from cells or tissues with RIPA buffer enhanced with protease inhibitors from Thermo Fisher Scientific (Cleveland, USA). The BCA protein assay kit from Solarbio was used to determine protein levels. Proteins in equal quantities underwent separation via SDS-PAGE and were then transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were subsequently blocked using either 5% nonfat milk or BSA for 2 h. Following the blocking phase, incubation with primary antibodies was performed, targeting NF-κB p65, its phosphorylated form p-NF-κB p65 (Sangon Biotech), GSDMD (Cell Signaling, Danvers, USA), FN1, NLRP3, and β-actin (Proteintech Group, Chicago, USA). The subsequent step involved a 2-h room temperature incubation with horseradish peroxidase-linked secondary antibodies. Detection of protein bands was achieved with enhanced chemiluminescence reagents (Millipore, Billerica, USA), and membrane images were captured with a Bio-Rad Universal Hood II Gel Doc System (Hercules, USA), and the results were quantified using ImageJ 1.53 K.
Statistical Analysis
Data are presented as mean ± standard deviation (SD). A two-tailed Student's t-test was used to evaluate differences between two groups. Comparisons among multiple groups were conducted through one-way Analysis of Variance, followed by Tukey's post-hoc test. Statistical significance was assigned at a two-sided P < 0.05. GraphPad Prism v8.3.0 (GraphPad Software, San Diego, USA) was used for the statistical analyses.
Results
ASP Mitigates Cardiac Damage in Rats with MI/R Injury
To evaluate the cardioprotective effects of ASP against MI/R injury, an in vivo rat model was established. Sprague-Dawley rats were assigned randomly to three experimental groups: Sham, MI/R, and MI/R + ASP. A comprehensive array of techniques, encompassing electrocardiography, echocardiography, TTC staining, and H&E staining, was employed to assess cardiac function, quantify infarct size, and examine histological alterations. Electrocardiographic evaluations (Figure 2A, B) demonstrated that the MI/R group experienced a notably higher ST-segment elevation compared to the Sham group. Nevertheless, ASP administration attenuated the elevation significantly. Based on echocardiographic assessments (Figure 2C-E), left ventricular EF and FS were reduced substantially in the MI/R group relative to in the Sham group. The changes were reversed significantly following ASP intervention. Analysis of TTC staining (Figure 2F, G) showed increased infarct size in the MI/R group in contrast to in the Sham group, whereas infarct size was considerably smaller in the MI/R + ASP group than in the MI/R group. H&E staining results (Figure 2H) revealed significant edema, partial rupture of myocardial fibers, cytoplasmic vacuolation, and increased inflammatory cell infiltration in rats subjected to MI/R injury, in contrast to the well-organized myocardial fibers in the Sham-operated rats. The myocardial structure changes were reduced significantly in the MI/R + ASP group. Collectively, the data demonstrate the successful establishment of a rat MI/R injury model, and further substantiate that pretreatment with ASP confers cardioprotective effects by alleviating MI/R injury in rats.
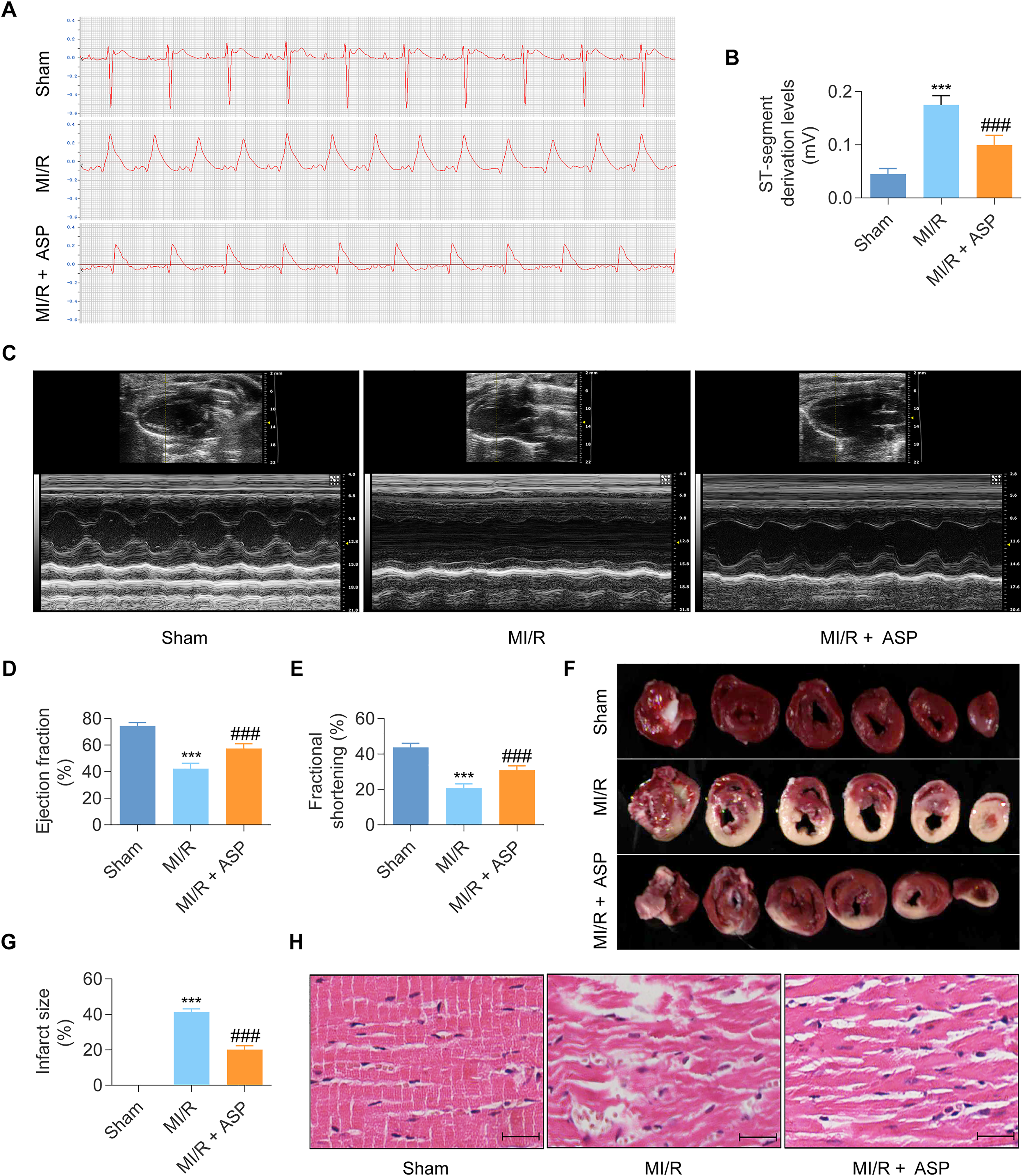
Angelica sinensis polysaccharide (ASP) confers cardioprotective effects in a rat model of myocardial ischemia/reperfusion (MI/R) injury. (A) Representative electrocardiogram (ECG) traces depict ST-segment elevation, an indicative hallmark of myocardial injury. (B) Quantitative analysis reveals that MI/R injury results in significant elevation of ST-segment levels compared to the Sham group, whereas the effect is attenuated by ASP pretreatment. (C) Representative echocardiographic records assess cardiac systolic function. (D, E) Echocardiographic assessment demonstrates marked reduction in left ventricular ejection fraction (EF) and left ventricular fractional shortening (FS) in the MI/R group, indicative of impaired cardiac function. ASP pretreatment preserves EF (D) and FS (E), suggesting improved cardiac contractility. (F) Triphenyltetrazolium chloride (TTC) staining delineates the myocardial infarct size (white regions). (G) Quantitative analysis reveals substantial increase in infarct size in the MI/R group, which is reduced significantly by ASP pretreatment. (H) Hematoxylin-eosin (H&E) staining illustrates myocardial architectural alterations. The MI/R group exhibits edema, partial rupture of myocardial fibers, cytoplasmic vacuolation, and increased inflammatory cell infiltration, whereas ASP pretreatment preserves myocardial structural integrity. Data are presented as mean ± standard deviation (n = 6 per group). Comparisons among groups were performed using one-way Analysis of Variance (ANOVA) followed by Tukey's post-hoc test for multiple comparisons. ***P < 0.001 versus the Sham group; ###P < 0.001 versus the MI/R group.
Transcriptomic Profiling Unveils Anti-Inflammatory Mechanisms as a key Mediator of ASP Cardioprotective Effects Against MI/R Injury
To elucidate the mechanisms underlying ASP cardioprotective effects against MI/R injury, transcriptomic profiling was performed on cardiac tissue samples from the Sham, MI/R, and MI/R + ASP groups. First, the gene expression profiles of the MI/R and Sham groups were compared (Supplementary Figure 1A) and 2642 DEGs were identified, among which 1594 genes were upregulated and 1048 genes were downregulated (Figure 3A and Supplementary Table 1). Second, the gene expression profiles of the MI/R + ASP and MI/R groups were compared (Supplementary Figure 1B), which revealed 888 DEGs, out of which 312 genes were upregulated and 576 genes were downregulated (Figure 3B and Supplementary Table 2). Finally, a Venn diagram analysis was performed to identify overlapping DEGs between the two comparisons and 715 shared cDEGs were observed (Figure 3C).

Venn diagram and functional analyses of differentially expressed genes (DEGs) identified by transcriptome sequencing in cardiac tissues from the sham, Myocardial ischemia/reperfusion (MI/R), and MI/R + Angelica sinensis polysaccharide (ASP) groups. (A) A volcano plot depicts DEGs in the MI/R group compared with the Sham group. Each data point represents a gene, with the x-axis displaying the log2 fold change (log2FC) in expression and the y-axis representing the negative logarithm of the p-value. DEGs with absolute log2FC ranking in the top 10 are labeled in the plot. Red and green data points indicate upregulated and downregulated genes, respectively, based on the specified threshold (abs(log2FC) > 1 and P < 0.05). (B) Volcano plot showing DEGs in the MI/R + ASP group compared with the MI/R group, with data point representation and thresholds similar to (A). (C) Venn diagram illustrating the distinct and overlapping DEGs among the three experimental groups. A total of 715 DEGs are co-regulated by MI/R and ASP treatments (common DEGs). (D) The top 20 biological processes are enriched for common DEGs based on Gene Ontology (GO) analysis. (E) The top 20 molecular functions are enriched for common DEGs based on GO analysis. (F) The top 20 cellular components are enriched for common DEGs based on GO analysis. (G) The top 20 enriched Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways among common DEGs are shown.
GO (Figure 3D-F) and KEGG (Figure 3G) enrichment analyses were conducted to explore the biological processes that the cDEGs were involved in. Notably, the cDEGs were involved mainly in the inflammatory response, cell death, PI3K-AKT, NF-κB, and MAPK signaling pathways. The transcriptomic findings highlight the anti-inflammatory properties of ASP as a key mediator of its protective effects against MI/R injury.
ASP Alleviates Myocardial Inflammation Injury in the MI/R-Injured Rats
To validate the transcriptomic evidence of ASP anti-inflammatory efficacy in the context of MI/R injury, we assessed inflammatory markers, myocardial cell death, and cardiac injury biomarkers in the rat MI/R injury model. ELISA was employed to quantify the serum levels of pro-inflammatory cytokines, including IL-1β, IL-18, and TNF-α. As shown in Figure 4A-C, MI/R injury resulted in significant elevation of the cytokine levels compared to the Sham group, indicating a robust inflammatory response. Remarkably, ASP pretreatment attenuated the increase in serum IL-1β, IL-18, and TNF-α levels substantially, suggesting its potent anti-inflammatory effects.

Angelica sinensis polysaccharide (ASP) mitigates excessive inflammation in a rat model of myocardial ischemia/reperfusion (MI/R) injury. (A-C) Enzyme-linked immunosorbent assay (ELISA) quantifies serum levels of inflammatory cytokines, including interleukin-1 beta (IL-1β, A), interleukin-18 (IL-18, B), and tumor necrosis factor-alpha (TNF-α, C). MI/R injury elevates these cytokine levels significantly compared to the Sham group, indicating a robust inflammatory response. ASP pretreatment substantially attenuates the increase in serum IL-1β, IL-18, and TNF-α levels, suggesting its potent anti-inflammatory properties. (D) Representative images of heart sections subjected to the terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay, which detects DNA fragmentation, a hallmark of cell death. (E) Quantitative analysis reveals a significantly higher percentage of TUNEL-positive cardiomyocytes in the MI/R group compared to the Sham group, indicative of increased cell death. ASP pretreatment reduces the number of TUNEL-positive cells markedly, suggesting its protective effects on cardiomyocytes against MI/R injury. (F, G) Serum concentrations of myocardial injury biomarkers, including lactate dehydrogenase (LDH, F) and cardiac troponin T (cTnT, G), are elevated substantially in the MI/R group compared to the Sham group. ASP pretreatment attenuates these elevations significantly, indicating its cytoprotective effects against MI/R injury. Data are presented as mean ± standard deviation (n = 6 per group). Comparisons among groups were performed using one-way Analysis of Variance (ANOVA) followed by Tukey's post-hoc test for multiple comparisons. ***P < 0.001 versus the Sham group; ###P < 0.001 versus the MI/R group.
Results from TUNEL staining (Figure 4D, E) indicated that the incidence of cardiomyocyte death was elevated markedly in the MI/R group compared to in the Sham group. ASP administration reduced the incidence of death among cardiomyocytes following MI/R injury significantly. Additionally, myocardial injury was assessed by measuring the serum levels of cTnT and LDH, which are sensitive biomarkers for myocardial cell damage. As illustrated in Figure 4F-G, MI/R injury increased serum cTnT and LDH levels markedly when compared to those in the Sham group, whereas ASP pretreatment attenuated the elevations significantly. Overall, the findings corroborate the transcriptomic evidence and underscore ASP therapeutic potential in attenuating the detrimental inflammatory responses associated with MI/R injury.
Bioinformatics Analysis Identifies Potential Therapeutic Targets of ASP in MI/R-Induced Injury
To further elucidate the molecular pathways underlying the anti-inflammatory effects of ASP against MI/R injury, bioinformatics analysis was conducted on the transcriptomic data to identify potential therapeutic targets. A PPI network was constructed using the 715 cDEGs regulated by both MI/R and ASP treatments (Figure 5A). Using the MCODE plugin in the Cytoscape software, the top module from the PPI network was identified (Figure 5B). The top 10 nodes with the highest degrees of connectivity in the PPI network were identified (Figure 5C). To further refine our list of candidate therapeutic targets, the external GEO dataset (GSE122020) was analyzed to identify genes that were differentially expressed significantly between the MI/R and Sham groups (|log2FC| > 1, P < 0.05). The Venn diagram (Figure 5D) was used to obtain the intersection of the results and identify candidate hub genes [FN1 and PTPRC (Protein tyrosine phosphatase receptor Type C)].

Bioinformatics analysis identifies the FN1/NF-κB/NLRP3 signaling axis as a potential therapeutic target of Angelica sinensis polysaccharide (ASP) in myocardial ischemia/reperfusion (MI/R) injury. (A) Protein-protein interaction (PPI) network is constructed from common differentially expressed genes (cDEGs) co-regulated by MI/R and ASP treatments, based on the STRING database. Red diamonds represent upregulated genes, while green circles represent downregulated genes. (B) The top-ranked module from the PPI network is obtained using the MCODE plug-in for Cytoscape, revealing a densely connected subnetwork of genes potentially involved in the ASP mechanism of action. (C) The top 10 genes with the highest node degrees, indicating their central roles and extensive connections within the PPI network. (D) Venn diagram illustrating the overlapping genes among different analytical approaches, including the top module genes identified through MCODE analysis, top genes with high node degrees, and differentially expressed genes (DEGs) from the GSE122020 dataset, which represents an independent transcriptomic study of MI/R injury. The overlapping genes represent candidates that are consistently identified as significant across multiple analyses and may play crucial roles in ASP-mediated cardioprotection against MI/R injury. (E) Validation of expression differences in the hub gene Fibronectin 1 (FN1) using human sample-based Gene Expression Omnibus (GEO) datasets (GSE97320 and GSE59867). The data show a significant difference in FN1 expression levels between AMI samples and controls, while Protein tyrosine phosphatase receptor Type C (PTPRC), initially considered a candidate hub gene, did not exhibit a significant difference and was therefore not further pursued. (F) Gene Set Enrichment Analysis (GSEA) revealing the enriched pathways associated with the key hub gene FN1. The analysis identifies significant enrichment in gene sets related to Diseases of programmed cell death, Cells and molecules involved in local acute inflammatory response, The NLRP3 inflammasome, and NF-κB signaling pathway. The findings suggest that the FN1/NF-κB/NLRP3 signaling cascade plays a pivotal role in mediating pyroptosis and inflammatory responses, thereby potentially exacerbating myocardial injury. Data are presented as mean ± standard deviation (SD). Comparisons among groups were performed using two-tailed Student's t-tests. *P < 0.05 versus the control group; **P < 0.01 versus the control group.
To determine the potential clinical applications of the candidate hub genes, the findings were validated using two GEO datasets (GSE97320 and GSE59867) based on human samples (Figure 5E). Our analysis revealed that only FN1 exhibited significant differential expression between the acute myocardial infarction and control groups (|log2FC| > 1, P < 0.05), whereas PTPRC gene expression did not exhibit a significant difference among the groups. Therefore, FN1 is considered a key hub gene and could serve as a therapeutic target of ASP in MI/R injury.
The identified FN1 gene, as a potential therapeutic target, was further investigated to understand its specific involvement in response to the agent. GSEA (Figure 5F) was conducted to determine significant associations between FN1 gene expression and gene sets related to programmed cell death, acute inflammatory response, NLRP3 inflammasome, and NF-κB pathway (all P < 0.05). Overall, the in silico findings indicate that the FN1/NF-κB/NLRP3 signaling axis may play a pivotal role in the mediation of the anti-inflammatory effects of ASP.
ASP Modulates the FN1/NF-κB/NLRP3 Pathway to Attenuate Inflammation Induced by MI/R in Rats
Subsequent in vivo experiments were undertaken to investigate the involvement of the FN1/NF-κB/NLRP3 pathway, which emerged as a potential therapeutic target through bioinformatics analysis. The immunohistochemical study results (Figure 6A, B) revealed that FN1 expression levels were notably higher in the MI/R group than in the Sham group. In contrast, FN1 levels in the ASP group were reduced considerably relative to the MI/R group.

Angelica sinensis polysaccharide (ASP) downregulates the FN1/NF-κB/NLRP3 inflammasome pathway in a rat model of myocardial ischemia/reperfusion (MI/R) injury. (A) Representative immunohistochemical images showing the expression levels of the Fibronectin 1 (FN1) in myocardial tissues from the Sham, MI/R, or MI/R + ASP groups. (B) Quantitative analysis of the average optical density of FN1 immunostaining, measured using ImageJ. MI/R injury results in significant upregulation of FN1 expression compared to the Sham group, which is attenuated by ASP pretreatment. (C) Representative Western blot bands depicting the protein levels of FN1, NF-κB p65, phosphorylated NF-κB p65 (p-NF-κB p65), NLRP3, N-terminal Gasdermin D (N-GSDMD), and β-actin (loading control) in myocardial tissue samples. (D-G) Quantitative analyses of Western blot data, showing the expression levels of FN1 (D), p-NF-κB p65 normalized to total NF-κB p65 (E), NLRP3 (F), and N-GSDMD (G) normalized to β-actin levels. MI/R injury leads to significant increase in the phosphorylation of NF-κB p65 and the expression of FN1, NLRP3 and N-GSDMD. ASP pretreatment substantially attenuates these alterations, suggesting its inhibitory effects on the FN1/NF-κB/NLRP3 inflammasome pathway. (H) Caspase-1 activity assay is performed on heart homogenates from different experimental groups. MI/R injury results in a significant increase in Caspase-1 activity compared to the Sham group, which is reduced markedly by ASP pretreatment. Caspase-1, a key effector protease activated by the NLRP3 inflammasome, serves as a functional marker of inflammasome activation and pyroptosis progression. Data are presented as mean ± standard deviation (n = 6 per group). Comparisons among groups were performed using one-way Analysis of Variance (ANOVA) followed by Tukey's post-hoc test for multiple comparisons. ***P < 0.001 versus the Sham group; ###P < 0.001 versus the MI/R group; ##P < 0.01 versus the MI/R group; #P < 0.05 versus the MI/R group.
The activation of Caspase-1 and the generation of N-GSDMD serve as markers of pyroptosis progression.8,10 Western blot assays (Figure 6C-F) indicated that FN1, p-NF-κB p65, NLRP3, and N-GSDMD levels were elevated considerably in the MI/R group when compared with in the Sham group. In contrast, ASP treatment decreased the levels of the proteins notably in comparison to the MI/R group. Furthermore, rats subjected to MI/R injury exhibited significantly elevated Caspase-1 activity compared to those in the Sham-operated group (Figure 6G). In contrast, the group treated with ASP in the MI/R context exhibited notable reduction in Caspase-1 activity relative to the MI/R group. Collectively, the results demonstrate that ASP alleviates MI/R-induced inflammation by inhibiting the FN1/NF-κB/NLRP3 pathway.
ASP Ameliorates the Pyroptotic Inflammation Damages Induced by H/R Exposure in H9c2 Cells
NLRP3 inflammasome-mediated cardiomyocyte pyroptosis represents a crucial biological process underlying the inflammatory injury induced by MI/R.8,10 To delineate whether ASP confers protection against MI/R-induced inflammatory injury by modulating cardiomyocyte pyroptosis, an in vitro model of H/R injury was established using H9c2 cells. Exposure to H/R notably reduced the viability of H9c2 cells (Figure 7A) and increased LDH release (Figure 7B) in the cells. However, treatment with ASP enhanced cell viability and reduced LDH release significantly. The addition of the inflammasome activator Nig to ASP-treated cells attenuated the protective effects partially, as evidenced by a reduction in cell viability and an increase in LDH release.
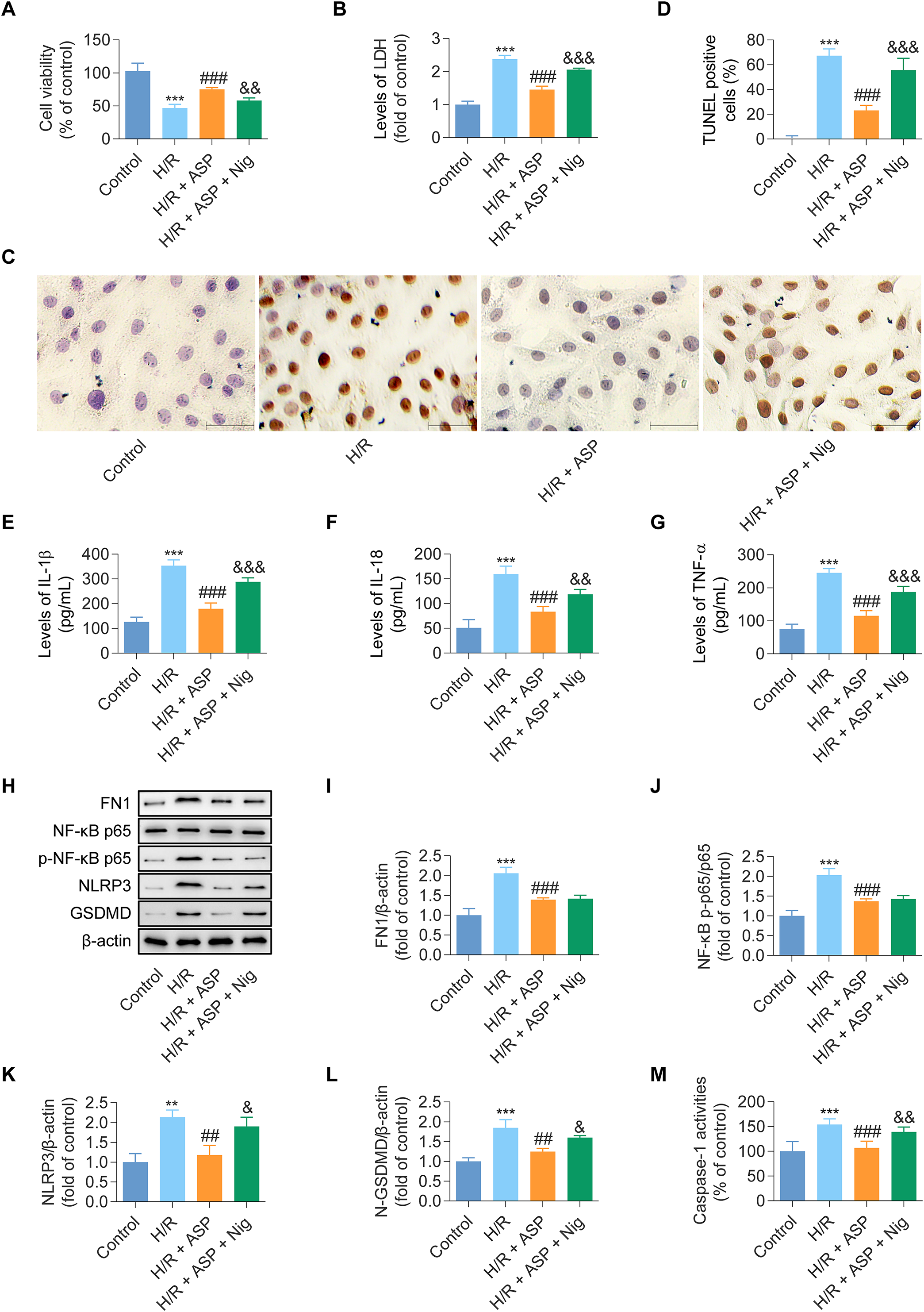
Angelica sinensis polysaccharide (ASP) mitigates pyroptotic inflammatory injury induced by hypoxia/reoxygenation (H/R) exposure in H9c2 cardiomyocytes. (A) Cell Counting Kit-8 (CCK-8) assay assesses viability in H9c2 cells subjected to H/R injury. H/R exposure results in significant decrease in cell viability compared to the normoxic control group, whereas ASP pretreatment substantially attenuates this reduction. Additionally, H9c2 cells were treated with nigericin (Nig), an inflammasome activator, which abrogates the protective effects of ASP on cell viability. (B) Lactate dehydrogenase (LDH) release assay evaluates cell membrane integrity. H/R injury leads to a marked increase in LDH levels, indicative of cell membrane damage, which is mitigated significantly by ASP pretreatment. However, Nig treatment results in a reversal of the decreased LDH levels in the ASP + MI/R group. (C) Representative images of H9c2 cells stained by the terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay, which detects DNA fragmentation, a hallmark of cell death (indicated by black arrows). (D) Quantitative analysis of the percentage of TUNEL-positive cells. H/R exposure results in significantly higher proportion of TUNEL-positive cells compared to the control group, indicating increased cell death. ASP pretreatment reduced the proportion of TUNEL-positive cells markedly. However, the cytoprotective effects of ASP were abrogated by Nig addition. (E-G) Enzyme-linked immunosorbent assay (ELISA) was used to quantify the levels of pro-inflammatory cytokines, including interleukin-1 beta (IL-1β, E), interleukin-18 (IL-18, F), and tumor necrosis factor-alpha (TNF-α, G). H/R injury resulted in significant elevation of the cytokine levels compared to the control group. ASP pretreatment substantially attenuated the increase in IL-1β, IL-18, and TNF-α levels. However, the anti-inflammatory effects of ASP were abrogated by the addition of Nig. (H) Representative Western blot bands depicting the protein levels of FN1, NF-κB p65, phosphorylated NF-κB p65 (p-NF-κB p65), NLRP3, N-terminal Gasdermin D (N-GSDMD), and β-actin (loading control) in H9c2 cell lysates. (I-L) Quantitative analyses of Western blot data, showing the expression levels of FN1 (I), p-NF-κB p65 normalized to total NF-κB p65 (J), NLRP3 (K), and N-GSDMD (L) normalized to β-actin levels. H/R injury leads to a significant increase in the expression of FN1, phosphorylation of NF-κB p65, and the levels of NLRP3 and N-GSDMD. ASP pretreatment attenuates these alterations substantially, suggesting its inhibitory effects on the FN1/NF-κB/NLRP3 inflammasome pathway. However, the addition of Nig abrogates the inhibitory effects of ASP on NLRP3 and N-GSDMD expression. (M) Caspase-1 activity assay is performed on H9c2 cell lysates from different experimental groups. H/R injury results in significant increase in Caspase-1 activity compared to the control group, which is reduced markedly by ASP pretreatment. The addition of Nig abrogates the protective effects of ASP, leading to further increase in Caspase-1 activity. Data are presented as mean ± standard deviation (n = 3 per group). Comparisons among groups were performed using one-way Analysis of Variance (ANOVA) followed by Tukey's post-hoc test for multiple comparisons. ***P < 0.001 versus the control group; ###P < 0.001 versus the H/R group; ##P < 0.01 versus the H/R group; &&&P < 0.001 versus the H/R + ASP group; &&P < 0.01 versus the H/R + ASP group; &P < 0.05 versus the H/R + ASP group.
The TUNEL assay results (Figure 7C, D) revealed that H/R increased the proportion of cells exhibiting positive TUNEL staining significantly in comparison to the control group. The effect was alleviated by ASP administration. Furthermore, Nig application negated ASP's protective impact on cells marked by TUNEL positivity. H/R cells displayed increased production of inflammatory cytokines (IL-1β, IL-18, and TNF-α) compared with that of controls (Figure 7E-G). In addition, ASP inhibited cytokine production significantly, whereas Nig addition to the ASP treatment increased cytokine production.
Western blot analysis (Figure 7H-L) revealed that FN1, phosphorylated NF-κB p65, NLRP3, and N-GSDMD levels were elevated markedly in the H/R group when compared to in the control group. ASP administration reduced the expression of the proteins in the H/R + ASP group notably relative to in the H/R group.
Similarly, ASP treatment reduced Caspase-1 activation by H/R significantly. Additionally, the inhibitory effect of ASP on NLRP3 inflammasome activation was reduced significantly by Nig treatment (Figure 7M). In summary, the findings demonstrate that ASP suppresses cardiomyocyte pyroptotic injury during MI/R through modulation of the FN1/NF-κB/NLRP3 pathway.
Discussion
ASP administration mitigated inflammatory response and enhanced preservation of cardiac function following MI/R injury. Subsequent transcriptomic sequencing and bioinformatic analyses provide insights into the potential molecular pathways contributing to ASP cardioprotective effects, identifying the FN1/NF-κB/NLRP3 signaling cascade as a key mediator. Complementary evidence from both in vivo and in vitro studies consistently substantiates that ASP confers cardioprotection against MI/R injury by inhibiting cardiomyocyte pyroptosis through the modulation of the FN1/NF-κB/NLRP3 pathway (Figure 8).

Schematic diagrams illustrating the mechanism of the effects of ASP in MI/R injury. Upon MI/R insults, the increased expression of FN1 promotes the phosphorylation of NF-κB p65, which primes the NLRP3 inflammasome. Then, activated NLRP3 inflammasome induces inflammation and pyroptosis, thereby contributing to cardiac damage. However, treatment with ASP inhibits cardiomyocyte pyroptosis by downregulating the FN1/NF-κB/NLRP3 pathway, which effectively mitigates inflammatory injury following MI/R.
Transcriptomic analysis has emerged as a potent analytical approach for comprehensively elucidating the intricate mechanisms underpinning the therapeutic effects of TCM. 30 In contrast to traditional candidate gene approaches, transcriptomic profiling offers an unbiased and comprehensive perspective, thereby enhancing the potential for unveiling novel genes and pathways implicated in the mechanisms of action. 30 Unlike hypothesis-driven methods that focus on pre-determined targets, transcriptomics enables the analysis of global gene expression changes, revealing the entire repertoire of genes and their intricate interaction networks influenced by the intervention. 30 This holistic approach facilitates the identification of key regulatory pathways and pivotal molecular nodes that may have been overlooked by conventional reductionist strategies. 30 Furthermore, the transcriptomic analysis results provide valuable insights for subsequent experimental validation, frequently offering an enhanced mechanistic understanding of the observed alterations in gene expression. 30 Recent research has effectively utilized transcriptomic analysis alongside experimental validation to explore the pharmacological mechanisms underlying different TCMs.27,31,32 Chen et al revealed that Panax notoginseng saponins enhance energy metabolism to improve cardiac function via the PPAR pathway in rats with acute myocardial infarction. 31 Salvianic acid A sodium safeguards against cardiac ischemic damage through the modulation of gene expression related to oxidative stress, inflammation, and apoptosis. 32 Additionally, Schisantherin A enhances cardiac performance post-myocardial infarction by influencing the regulation of three crucial fibrosis-related genes: Col3a1, Ctgf, and Lox. 27 Nonetheless, few investigations have employed transcriptomic analysis to elucidate the mechanisms of action underlying the effects of ASP in mitigating MI/R injury. This has hindered a comprehensive understanding of the extent to which the anti-inflammatory properties of ASP contribute to its effects on MI/R injury, as well as the key molecular pathways mediating ASP's anti-inflammatory actions.
In the present study, we establish the cardioprotective effects of ASP in the rat model of MI/R injury, corroborating previous reports.15,16 Zhang et al 15 demonstrate that ASP reduces infarct size in MI/R-injured rats significantly by alleviating oxidative stress, while Song et al 16 reveal that ASP improves cardiac function in rats with hypertensive heart disease by alleviating myocardial fibrosis, apoptosis, and oxidative stress. To comprehensively elucidate the key mechanisms underlying the protective effects of ASP against MI/R injury, we perform transcriptomic analysis. The results unveil that the anti-inflammatory properties of ASP play a pivotal role in mediating its cardioprotective effects against MI/R injury, a finding that is subsequently substantiated in our complementary in vitro and in vivo experimental studies. Our findings regarding the anti-inflammatory effects of ASP in the context of MI/R injury complement previous research15,16 and provide additional evidence supporting the therapeutic potential of ASP in mitigating MI/R-associated inflammatory injury.
NLRP3 inflammasome-mediated pyroptosis and the ensuing release of inflammatory cytokines are considered crucial instigators of the inflammatory cascade induced by MI/R.7,8,10 Research has demonstrated that the levels of NLRP3 inflammasome-associated components increase in response to MI/R damage. 7 Silencing of NLRP3 by RNA interference alleviates MI/R-induced inflammatory injury.33 Additional data have shown that NLRP3 knockout mice exhibit significantly reduced infarct size and improved cardiac function following MI/R injury compared with wild-type mice. 34 Using a Langendorff-isolated perfused heart model, Gastaldi et al showed that the NLRP3 inhibitor, IFN200, reduced myocardial inflammation and oxidative stress damage, thereby alleviating MI/R-induced cardiac dysfunction. 35 Clinical evidence indicates that colchicine, as a non-selective NLRP3 inhibitor, improves the prognosis of patients with acute myocardial infarction undergoing percutaneous coronary intervention. 36 In the present investigation, we employ bioinformatics analysis to elucidate the involvement of NLRP3-dependent pyroptosis in the anti-inflammatory effects exerted by ASP against MI/R injury. Subsequent experimental evidence demonstrates that ASP administration suppresses NLRP3-dependent pyroptosis in cardiomyocytes and mitigates the myocardial inflammatory response to ischemia/reperfusion, as substantiated by significantly diminished levels of NLRP3, GSDMD, active Caspase-1, and inflammatory cytokines. Furthermore, inflammasome activator Nig abrogates the protective effects conferred by ASP in H9c2 cells subjected to H/R injury. The present findings constitute the first report of ASP conferring improved cardiac function following MI/R by inhibiting NLRP3-dependent pyroptosis. This offers in-depth mechanistic insights into the anti-inflammatory actions of ASP and further corroborates that targeting NLRP3-dependent pyroptosis represents a promising therapeutic strategy for mitigating MI/R injury.
Regarding the mechanisms by which ASP inhibits NLRP3-dependent pyroptosis, our transcriptomic profiling and subsequent GSEA unveil FN1 as a pivotal hub gene associated with the NLRP3 inflammasome and NF-κB signaling cascade. Furthermore, we conduct verification using external GEO databases and show that the expression patterns of FN1 in different sample sources were consistent with our findings, further increasing the reliability and generalizability of our study results. FN1 is an extracellular matrix glycoprotein that exists in two subcategories: plasma FN1, primarily synthesized by liver hepatocytes, and cellular FN1, produced by various cell types, including myocytes, fibroblasts, chondrocytes, and synovial cells. 37 There is growing evidence that FN1 levels rise in the cardiomyocytes of both experimental rodents 38 and humans 39 within the first 24 h after MI/R injury. Gene ablation studies have demonstrated that FN1 impairs cardiac function and contributes to adverse cardiac remodeling by promoting inflammatory response.40,41 Mechanistic studies have reported that dysregulated FN1 expression activates the NF-κB signaling pathway in various pathological conditions, including tumors, 42 inflammatory diseases, 43 and MI/R injury. 40 To further elucidate the mechanism underlying FN1-mediated NF-κB activation, direct binding verification experiments have been performed. These experiments have demonstrated that FN1 binds to TLR4 on the cell surface, triggering the recruitment of adaptor proteins such as MyD88 and TRAF6.40,44 This interaction activates the IKK complex, which phosphorylates IκBα, leading to its ubiquitination and proteasomal degradation. The degradation of IκBα releases the NF-κB heterodimer (p50/p65) from its inhibitory complex and facilitates the subsequent phosphorylation of p65, enabling its nuclear translocation. 45 Once in the nucleus, activated NF-κB p65 binds to the promoter region of the NLRP3 gene, upregulating its expression. Crucially, NF-κB signaling is a prerequisite for both the priming and subsequent activation of the NLRP3 inflammasome.46,47 Our findings corroborate those of previous studies demonstrating that MI/R injury upregulates FN1 expression and consequently activates the NF-κB/NLRP3 signaling cascade.38,39 Notably, we demonstrate that by modulating the FN1/NF-κB/NLRP3 axis, ASP attenuates cardiomyocyte pyroptosis and exerts potent anti-inflammatory effects, thereby contributing to the mitigation of infarct size expansion and cardiac dysfunction following MI/R injury. These outcomes underscore the significant potential of ASP as a therapeutic candidate for mitigating pyroptosis in MI/R injury, and provide an insightful approach for the exploration of anti-inflammatory therapeutics.
Although the present study offers valuable insights, it is constrained by some limitations. Firstly, the use of cell and rodent models, divergent from MI/R patients’ clinical profiles, precludes direct extrapolation of findings to humans, necessitating judicious application to clinical settings. Secondly, while our investigation concentrated on the FN1/NF-κB/NLRP3 signaling pathway as the pivotal mechanism through which ASP exerts its effects, it did not extend to experimental validation of other biological mechanisms. Further research should investigate additional pathways and mechanisms to comprehensively elucidate ASP roles and interactions in the biological system.
Conclusions
By modulating the FN1/NF-κB/NLRP3 signaling pathway, ASP attenuates cardiomyocyte pyroptosis and alleviates the deleterious inflammatory sequelae, thereby exerting cardioprotective effects against MI/R injury. Our study not only elucidates the molecular mechanisms underlying ASP cardioprotective effects from a systems-level perspective but also highlights the therapeutic potential of targeting NLRP3-dependent pyroptosis in the amelioration of MI/R-associated inflammatory injury.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241279839 - Supplemental material for Angelica sinensis Polysaccharide Suppresses Pyroptosis in Myocardial Ischemia/Reperfusion Injury via the FN1/NF-κB/NLRP3 Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X241279839 for Angelica sinensis Polysaccharide Suppresses Pyroptosis in Myocardial Ischemia/Reperfusion Injury via the FN1/NF-κB/NLRP3 Pathway by Xiaowei Niu, Wen Jun Zhang, Fei Zhao, Jingjing Zhang, Shuwen Hu, Xue Bai, Zheng Zhang, and Ming Bai in Natural Product Communications
Supplemental Material
sj-pdf-2-npx-10.1177_1934578X241279839 - Supplemental material for Angelica sinensis Polysaccharide Suppresses Pyroptosis in Myocardial Ischemia/Reperfusion Injury via the FN1/NF-κB/NLRP3 Pathway
Supplemental material, sj-pdf-2-npx-10.1177_1934578X241279839 for Angelica sinensis Polysaccharide Suppresses Pyroptosis in Myocardial Ischemia/Reperfusion Injury via the FN1/NF-κB/NLRP3 Pathway by Xiaowei Niu, Wen Jun Zhang, Fei Zhao, Jingjing Zhang, Shuwen Hu, Xue Bai, Zheng Zhang, and Ming Bai in Natural Product Communications
Footnotes
CRediT Authorship Contribution Statement
Xiaowei Niu: Investigation, Formal analysis, Writing-original draft, Funding acquisition. WenJun Zhang: Investigation, Formal analysis, Writing-original draft. Fei Zhao: Investigation, Formal analysis, Revision. Jingjing Zhang: Investigation, Formal analysis, Revision. Shuwen Hu: Investigation, Formal analysis. Xue Bai: Formal analysis, Resources. Zheng Zhang: Conceptualization, Funding acquisition, Supervision, Writing-review & editing. Ming Bai: Conceptualization, Funding acquisition, Supervision, Writing-review & editing.
Data Availability Statement
All data analyzed during this study are included in this manuscript. Data will be made available on request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the National Natural Science Foundation of China (NO.82060807), the Outstanding Young Talents Project of Science and Technology Department of Gansu Province (No.24JRRA293), the Youth Doctoral Fund Project of the Department of Education in Gansu Province (NO.2022QB-011), the Special Fund for Civil-Military Integration Development of Gansu Province (NO.2060303), the Science and Technology Program of Gansu Province (20JR10RA677 and 23JRRA0937), the Scientific Research Project of Health Industry of Gansu Province (NO.GSWSKY2020-64), the Science and Technology Planning Project of Lanzhou City (NO.2020-ZD-72), and the Foundation for Scientific Research of the First Hospital of Lanzhou University (NO.ldyyyn2021-115 and NO.ldyyyn2017-30). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethical Approval
The study was approved by the Animal Care Committee of the First Hospital of Lanzhou University.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Research Council. The study protocol was reviewed and approved by the Animal Care Committee of the First Hospital of Lanzhou University.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


