Abstract
Cardiac hypertrophy (CH) is the main compensatory response to chronic heart stress and often progresses to a decompensation state potentially leading to heart failure. Phillyrin (PHI) is a novel compound derived from Forsythia, which has shown anti-inflammatory and anti-virus activities as well as renal protective effects on diabetic nephropathy. Therefore, we investigated the effects of PHI on CH induced by isoproterenol (ISO). Cardiac hypertrophy was induced by ISO in vivo, and the H9C2 cells were treated with ISO. PHI treatment alleviated CH in isoproterenol-induced mice in 7 and 14 days. Echocardiography showed that the PHI improved ISO-induced CH heart function and structure. PHI significantly decreased heart weight/body weight (HW/BW) and heart weight/tibia length (HW/TL) ratios and improved left ventricular (LV) function in ISO-treated mice. Hematoxylin and eosin staining revealed cardiomyocyte areas of the ISO group were significantly increased, and PHI was significantly reduced at 7 and 14 days, PHI-100 groups showed significantly better improvements than PHI-50. Sirius red staining indicated PHI significantly decreased collagen deposition in heart cross-sections induced by ISO, and PHI repressed ISO-induced cTn-I and NT-proBNP expression in mouse serum. In vitro data from H9C2 cells showed that PHI decreased cell areas and total cell protein levels in cells induced by ISO, whereas ANP, BNP, IL-6, and IL-1β expression was significantly inhibited by PHI. Also, PHI simultaneously inhibited P65 and P38 phosphorylation in vivo and in vitro. In conclusion, this study demonstrated the protective effect of PHI on CH in in vivo and in vitro, and this effect was related to the suppression of inflammation through the activation of the P38/NF-κB pathway.
Introduction
Cardiac hypertrophy (CH) 1 mainly arises from long-term pressure overload, which is a slow but effective compensatory function. Initially, such growth is an adaptive response to maintain cardiac function. Both physiological and pathological myocardial hypertrophy can lead to the enlargement of individual cardiomyocytes, but the characteristics of each type of hypertrophy are distinct. 2 Pathological CH is characterized by an abnormal increase in cardiac muscle mass in the left ventricle, resulting in cardiac dysfunction. CH increases myocardial oxygen demand and myocardial ischemia which ultimately decreases myocardial contractility and leads to heart disease. 3 The main clinical CH manifestations are chest pain, palpitations, and expiratory dyspnea. After disease development, heart failure becomes gradually manifested. 4
Currently, few specific drugs for CH treatment are available. Commonly used drugs include β-receptor blockers and valsartan. 5 However, these drugs must be combined with other diuretic drugs such as spironolactone, hydrochlorothiazide, etc.6,7 The usage of combined drugs was accompanied by long-term use side effects including hypotension, cough, and teratogenicity. 8 In recent years, more and more researchers have focused on the effects of traditional Chinese medicine on CH, and found some potential natural products, such as salidroside, 9 vinca alkaloids, 10 and several other cardiovascular medicines. 11
Phillyrin (PHI) is one of the main active phenylethanoid glycosides in the fruit of Forsythia. 12 Called Lianqiao in Chinese, F. forsythia fruit is traditionally used to treat pyrexia, hepatitis, gonorrhea, carbuncles, and erysipelas. 12 Pharmacological studies have shown that PHI exerts anti-inflammatory, anti-oxidative, and antiviral functionalities.13,14 Moreover, F. forsythia is a component of Tianwang Buxin Dan, a formula for treating heart failure in traditional Chinese medicine. Based on a network pharmacological investigation, Wang et al 15 showed that PHI improved left coronary artery ligation-induced cardiac fibrosis (CF) in rats, which indicated PHI could play roles in therapy on cardiac diseases, while the underlying mechanisms have remained unclear. To address this, we investigated the effects of PHI in isoproterenol (ISO)-induced CH in H9C2 cardiomyocytes. We also examined the molecular mechanisms underlying PHI protection toward CH in these cells. Also, an ISO-induced CH mouse model was generated to confirm the in vivo effects of PHI on CH.
Results
PHI Improves ISO-Induced CH Heart Function and Structure
ISO-induced CH was measured using heart weight/body weight (HW/BW) and heart weight/tibia length (HW/TL) ratios on days 7 and 14 of PHI treatments. As expected, CH was generated using subcutaneous ISO injections. Both HW/BW and HW/TL ratios were significantly higher in ISO animals than in controls on days 7 and 14. PHI administration significantly decreased these ratios when compared with ISO animals; significant effects were identified for 100 mg/kg PHI when compared with low concentration (Figure 1A to C). Data on day 14 were consistent with day 7 (Figure 1G to I).

The effects of Phillyrin (PHI) on cardiac indices and cardiomyocyte cell areas.
Anti-CH effects of PHI were also observed by H&E staining in hearts; cardiomyocyte area staining in ISO groups was increased when compared with controls while staining in day 14 hearts was significantly increased when compared with day 7 hearts. Cardiomyocyte cell areas in PHI-50 and PHI-100 groups were significantly reduced when compared with ISO groups on day 7 (Figure 1A and D). We also observed changes in cardiomyocytes on day 14 (Figure 1G and J).
Both cTn-I and NT-proBNP are important hypertrophy markers during CH. ELISA data showed these markers were significantly elevated upon ISO-induction when compared with control, however, PHI repressed this ISO-induced expression at days 7 and 14 (Figure 1E, F, K and L), with both 50 and 100 mg/kg PHI exerting effects.
PHI Improves Left Ventricular (LV) Function and Moderates LV Hypertrophy in ISO-Treated Mice
Echocardiography showed that LV, EF, and FS percentages were significantly decreased in ISO animals when compared with control. Both PHI-50 and PHI-100 inhibited EF and FS percentage reductions induced by ISO at 7 days (Figure 2A and B). Furthermore, LV mass, LVAW, and LVPW values were significantly increased in ISO animals when compared with control (Figure 2C to E), suggesting ISO stimulation had induced CH (7 days). Cardiac function indices were significantly lower after PHI (50 and 100 mg/kg) intervention, suggesting PHI improved heart systolic and diastolic functions, however, no dose-dependent therapeutic effects were observed. After 14 days of PHI intervention (50 and 100 mg/kg), cardiac function indices were significantly reduced when compared with the ISO group (Figure 2H to J). However, both EF and FS percentages were not significantly decreased by ISO at 14 days (Figure 2F and G). This observation may have been related to changes in cardiac function during late ventricular remodeling.

The effects of Phillyrin (PHI) on cardiac function by echocardiography
PHI Improves CH and Collagen Deposition Induced by ISO
Sirius red staining indicated significant collagen deposition in heart cross-sections in ISO animals. In contrast, collagen deposition in PHI groups was significantly decreased when compared with ISO animals (7 and 14 days, Figure 3A to C). Thus, PHI exerted anti-hypertrophic and anti-fibrotic effects in cardiac tissue.

The effects of Phillyrin (PHI) on in vivo cardiac hypertrophy (CH) pathology.
PHI Reduces Phospo-38-MAPK and Phospho-65-NF-κB Levels During CH
Western blotting showed significant increases in phospho-NF-κB P65(P-P65) and phospho-P38 P-P38) levels in the ISO group. However, PHI significantly decreased these levels when compared with the ISO group (7 days, Figure 4A to C). These PHI effects were concentration-dependent, and PHI-100 showed better inhibitory potency than PHI-50. Data at 14 days were consistent with day 7 (Figure 4D to F).
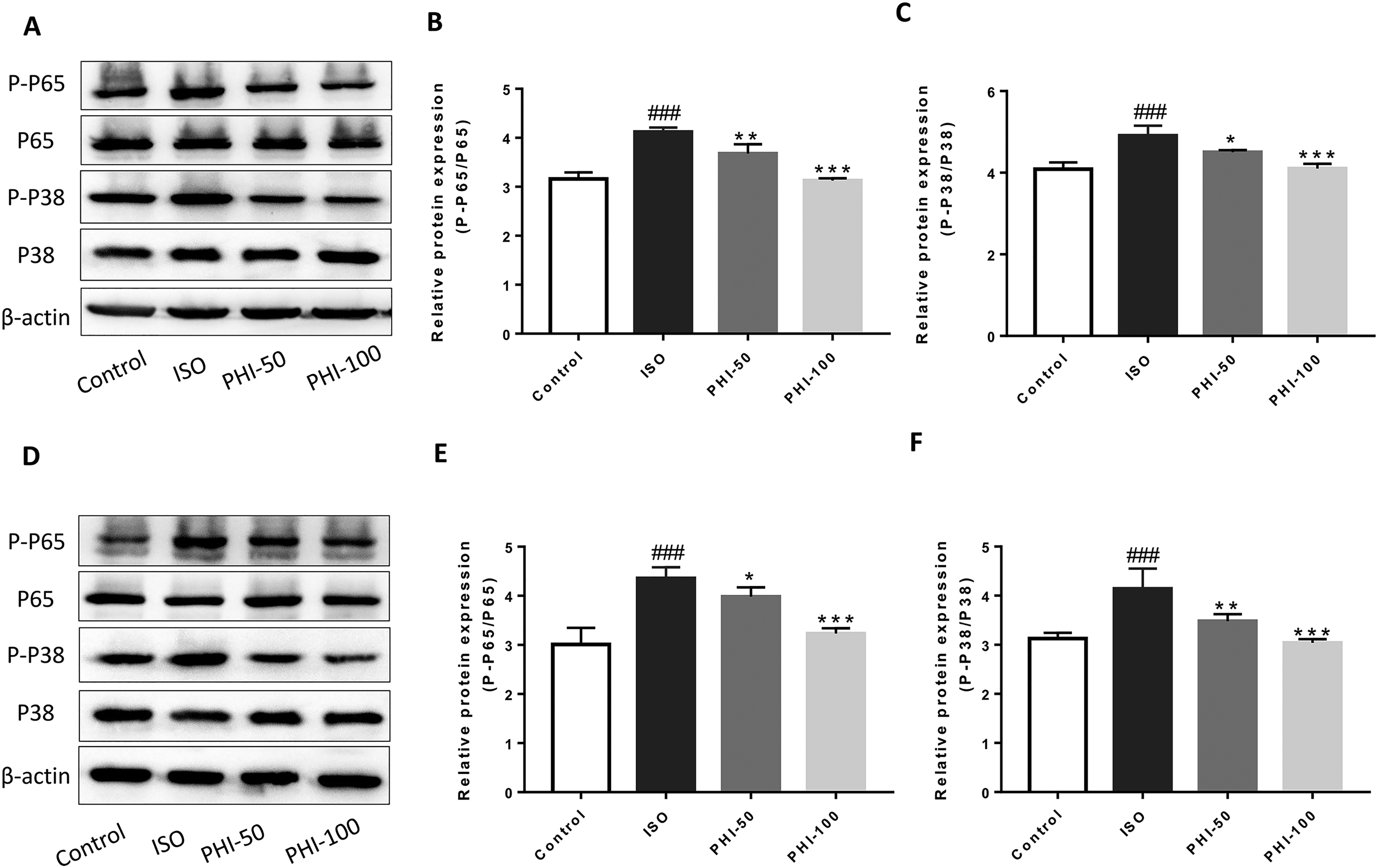
The effects of Phillyrin (PHI) on phospho-P38 and phospho-P65 levels.
PHI Prevents ISO-Induced Cardiac Hypertrophy In Vitro
PHI had no effects on proliferation in the 0.1 to 100 μM range (Figure 5A). We observed that ISO significantly induced H9C2 hypertrophy, whereas PHI significantly alleviated this as indicated by increased cell surface areas (Figure 5B and D). Protein assays also showed that ISO increased total protein levels in H9C2 cells, consistent with cell surface area data (Figure 5C). Quantification of the cardiac hypertrophy genes, ANP, and BNP by qRT-PCR showed that ISO significantly increased these levels, whereas PHI significantly inhibited their expression (Figure 5E and F). Also, elevated IL-1β and IL-6 mRNA levels were observed in H9C2 cells when induced by ISO, whereas PHI treatment significantly suppressed these levels (Figure 5G and H). Western blotting was used to detect P38, P65, P-P38, and P-P65 expression in H9C2 cells treated with and without ISO, and ISO + PHI. ISO stimulation increased P-P38 and P-P65 expression levels but had no effects on P38 and P65 expression. When compared with this group, P-P38 expression in H9C2 cells was significantly reduced in PHI (1, 3, and 10 μM) groups, with protein levels significantly decreasing as PHI concentration increased (Figure 5I and J). P-P65 expression was increased in the ISO group when compared with controls, whereas PHI diminished this increase (Figure 5I and K).

Different Phillyrin (PHI) doses inhibit isoproterenol (ISO)-induced H9C2 hypertrophy.
Discussion
Globally, CH is a major cause of morbidity and mortality 16 and is a common pathophysiological component of cardiac remodeling in several cardiovascular diseases, including, valvular heart disease and hypertension.17–21 ISO is a β-adrenoceptor agonist widely used to rapidly induce CH.22,23 The ISO dose used in this study was taken from previous studies.24–26 Thus, we used ISO to stimulate CH in mice for 7 and 14 days, and observed that changes in cardiac indices, HW/BW ratios and cardiomyocyte size at day 7 were no different from day 14. However, Sirius red staining indicated that cardiac collagen deposition at 14 days was significantly more serious than at 7 days. Thus, we speculated that collagen deposition in the 14-day ISO-induced model may have been due to pathological ventricular remodeling. However, echocardiography showed significant improvements in cardiac function upon PHI treatment. Thus, PHI reduced CH and ventricular remodeling in cardiac myocytes. As expected, cardiac pathology showed that cardiomyocyte areas had significantly increased (7 and 14 days) in the ISO group, whereas PHI significantly inhibited this increase.
Previous studies identified relationships between inflammatory reactions and CH; both P38 and NF-κB pathways were involved in CH. 25 As an important traditional Chinese medicine, Forsythia suspensa exerts generalized anti-inflammatory effects on infectious and inflammatory diseases.26–28 PHI is an active Forsythia suspensa component and ameliorates symptoms in several animal disease models, including a lipopolysaccharide-induced calvaria osteolysis model and a controlled cortical impact model of TBI (traumatic brain injuries).29,30 Molecular alterations were previously shown by inhibiting p38 and p65 phosphorylation in MAPK and NF-κB pathways.31,32 Therefore, to investigate the mechanistic impact of PHI on CH, we investigated P38 and NF-κB alterations induced by PHI. ISO increased P38 and NF-κB P65 phosphorylation levels, whereas PHI significantly decreased these levels in a concentration-dependent manner. Furthermore, elevated serum troponin cTn-I/TNNI3 and NT-proBNP levels were observed in ISO-induced mice, whereas PHI inhibited this expression. Consistently, PHI improved ISO-induced CH across both experimental models. Therefore, P38 and NF-κB P65 phosphorylation inhibition could be a potential mechanism whereby PHI exerts its anti-CH effects.
During clinical therapy for CH, valsartan mechanisms reduce blood pressure and inhibit NF-κB signaling.33–35 In this study, we observed that PHI had no effect on blood pressure in ISO-induced CH mice (data not shown), however, other significant CH improvements were identified. Our data indicated that reductions in inflammatory reactions may underpin the pharmacological effects of PHI toward CH. Also, PHI significantly inhibited MAPK and NF-κB inflammatory signaling pathways. However, as a natural drug extract, high PHI concentrations and low solubility restrict its clinical use, therefore, improved structural modifications are required in future research.
Conclusions
PHI inhibited ISO-induced CH in in vivo and in vitro models, and improved impaired LV function. PHI partially exerted its pharmacological effects by blocking P38 MAPK and NF-κB P65 signaling pathways.
Methods
Chemicals and Reagents
PHI (98% purity) was purchased from Chengdu Rueben's Biotechnology Co (Chengdu, China). ISO (>99.0% purity) was purchased from MedChemExpress (New Jersey, USA). The bicinchoninic acid (BCA) protein assay kit was purchased from Shanghai Beyotime Biotechnology (Shanghai, China). The following primary antibodies were used: P38 MAPK (8690), phospho-P38 MAPK (4511), NF-κB P65 (8242), and phospho-NF-κB P65 (3033) were purchased from Cell Signaling Technology (Boston, USA), β-actin (8H10D10) mouse mAb (CST, 3700, Boston, USA), horseradish peroxidase (HRP)-labeled goat anti-mouse IgG (H + L) (Beyotime), and HRP-labeled goat anti-rabbit IgG (H + L) (Beyotime). Bovine serum albumin (Thermo Fisher, USA). Ammonium persulfate (APS, Beyotime). Fetal bovine serum (BI, Shanghai, China). Penicillin/streptomycin (Thermo Fisher, USA). Enzyme-linked immunosorbent assay (ELISA) kits for mouse troponin I type 3(NNI3/cTn-I, E-EL-M1203c) and mouse N-terminal pro-brain natriuretic peptide (NT-proBNP, E-EL-M0834c) were purchased from Elabscience Biotechnology Co, Ltd (Wuhan, China). The PrimeScript RT reagent kit came from TaKaRa, Otsu, Japan.
Animals
Male C57BL/6 mice (age: 6-9 weeks, weighing 22-26 g) were purchased from the Animal Center of the Third Military Medical University (Chongqing, China). Animal studies were approved by the Institutional Animal Care and Ethics Committee of the university (Permit Number: SYXK-PLA-20190 021). Animals were adaptively fed before study commencement and were housed under the following conditions; temperature: 23 °C ± 2 °C and relative humidity: 45% to 65%. Animal studies were performed in accordance with protocols approved by our university.
H9C2 rat Cardiomyocyte Culture
H9C2 rat cardiomyocytes were provided by the Department of Pharmacology, Medical College of Army Medical University, and cultured in Dulbecco's Modified Eagle Medium plus 10% FBS and 1% penicillin/streptomycin. Cells were grown to near confluence in 75 cm2 cell culture flasks at 37 °C in 5% CO2 at 95% relative humidity. 16 Once or twice a week, cells were passaged after trypsin-EDTA digestion and resuspended in a complete medium in a 1:3 ratio. Cells were routinely sub-cultured to 80% confluence before studies.
PHI cytotoxicity was evaluated using a CCK8 assay kit (AbMole, Houston, USA) at 24 and 48 h (PHI concentration: 0.1-100 μmol). Then H9C2 cells were divided into 5 groups: control, ISO (20 μmol), PHI (1 μmol), PHI (3 μmol), and PHI (10 μmol). After culturing for 24 h, cultures were stimulated with ISO (20 μmol) for 24 h to induce a CH model. Then, cultures were stimulated with different PHI concentrations for 24 h. Cells were fixed in 4% paraformaldehyde, stained in 0.1% crystal violet, and invaded areas assessed using ImageJ.
Study Design
This study included both animal and cell investigations. Animal experiments were divided into 2 periods, 7 and 14 days. Male C57BL/6 mice were randomly divided into 4 groups (N = 12). (1) Control animals received 0.9% normal saline daily via intraperitoneal injection. (2) The ISO group received 10 mg/kg ISO in 0.9% normal saline via daily subcutaneous injection. (3) PHI-50 and PHI-100 groups received 50 mg/kg and 100 mg/kg PHI via intraperitoneal injection while taking ISO. Mice in each group were sacrificed on days 7 and 14, and there were 6 mice in each group in each time period. Heart and blood samples were taken; hearts were washed in phosphate-buffered saline (PBS), weighed, and stored at −80 °C, while a small heart portion was stored in formaldehyde for histopathology. Blood samples were stored at 4 °C overnight for analyses.
CH Induction
In vivo CH was induced using ISO (10 mg/kg) subcutaneous injections 17 for 7 and 14 days, and then mice were intraperitoneally injected with PHI (50 and 100 mg/kg) or 0.9% sterile normal saline accordingly. ISO (10 mg/kg) and PHI (50 and 100 mg/kg) were dissolved in 0.9% sterile saline and prepared before use.
Echocardiography
After 7 and 14 days of ISO stimulation and PHI administration, the cardiac structure and function were detected using Vevo 2100 high-resolution digital imaging system (Visual Sonics, Canada) equipped with an MS400 Micro Scan Transducer. Mice were depilated, anesthetized with isoflurane, and maintained with 1% isoflurane. The following indexes were detected: fractional shortening (FS), ejection fraction (EF), and other cardiac functional parameters, including left ventricular anterior end-systolic wall (LVAWs), left ventricular anterior end-diastolic wall (LVAWd), left ventricular posterior end-systolic wall (LVPWs), left ventricular posterior end-diastolic wall (LVPWd), left ventricular internal end-systolic diameter (LVIDs), left ventricular internal end-diastolic diameter (LVIDd). 18
Morphological Heart Analysis
HW/BW and HW/BL ratios were used to measure CH.19,20 Mice were sacrificed by cervical dislocation at days 7 and 14 for histomorphology, cardiac, biomechanical, histological, and molecular evaluations. Mouse hearts were also removed and sizes recorded and cardiac indices calculated using HW.
Quantifying serum Cytokine Levels Using ELISA
Blood was collected from the eyeballs of anesthetized mice and separated by centrifugation to generate serum. TNNI3/cTn-I and NT-proBNP cytokine levels were then determined by ELISA, according to the manufacturer's instructions.
Assessing Cardiac Injury and Collagen Deposition Using Histopathology
A portion of heart tissue from groups was fixed in 4% paraformaldehyde for routine histopathology. Tissues were fixed and embedded in paraffin for H&E and Sirius red staining. Tissue sections were 50 μM. Morphological changes were observed under light microscopy (Olympus BX43, Japan) at 40 × magnification. Images were captured using a digital camera (Japan).
Western Blotting
To extract proteins from H9C2 cells and 50 mg heart tissue, RIPA lysate solution containing 1% PMSF (phenylmethanesulfonyl fluoride) and 1% phosphatase inhibitors was added to samples for 30 min on ice. Protein concentrations were determined using a BCA kit, after which proteins in 1 × sodium dodecyl sulfate-polyacrylamide electrophoresis (SDS-PAGE) loading buffer were boiled for 6 min. Proteins were separated by SDS-PAGE and transferred to polyvinylidene difluoride membranes, then blocked in 5% BSA for 1.5 h. Membranes were incubated overnight at 4 °C with primary antibodies (β-actin, P38, P-P38, P65, and P-P65, 1:1000). The next day, membranes were washed 3 times in TBST (Tris-HCl buffer salt solution) and incubated with HRP-labeled secondary antibodies for 45 min at room temperature. Membranes were then washed 3 times in TBST for 10 min each, and immunoreactive bands were visualized using enhanced chemiluminescence (Shanghai, China).
Quantitative Real-Time PCR (qRT-PCR)
We performed qRT-PCR to evaluate ANP, BNP, IL-6, and, IL-1β mRNA expression in H9C2 cells. Total RNA was isolated using Trizol (Sigma, Louis, MO, USA) and reverse transcribed using a Prime Script RT reagent kit. Primer sequences were: rat ANP, forward 5′-TCTTCCTCGTCTTGGCCTTT-3′ and reverse 5′-CCAGGTGGTCTAGCAGGTTC-3′, rat BNP, forward 5′-TGGGAGGTCACTCCTATCCT-3′ and reverse 5′-GGCCATTTCCTCCGACTTT-3′, rat IL-6, forward 5′-CTGCAAGAGACTTCC ATCCAG-3′ and reverse 5′-AGTGGTATAGACAGGTCTGTTGG-3′, rat IL-1β, forward, 5′-ATGGCAGAAGTACCTAAGCTC-3′ and reverse 5′-TTAGGA AGACACA AATTGCATGGTGAAC-3′, rat β-actin forward 5′-ATGCCGACAGG ATGCAGAA-3′, and reverse 5′-GCTGATCCACATCTGCTGGA-3′.
Statistical Analysis
Data were analyzed using the mean ± standard deviation (SD). Graphpad Prism 7 and Origin 2018 were used for statistical analyses and graphics. We used a one-way analysis of variance and Dunnett range post-hoc comparisons to determine significant differences. A P < .05 value indicated statistical significance.
Footnotes
Acknowledgments
We thank Army Medical University for the experimental platform provided.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundations of China (No. 81673427, 81973319).
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Not applicable.
