Abstract
Christmas Rose (Helleborus niger L.) is an alpine plant belonging to the Ranunculaceae family. Typical for this popular ornamental plant are white flowers that appear during winter. Root extracts contain numerous medicinally active ingredients and potent cardiac glycosides. Recent findings indicate that Helleborus root extracts possess the potential for cancer treatment and inhibit the proliferation of tumor cells. In 2022, we published a practical extraction method of ( + )-ranucoside
Keywords
Introduction
The Christmas Rose (Helleborus niger L.) 1 is a perennial flowering plant belonging to the subspecies Helleborus of the Ranunculaceae family. Its main distribution area includes the Eastern European Alps, where it can be found up to an altitude of 1800 m. Christmas Rose is also a popular ornamental plant in horticulture and various cultivars have been generated. It has a very early flowering period, ranging from December to April and its white flowers and green leaves often appear among snow and ice. Therefore, this alpine flower has been a symbol of life and death since middle age, and still today. 2
A typical feature of the species H niger L. is its black rhizome from which it got its scientific name. Rhizome and roots (Radix Hellebori nigri) are used as medicine in allopathy and homeopathy. 3 Furthermore, pharmacological studies revealed that the root extracts contain numerous medicinally active ingredients4–7 as well as the powerful cardiac glycoside hellebrin8–11 which possesses similar pharmacological properties to those of the Digitalis glycosides.12–14 Recent findings indicate that Helleborus root extracts are safe and possess the potential for cancer treatment. 15 Furthermore, extracts of roots and the whole plant have been shown to inhibit the proliferation of tumor cells.16,17
In the 16th century, the Swiss physician and alchemist Paracelsus described Christmas Rose leaves as a dementia preventative par excellence.
18
But despite these early accounts of their pharmacological potential, the aboveground parts of the plant have been little studied since then. In 1974, (+)-ranucoside

The toxic flower Helleborus niger L. (Christmas Rose) blooms in winter and belongs to the Ranunculaceae family. Its aboveground parts contain the spiroacetal glycoside ( + )-ranuncoside
Based on our previously developed efficient and stereo-controlled synthetic routes to novel bicyclic motifs of spiroacetal natural products
24
as well as of tricyclic spiroacetal domain derivatives of the plant glycoside ( + )-ranuncoside
The synthesis is biomimetic, as it is an analogue to the biosynthesis of ( + )-ranuncoside
Results and Discussion
Retrosynthesis
The retrosynthesis of ( + )-ranuncoside
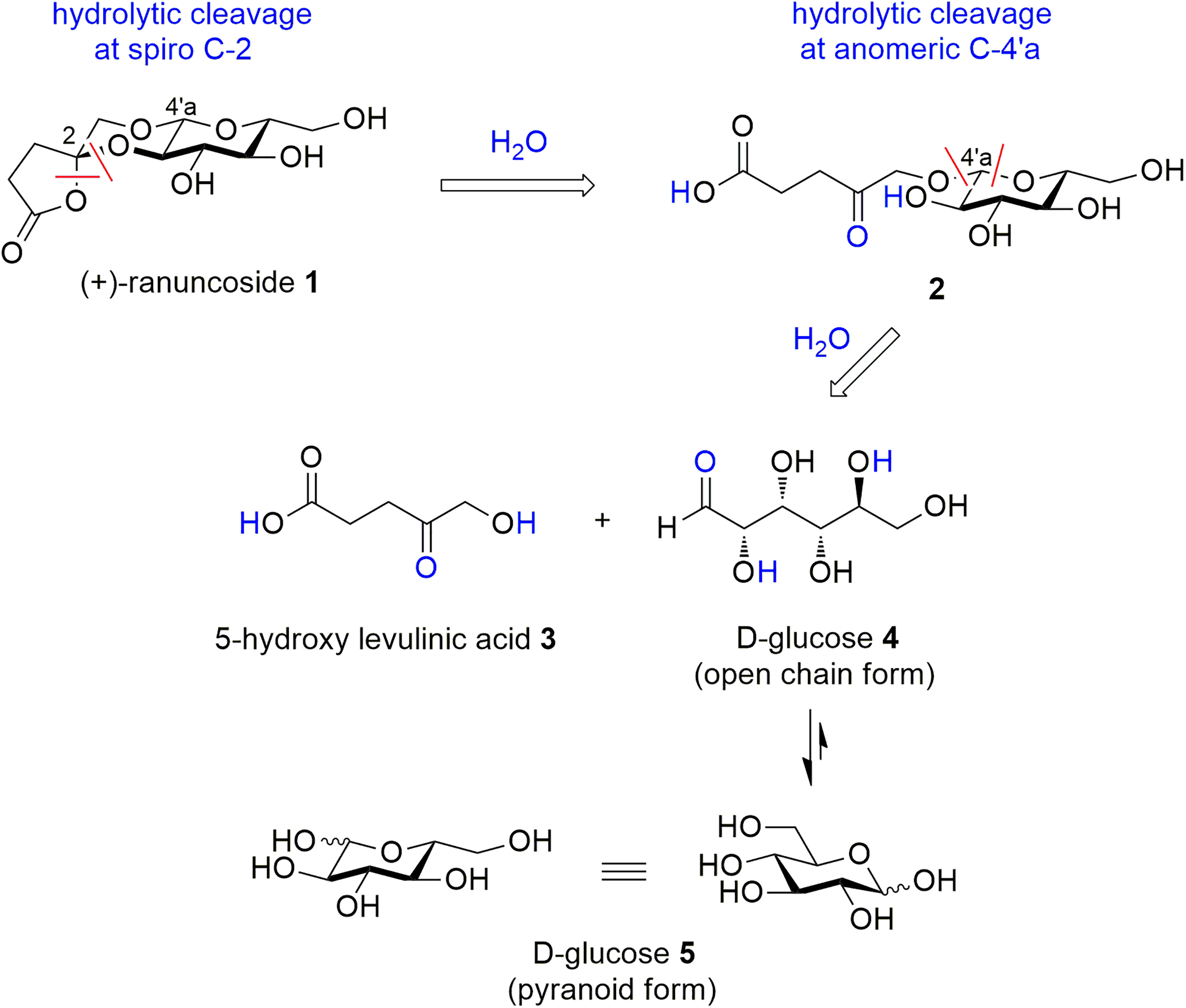
Retrosynthesis of ( + )-ranuncoside
Retrosynthetic cleavage of 2 bonds at the spiro center at carbon C-2 (red lines in Scheme 1) followed by the addition of 1 molecule of water (blue color in Scheme 1) forms the glycoside
Synthesis of the Levulinic Acid–Derived Starting Material 8
Starting from low-cost platform chemical levulinic acid

Synthesis of the glycosylation acceptor, the 5-hydroxylevulinic acid methyl ester
The further reaction of bromide
Altaicadispirolactone (1,6,9,13-tetraoxadispiro[4.2.4.2]tetradecane-2,10-dione) 11
The bicyclic spiro part of altaicadispirolactone
From a historical point of view, it is interesting that in 1962 Rappe
42
published the correct structure (without stereochemistry) of
The name altaicadispirolactone has been reported much later by Li and Li
44
according to the isolation of (5R,8R)-1,6,9,13-tetraoxadispiro[4.2.4.2]tetradecane-2,10-dione (altaicadispirolactone) from Anemone altaica, a flower native to the Altai Mountains (Central Asia). They described the synthesis of (±)-altaicadispirolactone by refluxing 5-hydroxylevulinic acid
Synthesis of the D-Glucose-Derived Starting Material 13
In 1901, the first synthesis of acetobromo glucose

By reacting D-glucose
Here, we present 2 laboratory reaction protocols for the synthesis of 1,2,3,4,6-penta-acetyl-β-D-glucopyranose
Biomimetic Synthesis of ( + )-ranuncoside 1
The crucial step in our total synthesis was the stereoselective β-glycosylation of 5-hydroxylevulinic acid methyl ester

Synthesis of ( + )-ranuncoside
Further selective deprotection of the acetyl-protecting groups in the sugar portion under Zémplen conditions with catalytic amounts of sodium methoxide in methanol succeeded as expected without any problems. The yield of the 5-(β-D-glucopyranosyloxy)levulinic acid methyl ester
The observed 2 carbonyl shifts in the 13C NMR spectrum at 173.71 and 208.35 ppm for an ester
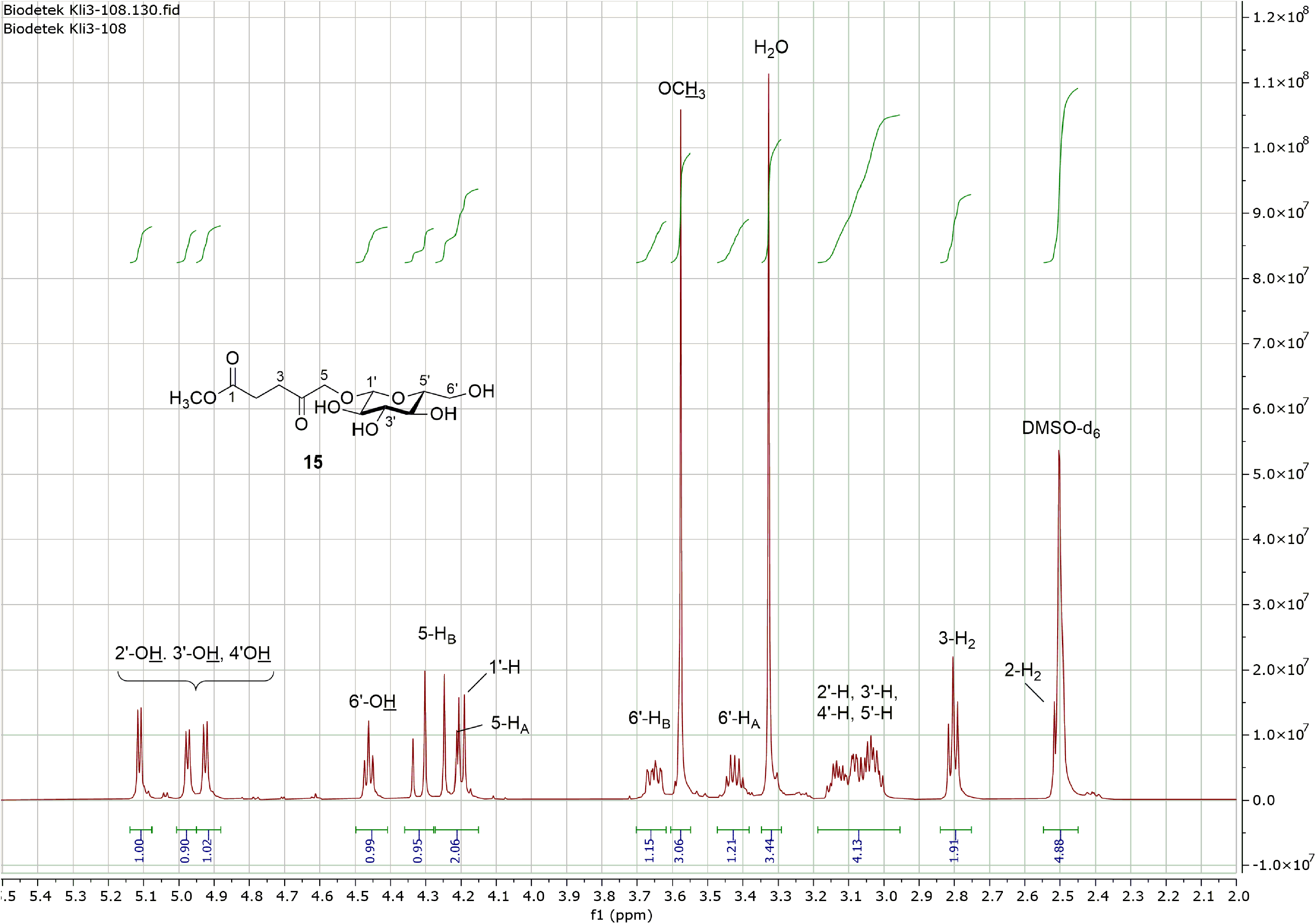
500 MHz 1H NMR spectrum of 5-(β-D-glucopyranosyloxy)levulinic acid methyl ester
The successful saponification of the methyl ester to the carboxylic acid was unequivocally evidenced by the absence of an ester methyl group in the 1H and 13C NMR spectra. By means of 13C NMR data, it could be excluded that the open-chain compound
The structure of
The last reaction step, the generation of the 5-membered butanolide ring, succeeded in an acidic environment with p-toluenesulfonic acid and tetrahydrofuran as solvent under relatively drastic reaction conditions (reflux 4 days). The natural product ( + )-ranuncoside
Conclusions
The Christmas Rose (H niger L.) 1 belongs to the subspecies Helleborus of the Ranunculaceae family and its rhizome and roots contain numerous medicinally active ingredidents4–7 including potent cardiac glycosides, which possess similar pharmacological properties to those of the Digitalis glycosides.12–14
Recently, we published a practical extraction method of ( + )-ranucoside
A characteristic feature of a retrosynthetic approach was the cleavage of the central dioxane ring at (a) the spiro center (C-2) and (b) at the anomeric center (C-4′a) and with respective water addition. This resulted in 5-hydroxylevulinic acid
Glycosylation of 5-hydroxylevulinic acid methyl ester
This is to our knowledge, the first report of a total synthesis of
Experimental
TLC was performed on POLYGRAM® SILG/UV254 (Macherey Nagel & Co.). Preparative chromatographic separations were carried out on columns with Merck silica gel 60 (40-63 μm). Melting points were determined on a Büchi Tottoli apparatus and are uncorrected. Specific optical rotations were determined on a Perkin-Elmer Polarimeter 241 in 1 dm cuvettes at a wavelength of 589 nm. NMR spectra were measured on a Bruker 300 MHz spectrometer with Avance-II console and BBO probe as well as 500 MHz spectrometer with DPX console and BBFO probe at 303 K using TMS as internal reference or by calibration with the shift of the solvent of the sample. 54 The abbreviation of the multiplicities of the shifts is indicated as s for singlet, d for doublet, t for triplet, q for quartet, sxt for sextet, oct for octet, m for multiplet, br for broad, and p for pseudo. Mass spectra were run on a Bruker Impact II spectrometer.
Methyl 5-bromo-4-oxopentanoate (5-Bromolevulinic Acid Methyl Ester) 7
Bromine (16.1 mL, 50.4 g, 0.32 mol) was added dropwise to a solution of levulinic acid
Methyl 5-hydroxy-4-oxopentanoate (5-Hydroxylevulinic Acid Methyl Ester) 8
Sodium formate (8.95 g, 131.6 mmol) was added to a solution of 5-bromolevulinic acid methyl ester (25 g, 119.6 mmol) in ethanol (99%, 161 g) and water (28 mL), and the solution was heated to reflux. The starting material was no longer visible by TLC after a reaction time of 3 h, and 2 product spots appeared at lower positions. After 18 h of reflux, only the more polar product of the 2 remained visible (Rf in n-hexane/ethyl acetate 6:4: starting material ∼0.8; intermediate product ∼0.45; product ∼0.2). After removal of the ethanol by distillation under vacuum, the residue was mixed with water (100 mL), the pH set to ∼5, and the residue extracted with ethyl acetate (10 × 30 mL). After the solvent was removed by distillation, 16.0 g of crude product was obtained. The product was purified from the few less polar contaminants by column chromatography on silica gel using n-hexane/ethyl acetate 7:3 as eluent: yield 12.85 g (74%) of chromatographically pure
1H NMR (500 MHz, CDCl3) δ 2.67 (br s, 4H, 2-H2, 3-H2), 3.66 (s, 3H, C
13C NMR (125.8 MHz, CDCl3) δ 27.59 (C-2), 32.94 (C-3), 52.11 (
(5R*,8S*)-1,6,9,13-Tetraoxadispiro[4.2.4.2]tetradecane-2,10-dione (meso-altaicadispirolactone) 11
A solution of potassium hydroxide (61 mg, 1.05 mmol) in methanol (5 mL) was added to a solution of 5-hydroxylevulinic acid methyl ester
1H NMR (500 MHz, DMSO-d6) δ 2.05 (m, 2H, 4-HA, 12-HA), 2.13 (m, 2H, 4-HB, 12-HB), 2.56 (ddd, 2H, 3-HA, 11-HA), 2.71 (pt, 2H, 3-HB, 11-HB), 3.88 (d, 2H, 7-HA, 14-HA), 3.94 (d, 2H, 7-HB, 14-HB); J3A,4A = J11A,12A = 10.60, J3A,4B = J11A,12A = 10.30, J3B,4A = J11B,12A = 2.70, J3B,4B = J11B,12B = 10.30, J3gem = J11gem = 18.15, J7gem = J14gem = 12.42 Hz.
13C NMR (125.8 MHz, DMSO-d6) δ 26.90 (C-3, C11), 29.09 (C-4, C12), 64.97 (C-7, C-14), 102.50 (C-5, C-8), 175.48 (C-2, C-10).
1,2,3,4,6-Penta-O-acetyl-β-D-glucopyranose 12
Water-free D-glucose (180.2 g, 1.0 mol) was stirred into acetic acid (180 mL). After addition of water-free sodium acetate (90.1 g, 1.1 mol), the suspension was heated to 90 °C under stirring. Then, acetic anhydride (630 mL, 6.7 mol) was added over 1 h under stirring and initial cooling. Stirring was continued for another 1 h at ca. 95 °C. The mixture was then cooled to ca. 70 °C and a solution of sulfuric acid (0.8 mL) in water (1.4 L), pre-warmed to ca. 70 °C, was slowly added under vigorous stirring. The solution was cooled whereby the product started to form crystals. Stirring was continued for another 1 h at ca. 10 °C. The crystallized product was then filtered by suction with a Büchner funnel and washed with water (3 × 200 mL). To obtain the pure β-compound, the wet product was recrystallized from ethanol (2 L). Upon drying in a vacuum oven at 60 °C, the pure pentaacetyl-β-D-glucose
1H NMR (500 MHz, CDCl3) δ 2.00 (s, 3H, 3-C
13C NMR (125.8 MHz, CDCl3) δ 20.72 (2-
2,3,4,6-Tetra-O-acetyl-α-D-glucopyranosyl bromide (Acetobromo-α-D-glucose) 13
Pentaacetyl-β-D-glucose
1H NMR (500 MHz, CDCl3) δ 2.02, 2.03, 2.08, 2.09 (s, 12H, 4 C
13C NMR (125.8 MHz, CDCl3) δ 20.69, 20.76, 20.79, 20.80 (4x
Methyl 4-oxo-5-[(2,3,4,6-tetra-O-acetyl-β-D-glucopyranosyl)oxy]pentanoate, [2,3,4,6-Tetra-O-acetyl-5-(β-D-glucopyranosyloxy)levulinic acid methyl ester] 14
Mercury cyanide (2.2 g, 17.4 mmol) was added to a solution of 5-hydroxylevulinic acid methyl ester
The reaction mixture was then added to a saturated sodium bicarbonate (NaHCO3) solution (200 mL) and stirred well for 15 min. The organic phase was separated and consecutively washed with water (100 mL) and potassium iodide solution (15%, 100 mL). Drying (Na2SO4) and removal of the solvent in vacuo left a crude product containing polar and less polar by-products. It was purified by column chromatography on silica gel and dichloromethane/methanol 99.5:0.5 as eluent. The mixed fractions were subjected to a second separation (methyl tert-butyl ether/n-hexane 60:40). The combined product fractions were recrystallized from ethanol: 2.5 g (60%) of
MS: HR-ESI 477.15990 [M + H]+ (calcd. for C20H28O13 + H, 477.16027).
1H NMR (500 MHz, CDCl3) δ 2.00, 2.01, 2 × 2.08 (4 C
13C NMR (125.8 MHz, CDCl3) δ 20.74, 20.75, 20.84(2x), (4
Methyl 5-(β-D-glucopyranosyloxy)-4-oxopentanoate, [5-(β-D-glucopyranosyloxy)levulinic acid methyl ester]15
The tetra-O-acetyl-5-(β-D-glucopyranosyloxy)levulinic acid methyl ester
MS: HR-ESI 331.09982 [M + Na]+ (calcd. for C12H20O9 + Na, 331.09995).
1H NMR (500 MHz, DMSO-d6) δ 2.50 (t, 2H, 2-H2) overlapped by DMSO-d6, 2.80 (t, 2H, 3-H2), 3.02 (m, 1H, 2′-H), 3.05 (m, 1H, 4′-H), 3.08 (m, 1H, 5′-H), 3.13 (m, 1H, 3′-H), 3.33 (s, H2O), 3.42 (m, 1H, 6′-HA), 3.58 (s, 3H, OC
1H NMR (500 MHz, DMSO-d6, D2O) δ 2.50 (br s, 2H, 2-H2), 2.74 (br s, 2H, 3-H2), 3.04 (m, 2H, 2′-H, 4′-H), 3.11 (m, 1H, 5′-H), 3.16 (pt, 1H, 3′-H), 3.41 (dd, 1H, 6′-HA), 3.54 (s, 3H, OC
13C NMR (125.8 MHz, DMSO-d6) δ 27.81 (C-2), 34.40 (C-3), 52.53 (
3-((2S,4aR,6R,7S,8S,8aR)-2,7,8-trihydroxy-6-(hydroxymethyl)hexahydro-6H-pyrano[2,3-b][1,4]dioxin-2-yl)propanoic acid 17
A solution of lithium hydroxide hydrate (1.1 eq., 0.174 mmol) in water (3 mL) was added to a solution of ester
MS: HR-ESI 317.08408 [M + Na]+ (calcd. for C11H18O9 + Na, 317.08430).
1H NMR (500 MHz, DMSO-d6) δ 2.41 (t, 2H, 3-H2), 2.71 (br s, 2H, 2-H2), 3.02 (m, 1H, 8′a-H), 3.04 (m, 1H, 7′-H), 3.09 (m, 1H, 6′-H), 3.13 (m, 1H, 8′-H), 3.41 (dd, 1H, 6′′-HA), 3.64 (d, 1H, 6′′-HB), 4.18 (d, 1H, 4′a-H), 4.20 (d, 1H, 3′-HA), 4.32 (d, 1H, 3′-HB), 3.30 and 5.50 (each br s, 2′-O
13C NMR (125.8 MHz, DMSO-d6) δ 27.42 (C-3), 33.52 (C-2), 61.05 (C-6′′), 69.96 (C-7′), 73.12 (C-3′), 73.36 (C-8′a), 76.61 (C-8′), 77.05 (C-6′), 102.70 (C-2′, C-4′a), 174.12 (C-1).
( + )-Ranuncoside Monohydrate, [(2R,4ʹaR,6ʹR,7ʹS,8ʹS,8ʹaR)-7ʹ,8ʹ-Dihydroxy-6ʹ-(Hydroxymethyl)hexahydro-3H-Spiro[furan-2,2ʹ-Pyrano[2,3-b][1,4]dioxin]-5(4H)-One - Hydrate (1 )]
p-Toluenesulfonic acid monohydrate (20 mg) was added to a solution of pyranodioxane
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221145919 - Supplemental material for Biomimetic Total Synthesis of ( + )-Ranuncoside, a Unique Tricyclic Spiroacetal Glycoside of Christmas Rose (Helleborus niger L.)
Supplemental material, sj-docx-1-npx-10.1177_1934578X221145919 for Biomimetic Total Synthesis of ( + )-Ranuncoside, a Unique Tricyclic Spiroacetal Glycoside of Christmas Rose (Helleborus niger L.) by Eckehard Cuny, Jörg Fohrer and Franz-Dietrich Klingler in Natural Product Communications
Footnotes
Acknowledgements
The author EC thanks Prof. Dr Michael Reggelin for the opportunity to work in his group. The author FDK thanks Dr Vibhuti Klingler-Dabral for providing the facilities for product synthesis and proofreading of the manuscript.
Author Contributions
FDK performed the synthesis of ( + )-ranuncoside. All 3 authors performed the structural elucidation. EC prepared the figures and schemes and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
