Abstract
The use of medicinal herbs as remedies reaches back to the Stone Age, and their importance as a source of drugs has continuously increased since then. Herbal ingredients can serve as active pharmaceuticals themselves or as lead substances for the development of synthetic pharmaceuticals with less toxicity, higher effectiveness or with new properties. To date, only 6% of the ∼600,000 plants on earth have been tested pharmacologically. Among these, the medicinal plant Helleborus niger L. (Christmas rose) is especially promising because its leaves contain ( + )-ranuncoside
Introduction
Plants were the first food of humans and it can be assumed that prehistoric people could differentiate between numerous edible and toxic plants and used medicinal herbs for healing purposes.1,2 Early discoveries of drugs in Europe reach back to the Stone Age, evidenced by a 5700 year old piece of chewed birch pitch from Betula pendula found in Denmark. 3 Another example are two dried fragments of the polypore fungus Fomitopsis betulina presumed to be used for prehistoric medical purposes, which were found among the belongings of the 5300 year old glacier mummy named Ötzi the Iceman, discovered in 1991 in the Ötztal Alps near the Austrian–Italian border. 4 Until the middle of the 18th century medicinal plants were the only source of organic drugs. Since then, the importance of medicinal plants has increased continuously. Around 50% of modern drugs depend on biogenic based compounds, and 10% of all drugs are pure medicinal plants.5,6
Helleborus species (Ranunculaceae) have been used as medicinal plants for over 2000 years, eg in ancient Greece, owing to their ingredients with potent pharmaceutical properties.7,8 Paracelsus, a seminal physician and alchemist of the 16th century, valued the leaves and roots of Helleborus niger L. (black hellebore or Christmas rose (Figure 1 [a]) as an effective remedy for numerous diseases.8-10 Even to this date, extracts of the rhizome and other subterranean parts of this plant are widely used in traditional medicine for a diverse range of symptoms and diseases.8,11,12 Helleborus niger extracts are also used in anthroposophic medicine, 13 and clinical uses include the concomitant treatment of oncological diseases. 14

The poisonous flowering plants (a) Helleborus niger L. (black hellebore, Christmas rose) and (b) Helleborus foetidus L. (stinking hellebore) are native to mountainous areas of Central and Southern Europe, and (c) Ranunculus repens L. (creeping buttercup) is native to Europe, Asia, and northwestern Africa. They all belong to the Ranunculaceae family and contain the spiroacetal glycoside ( + )-ranuncoside
Helleborus niger extracts contain numerous active ingredients8,15-17 such as hellebrin18-21 and helleborein,22,23 which are powerful cardiac glycosides with pharmacological properties similar to those of the Digitalis glycosides.24-26 Moreover, a recent preclinical evaluation showed that black hellebore extracts are safe and have potential for cancer treatment. 27 This finding was supported by two independent studies, which both found that the proliferation of tumor cells can be inhibited by Helleborus niger root and whole plant extracts.28,29
The chemistry of the active ingredients in the above-ground parts of Helleborus niger has been investigated in a few studies. In 1973, Martinek
30
isolated a crystalline compound from the ethanol extract of dried stems, leaves, and flowers of Helleborus niger L. which had been collected in the Austrian Alps (South Carinthia). In a subsequent paper by Martinek,
31
the ingredient was identified as ( + )-ranuncoside
Vitalini et al
34
investigated the ingredients in the leaves of Helleborus niger L. as well, but with plants collected in the mountain woods of Valestino (Brescia, Italy). Interestingly, in contrast to Martinek, they did not find the highly crystalline ( + )-ranuncoside

Isolated natural products from the alcohol extract of dried leaves of christmas rose (Helleborus niger L.): ( + )-ranuncoside
The spiroacetal ring system is responsible for the biological activity of a multitude of natural products. 38-40 For that reason, we previously developed efficient and stereo-controlled routes to novel bicyclic motifs of spiroacetal natural products41,42 as well as tricyclic spiroacetal domain derivatives of the plant glycoside ranuncoside.
43
With regard to potentially novel antibiotics and selective anticancer agents, we needed ( + )-ranuncoside
Since (a) a chemical synthesis of
Results and Discussion
The history of isolation and structure clarification of ranuncoside
Isolation of ( + )-Ranuncoside 1 from Christmas Rose
There are many different techniques available for extraction and isolation of natural products.
44
Generally, the plant material has to be pre-prepared carefully prior to extraction of phytochemicals. For a selective and efficient extraction of a polar substance like the glycoside ( + )-ranuncoside
To achieve a defined solid plant material, the green parts (leaves, stems, flowers) of the plant were dried in a vacuum drying oven to a constant weight and ground to a fine powder. The drying is very important in order to achieve a deep penetration of the water immiscible solvent in the first step. The small particle size leads to a much higher surface area which is of crucial importance for the solid-liquid extraction efficiency. In order to remove the nonpolar compounds, we used classical Soxhlet extraction with chloroform (trichloromethane) as solvent, given that ( + )-ranuncoside
After removing chlorophyll, carotenoids, and other nonpolar components, the Soxhlet extraction was simply continued by changing the solvent to an ethanol/water mixture. In this step, the ( + )-ranuncoside
With this simple and efficient laboratory process we were able to extract over 500 mg of the spiroacetal-glycoside ( + )-ranuncoside
Structural Elucidation of ( + )-Ranuncoside 1
( + )-Ranuncoside
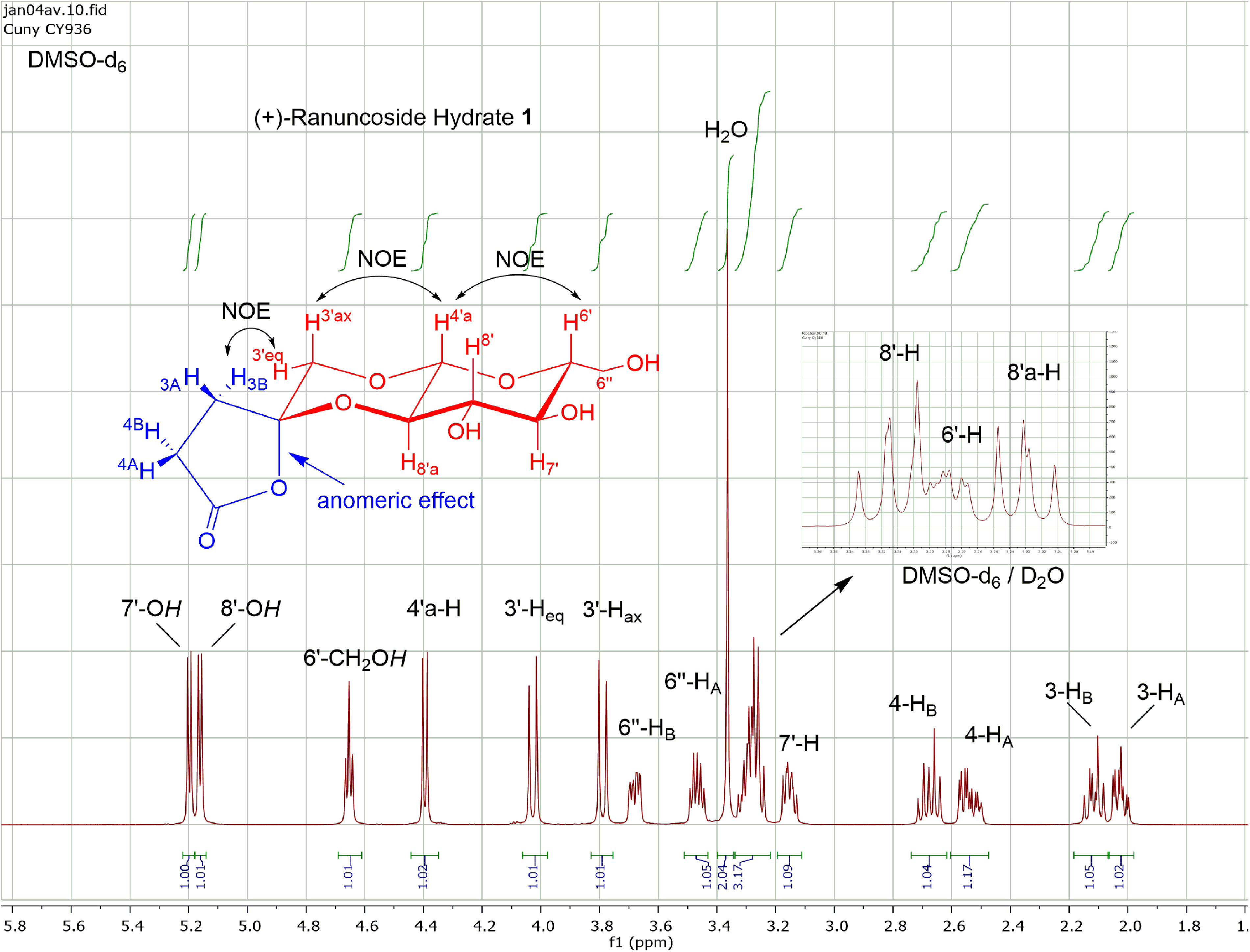
500 MHz 1H NMR spectrum of ( + )-ranuncoside
Systematic name and atomic numbering were made using SciFinder and ChemDraw. Confusingly, the name ranuncoside was, after the first naming by Tscheche et al, 32 later also used for two entirely different structures: (a) pentacyclic triterpenoid glycosides from Hydrocotyle ranuncoloides (Apiaceae) 47 and (b) an olefinic glycoside from Ranunculus muricatus (Ranunculaceae) with unsolved stereochemistry in the hexose moiety. 48
The structure of ( + )-ranuncoside
In the standard work “Stereochemistry of Carbohydrates” by Stoddart,
49
the conformational analysis of furanoid rings is described in detail. Against this background, the observed large geminal coupling constants of 13.4 (3-CH2) and 17.5 Hz (4-CH2), the size of the remaining coupling constants of 3.5, 9.5, 9.6 and 9.8 Hz indicate a flattened half-chair conformation of the five-membered ring. In combination with the x-ray structure analysis data (see below), a 3T2 twist (half-chair) conformation of the furanoid ring is present in solution, too. In an additional Newman-projection formula, the arrangement of all 4 protons is clearly presented (see structures

Partial structures
As expected, the 2 protons of the methylene group in the central dioxane portion 3′-Hax and 3′-Heq appear as 2 sharp doublets (3.78 and 4.02 ppm) and with large geminal coupling (12.7 Hz), respectively. Based on the smaller coupling constant of 7.3 Hz and the chemical shift at δ = 4.38 ppm, the other doublet could be assigned to the full acetal proton 4′a-H at the annulation position of dioxane and pyran ring. In terms of its NOE interaction with 3′-Hax (3.78 ppm), both dioxane protons at C-3′ (3′-Hax and 3′-Heq) can be assigned. From the pyran ring protons with usual stereochemistry of β-
The appearances of the shifts at δ = 3.46 (6′′-HA) and δ = 3.67 ppm (6′′-HB) with 1H-ddd are both typical for the exocyclic 6′-CH2OH group of the pyran ring. The shifts of the 4 hydroxyl groups are well separated: 2 near sharp doublets at δ = 5.21 (4.5 Hz) and δ = 5.25 (5.4 Hz) for 7′-OH and 8′-OH, the triplet at δ = 4.70 (5.7 Hz) for the 6′-CH2OH group and the big sharp singulet at δ = 3.39 for 1 molecule of water.
The 13C NMR spectral data of ( + )-ranuncoside
The structural elucidation of ( + )-ranuncoside
For better clarity, in the structure of
Conclusions
Medicinal plants have a long medical-cultural tradition which reaches far back into human history. With the recent trend of returning to natural medicines, they are now again of great interest. The main reasons for this are (a) the cheap source of raw materials for drugs and (b) the vast amount of different ingredients which could serve as lead substances for the development of pharmaceuticals with lower toxicity, greater efficiency or with new properties. A candidate that meets both requirements is ( + )-ranuncoside
Since the natural substance ( + )-ranuncoside
( + )-Ranuncoside
As shown by the observed NOE interaction between H-3′ax of the dioxan ring and H-4′ of the pyran ring the trioxa decalin ring system has cyclohexane-type all-chair conformation and trans-configuration. Thus, it is rigid, as in steroids with trans-decalin partial structure. The β-
Because of the precise knowledge of the structure and conformation of ( + )-ranuncoside
Experimental
TLC was performed on POLYGRAM® SILG/UV254 (Macherey Nagel & Co.). Preparative chromatographic separations were carried out on columns with Merck silica gel 60 (40-63 μm). Melting points were determined on a Büchi Tottoli apparatus and are uncorrected. Specific optical rotations were determined on a Perkin-Elmer Polarimeter 241 in 1 dm cuvettes at a wavelength of 589 nm. NMR spectra were measured on a Bruker 300 MHz spectrometer with Avance-II console and BBO probe as well as 500 MHz spectrometer with DPX console and BBFO probe at 303 K using TMS as internal reference or by calibration with the shift of the solvent of the sample. 52 The abbreviation of the multiplicities of the shifts are indicated as s for singlet, d for doublet, t for triplet, q for quartet, sxt for sextet, oct for octet, m for multiplet, br for broad and p for pseudo. Mass spectra were run on a Bruker Impact II spectrometer.
( + )-Ranuncoside Hydrate, [(2R,4′aR,6′R,7′S,8′S,8′aR)-7′,8′-Dihydroxy-6′-(Hydroxymethyl)hexahydro-3H-Spiro[furan-2,2′-Pyrano[2,3-b][1,4]dioxin]-5(4H)-One - Hydrate (1 )]
Isolation of ( + )-ranuncoside
TLC analysis confirmed that the mother liquor still contained considerable quantities of the product, but it was not isolated by a second chromatography.
1H NMR (500 MHz, DMSO-d6) δ 2.02 (ddd, 1H, 3-HA), 2.09 (ddd, 1H, 3-HB), 2.53 (ddd, 1H, 4-HB), 2.66 (ddd, 1H, 4-HA), 3.14 (ddd, 1H, 7′-H), 3.27 (m, 3H, 6′-H, 8′-H, 8′a-H), 3.35 (s, H2O), 3,46 and 3,67 (each 1H-ddd, 6-HA and 6-HB of 6′-CH2OH), 3.77 (d, 1H, 3′-Hax), 4.01 (d, 1H, 3′-Heq), 4.38 (d, 1H, 4′a-H), 4.64 (pt, 1H, 6′-CH2OH), 5.17 (1H-d, 8′-OH), 5.14 (1H-d, 7′-OH);
J3gem = 13.4, J3A,4A = 9.8, J3A,4B = 3.5, J3B,4A = 9.6, J3B,4B = 9.5, J4gem = 17.5, J3′gem = 12.7, J4′a,8′a = 7.5, J6′, 6′′A = 5.8, J6′, 6′′B = 2.0, J6′′A, 6′′OH = 5.7, J6′′B, 6′′OH = 5.8, J6′′gem = 12.0, J6′,7′ = 9.9, J7′, 7′OH = 5.7, J7′, 8′ = 8.2, J8′, OH = 5.2 Hz.
NOE: 4′a-H (4.38 ppm) → 3′-Hax (3.78 ppm), 4′a-H (4.38 ppm) → 6′-H (3.28 ppm) and 3′-Heq (4.02 ppm) → 3-HB (2.09 ppm).
1H NMR (500 MHz, DMSO-d6, D2O) δ 3.15 (dd, 1H, 7′-H), 3.23 (dd, 1H, 8′a-H), 3.28 (ddd, 1H, 6′-H), 3.32 (dd, 1H, 8′-H), 3.46 (dd, 1H, 6′′-HA), 3.66 (dd, 1H, 6′′-HB); J6′, 6′′A = 5.8, J6′, 6′′B = 2.0, J6′′gem = 12.1, J6′,7′ = 9.5, J7′, 8′ = 8.4, J8′,8a = 9.7 Hz. Differences to the spectrum above are (1) the absence of the shifts for the protons of the hydroxyl groups, (2) the splitting of the multiplet at 3.27 ppm and (3) the omission of the coupling constants of the hydroxyl groups.
13C NMR (125.8 MHz, DMSO-d6) δ 27.09 (C-4), 28.77 (C-3), 60.62 (6′-CH2OH), 69.32 (C-3′), 70.33 (C-7′), 72.72 (C-8′), 74.47 (C-8′a), 78.66 (C-6′), 96.91 (C-4′a), 103.63 (C-2), 175.55 (C-5).
MS: HR-ESI 277.0918 [M + H]+ (calcd. for C11H17O8+, 277.0918).
x-ray structural analysis: The crystal data and structure refinement are depicted in Table 1.
Crystallographic Data for the Crystal Structure Determination of the Monohydrate of ( + )-Ranuncoside

The atomic coordinates, bond length, anisotropic displacement parameters, hydrogen coordinate, torsion angles, and hydrogen bonds have been published as a CSD Communication and has a DOI and full CCDC citation: DOI: 10.5517/ccdc.csd.cc28xm50; CCDC Number: 2112872. It is publicly available through http://www.ccdc.cam.ac.uk/conts/retrieving.html [or from the Cambridge Crystallographic Data Centre, 12, Union Road, Cambridge CB2 1EZ, UK; Fax: + 44 1223/336 to 033; E-mail: deposit@ccdc.cam.ac.uk].
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211069456 - Supplemental material for Efficient Isolation and Structure Analysis of (+)-Ranuncoside, a Unique Tricyclic Spiroacetal Glycoside, from Christmas Rose (Helleborus niger L.)
Supplemental material, sj-docx-1-npx-10.1177_1934578X211069456 for Efficient Isolation and Structure Analysis of (+)-Ranuncoside, a Unique Tricyclic Spiroacetal Glycoside, from Christmas Rose (Helleborus niger L.) by Eckehard Cuny and Franz-Dietrich Klingler in Natural Product Communications
Footnotes
Acknowledgments
The author (EC) thanks Prof. Dr Michael Reggelin for the opportunity to work in his group, Ms. Sabine Foro for providing the x-ray data and Dr Jörg Fohrer for the helpful discussion of the NMR spectra. The author (FDK) thanks Dr. Vibhuti Klingler-Dabral for providing the facilities for product isolation and proofreading of the meanuscript.
Author Contributions
FDK performed the isolation of (+)-ranuncoside. Both authors performed the structural elucidation. EC prepared the figures and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
