Abstract
Introduction
Currently, the mortality rate of patients with lung cancer ranks the highest in the world. 1 According to a 2020 report by the China Cancer Registry, of the approximately 4.57 million new cancer cases diagnosed in China each year, as many as 1.13 million are lung cancer cases, of which approximately 80%–85% are nonsmall cell lung cancer (NSCLC). 2 Current treatments for NSCLC include molecular targeted therapy, immunotherapy, chemotherapy, and radiotherapy. Although major breakthroughs have been made in the treatment of NSCLC, the problem of drug resistance has become increasingly prominent after treatment with currently used drugs, and the development of new therapeutic targets and drugs is imminent. 3 Traditional Chinese medicine has advantages for use in multitarget therapy and can compensate for gaps in therapy related to drug resistance; an increasing number of studies have confirmed its effectiveness.
Tetrastigma hemsleyanum Diels et Gilg (T hemsleyanum), family Vitaceae, is a unique and valuable Chinese herbal medicine, mainly distributed and grown in the eastern, central, southern, and southwestern provinces of China. 4 As a commonly used traditional Chinese medicine, T hemsleyanum has the therapeutic effects of clearing away heat and detoxifying, promoting blood circulation, and relieving pain. It can be used alone or in combination with other drugs to treat pneumonia, hepatitis, and various cancers. 5 Recent pharmacological studies have shown that T hemsleyanum has antiinflammatory, antiviral, antitumor, and immunomodulatory effects, which may be attributed to flavonoids, polysaccharides, terpenoids, and alkaloids. 6 It has been reported that the most important substances in T hemsleyanum ethanolic extract (Te-EtOH) are flavonoids, including proanthocyanidins, quercetin, and rutin. 6
In our previous studies, we proved that Te-EtOH overcomes gefitinib resistance in NSCLC cells by inhibiting the protein kinase B (AKT)/extracellular signal-regulated kinase/Signal transducer and activator of transcription 3 signaling pathways and that these are related to abnormal glycolysis of tumor cells. 7 Therefore, we speculate that Te-EtOH inhibition in NSCLC is closely related to glycolysis. Studies have shown that tumor and normal cells use energy in different ways. Under the condition of a sufficient oxygen supply, normal cells acquire energy through oxidative phosphorylation in mitochondria, and the glycolytic pathway is activated to supply energy only when oxidative phosphorylation is insufficient; in contrast, tumor cells primarily use sugar under most circumstances. Energy can be acquired from glycolysis under adverse conditions. 8 However, there are few studies on the antitumor effects of Te-EtOH on regulation of tumor cell energy metabolism.
Phosphatidylinositol 3-kinase (phosphoinositide 3-kinase [PI3K]) is an important signal transduction molecule involved in the regulation of cell proliferation, differentiation, and apoptosis. It is also one of the most important protein molecules regulating the abnormal glycolysis of tumor cells. PI3K can enhance the ability of cells to take up glucose and the activities of hexokinase (HK) and phosphofructokinase (PFK) by activating the downstream kinase AKT and promoting cell glycolysis. Abnormal activation of the PI3K/AKT pathway is one of the important mechanisms for inducing tumor aerobic glycolysis 10. As a downstream target of the PI3K/AKT pathway, hypoxia-inducible factor 1α (HIF-1α) is considered to be the most important glycolytic transcription factor. In most cases, due to the massive proliferation of tumor cells, relatively high hypoxia is generated, which indirectly induces the high expression of HIF-1α in tumors. 9 Highly expressed HIF-1α combined with activation of the hypoxia response element in the promoter region of sugar metabolism genes lead to upregulated glycolytic enzymes at the transcriptional level and promote the multistep glycolysis process. 10 On the other hand, pyruvate dehydrogenase kinase 1 (PDK1) can upregulate and inhibit pyruvate dehydrogenase expression under the regulation of HIF-1α, thereby affecting the tricarboxylic acid (TCA) cycle dependent on pyruvate. 11 Nonetheless, it is unclear whether the effect of Te-EtOH on NSCLC cells is related to the PI3K/AKT/HIF-1α signaling pathway.
In this study, we examined the energy metabolism of Te-EtOH in NSCLC cell lines and in a hypoxic cell model, as well as the underlying potential mechanism(s). Our data suggested that Te-EtOH exerted antitumor effects in vitro by regulating energy metabolism and angiogenesis in NSCLC cells.
Results and Discussion
Te-EtOH suppressed NSCLC cells viability. To reveal the inhibitory effect of Te-EtOH on NSCLC cells, we used Cell Counting Kit-8 (CCK-8) to analyze the effects of different doses of Te-EtOH (0.001, 0.01, 0.025, 0.05, 0.1, 0.2, 0.4, and 0.8 mg/mL) on A549, H1299, and normal lung epithelial cells BEAS-2B for 24 h. The results showed that the Te-EtOH-inhibited NSCLC cell growth in a dose-dependent manner (Figure 1A and B), but had a weak effect on BEAS-2B cells (Figure S1). In addition, a CCK-8 experiment was performed to detect the effect of 0, 0.01, 0.1, and 0.2 mg/mL Te-EtOH on the viability of A549 and H1299 cells treated for different periods of time. Our data indicated that Te-EtOH-inhibited NSCLC cell growth in a time-dependent manner (Figure 1C and D). To verify the effect of Te-EtOH on the morphology of NSCLC cells, microscopic observation showed that A549 cells treated with different concentrations of Te-EtOH were round and granular (Figure 2A). Apoptotic cells were observed by fluorescence microscopy after Annexin V and Hoechst staining (Figure 2B); flow cytometry results were similar (Figure 2C and D). Annexin V red Reagent for apoptosis was observed in 3D spheroids treated with Te-EtOH. The apoptosis rate of A549 and H1299 cells in the 3D spheroids treated with 0.1 mg/mL and 0.2 mg/ml Te-EtOH was increased significantly in comparison to the control (Figure 2E and F). Therefore, these results indicated that Te-EtOH suppressed the NSCLC cells viability.
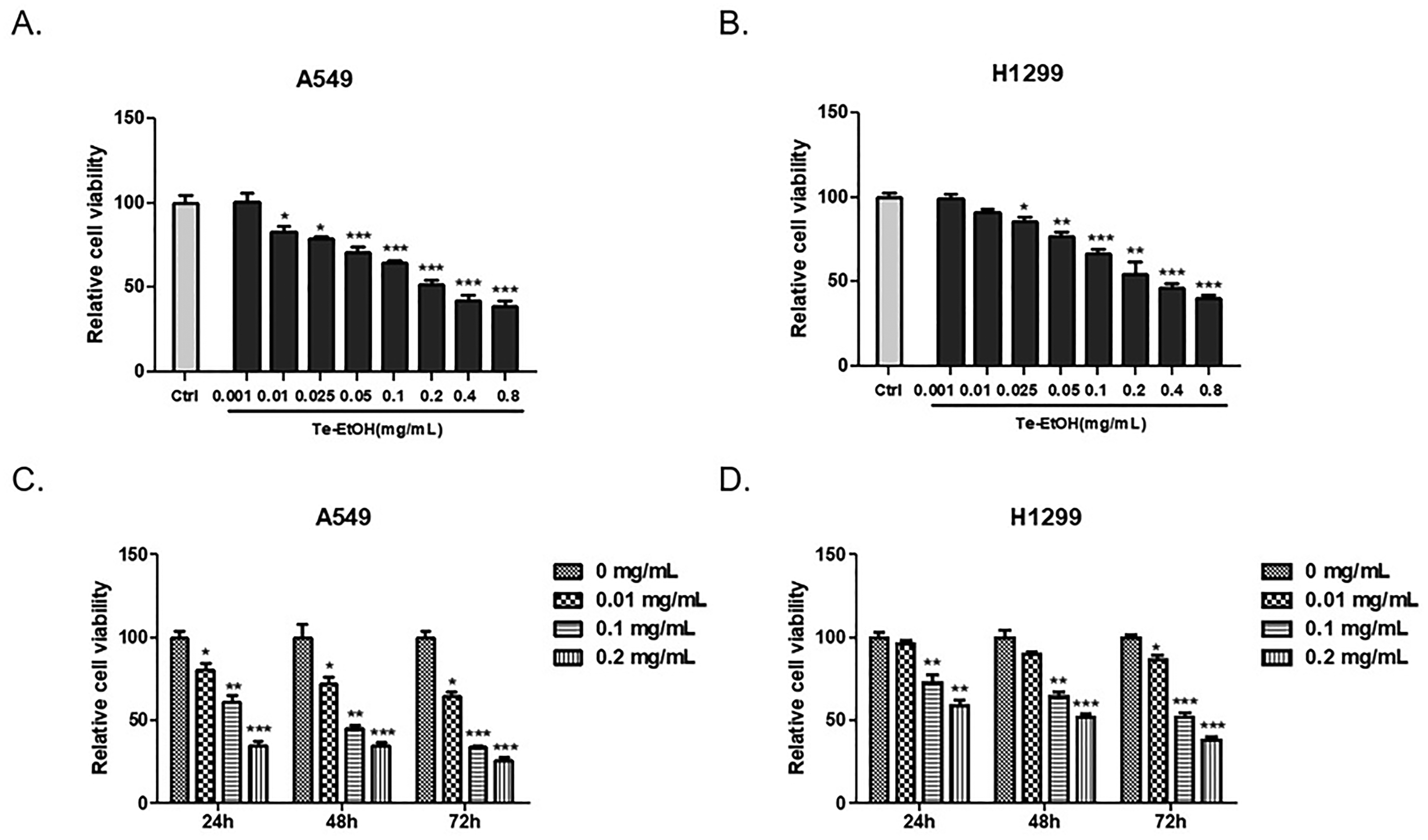
Te-EtOH suppressed the lung cancer cells' viability. (A, B) CCK-8 assays were performed to determine the viability of A549 and H1299 cells treated with 0 (control), 0.001, 0.01, 0.025, 0.05, 0.1, 0.2, 0.4, and 0.8 mg/mL Te-EtOH for 24 h. (C, D) A549 and H1299 cells were incubated with 0 (control), 0.01, 0.1, and 0.2 mg/mL Te-EtOH for 24, 48, and 72 h, and cell viability was measured by CCK-8 assay. The right panel represents the integration of fluorescence intensity/area. The mean ± SD from 3 technical replicates is shown. *P < .05, **P < .01, ***P < .001 versus Ctrl.

Detection of apoptosis in Te-EtOH-treated A549 cells. (A) Morphological alterations of A549 cells treated with 0 (control), 0.01, 0.1, and 0.2 mg/mL for 24 h. (B) A549 cells were stained by Annexin V and Hoechst and observed under a fluorescence microscope. (C, D) Flow cytometry analysis of Te-EtOH-treated A549 cells. (E, F) Representative signals for Annexin V in A549 3D spheroids. The right panel represents the integration of fluorescence intensity/area. The mean ± SD from 3 technical replicates is shown. *P < .05, **P < .01, ***P < .001 versus Ctrl.
Te-EtOH inhibits glycolysis, glucose consumption, lactate release, and adenosine triphosphate (ATP) generation level in A549 and H1299 cells. To study further the proapoptotic and antiproliferation effects of Te-EtOH, we took the cell samples incubated with and without Te-EtOH for transcriptome sequencing. The results showed that compared with the control group, the Te-EtOH group had 3581 upregulated genes and 4757 downregulated genes (Figure S2A Heat map and Figure S2B Volcano map). Furthermore, KEGG enrichment analyses were performed to further understand the function of the differentially expressed genes. Differential genes were significantly enriched in endocytosis, autophagy, mitogen-activated protein kinase (MAPK) signaling, and metabolism pathways (Figure S2C). It has been reported that Te-EtOH has the function of regulating autophagy and the MAPK signaling pathway,7, 12 but there is no related report on the metabolic signaling pathway. Based on transcriptome analysis, we postulated that the apoptotic effects of Te-EtOH were via regulating metabolism. Therefore, we explored the effect of Te-EtOH on the metabolism of H1299 and A549 cells. As shown in Figure 3A to C, our data confirm that, compared with their effects on the control group, Te-EtOH (0.1 and 0.2 mg/mL) significantly reduced glucose uptake and ATP and lactic acid (LA) accumulation. Next, performing Western blot analysis, we determined the key enzymes of glycolysis (HK2, lactate dehydrogenase A [LDHA], and PDK1) and the expression of glucose transporter 1 (GLUT1) after Te-EtOH treatment. The Western blot analysis results showed that, compared with those of the control group, the protein expression levels of HK2, LDHA, PDK1, and GLUT1 in the Te-EtOH group were decreased in a dose-dependent manner (Figure 3D). These results indicated that Te-EtOH inhibited glycolysis and glucose transport in these NSCLC cells.

Te-EtOH inhibited glycolysis, glucose consumption, lactate release, and ATP generation in A549 and H1299 cells. (A–C) Glucose consumption, lactate release, and ATP generation levels were measured in A549 and H1299 cells treated with Te-EtOH (0.01, 0.1, 0.2 mg/mL) or the control for 24 h. (D) Western blotting was performed to measure the expression of marker proteins encoded by glycolysis-related genes (GLUT1, HK2, LDHA, and PDK1) in A549 cells. The mean ± SD from 3 technical replicates is shown. *P < .05, **P < .01, ***P < .001 versus Ctrl.
Te-EtOH inhibited glycolysis in A549 cells by inhibiting the PI3K/AKT/HIF-1α signaling pathway. Many studies have indicated that the PI3K/AKT pathway plays essential roles in energy metabolism and tumor growth. PI3K can enhance the ability of cells to take up glucose, affect the activities of HK and PFK by activating the downstream kinase AKT and promote cell glycolysis. 13 As a downstream target of the PI3K/AKT pathway, HIF-1α is considered to be the most important glycolytic transcription factor. Therefore, we also tested whether Te-EtOH exerts its antitumor function by regulating this pathway. Western blot analysis results showed that, compared with that in the control group, the protein expression of p-AKT and HIF-1α in the Te-EtOH group was decreased significantly in a dose-dependent manner in A549 cells (Figure 4A). Subsequently, we explored whether stimulating the activation of the PI3K/AKT/HIF-1α pathway could change the attenuation of cellular glucose metabolism caused by Te-EtOH. We added 10 ng/mL epidermal growth factor (EGF) to trigger activation of the PI3K/AKT/HIF-1α signaling pathway. Microscopic observation analysis (Figure 4B) and fluorescence microscopy after Annexin V and Hoechst staining (Figure 4C) showed that EGF promoted the proliferation of A549 cells, but could be inhibited by 0.2 mg/mL Te-EtOH. As shown in Figure 4D, added EGF protein in A549 cells without drug treatment increased the activation of the PI3K/AKT/HIF-1α signaling pathway and the expression of glycolysis-related proteins such as HK2, LDHA, PDK1, and GLUT1. When treated with Te-EtOH, EGF protein overexpression inhibited the decrease in PI3K/AKT/HIF-1α activation and glycolysis-related protein expression induced by Te-EtOH. These results indicated that Te-EtOH could effectively inhibit glycolysis in A549 cells by inhibiting the PI3K/AKT/HIF-1α signaling pathway.

Te-EtOH inhibited glycolysis in A549 cells by inhibiting the PI3K/AKT/HIF-1α signaling pathway. (A) Western blotting was performed to test the protein levels of PI3K, AKT, p-AKT, and HIF-1α in A549 treated with Te-EtOH (0.01, 0.1, 0.2 mg/mL) or the control. (B) Microscope observation analysis and (C) fluorescence microscope after Annexin V and Hoechst staining were used to detect the proliferation and apoptosis of A549 cells. (D) The upregulation of the PI3K/AKT/HIF-1α pathway activated by 10 ng/ml EGF increases the level of glycolytic metabolism that had been inhibited by 0.2 mg/mL Te-EtOH.
Te-EtOH inhibited the growth of NSCLC cells by suppressing HIF-1α-dependent glycolysis and angiogenesis. Given the importance of HIF-1α in tumor growth and that vascular endothelial growth factor (VEGF) is a key and specific regulator of angiogenesis, which is promoted by HIF-1α under hypoxic conditions, we also explored the effect of Te-EtOH on the expression of HIF-1α, VEGF, and metabolic capability under hypoxic conditions. CoCl2, a hypoxia mimetic agent, was used to induce hypoxia in A549 cells. As shown in Figure 5A, CoCl2 significantly upregulated the expression of HIF-1α and VEGF, which had been inhibited by Te-EtOH. In addition, we also found that Te-EtOH inhibited hypoxia-induced glycolysis, including the activities of HK (Figure 5B), pyruvate kinase (PK; Figure 5C), lactate dehydrogenase (LDH; Figure 5D), and lactate production (Figure 5E). Collectively, our data revealed that Te-EtOH inhibited the growth of NSCLC cells by suppressing HIF-1α-dependent glycolysis and angiogenesis.
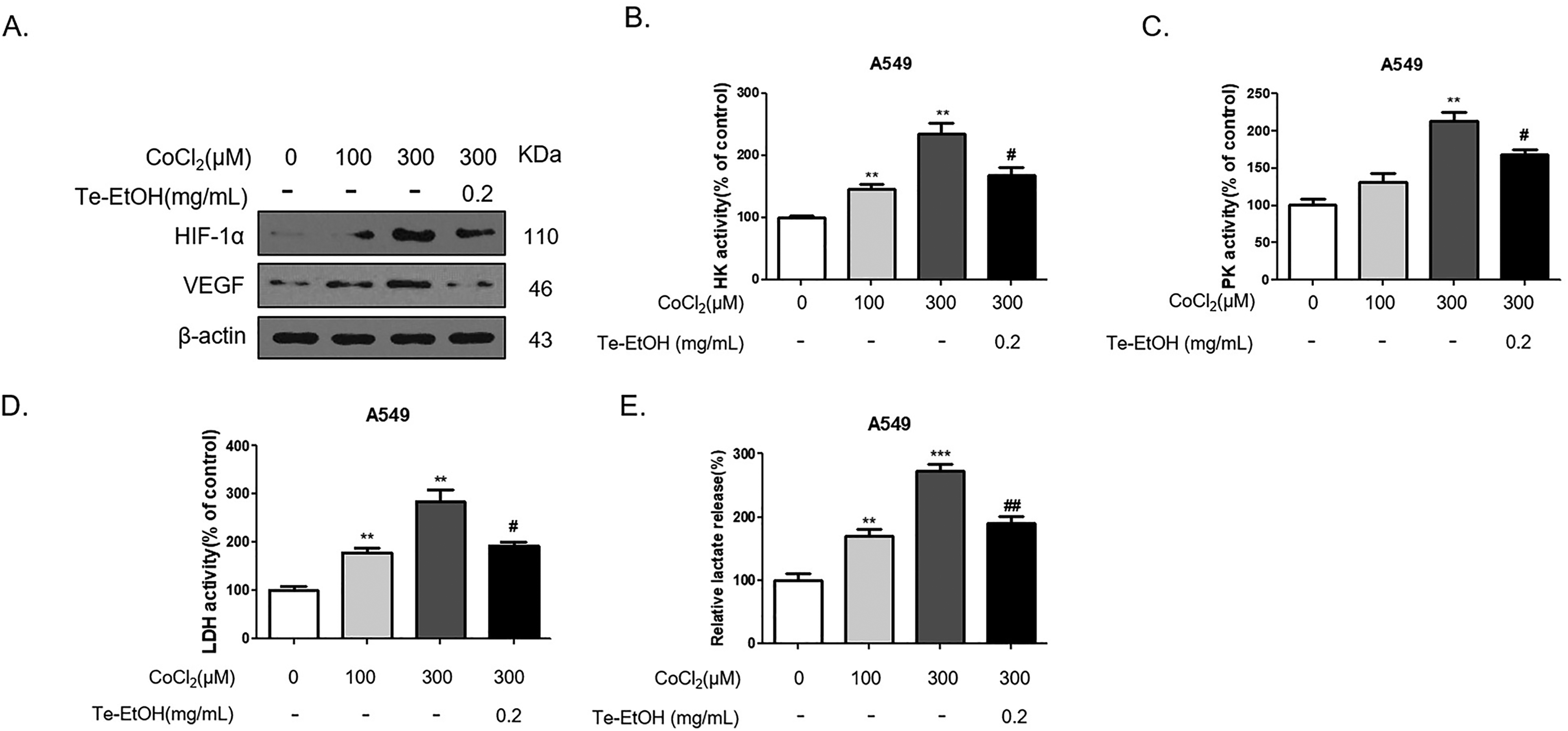
Te-EtOH inhibited hypoxia-induced expression of HIF-1α, VEGF, and metabolic capability in A549 cells. (A) Western blotting was used to determine the protein levels of HIF-1α and VEGF in the normal group and CoCl2-induced hypoxia group. (B–E) The activities of HK, PK, LDH, and lactate production were detected by colorimetric assay using a specific test kit. The mean ± SD from 3 technical replicates is shown. *P < .05, **P < .01, ***P < .01 versus the Ctrl; #P < .05,## P <0.01 versus group of 300 µM CoCl2.
Lung cancer is one of the most common malignant tumors with an extremely high mortality rate. NSCLC is the most common type of lung cancer, especially in Asia. 1 The resistance of NSCLC cells to apoptosis is a major obstacle to successful antitumor therapy. Therefore, the current focus of research is the development of new complexes which can improve the ability of treatments to induce NSCLC cell apoptosis. Numerous previous studies have demonstrated that Te-EtOH displays unique antioxidant capacity in vitro and in vivo as therapeutic adjuvants for multiple cancer types, including prostate, breast, lung, and colorectal.14–19 However, reports on the inhibitory effect of Te-EtOH on NSCLC are rare. In this study, we discovered the potential antitumor effect of Te-EtOH on NSCLC cells. As described previously, the most important substances in Te-EtOH are flavonoids. 6 Studies have shown that flavonoids play an important role in the prevention and treatment of lung cancer through multiple molecular mechanisms of action, including inhibition of activation of key transcription factors to modulate carcinogen-metabolizing enzymes, specific cell cycle arrest, activation of apoptosis, and modulation of cell signaling pathways. These would directly inhibit the development of lung cancer. 15 The alkaloids in Te-EtOH can inhibit or destroy the growth of tumor cells by interfering with mitosis. 20 Other substances, such as polysaccharides and steroids, have rarely been reported in antitumor research.
Many tumor cells can increase glucose uptake and aerobic glycolysis. Glycolysis typically leads to the production of lactic acid and inhibition of the mitochondrial oxidative phosphorylation (TCA) cycle dependent on citric acid in cancer cells. This significant metabolic change, called the Warburg effect, gives tumor cells an advantage under hypoxic conditions. 21 Therefore, the specificity of tumor cells for glycolysis makes them more or less sensitive to treatment with specific glycolysis-target inhibitors, such as the glycolysis inhibitor 2-deoxyglucose (2-DG), which interferes with the function of the glycolysis-initiating enzyme HK. Hence 2-DG has been studied as a promising therapeutic compound targeting tumor metabolism changes. Some evidence suggests that the treatment of NSCLC may be realized by targeting glycolysis-related proteins. For example, a study showed that NSCLC cells treated with the glycolysis inhibitor 2-DG exhibited mitochondrial respiratory defects and increased apoptosis rates. 22 In this study, we found that Te-EtOH inhibited glycolysis and glucose transport in NSCLC cells. We also studied the regulatory mechanism of Te-EtOH on glycolytic metabolism. We found that inhibition of glycolytic metabolism induced by Te-EtOH was related to the inhibition of PI3K/AKT signaling pathway activation.
PI3K/AKT signaling pathway activation mainly promotes cell survival and proliferation, but it can also promote glucose uptake, glycolysis, and enzyme activity in the pentose phosphate pathway. 23 On the one hand, the PI3K/AKT signaling pathway promotes the expression of GLUT1 and GLUT3 and is distributed in clusters on the tumor cell membrane. On the other hand, the PI3K/AKT signaling pathway increases the expression and activity of HK Ⅱ and promotes its binding to mitochondria through the PI3K/AKT/mammalian target of rapamycin pathway. 24 AKT can also significantly regulate the expression of glycolytic enzymes such as 6-PFK-2, and glycokinase is translocated to the outer mitochondrial membrane, which promotes glucose phosphorylation and increases glucose influx into the cellular metabolism machinery. 25 In addition, the PI3K/AKT signaling pathway can also increase the expression of other glycolytic enzymes such as 6-PFK1. Therefore, PI3K/AKT signaling pathway activation is one of the most important mechanisms of abnormal glucose metabolism in tumor cells.
As a downstream target of the PI3K/AKT pathway, hypoxia-inducible factor-1α (HIF-1α) is considered to be one of the most important glycolytic transcription factors and an important cancer-related molecule involved in cellular metabolic reprogramming. The most important feature of tumors is the uncontrolled proliferation of cells and the enlargement of the tumor body. 26 As the blood supply cannot keep up with the growth rate of tumor tissues, a hypoxic microenvironment is formed in the tumor. In fast-growing tumor cells, the oxygen supply cannot meet the demand of mitochondria to produce ATP. Tumor cells then upregulate glycolysis to compensate for the lack of oxidative phosphorylation capacity and avoid death caused by hypoxia. 27 HIF plays an important role in this process. HIF-1 is a nucleoprotein with transcriptional activity with a fairly broad spectrum of target genes, including approximately 100 target genes related to hypoxia adaptation, inflammation development, and tumor growth. When HIF-1 binds to a target gene, the body produces a series of responses through triggered transcription and posttranscriptional regulation. Although some lead to adaptive compensatory functions, these responses often cause pathological damage to the body, such as hypoxic pulmonary hypertension, and tumors show accelerated growth. 28 HIF-1 is closely related to the Warburg effect in tumors.
In addition, it has been confirmed that under hypoxic conditions, HIF-1α is activated and regulates transcription factors that participate in tumor neovascularization, such as VEGF.29, 30 The mechanism may involve hypoxia-driven angiogenesis, hypoxia activation of the PI3K/AKT pathway, or the prevention of HIF-1α posttranslational hydroxylation and subsequent degradation of HIF-1α, causing it to accumulate. HIF-1α is then transferred to the nucleus where it forms part of an initiation complex that initiates the transcription of target genes and leads to increased levels of corresponding protein products, including the increased expression of VEGF, which promotes tumor angiogenesis. 31 The overexpression of VEGF mainly acts through the receptors VEGF receptor-1 and VEGF receptor-2. When VEGF binds to one of its receptors, signal transduction is activated, which leads to endothelial cell proliferation, migration, and angiogenesis. The new blood vessels that thus form provide nutrients and oxygen to tumors, remove metabolic waste products, stimulate tumor growth, and accelerate tumor metastasis. 32 Recent studies have shown that hypoxia/HIF-1α can directly or indirectly (through adenosine, lactic acid, or acidosis) promote the expression of VEGF and activate its receptors, thereby promoting tumor cell evasion of immune surveillance. 33 Furthermore, HIF-1α can participate in the regulation of angiogenesis through VEGF and platelet-derived growth factor, enhance the transcriptional activity of Notch, mediate a tumor metabolism pathway (glucose, lipid, and amino acid metabolic pathways), and exert a tumor-promoting effect through immunosuppression. 34 These studies indicate the possibility of obtaining better results in the treatment of tumors through the HIF-1α/VEGF pathway. HIF-1α and VEGF may be the bases for breakthroughs in the study of the mechanisms of tumor invasion and metastasis, and they may enable the development of additional effective tumor treatment drugs, thereby improving the outcomes of tumor treatment. However, there are some limitations of our study that need further investigation. First, the effect of different main components from Te-EtOH on NSCLC cells still needs to be investigated to further identify the core monomer components that inhibit the glycolysis and angiogenesis of NSCLC cells. Also, further clarification is necessary to determine the effect of Te-EtOH on the tumor growth of NSCLC cells in vivo. In addition, whether there are other mechanisms involved in the inhibitory effect of Te-EtOH on NSCLC cells still needs further research. Therefore, there is still work to be done on how Te-EtOH affects the response to NSCLC.
Conclusions
In this study, we found, for the first time, that Te-EtOH can reduce the level of glycolytic metabolism in NSCLC cells. Furthermore, we have illuminated the mechanism behind the association of Te-EtOH inhibition of HIF-1α-dependent glycolysis and VEGF expression in NSCLC cells.
Materials and Methods
Cell culture and treatment. A549 and H1299 cell lines were purchased from the Chinese Academy of Sciences, and the normal lung epithelial cell line (BEAS-2B) from Procell. The A549 cell line was cultured in F-12K medium (Gibco, 21127022), the H1299 cell line in RPMI 1640 (Gibco, 11875093), and the BEAS-2B cell line in Dulbecco’s Modified Eagle’s Medium (Gibco, 11965092); all cell lines were supplemented with 10% fetal bovine serum (HyClone, sh30084.03), 100 μg/mL streptomycin and 100 U/mL penicillin (Gibco, 15070063). The cells were cultured and grown in a humidified incubator at 37 °C in 5% (v/v) CO2. In addition, 10 ng/mL EGF (Sigma-Aldrich, SRP 3027) was added to trigger activation of the PI3K/AKT/HIF-1α signaling pathway for 8 h in A549 cells.
Preparation of Te-EtOH
Cell viability assay. Cell viability was detected using the CCK-8 kit (Beyotime Biotechnology, C0038), according to the manufacturer's instructions. Approximately 6 × 103 A549 and H1299 cells were seeded per well in a 96-well plate. After treatment with different concentrations of Te-EtOH for 24, 48, and 72 h, the cells were incubated with 10 μL of CCK-8 at 37 °C for 1 h, and the absorbance was measured at OD450.
Cell death assay. The A549 cells were seeded into 96-well culture plates at a density of 1 × 104 cells/well. After overnight culture, they were treated with different concentrations of Te-EtOH and incubated at 37 °C for another 24 h with 5 µg/mL Annexin V-CF594 (Biotium, 29011R) and 10 µg/mL Hoechst (Solarbio Science & Technology, C0020). Optical and fluorescent images were captured using a fluorescent microscope (Nikon Corporation).
Flow cytometry. Apoptosis of A549 cells was assessed with an Annexin V-APC/7-AAD Apoptosis Detection Kit (Keygen Biotech, KGA1025). A549 cells treated with Te-EtOH for 24 h were harvested, then resuspended in 500 µL of 1 × binding buffer, and the resuspended cells were incubated with 5 µL Annexin V-APC and 5 µL 7-AAD in the dark for 5 min before using a CytoFLEX flow cytometer (Beckman) for detection.
Three-Dimensional Cell Culture and Treatment. A549 cells (3000 cells/well) were centrifuged at 1000 r/min for 10 min to facilitate clustering of the cells into the wells and form cell spheroids in ultralow attachment surface round bottom 96-well plates (Corning Inc., 7007). After 4 days, the 3D cultured cells were treated with 0.1 and 0.2 mg/mL Te-EtOH for another 3 days. Then the apoptotic cells of the spheroids were determined using Annexin V, CF594. Optical and fluorescent images were captured using a fluorescent microscope (Nikon Corporation). Images were processed and analyzed using ImageJ software.
Transcriptome sequencing. A549 cells treated either with or without Te-EtOH (0.1 mg/mL) for 24 h were collected in 10 cm plates. Then, the cell samples were used for quality assessment, library construction, and transcriptome sequencing analysis (Shanghai Sequen Gene-Tech Co.).
Glucose consumption test. The concentration of glucose was measured using a glucose assay kit (Sigma-Aldrich, GAGO20), following the manufacturer's instructions. After 24 h of treatment with Te-EtOH, the cell samples were mixed with the glucose assay reagent and incubated for half an hour at 37 °C, and the absorbance at 540 nm was measured. The amount of glucose consumed was then determined by subtracting the amount of glucose remaining at the designated time point from the initial glucose content of the sample.
HK, PK, LDH activities, lactate, and ATP content measurements. The cells were seeded in 6-well plates and treated with Te-EtOH for 24 h. Then, they were harvested using 0.25% trypsin digestion and the protein concentration was determined by the bicinchoninic acid assay (BCA) method. HK assay kit (Solarbio, BC0740), PK activity assay kit (Solarbio, BC0540), LDH activity detection kit (Solarbio, BC0680), lactate assay kit (Nanjing Jiancheng Bioengineering Institute, A019-2-1) and ATP assay kit (Beyotime Biotechnology, S0026) were used to detect HK, PK, LDH activities, lactate, and ATP contents according to the manufacturer's methods. Relative values were calculated by normalizing the activities of HK, PK, LDH, lactate, and ATP in the control group to 1.0.Western blot. The total protein in the cell samples was extracted, and the protein concentration was determined by the BCA method. Protein electrophoresis was performed on a denaturing sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel and then transferred to poly(vinylidene fluoride) membranes (Millipore). After that, the membranes were blocked with 5% fat-free milk for about 2 h, washed with 1 × phosphate-buffered saline containing 0.1% Tween-20 (PBST), and incubated with primary antibodies (antibodies against PDK1 (Proteintech, 10026-1-AP), GLUT1 (Abcam, ab115730), LDHA (Proteintech, 19987-1-AP), PI3K (Proteintech, 20584-1-AP), AKT (Proteintech, 20584-1-AP), p-AKT (Proteintech, 20584-1-AP), HIF-1a (Proteintech, 20584-1-AP), VEGF (Proteintech, 66828-1-IG), and β-actin (Boster, BM0627)) for another 2 h at room temperature. Subsequently, the membranes were washed 3 times with 1 × PBST and incubated with horseradish peroxidase-conjugated secondary antibody for 1 h at room temperature. Finally, immunoreactive products were visualized by enhanced chemiluminescence (Thermofisher), as described in our previous publications. 7
Statistical analysis. GraphPad Prism 8.0 (GraphPad Software, Inc.) was used to analyze the statistics of the results, and the data were expressed as means ± standard deviation from 3 independent experiments. A two-sided Student's t-test was used to determine the significance between the 2 groups, and *P < .05 was considered significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221142796 - Supplemental material for Tetrastigma hemsleyanum Ethanolic Extract Inhibited the Growth of Nonsmall Cell Lung Cancer Cells by Suppressing Hypoxia-Inducible Factor-1α-Dependent Glycolysis and Angiogenesis
Supplemental material, sj-docx-1-npx-10.1177_1934578X221142796 for Tetrastigma hemsleyanum Ethanolic Extract Inhibited the Growth of Nonsmall Cell Lung Cancer Cells by Suppressing Hypoxia-Inducible Factor-1α-Dependent Glycolysis and Angiogenesis by Zhiqiang Wu, Bin Xu, Qin He, Zhuyuan Hu and Zhiyi Yu in Natural Product Communications
Footnotes
Acknowledgments
We extend our gratitude to Shishi Zhou and Caijun Dai, affiliated with Jinhua Hospital, Zhejiang University School of Medicine, for sharing their ideas with us on the design of the study and data analysis during this research.
Authors’ Contributions
Concept of the study and supervision by Zhiyi Yu. Data collection: Zhiqiang Wu, Bin Xu, Qin He, and Zhuyuan Hu. Design of the study: Qin He, Bin Xu, and Zhiyi Yu. Statistical analysis: Qin He, Zhuyuan Hu, Zhiqiang Wu, and Zhiyi Yu. Analysis and interpretation of the data: Zhiyi Yu. Drafting the manuscript: Zhiqiang Wu, Bin Xu, Qin He, Zhiyi Yu, and Zhuyuan Hu. Critical revision of the manuscript: Zhiyi Yu, Zhiqiang Wu, Zhuyuan Hu, and Bin Xu.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Youth Scientific Research Fund Project of Jinhua Central Hospital (No. JY2019-1-01) and the Major (Key) Science and Technology Research Project of Jinhua City (Nos. 2018-3-017 and 2021-3-0093).
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
