Abstract
The lichen host,

Cancer is one of the leading causes of death worldwide. Chemotherapy has been the choice for cancer treatment for many years, but this can also affect normal cells and create many undesirable side effects and has the potential to develop resistance. Therefore, alternative medicines of natural origin are gaining attention in cancer research. Among them, fungal metabolites have gained much attention in the scientific community as a source of novel bioactive compounds. Endolichenic fungi are a diverse group predominantly inhabiting asymptomatically in the interior of lichen thalli. These fungi produce a broad spectrum of secondary metabolites for their survival in order to overcome their local challenges. Since the first report of metabolites of an endolichenic fungus,
1
176 compounds have been isolated, of which 104 were identified as new compounds
2
; several of these with antioxidant and antibacterial properties have been reported.
3
Kannangara et al
4
isolated several endolichenic fungal strains with antifungal properties from lichens found in Hakgala montane forest in Sri Lanka. Five novel bioactive polyketides with antioxidant properties have been isolated and characterized from the endolichenic fungi,
Results and Discussion
Isolation and Identification of Fungal Strains
The endolichenic fungus isolated from the lichen host,
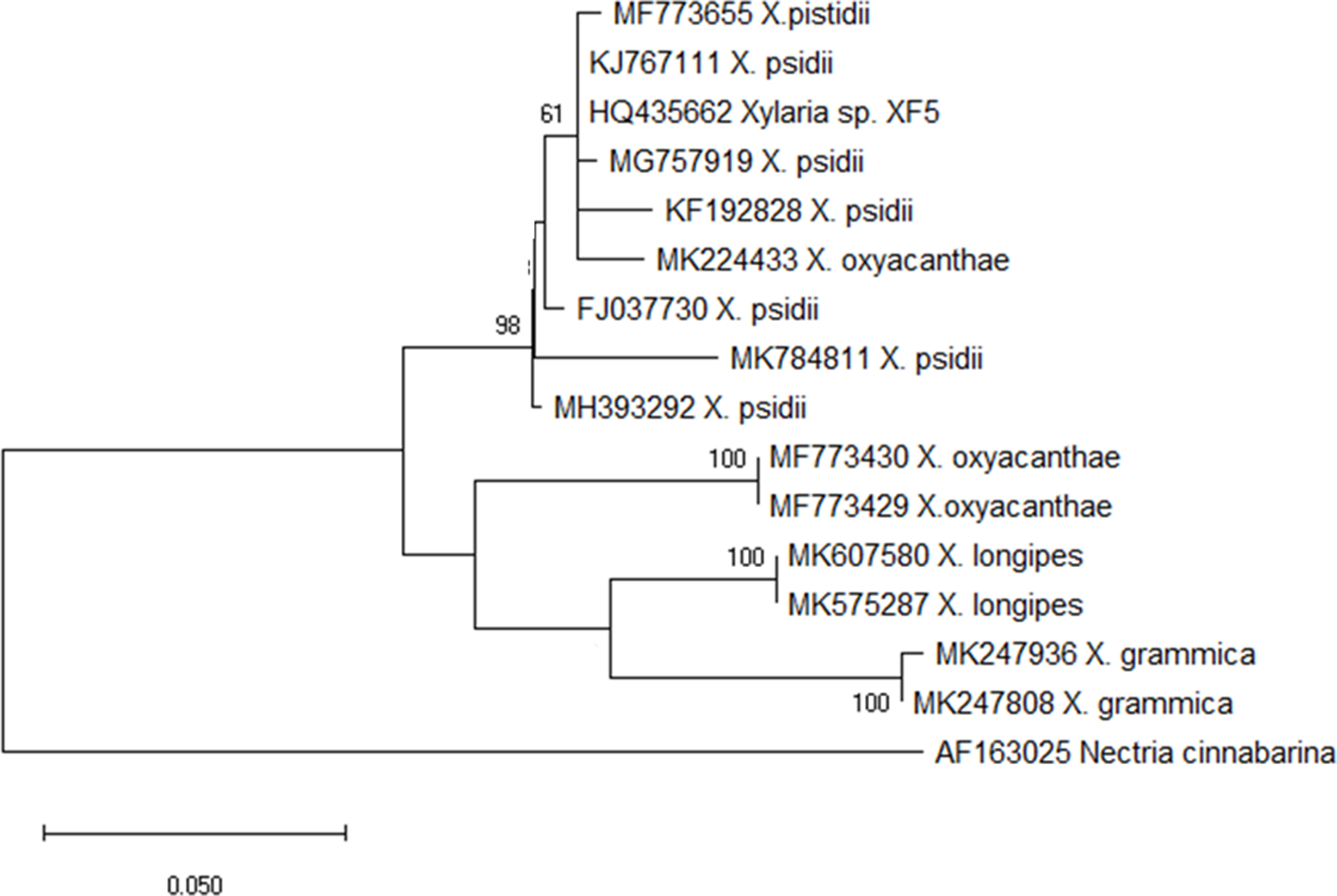
Maximum likelihood tree with the highest log likelihood (−2045.87) is shown. The percentage of trees in which the associated taxa clustered together is shown next to the branches in bootstrap values. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. The sample used in the current study is shown in GenBank accession number MF773655.
Bioassay-Guided Isolation of the Bioactive Compounds of Xylaria psidii and Their Cytotoxicity
The cytotoxic activity of the crude extract of

Cytotoxicity of (A) EtOAc extract and fractions obtained from the EtOAc extract of
Percentage Cell Survival of Lung Cancer Cells (NCI-H292) and IC50 Values.

IC50 values obtained for the crude/subsequent fractions isolated from the endolichenic fungus,
Structure Elucidation of Compound 1
Bioassay-guided fractionation of the anticancer active EtOAc extract of
The presence of 2 six-membered aromatic rings in compound
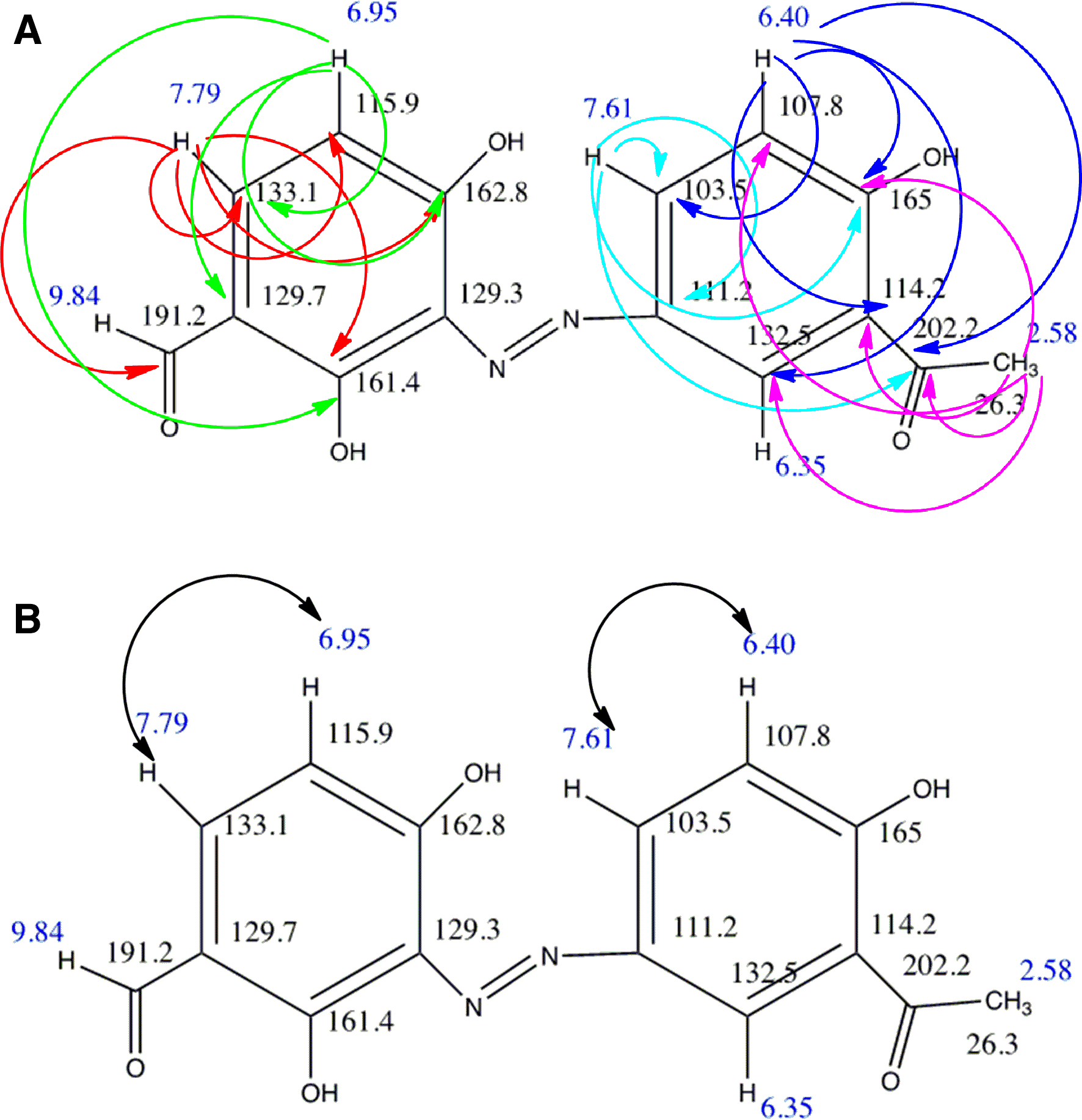
(A) Key HMBC and (B) DQF-COSY correlations for compound 1.
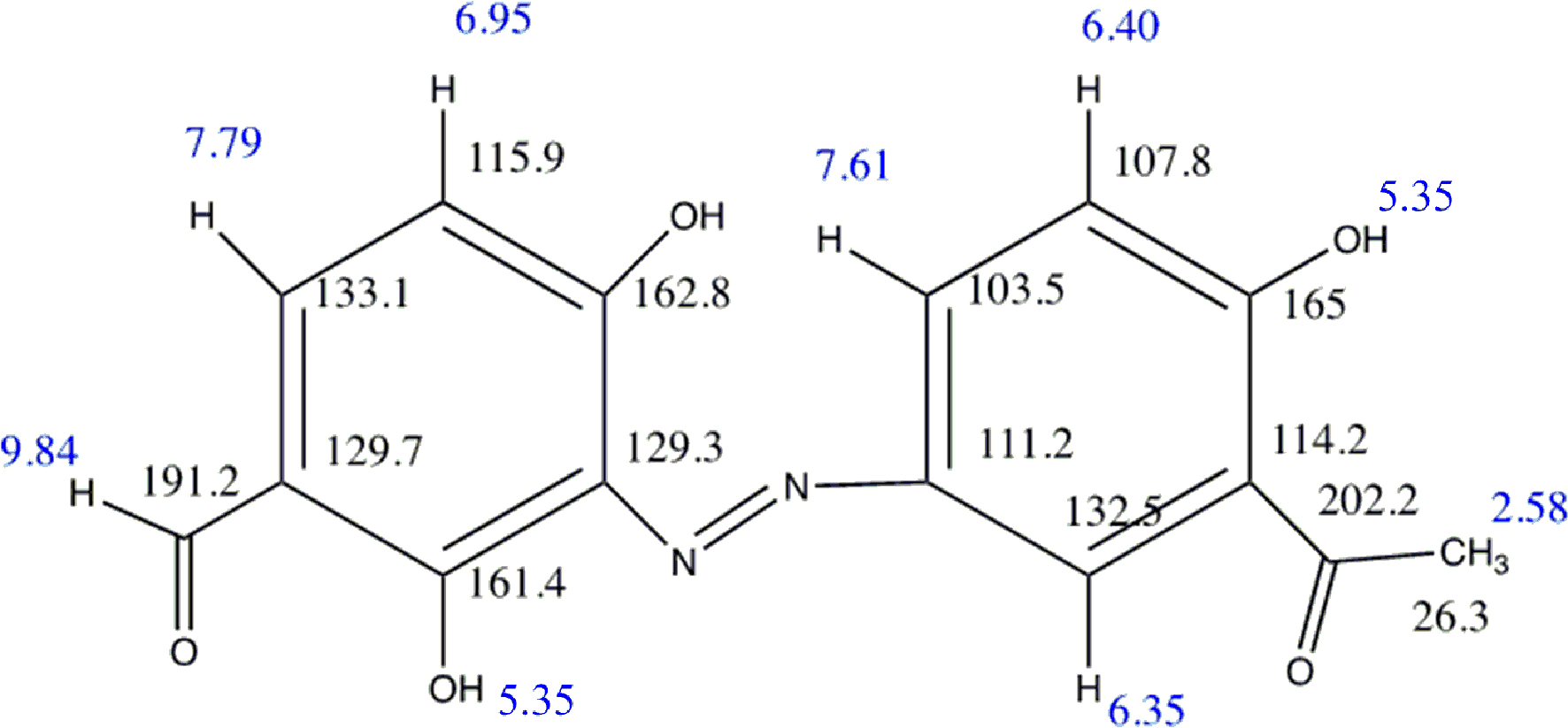
Structure of compound 1.
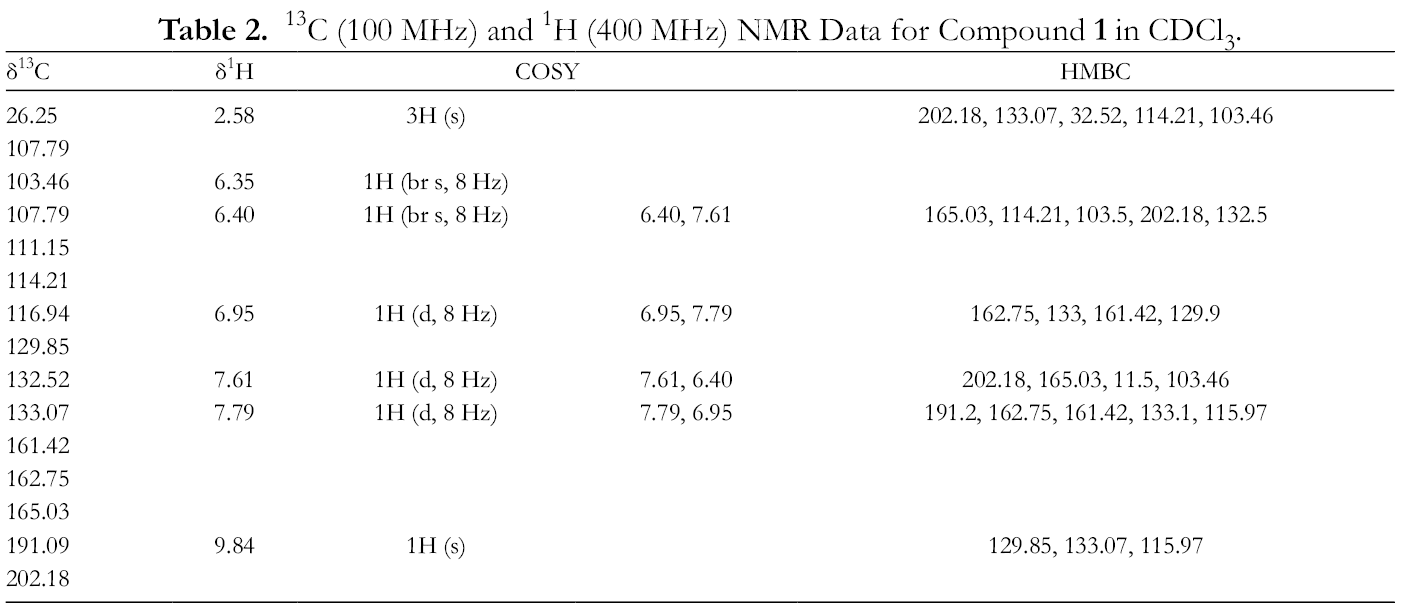
13C (100 MHz) and 1H (400 MHz) NMR Data for Compound 1 in CDCl3.
The genus
Conclusion
Compound
Methodology
Isolation and Identification of Fungal Strain and Molecular Phylogeny
The lichen host,
Molecular identification of the isolated pure cultures of the endolichenic fungi were carried out as described in Maduranga et al.
9
Total genomic DNA was isolated from selected pure cultures, and the nuclear robosomal interanl transcribed spacer region (rDNA-ITS) region was amplified using polymerase chain reaction (PCR); the PCR products were subjected to Sanger dideoxy sequencing at the Genetech Institute, Colombo, Sri Lanka. Sequences were manually edited using BioEdit sequence Alignment Editor (Version 7.2.5). Using Basic Local Alignment Search Tool (BLAST), the sequences were compared with the sequences at the National Center for Biotechnology Information (NCBI) database. Based on the morphology and molecular characteristics,
Preparation of EtOAc Extract of Xylaria Psidii and Determination of Anticancer Activity
A seed culture of
The NCI-H292 cell line, obtained from a 32-year-old female patient with mucoepidermoid pulmonary carcinoma, was used for the cytotoxicity assay. It is an adherent epithelial lung tissue of
The cytotoxic activity of the crude extract was determined in a flat bottom 96-well microtiter plate using the sulforhodamine B (SRB) assay against the NCI-H292 lung cancer cell line according to the method described by Samarakoon et al. 11 The percentage of cells surviving was determined after 24 hours by measuring the optical density at 540 nm, using a microplate reader (Synergy, Micro Plate Reader, United Kingdom). Three replicates were carried out for each experiment. IC50 values of the crude extracts were calculated using GraphPadPrism (version 6.0) software package.
Isolation of Cytotoxic Compounds in the EtOAc Extract
A portion (1.32 g) of the crude extract was partitioned with
Bioassay-Guided Isolation of the Cytotoxic Compounds in the MeOH Fraction
A portion (48.5 mg) of the MeOH fraction was subjected to silica gel column chromatography. The column was eluted with CH2Cl2 followed by increasing amounts of MeOH in CH2Cl2 and finally 100% MeOH. The resulting fractions were pooled on the basis of their TLC profiles to obtain 5 fractions. These were further purified either by silica column chromatography as described above or preparative TLC (9 × 20 cm, stationary phase: 60 F-254, Fluka, Germany) depending on the yield of each fraction, with the solvent system, 3% MeOH in CH2Cl2. Chromatographic separation was repeated on subsequent fractions until the isolation of pure compounds (Supplemental Figure S1) and their cytotoxic activity was determined. Only 1 of them (compound
Determination of the Structure of Compound 1
The structure of the compound was determined by 1H-NMR and 13C-NMR spectrophotometry in combination with DQF-COSY, HMBC, and HSQC correlations. HRMS was used to obtain the molecular mass of the compound.
Supplemental Material
Supplementary Material 1 - Supplemental material for A Novel Cytotoxic Compound From the Endolichenic Fungus, Xylaria psidii Inhabiting the Lichen, Amandinea medusulina
Supplemental material, Supplementary Material 1, for A Novel Cytotoxic Compound From the Endolichenic Fungus,
Footnotes
Acknowledgment
We wish to thank Prof. Kiran Kalia, Director (NIPER-A) and Dr Abhijeet S. Kate, Assistant Professor, National Institute of Pharmaceutical Education & Research, Ahmedabad for providing HRMS data of the pure compound.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Council (Grant no. NRC/15-08) and the Ministry of Science Technology and Research (MSTR/TRD/AGR/03/02/07).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
