Abstract
Objectives
Blood vessels provide convenient conditions for tumor cell proliferation and distant metastasis. Antiangiogenesis and proapoptosis of tumor cells have become critical means of clinically treating tumors. This study aimed to investigate the antiangiogenic and pro-apoptotic abilities of α-solanine.
Methods
This study used the CCK8 method to select appropriate drug concentrations. Next, the antitumor mechanism of α-solanine under hypoxia was investigated using wound healing, transwell, cellular immune, and Western blot assays. Finally, a reverse validation was performed by adding MHY1485, an mammalian targets of rapamycin (mTOR) activator.
Results
α-Solanine significantly decreased B-cell lymphoma2-related protein, n-Ca, vascular endothelial growth factor, MMP2, and MMP9 expression in cells. In addition, Bax, p53, TIMP1, and E-Ca expressions were upregulated. However, this study found that adding MHY1485 significantly attenuated the effects of α-solanine. These results suggest that α-solanine may achieve these functions by regulating the PI3K/Akt/mTOR signaling pathway.
Conclusion
α-Solanine is highly resistant to angiogenesis and promotes apoptosis of M2 tumor-associated macrophages. These effects may depend on the PI3K/Akt/mTOR signaling pathway. The present study demonstrated that α-solanine has a strong anticancer effect under hypoxic conditions.
Introduction
Cancer is a major global public health problem that critically threatens human health with increasing incidence and mortality. Approximately 10 million people die of cancer annually, resulting in huge population losses and a heavy economic and medical burden. With the aging of the population, this trend has become more obvious. 1 Although chemotherapy is an effective treatment strategy for tumors, chemotherapy drugs have severe cytotoxic effects and are prone to drug resistance in tumors. 2 Relevant research institutions in Chinese herbal medicine have identified numerous herbal preparations that produce satisfactory clinical and therapeutic effects on tumors. Consequently, many studies have focused on natural products in developing antitumor drugs.
Hypoxia is a characteristic of almost all solid tumors that causes immune impairment, modifies tumor metabolism, and promotes tumor progression.3,4 Hypoxia develops as an adaptive response during cancer progression, impedes drug penetration into tumor regions, manages immune responses, and directs cellular plasticity. These abilities lead to drug resistance, recurrence, and metastasis.5–7 Hypoxia is mainly regulated by the activity of hypoxia-inducible factor-1α (HIF-1α), which is involved in several biological processes related to tumor immunity. 8 Hepatocellular carcinoma (HCC) is prevalent cancer and profound vascular disease. HCC has significantly higher serum levels of HIF-1α than benign liver diseases. 9 Hypoxia is also a common element in the tumor microenvironment (TME) of pancreatic ductal adenocarcinoma, a highly deadly cancer. 10 HIF-1α can promote invasion and metastasis of pancreatic cancer cells and their resistance to chemotherapy.11,12 Therefore, the present study was conducted under hypoxia to create realistic conditions mimicking the microenvironment of solid tumors and closely monitor the expression of HIF-1α.
The growth and metastasis of solid tumors depend on tumor angiogenesis, with blood vessels providing convenient conditions for tumor cell proliferation and distant metastases. Antiangiogenesis has become an essential means of clinical treatment of tumors. Tumor angiogenesis involves the dissolution of the vascular matrix membrane, increase in vascular permeability, and degradation of the extracellular matrix. Compared to normal angiogenesis, tumor blood vessels tend to be unevenly distributed and form a disorderly and distorted vascular network, leading to insufficient blood supply and high vascular permeability. 13 Vascular endothelial growth factor (VEGF), a regulatory cytokine, is a key promoter of angiogenesis.6,14 VEGF and HIF-1α are the strongest angiogenesis promoters associated with each other. HIF-1α activates VEGF to promote angiogenesis in cancer. 9 VEGF can promote tumor dedifferentiation and epithelial mesenchymal transition (EMT) transition through the phosphorylation of AKT, ERK1/2, and other signaling pathways. It also enhances tumor migration and survival ability and plays a significant role in the function and self-renewal of tumor stem cells, increasing the degree of malignancy of tumors. 15 MMP2 and MMP9 are important members of the MMPs family that play a key role in degrading extracellular matrix mediated by cancer cells and promoting tumor angiogenesis by hydrolyzing matrix components. 16 EMT is a temporary and reversible process that transitions from epithelial cells to mesenchymal cells to form mesenchymal cells such as fibroblasts. It is regarded as an important step involved in tumorigenesis and metastasis. Existing studies suggest that EMT plays an important role in the angiogenesis process. It has been found that EMT occurs with the above-mentioned component changes. In pancreatic cancer, hypoxia can promote EMT of pancreatic cancer cells, maintain the stemness of pancreatic cancer cells, and increase the expression of microenvironmental components such as VEGF. 17 B-cell lymphoma2-related protein (Bcl-2) is one of the earliest members of the BCL-2 protein family that regulates apoptosis, and it is also a drug target related to apoptosis. Bax itself forms a homodimer, while Bcl-2 and Bax form a heterodimer. When there is more Bax in the cell, the homodimer dominates and is prone to apoptosis, while when there is more Bcl-2, the heterodimer increases and the apoptotic tendency is weakened. The Bax/Bcl-2 ratio was used to determine whether the cell had entered apoptosis. It has been found that Bax expression is down-regulated and Bcl-2 expression is up-regulated in human colon cancer cell lines. 18 Bcl-2 and Bax affect the sensitivity of tumor cells to a variety of apoptotic stimuli by forming homodimers or heterodimers, and their ratio is key to ultimately determining cell survival or death. Therefore, this study examined the expressions of these vascular and apoptosis factors to determine the antitumor efficacy of α-solanine.
Solanine, a naturally occurring steroidal alkaloid, was first identified in 1820 and has since been predominantly found in plants such as potatoes and tomatoes. 19 Solanine can be divided into α-solanine, β-solanine, and γ-solanine, with α-solanine having the highest content. Solanine induces apoptosis through the mitochondrial pathway. 20 Shen et al 21 found that α-solanine can inhibit the invasion of human prostate cancer cells, which may be related to its ability to reduce MMP-2/9 expression, up-regulate the miR-138 expression, down-regulate miR-21 gene expression, and inhibit the phosphorylation of PI3K, Akt, ERK, and other proteins. α-Solanine has a wide range of antitumor effects. Therefore, researchers have begun to study using solanine and α-solanine to treat tumors. By examining the biological activity of α-solanine, this study further researched the effects of α-solanine on the apoptosis and angiogenesis of M2 tumor-associated macrophages (TAMs) and its related mechanism. This research is significant for developing current drugs based on α-solanine.
Materials and Methods
Reagents. α-Solanine (Shanghai Yi En Chemical Technology Co., Ltd, R128770) solution was prepared in dimethyl sulfoxide. MHY1485 was purchased from MedChemExpress (HY-B0795), IL-4 from PeproTech EC Ltd, fetal bovine serum (FBS) from Sigma-Aldrich, and Trypsin and Dulbecco's modified Eagle's medium (DMEM) from Gibco. Anti-HIF-1 alpha (cat. no. A11945), anti-VEGF (cat. no. A12303), anti-MMP9 (cat. no. A0289), anti-MMP2 (cat. no. A11144), anti-p-AKT1 (cat. no. AP0637), anti-mammalian targets of rapamycin (mTOR) (cat. no. A11355), anti-p-mTOR (cat. no. Ap0115), anti-p-PI3K (cat. no. Ap0854), anti-N-Ca (cat. no. A19083), anti-E-Ca (cat. no. A20798), anti-TIMP1 (cat. no. A1389), and anti-β-actin (cat. no. AC004) were obtained from Abclonal. p53 (cat. no. BF8013) was purchased from Affinity Biosciences, and Bcl-2 (cat. no. K003505P) and p53 (cat. no. K008076P) from Solarbio.
Cell culture. The mouse macrophage cell line RAW264.7 was purchased from Prisill Life Technologies Ltd The cells were cultured in a DMEM medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin. When the cell density reached approximately 80%, the cells were directly blown on the culture's flask wall with a pipette, and experimental treatments were performed.
Treatment of cells. The experiment was divided into 4 groups. (a) Group A: The control group; (b) Group B: The cells were induced with IL4 (10 ng/mL) to become M2-like TAM; (c) Group C: 1 µg/mL α-solanine was added after the cells were induced into M2-like TAM with IL4; and (d) Group D: 3 µg/mL α-solanine was added after the cells were induced into M2-like TAM with IL4. The groups were placed in a hypoxic incubator (37 °C, 1% O2) for 24 h. After that, the mTOR activator MHY1485 was added to co-culture with M2 cells for reverse validation. At this stage, the cells were divided into 3 groups: (a) M2 Group: Cells were induced into an M2-like TAM with IL4; (b) drug treatment group: 3 µg/mL α-solanine was added after the induction of cells into M2-like TAM with IL4; and (c) Inhibitory Group: Cells were induced to M2-like TAM with IL4, and 10 µmol/L of MHY1485 was added. The groups were placed in a hypoxic incubator (37 °C, 1% O2) for 24 h.
Cell viability assay. After calculating the concentration of digested cells, this study inoculated the cells into a 96-well plate, with a cell count of about 1 * 104 per hole and then cultured in an incubator. When the cell density reached approximately 80%, the culture solution was modified to serum-free DMEM. At this stage, different concentrations of α-solanine (0.5, 1, 2, 3, 4, 6, 8, 10, or 12 µg/mL) were added. After 24 h, 10 µL of CCK8 solution was added to each hole and incubated for 2 h. Finally, the absorbance was measured for each hole in the enzyme marker.
Scratch test. The cells were seeded into 6-well plates. When the cell density reached approximately 90%, 3 straight lines were drawn perpendicular to the mark using a sterilized white lance. The cells were washed thrice with PBS and treated with serum-free media and corresponding drugs. At this stage, pictures were taken directly under the microscope. The cells were then placed in a hypoxic incubator, cultured for another 24 h, and photographed again under a microscope.
Cell invasion and migration assays. The Biozellen three-dimensional Cell Culture Hydrogel Kit (No: B.P.00002.2) was used for the invasion assay. After the cells were centrifuged, they were blown down to homogeneity using serum-free culture media, and 100 μL of culture medium containing cells was added to each Transwell chamber. In migration and invasion assays, the samples corresponded to cell numbers of 5 × 103 and 5 × 104, respectively. The corresponding drugs were then added to the upper chamber. Finally, 600 μL of the finished culture medium containing 10% serum was added to the lower chamber and incubated in a hypoxic incubator for 24 h. The samples were washed with PBS the next day, and 4% paraformaldehyde was used for cell fixation. Subsequently, the samples were stained again with hematoxylin crystals and photographed.
Flow cytometry. The Annexin V-FITC/PI apoptosis detection kit was purchased from KGI Bio (cat. NO: KGA107). After cell treatment, pancreatin without EDTA was used to digest the cells. After that, we washed twice with PBS, followed by centrifugation (800 g centrifugal force, 5 min), and collected about 50,000 cells/group. After adding 500 µL Annexin V-FITC, 5 µL propidium iodide was added to the mix. They were then allowed to react in the dark at room temperature for 10 min. After that, we used flow cytometry for up-machine detection.
Western blot analysis. The samples were placed on ice and washed thrice with PBS. After the last wash, the cell lysate comprising 200 μL RIPA, 25 μL PMSF, and 2 μL phosphatase inhibitor was added to the samples. After centrifugation, the supernatant was extracted. Protein concentrations in each group were determined according to the instructions of the BCA kit (Abclonal, RM00005). These were then electrified at 80 V for 30 min and 120 V for 65 min. After cutting the glue, these were transmembraned at 200 mA. Subsequently, the primary antibodies were blocked and incubated. Antibodies of Bcl-2, n-Ca, VEGF, MMP2, MMP9, Bax, p53, TIMP1, E-Ca, HIF-1ɑ, p-AKT1, mTOR, p-mTOR, and p-PI3K had concentrations of 1:1000 and the β-actin concentration was 1:10 000. After incubating the primary antibody for 1 day, the corresponding secondary antibody was incubated and exposed.
Immunofluorescence staining. First, samples were mixed with paraformaldehyde. Subsequently, 0.5% Triton X-100 was added to the samples for 20 min. After another PBS wash, 5% goat serum was added for blocking, after which each sample was then incubated with the corresponding primary antibody and stored overnight at 4 °C. The next day, the corresponding fluorescent secondary antibody was added and incubated for an additional hour. After washing the samples, a drop of DAPI solution was added that reacted for 5 min. At this time, we covered the glass slide with an antifluorescence quenching agent. Finally, we took pictures of each group under an inverted fluorescence microscope.
Statistical analysis. SPSS software (version 26.0) was used to process all data. The representative curves in the pictures are displayed as the mean ± standard deviation. A 1-way analysis of variance test was used to evaluate the differences between groups. P < .05 indicated that the difference between groups was statistically significant. All experiments were repeated thrice.
Results
The Cytotoxic Effects of α-Solanine and its Impact on Cell Invasion and Migration Under Hypoxia
CCK8 assay was used to study the effects of different concentrations of α-solanine on cell viability. The results showed that the cell survival rate was not significantly reduced when the concentration of α-solanine was 0-3 μg/mL. However, a significant reduction and toxic effect on cells occurred when the concentration was greater than 4 μg/mL (Figure 1A). Therefore, 1 and 3 μg/mL concentrations were selected for subsequent correlation experiments. In the scratch test, the number of migrating cells in Group B was significantly more than in Group A. In addition, the number of cells in Groups C and D was considerably lower than in Group B. These observations indicated that α-solanine could effectively inhibit cell migration (Figure 1B). The Transwell experiment showed that the number of invading and migrating cells was significantly larger in Group B than in Group A, consistent with the M2-like TAMs feature. After adding α-solanine, cell invasion, and migration were significantly downregulated (Figures 1C-F). These experiments established the optimal concentration of α-solanine, demonstrated its antimigration and anti-invasion abilities, and laid a foundation for additional research on its action mechanism.

Determination of ɑ-solanine toxicity and its effect on cell migration and invasion under hypoxia. (A) RAW264.7 cells were treated with different concentrations of α-solanine. (B) Images of each group at 0 and 24 h in the scratch test. (C) Representative image of RAW264.7 cells in Transwell assay using a magnification of 200×. + indicates the addition of 1 µg/mL α-solanine, and ++ indicates the addition of 3 µg/mL α-solanine.
Under Hypoxic Conditions, α-Solanine Regulated Angiogenesis and Apoptotic Factors
M2-like Tams significantly promote angiogenesis compared to macrophages in hypoxia. 22 MMP2, MMP9, TIMP1, E-Ca, N-Ca, and VEGF are associated with tumor cell angiogenesis. The Western blot experiment showed that MMP2, MMP9, N-Ca, and VEGF expression levels in M2-like TAM were higher than in macrophages (Figure 2). However, their expression levels were significantly reduced after treatment with α-solanine. In addition, when cells were treated with α-solanine, TIMP1 and E-Ca expressions were significantly increased. Cellular immunofluorescence experiments also confirmed that MMP2 expression was inhibited (Figure 3A). In contrast, E-Ca expression was significantly increased after adding α-solanine, consistent with Western blot results (Figure 3B). Regarding apoptosis, Bcl-2 expression was significantly inhibited, while Bax and p53 expressions were significantly increased after the addition of α-solanine. Simultaneously, flow cytometry also suggested that solanine could promote apoptosis (Figure 2A). Therefore, α-solanine can substantially inhibit the proangiogenic, proproliferation, and antiapoptosis abilities of M2-like Tams.

α

Effect of α-solanine on MMP2, E-Ca and HIF-1ɑ under hypoxic conditions. Effects of α-solanine on (A) MMP2, (B) E-Ca, and (C) hypoxia-inducible factor-1α (HIF-1ɑ) in cellular immunofluorescence experiments at a magnification of 200×. + indicates the addition of 1 µg/mL α-solanine, and ++ indicates the addition of 3 µg/mL α-solanine.
α-Solanine Regulated PI3K/AKT/mTOR Signaling Pathway Under Hypoxic Conditions
In this study, we first assumed that α-solanine might inhibit the proangiogenic and antiapoptotic abilities of tumor cells by regulating the PI3K/AKT/mTOR signaling pathway. In the results, it could be distinctly observed that HIF-1α, p-PI3K, and p-AKT in this pathway were regulated by α-solanine (Figures 3C and 4). In addition, the antiangiogenic and proapoptotic abilities of α-solanine were significantly inhibited after adding the mTOR agonist MHY1485 (Figure 5). Therefore, this study clarified that α-solanine inhibits angiogenesis and promotes apoptosis in M2-like TAM cells by regulating the PI3K/AKT/mTOR signaling pathway.

Regulation of PI3K/AKT/mammalian targets of rapamycin (mTOR) signaling by α-solanine under hypoxic conditions. (A) Graphical representation of bands for various purposes. (B-F) Line plots of relative expressions of various target bands. *P < .05 versus Group A; #P < .05 versus Group B. + indicates the addition of 1 µg/mL α-solanine, and ++ indicates the addition of 3 µg/mL α-solanine.
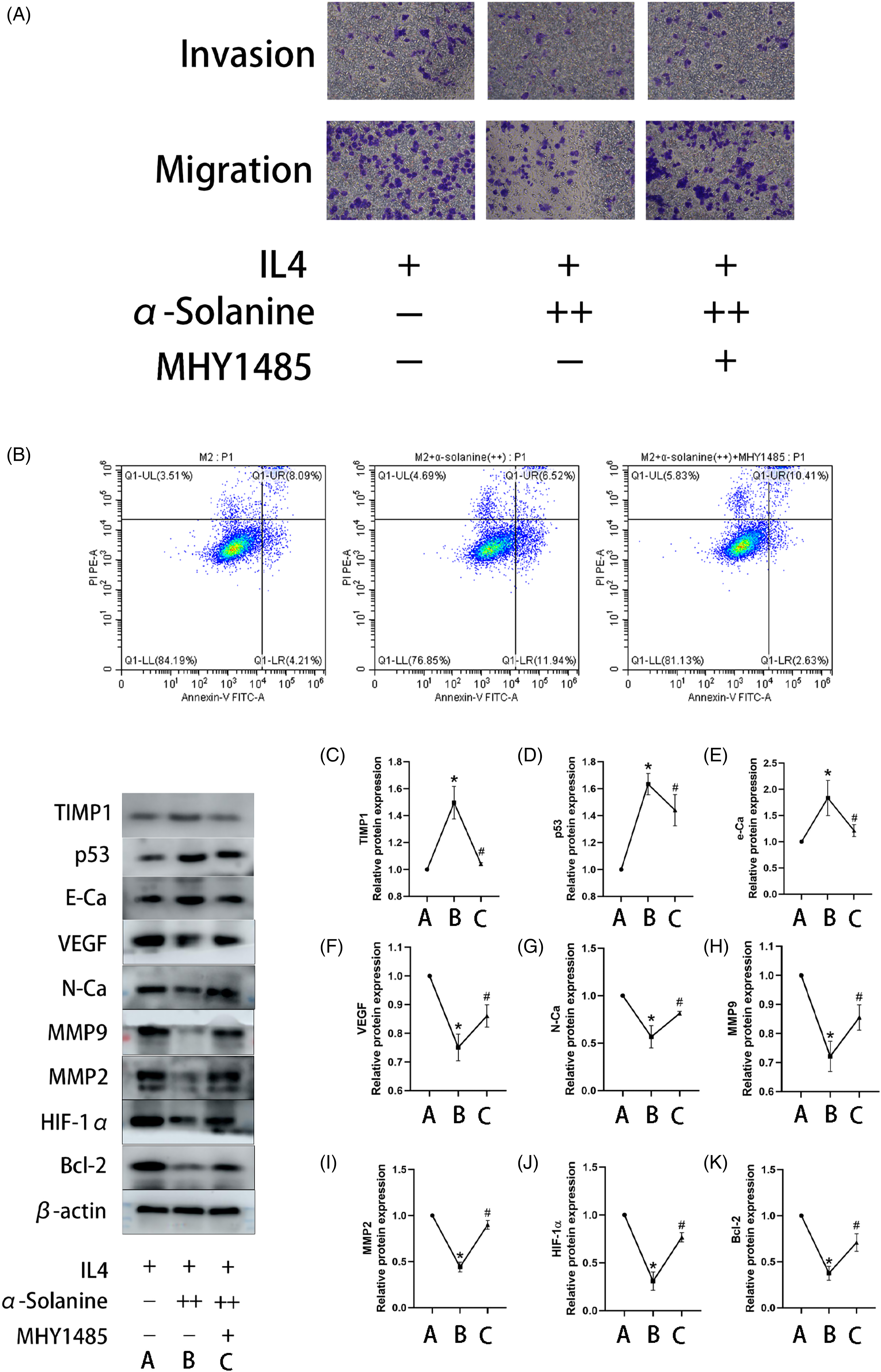
MHY1485 inhibited the anticancer ability of ɑ-solanine under hypoxic conditions. (A) Representative image of RAW264.7 cells in the Transwell assay at a magnification of 200×. (B) Flow plots of apoptosis in each group. (C-G) Line plots of relative expressions of various target bands. *P < .05 versus Group M2; #P < .05, versus Group M2+ α-solanine. ++ indicates the addition of 3 µg/mL α-solanine.
Discussion
Macrophages infiltrated in tumor tissues are called TAMs, which are highly plastic and heterogeneous and can adjust their functional status with changes in the TME. 23 In the early stages of tumor development, proinflammatory cytokines in TME can promote the polarization of TAMs to M1 type, killing tumor cells. As the tumor develops, tumor cells can promote the polarization of TAMs to M2 type by releasing CSF1. HIF-1α and NF-ĸB activations occur during cancer invasion and are positively correlated with the release of granulocyte-macrophage colony-stimulating factor (GM-CSF) from the tumor. GM-CSF helps the tumor to recruit other M2 cells, promoting tumor progression. 24 Class M2 TAMs play an essential role in the biological activity of tumors. Therefore, regulating M2 TAMs is an effective strategy for treating tumors. 25 This study investigated the properties of macrophages by inducing their conversion into M2 macrophages via IL4 and found that M2-like TAMs possessed potent tumor-promoting activity.
Angiogenesis, the process by which new capillaries emerge from pre-existing blood vessels, is essential for the growth and metastasis of many solid tumors. 6 When proangiogenic molecules outnumber antiangiogenic molecules, angiogenic mechanisms are activated. 26 Among the identified proangiogenic factors, VEGF is a key mediator that regulates the growth and maintenance of pathological blood vessels. 27 It is also a potent vasodilator that enhances the mobilization of bone marrow-derived endothelial progenitor cells. As revascularization is associated with malignant tumor aggressiveness, it is reasonable to delay tumor growth and metastasis by inhibiting angiogenesis. Antiangiogenic therapy has been successfully applied to colorectal cancer and other tumor types.28,29 Since its approval in 2004, bevacizumab, a humanized monoclonal antibody that binds VEGF-A, has been used as the first-line therapy in colorectal, non-small-cell lung, metastatic breast, and metastatic renal cell cancers. It is also used as the second-line therapy in glioblastoma multiforme. 30 Other anti-angiogenic drugs approved by the Food and Drug Administration are multitargeted tyrosine kinase inhibitors. These include sunitinib, sorafenib, and pazopanib, which target VEGF receptors, particularly VEGFR-2. 31 Studies have found that 100 μM capsaicin induces EMT in colorectal cancer SW480 cells, activates AKT/mTOR and STAT3 pathways, and induces increased expression of MMP2 and MMP9 in the TME. 32 Angiogenesis and EMT play an important role in tumor invasion, recurrence, and metastasis. EMT is the initial step in tumor invasion and metastasis. Angiogenesis provides another possibility for tumor cells to metastasize beyond the classical vascular pathway. Changes in the microenvironment underlie the above 2 tumor biological processes, which are closely related to each other. Deep exploration of their relationship in order to elucidate the role of EMT in angiogenesis and provide a solid theoretical and practical basis for targeted EMT inhibition of angiogenesis in oncology will better serve clinical oncology and benefit the majority of patients. Therefore, blocking angiogenesis inhibits tumor growth and provides survival benefits for cancer patients. 33 The present study found that the expression levels of MMP2, MMP9, E-Ca, N-Ca, and VEGF were significantly decreased after the α-solanine treatment, while their antagonists TIMP1 and E-Ca were significantly up-regulated. These results also revealed that α-solanine has a favorable antiangiogenic effect.
The fundamental feature of the vast majority of tumors is the rapid proliferation of tumor cells. Enlarged solid tumors deplete the body of nutrients and create compaction in their vicinity. Therefore, the control of cell proliferation is currently a hot topic in cancer treatment. 34 Studies have shown that blocking G2/M phase cell cycle progression by increasing p21/cip1 protein levels and down-regulating Cdc2, cyclin B1, and Cdc25C protein levels can inhibit tumor proliferation. 35 Some researchers have found that by reducing the expression of IGF-1 (insulin-like growth factor-dependent protein kinase) in breast cancer cells, antitumor proliferation can arrest the cell cycle and induce apoptosis. 36 Apoptosis is a type of programmed cell death controlled by genetic regulation. Vukelic et al 37 found that apoptosis can be induced by activating the ERK signaling pathway. Therefore, inhibiting cell proliferation and inducing apoptosis is an effective strategy for treating tumors. 38 The present study found that M2-like TAM with α-solanine could inhibit Bcl-2 expression and up-regulate Bax and p53 expressions, inhibiting tumor cell proliferation and promoting tumor cell apoptosis.
Recent studies have found that mTOR-related pathways are complex and extensive, and the abnormal regulation of multiple elements is closely associated with the occurrence and development of tumors. 39 As a hub for aggregating multiple signaling pathways, mTOR has 2 pathways that integrate upstream signal transmission: PI3K/AKT dependent and independent pathways. 40 Jung et al 41 showed in a liver cancer study that VEGF up-regulated the PI3K/Akt/mTOR signaling pathway by binding to its corresponding receptors, causing intracellular enzyme reaction and increasing vascularization. It has also been proven that RET gene overexpression can activate the PI3K/Akt/mTOR signaling pathway, which further induces HSF1 expression and angiogenesis. 42 The present study found that α-solanine inhibited HIF-1ɑ, p-PI3K, p-AKT, mTOR, and p-mTOR expression. In addition, the antiangiogenesis, antiproliferation, and pro-apoptosis effects of α-solanine might be achieved by regulating the PI3K/Akt/mTOR pathway.
Chinese herbs play an essential role in traditional Chinese medicine. 43 More attention has been paid to developing TCM, exploring its mechanism of action, and providing a scientific basis for its use. α-Solanine is a steroidal glycoside alkaloid present in most Chinese herbal medicines. Several studies have demonstrated that it has a strong antitumor effect. 44 This study selected its antiangiogenesis, proliferation inhibition, and apoptosis promotion directions for initial exploration, confirming its strong antitumor effect and providing a basis for developing subsequent derivatives.
Conclusions
α-Solanine is highly resistant to angiogenesis and promotes apoptosis of M2 TAMs. These effects may depend on the PI3K/Akt/mTOR signaling pathway. The present study demonstrated that α-solanine has a strong anticancer effect under hypoxic conditions.
Footnotes
Acknowledgements
The authors thank the Department of Medicine, Taizhou University for providing facilities and technical support.
Author Contributions
JH and BF handled the conceptualization of the study. JH, LP, XW, MJ, SZ, HZ, and BF contributed to the methodology of the study. JH, SZ, HZ, and HF contributed to the investigation of the study. LP, MJ, HZ, HF, and BF contributed to the visualization of the study. Funding acquisition was done by BF. Project administration was handled by FB. Supervision was done by JH, HF, and BF. Writing—original draft was done by JH, XW, and BF. Writing—review & editing was done by JH, XW, and BF. All listed authors have made a significant scientific contribution to the research in the manuscript, approved its claims, and agreed to be authors.
Data Availability Statement
We have included a data availability statement in our manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wenling Social Development Science and Technology Project, The Taizhou Municipal Science and Technology Bureau, (grant number 2021S00003, 21ywb112, 22ywb86).
