Abstract
Background
Lung cancer is the most common cause of cancer mortality around the world, and non-small cell lung cancer (NSCLC) represents 80% of the lung cancer subtypes. Various factors, including gene alterations and changes in an individual's lifestyle, are potential causes of the disease. Many FDA-approved drugs have been used to treat the disease, but their success rate is still not halfway to the mark. Nano-based delivery provides better biocompatibility and targeted delivery without compromising the effectiveness of the drugs.
Objectives
The objectives of this systematic literature review are to find the current epidemiology and status of lung cancer TNM classification. The molecular targets of NSCLC and how dual drug nanoformulations can manage NSCLC.
Methods
We shortlisted articles from the Google Scholar literature database based on relevant filters and inclusion and exclusion criteria. Further screening was performed, which resulted in papers being excluded because they poorly aligned with NSCLC or dual drug nanoformulation. Articles addressing epidemiology, TNM classification, molecular targets, and dual drug nanoformulations were included.
Results
We have summarized the studies done on dual drug nanoformulation developments. We primarily discussed epidemiology and the alterations that cause the onset of the disease. Next, we discussed the types of nanoparticles used in targeted delivery and dual drug nanoformulations.
Conclusion
Dual drug nanoformulations would give a synergistic anti-cancerous effect, which tends to give a better result on the disease and reduce individual drug-related toxicity, suppressing multi-drug resistance through different mechanisms of action and reducing the dose of each agent. Preclinical trials, including in silico, in vitro, and in vivo studies on mouse models, have shown them to be a better alternative to conventional methods. Given their accessibility and therapeutic benefits, these nanoformulations offer a promising approach to managing NSCLC.
Introduction
Non-small cell lung cancer (NSCLC) is the most prevalent type of lung cancer. Lung cancer is the third most common type of cancer, being a leading cause of 2 million deaths worldwide 1 of which NSCLC accounts for 80–85% (small cell lung cancer accounts for 15-20%) and has a survival rate of about 18.6% in 5 years survival. 2 NSCLC is further divided into subtypes: adenocarcinoma, squamous, and large cell carcinoma. It is found in smokers’ patients whether adenocarcinoma arises due to exposure to carcinogenic (asbestos, radon, etc) substances. 3 Changes in daily lifestyles, smoking, epigenetics (genetic mutations, downstream pathway dysregulation, etc), histological modifications, etc led to a rise in the number of cancer patients; the survival rate is low in advanced stages of the disease despite having innovative strategies because of drug resistance adaptions in patient. 2
NSCLC tumors arise from different multilayered complex mechanisms due to their various impaired signaling pathways and proliferations, such as epidermal growth factor receptor (EGFR)-mediated pathways and their mutations, such as T790M, V-raf murine sarcoma viral oncogene homolog B1 (BRAF), V-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog (KRAS), etc and many have been studied; some are yet to be discovered. 4 Carcinogenesis arises due to the somatic mutations in oncogenic driver genes, which include the Ras/mitogen-activated protein kinase (MAPK) pathway, Anaplastic Lymphoma Kinase (ALK)/Rearranged During Transfection (RET)/ROS Proto-Oncogene 1 (ROS1)/Neurotrophic Tyrosine Receptor Kinase (NTRKs) fusions, and the KRAS G12C mutation hotspot. All of these can be cured using the target therapies. Other molecular drivers include KRAS/BRAF, Mesenchymal-Epithelial Transition factor (MET), Tumor Suppressor Genes (TP53/STK11/KEAP1), Tumor mutation burden (TMB), Circulating Tumor DNA (ctDNA). 5
Available Treatment Options and Drawbacks
Ongoing conventional therapeutics being used are combinations of chemotherapy (platinum-based), radiotherapy, adjuvant chemotherapy, angiogenesis suppression, plant-based alkaloids, Abraxane used in combination with Atezolizumab, 6 etc They all may have some side effects, including organ damage, neurotoxicity, drug resistance, heart disease, lung inflammation, pneumonitis, 7 Peripheral neuropathy, intestinal damage, nausea, hair loss, bone marrow issues, etc 3 All these side effects and drug effectiveness depend on the Prognostic Factors of a Patient: Age, gender, performance status, smoking/alcohol coverage, nutritive and morphometric parameters, etc5,8 Also, their tumor target inability and drug resistance mechanisms have significant limitations, which spurred the interest of researchers to evolve innovative treatment strategies. One such promising approach that is now emerging is dual drug nano-formulation using nanotechnology-based systems, which enhance water solubility and cellular absorption of drugs to reach target sites and minimize their effect on other cells. It also offers the advantage of dose reduction. They can potentially improve treatment outcomes, reduce drug resistance, and minimize the side effects associated with chemotherapy. 2 However, it is not easy to utilize dual-drug nanoparticle formation as drugs have diverse physicochemical and kinetic properties. 9 Dual drug nano-formation requires three remarkable basic strategies to overcome drug resistance, to identify the down-regulating activity of drug-using inhibitors, to develop new techniques that can resist against resistance, and to combine two or multiple drugs using nanocarriers in which one should improve the efficacy of the second drug. 10
Nanoparticles (NPs), such as polymeric nanoparticles, are often used, as they can control and sustain the drug release at the target site. 1 Niosomes are also used as nanocarriers (spherical shaped); they are like liposomes comprised of hydrophilic heads and lipophilic tails. They can carry both hydrophobic and hydrophilic therapeutic agents at the same time. They are cost-effective but have a limitation of drug leakage, which could be overcome by surface functionalization with the help of polymeric materials such as chitosan, Polyethylene glycol (PEG), etc 2
Nanoparticles
Target delivery is often facilitated by the conjugation of anticancer drugs with targeting moieties that bind cancer cells. Usually they selectively target EGFR, FA receptors, transferrin receptors, and other receptors present in lung cancer cells. Nanocarriers, such as liposomes, polymeric nanoparticles (such as amphiphilic copolymers), micelles, solid lipid nanoparticles (SLNs), nanostructured lipid carriers (NLCs), cyclodextrins, dendrimers, chitosan-based NPs, exosomes, cell membrane-camouflaged NPs, nonviral NP, 6 hybrid lipid or lipid-based nanoparticles, are used; their composition can be modified and improvised to boost their efficacy 3 and make them personalized target therapies against specific heterogeneous cancer sites over an extended period. 6
Dual-Drug
Multiple drugs delivered by nano-carriers must be in appropriate ratios and doses; only then can they be effectively used to enrich the effect of drugs and minimize their toxicity in patients. 11 The cisplatin drug has been used as first-line therapy but has resistance limitations; to overcome the situation, epigallocatechin gallate (EGCG), a type of polyphenol extracted from tea is now combinatory used with cisplatin encapsulated under Self-assembling gelatin/EGCG nanoparticles (GE); they kill cancer cells by enhancing toxicity in the tumor cell environment. 12 EGFR inhibitor (Icotinib) and doxorubicin (DOX), erlotinib and cisplatin efficiently induce apoptosis in erlotinib-resistant cells, 11 Gemcitabine (GEM) and paclitaxel (PTX), 13 Paclitaxel (PTX) and triptolide (TL) reduces drug resistance and have high therapeutic effect with the least toxicity side effects, 14 Doxorubicin (DOX) and cis-platinum (CDDP) loaded in Methoxy poly(ethylene glycol)-poly(ethylenimine)-poly(l-glutamate) (mPEG-OEI-PLG) copolymers targets EGFR shows enhanced anti-tumor efficacy with no side effects, 15 Cisplatin and demethoxycurcumin loaded in CHC nanoparticles undergo biofunctionalization with CD133 showed boosted and effective results in uptake of drug by lung cancer cell for the therapeutics, 16 Cisplatin and etoposide also increases efficacy of lung cancer treatment and does not cause any other toxicity, 17 Erlotinib (ELTN) and fedratinib loaded into poly(ethylene glycol) (PEG)-poly(lactic acid) (PLA) found to be stable and efficient in acidic environment of tumor cell, also it down-regulates the protein expression of JAK2/STAT3 signaling pathway, which facilitates the reversal of erlotinib resistance of the drug, 18 etoposide (ETP) combined with berberine (BER) showed high cytotoxicity against A549 lung cancer cells when used as dual drug in hydrophilic albumin nanoparticles (HSA NPs), 19 Metformin and artemisinin (ART) into PEGylated niosomal nanoparticles (Nio-NPs) showed reduced survival of lung cancer cells by arresting their cell cycle, 20 Gemcitabine (GEM) and 5-fluorouracil (5FU) loaded PEG-PLGA-based nanoformulations showed drug stability and enhanced performance, 21 Methotrexate (MTX) and pemetrexed (PMX) co-loaded chitosan nanoparticles (CNPs) facilitates blood circulation and showed positive anti-tumor response, 18 Gemcitabine and platinum, cisplatin along with gemcitabine and bevacizumab are currently under the clinical trials for advanced NSCLC treatments, 22 Bevacizumab-coated gefitinib-loaded nanoparticles (BCGN) used for tumor angiogenesis inhibition and EGFR phosphorylation it was found that they significantly enhance efficacy of drug by tumor growth inhibition, 23 Nivolumab and pembrolizumab, Atezolizumab and durvalumab used against Tumor PD-L1 expression in lung cancer tumor cells, Pembrolizumab, carboplatin and pemetrexed/pembrolizumab, Carboplatin and paclitaxel/ nab-paclitaxel depending on the histology is used against cancer target sites. 24
Rationale
Historically, oral drug delivery methods have not been feasible for the treatment of lung cancer due to the inability of the therapeutic to penetrate lung tumor sites and achieve efficient therapeutic concentration even at high administered doses. Therefore, the size, shape, and surface charge of nanoparticles provides an avenue for the development of novel routes of administration for anticancer agents. Combination therapy is effective due to its synergistic action, reduced toxicity, and negligible drug resistance. The potential of enhanced therapeutic effectiveness, responsiveness to the stimuli, and scale-up. Furthermore, because of in-vivo toxicity study and use of biomarkers, the overall effectiveness of introducing innovative combinations of nano-formulations into clinical settings is likely to increase. Understanding the molecular mechanism of non-small lung cell cancer and the changes that have been brought about due to the targeted drug delivery will also be assessed in this review.
Review Questions
This review was intended to provide comprehensive insight for future researchers by answering the multiple sub questions:
What is the epidemiology and current state of the TNM classification for lung cancer? What are the molecular targets for NSCLC? What are the different types of nanoparticles used in the targeted delivery? How dual drug nanoformulations can be used for the management of NSCLC?
Epidemiology
According to Global Cancer Observatory Statistics 2022, lung cancer remained at the top of the list for the highest number of cases among both sexes, having a frequency of 12.4% (total cancer cases), followed by breast, colorectal, and other types of cancer (Figure 1). Among men, it is the leading cause of cancer-related incidence. In women, it is the second major cause of cancer incidence, preceding breast cancer. The incidence as well as mortality is highest for lung cancer among other cancer types (Table 1). The mortality rate of 18.7% is double the second most common cancer-related death (colorectum cancer 9.3%).

Distribution of cases for the 5 most common cancers in 2022 for (Top left) MALES, (top right) females, and (Below) Both sexes. for each sex, the area of the pie chart reflects the proportion of the total number of cases. (GLOBOCAN 2022. Global Cancer Observatory (http://gco.iarc.fr/) International agency for research on cancer 2022.).
Number of Mortalities in Top 5 Cancers (http://gco.iarc.fr/).

TNM Classification
The term TNM staging was hypothesized by Pierre Denoix (1943-1952). It was accepted by the Union for International Cancer Control (UICC) as a Tumor Nomenclature and Statistics committee in 1953. In 1996, the International Association for the Study of Lung Cancer (IASCL) was developed due to the need for an international database for the external validity of future TNM editions. IASCL along with Cancer Research and Biostatistics (CRAB), worked on the collection and analysis of lung cancer and its TNM staging and updates, which collected data on lung cancer (∼200 000) for TNM stage classification. They published many editions; after undergoing vital revisions for their classification, they launched the seventh edition, published in 2009, based on 81 496 patients’ data. Due to some limitations, IASLC launched a new edition, ie, the eighth, in 2017, edition data collection (77 156) for this edition was performed between 1999 and 2010 from 16 different countries which additionally incorporated mesothelioma, thymic malignancy and esophageal cancer which was found to be internationally effective in 2018.
In the eighth edition, they reclassify the different stages of NSCLC (lung cancer) depending on the prognostic data; changes were found in tumors based on their size and other non-quantitative descriptors and metastases based on their intra-thoracic and extra-thoracic subclassification, whereas no changes in nodes were found (Table 2). The rate of survival of the patient depends on the size of the tumor; 7 cm is considered under the T4 stage, where diaphragm tumor invasion is found.
Differentiation Between Seventh and Eighth Editions of TNM Classifications. 25
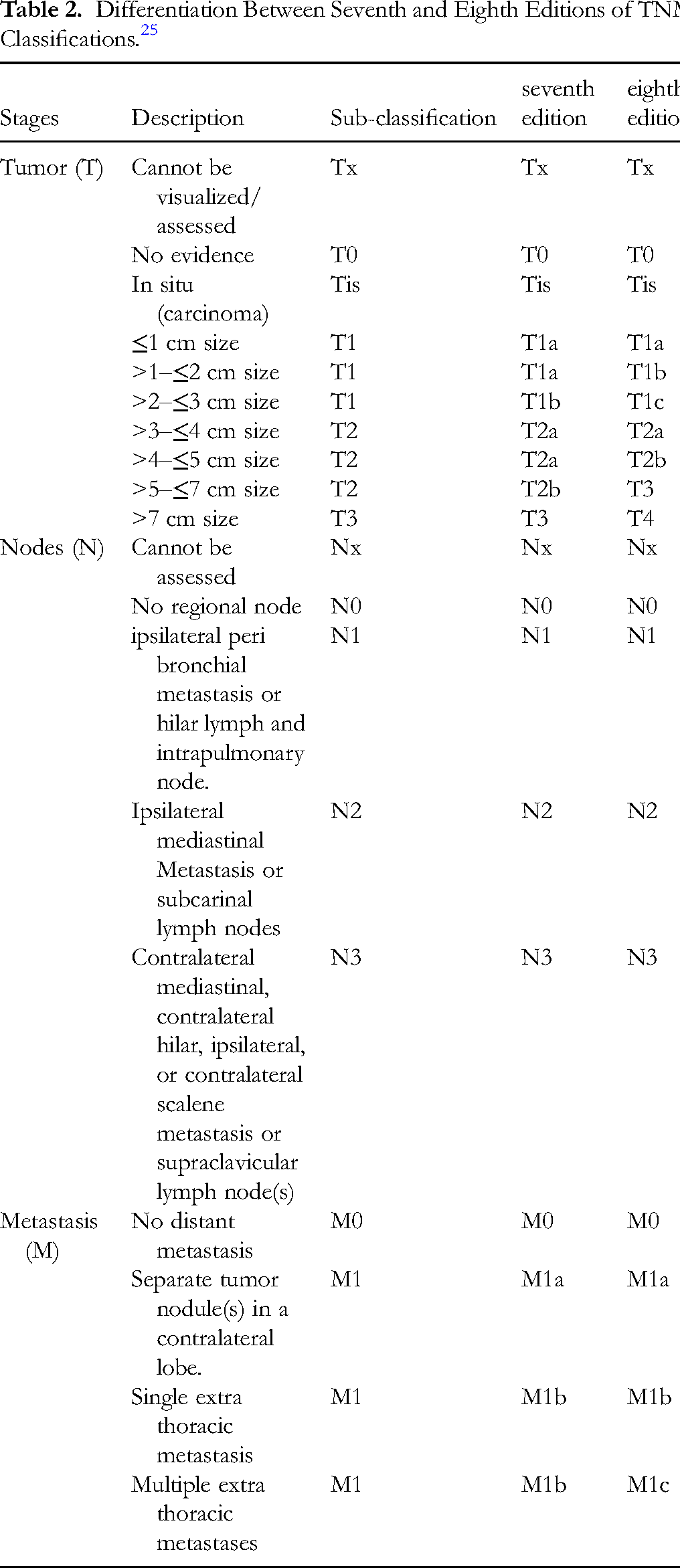
This latest (eighth) edition provides better predictions for patients’ survival during the treatment with rich data availability (Table 3), which helps in aiding better therapy/novel trials, and it also highlights the specific roles of clinical, radiologic, and pathological staging along with the importance of multidisciplinary meetings in patients staging in lung cancer management.
Given Below is the Table for Stage-Based Editions (Seventh and Eighth) of TNM Classification.

Limitations of the Eighth Edition
Data design— Enough data is not available to design TNM classification.
Geographical bias— Data considered does not represent all regions equally. Regions like Japan are overexpressed, and others are under expressed (North America, Africa, India, and China)
Bias- Data collection is biased, as surgical patients’ data was more than the radiotherapy and chemotherapy data of the patients, ie, non-surgical data (∼4.7%).
Variability in measurement- tumor size measurement is not stressed much, ie, inter-, and intra-observer variability was found exclusively in the case of sub-solid nodules or smaller lesions. 25
Methodology
Search Strategies
The Google Scholar literature database was used to explore and extract the relevant articles and information regarding this domain. We have used various keywords like “Dual Drug for the Management of NSCLC,” “Dual Drug Nanoformulations in NSCLC,” “Dual Drug-Loaded Nanoparticles in NSCLC,” “Dual drug nanoformulations for the management of NSCLC”. PRISMA guidelines 26 were carefully followed while the literature search was being conducted.
Inclusion Criteria
Studies were selected based on their focus on dual drug nanoformulations which have been used for preclinical studies. This included original research or comprehensive reviews that explored molecular targets for NSCLC, different nanoparticles used for targeted delivery and the dual drug nanoformulations. Articles addressing Epidemiology, TNM classification, Molecular targets and dual drug nanoformulations were included. Studies which were specifically targeting dual drug nanoformulations for the management of NSCLC were also prioritized.
Exclusion Criteria
Exclusion criteria filtered out studies that did not primarily address the dual drug nanoformulations and were not peer-reviewed, such as conference abstracts, opinion pieces or editorials, and those unrelated to the NSCLC. After the execution of the search query, the titles and abstracts of the retrieved articles were independently evaluated by authors to identify possibly pertinent studies. We omitted publications that were not directly pertinent to the review's subject, lacked significant data or analysis, or were not accessible in full text. Subsequent to the preliminary screening, we meticulously evaluated the remaining publications for methodological rigour and their contributions to the field by comprehensively reviewing the full texts.
For study selection, first evaluation of titles and abstracts was conducted according to each eligibility requirement, followed by a review of the full-text article if a conclusive determination could not be reached. First two authors conducted the study review and selection, and in case of any discrepancies, it was resolved by the third author.
Data Extraction
Initially, we shortlisted 400 articles based on relevant filters, and inclusion and exclusion criteria. Further screening was performed, which resulted in 200 papers being excluded based on not optimally aligning with NSCLC or dual drug nanoformulation. After reading the abstract of these 200 papers, at the end 64 papers were selected, and others were excluded because they did not have optimal information. Full-text understanding of these papers was performed for this review article as shown in Figure 2. The selected ones were carefully examined by reading the full publications, respectively, and information was efficiently utilized in this review paper.
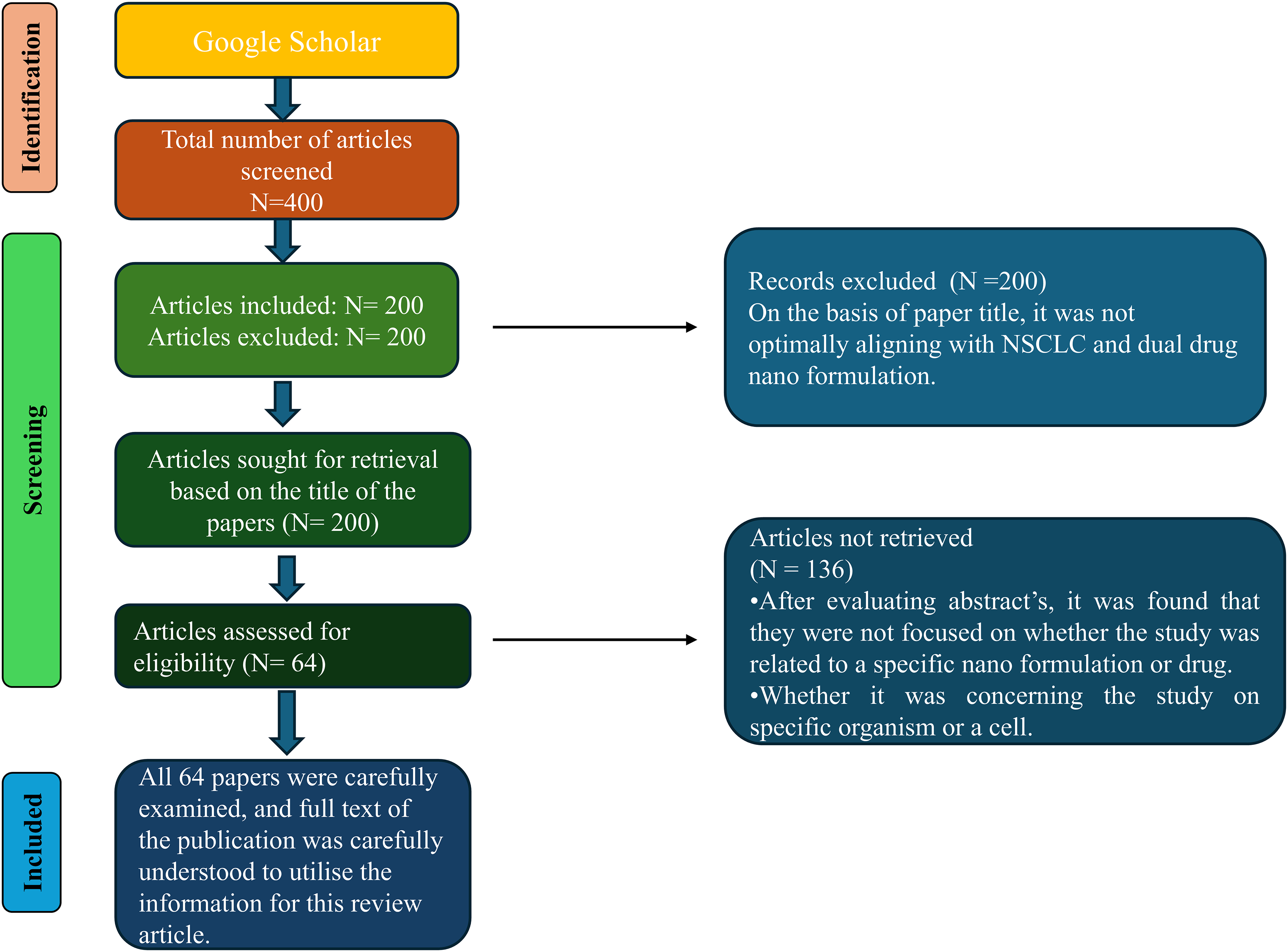
Illustrates the selection method and the criteria used for screening the articles.
Risk of Bias
This systematic literature review reveals methodological variances in the in vitro studies, including the use of diverse nanoparticles, assays, and cell lines to assess the anticancer efficacy of dual drug nanoformulations. The in vivo research referenced in this review did not provide information on sample size calculation or randomisation procedures. Additionally, two independent evaluators performed the study review and selection. The reviewers’ decisions were analysed, and any inconsistencies were resolved by a third reviewer.
Factors Triggering Lung Cancer
Environmental and Lifestyle Factors
Smoking Cigarettes
Enhanced inflammatory responses are found in the patients who are exposed to smoking facilitated by the release of inflammatory mediators and GF (growth factors), ie, TGF-β, EGFR, IL-1, IL-8, and G-CSF (granulocyte colony-stimulating factors). This effect remains conserved for a long time and results in chronic and pathological changes that create a microenvironment for neoplastic alterations in lung cells.
Hazards: Air Pollutants
Air pollutants facilitate carcinogenic mutations and affect the human respiratory system through gases (SO2, NOx, CO), heavy metals, and particulate matter (PM). IARC (the International Agency for Research on Cancer) states that prolonged exposure to air pollution and particulate matter (Group 1 carcinogens) facilitates lung cancer development. 27
Molecular Alteration Drivers
EGFR
EGFR (ERBB1) belongs to the ERBB family of tyrosine kinases, which is comprised of other members like ERBB2 (HER2), ERBB3, and ERBB4. They are usually found in lung cancer patients, 27 with a mutation rate of 13% to 44% among East Asians,5,28 and arise due to mutational activation, amplification, and ligand simulations. Deregulation of the ERBB pathway is promoted by the gene fusion of ERBB3 and NRG1 (ligand), which facilitates NRG1 EGF-like domain expression on the cell surface, resulting in heterodimer formation (ERBB2-ERBB3) mediates cell proliferation and its survival, which activates PI3K-AKT and MAPK pathways (by phosphorylation), further facilitates for cancer development. Whereas in lung adenocarcinoma, CD74-NRG1 fusions are found, which activate ERBB3, leading to cancer progression in non-smokers. 29
EGFR is a receptor tyrosine kinase; it has an extracellular domain where the ligand binds, a membrane transverse domain, and a cytoplasmic domain where tyrosine kinase is present. Ligation binding takes place at the ligand binding domain. There are conformational changes in the EGFR that promote dimerization, which further induces autophosphorylation in tyrosine kinase residue, stimulates the Ras/Raf/MAPK and PI3K/Akt/mTOR downstream signaling pathways, and controls cell survival, their proliferation, and migration.
EGFR mutations include the deletion of exon 19, which enhances prognosis levels due to the conformational shift in EGFR structure resulting in the tightening of the ATP binding cleft, allowing their prolonged ligand activation 30 and facilitating lower disease-free survival (DFS) 5 compared to mutation L858R. This is caused because of the substitution action of leucine-to-arginine residue in exon 21, which stabilizes the activation loop and promotes prolonged receptor activation. 30
For the treatment, ERBB2-ERBB3, PI3K-AKT, and MAPK pathways are being targeted in cancers driven by NRG1 fusions using combinational therapies. 29 EGFR mutations are of substantial risk, as they facilitate distant micro-metastasis formation, which facilitates a higher risk of recurrence after treatment by tyrosine kinase inhibitors (TKIs). 5 This resistance mechanism in EGFR is mediated by a mutation (threonine-to-methionine) on exon 19 at codon 790, which enhances ATP affinity and reduces first-generation EGFR TKI potency that is used during the treatment. 29
Treatments and Effectiveness
First-generation medicines (for example, erlotinib, gefitinib, etc) work by inhibiting the auto-phosphorylation mechanism by blocking the ATP-binding site, which prevents downstream signaling pathway activation, but due to the resistance mechanism, their efficacy is reduced; they also have stomatitis, paronychia, and diarrhea-like side effects after the intake.
Second-generation medicines (for example, afatinib, dacomitinib, etc) are ERBB family inhibitors that bind EGFR, HER2, and HER4 receptors, blocking these receptors irreversibly and preventing downstream signaling activation, but these drugs are less effective compared to first-generation TKIs and have toxicities.
Third-generation medicines (for example, osimertinib) are used, which bind T90M mutations that provide a resistance mechanism against other drugs. These drugs bind cysteine 797 residues of the EGFR ATP-binding domain and irreversibly prevent further downstream signaling, but now new mutations (cysteine-to-serine (C797S), L798I, L718Q, L692V, E709 K, L844V, and G796D) are arising, providing resistance to these drugs. 31
To tackle the scenario, combination therapies (EGFR TKIs with chemotherapy, EGFR TKIs with immunotherapy, and anti-angiogenic therapies) are now being used against multiple pathways promoting resistance mechanisms in patients through secondary site mutations (EGFR-T790M, EGFR-C797S, etc) by altering drug binding, gatekeeper mutations (EGFR-T790M), solvent-front mutations (EGFR-G796S/R), off-target resistance (downstream signaling pathways), and other mutations (EGFR-T854A). However, it is still crucial to develop strategies to overcome these resistant pathways for better treatments and survival.30,32
KRAS
KRAS is an RAS family (rat sarcoma oncogenes) gene that regulates cell signaling pathways, survival, and proliferation. KRAS mutations are responsible for 85% of RAS mutations in humans. There are 35% of KRAS mutations found in NSCLC patients. The most common p. G12C variant (APPROX. 13%) arose due to a single-nucleotide mutation (glycine replaced by cysteine) at codon 12 and is the most recurrent variant in NSCLC, found to be widespread in adenocarcinoma (prevalence of 13%). Other mutations include G12V (21%) and G12D (17%). Smokers are found to have a high incidence of KRAS mutations, whereas EGFR and HER2 mutations are found in non-smokers. Mutations in these various molecular mechanisms and pathogenic pathways drive lung cancer. 27 There is no clear prognosis value found for KRAS mutation in NSCLC, but it may negatively show its worse prognosis impact on stage 1 tumors. 33
Targeted therapies used in KRAS mutations are Adagrasib and Sotorasib, both of which are FDA-approved drugs that work as KRASG12C mutation inhibitors, which have 37.1%–42.9% ORR Sotorasib Irreversibly binds KRAS and traps it, which inhibits it from covalently binding with inactive GDP-bound conformation pocket, hence KRAS remains inactive and that hinders KRAS oncogenic signaling, whereas Adagrasib has a longer half-life than Sotorasib. Several ongoing studies are working on targeting downstream signaling pathways (MEK inhibitors).
Combinational strategies of sotorasib and adagrasib (KRAS inhibitors), along with EGFR inhibitors, SHP's (src homology-2 domain-containing phosphatase-2) inhibitors, 33 and immune checkpoint inhibitors are being tested to assess their effectiveness against KRAS mutations in NSCLC.
Other oncogenic drivers that activate MEK inhibitor resistance must be targeted by combinational therapy using MEK inhibitors, PI3 K or FGFR1 inhibitors, YAP1 inhibitors, and CDK4/6 inhibitors.30,34
BRAF
Somatic BRAF gene mutations occur in almost 3%–8% of lung adenocarcinomas, and they are destructively metastatic; still, their impact on localized NSCLC is not noticeably clear as they are more commonly involved in colon cancers. 5 BRAF-V600E is the most common mutation (approx. 50%) found in BRAF, and other mutations, such as BRAFG469A/V, BRAFD594G, and Non-V600E Mutation (approx. 50%) are also found.
Targeted FDA-approved drugs against BRAF are vemurafenib and dabrafenib, which work on the BRAF-V600E mutation, which is the most common mutation that further activates MEK-ERK downstream signaling. However, because of the resistance mechanism, such as secondary mutations, activation of alternative signaling pathways, etc, inhibitors are not effective for the combinational therapy being used, which includes MEK (Trametinib) and BRAF (Dabrafenib) inhibitors for better outcomes. Another oncogenic driver is BRAF dimerization, which develops an active resistance mechanism against MAK inhibitors. For dimerization, RAF inhibitors are made that can effectively inhibit RAF heterodimer-induced activation of MEK–ERK signaling.30,35
ALK
The ALK (Anaplastic Lymphoma Kinase) gene rearrangement is found in approximately 7% of NSCLC patients, these alterations arise due to the fusion mechanism of ALK kinase domains and their N-terminal partners (echinoderm microtubule-associated protein-like 4 (EML4)), which activate ALK and lead to their overexpression, and can contribute to both EGFR and ALK TKIs resistance. 30 ALK genes and mutations show multivariate results in different analyses. Some paper results show ALK rearrangement is not to be considered as an independent prognostic factor; some states positive ALK patients were found to have the least chance of DFS in stage 3 patients. 5
Targeted FDA-approved drugs being used are crizotinib (a first-generation ALK inhibitor), ceritinib, alectinib, and brigatinib (second-generation inhibitors). Second-generation inhibitors have good potency, CNS penetration, and, most importantly, effective secondary mutation resistance (ALK-G1202R, ALK-G1269A, and αC helix ALK-F1174C). ALK and ROS1 inhibitors are also found but have resistance limitations. For that, lorlatinib has been developed and might be effective against these mutations.30,36
MET
MET alterations in the signaling pathway arise due to the exon 14 skipping mutation; it is found in lung adenocarcinomas in a low percentage (1-2%), and it does not lead to the prognosis in localized NSCLC. 5 Normally, MET (a proto-oncogene) encodes a tyrosine kinase receptor, which regulates cell survival, migration, and invasion when bound with HGF, but mutations, fusions, and amplification deregulate the tyrosine kinase activation and promote ligand-independent downstream signaling. It was found that MET copy numbers are higher in patients having MET amplification. MET amplification occurrence is 1%–5% in untreated NSCLC and 5%–20% in EGFR-mutant tumors. 33
Targeted drug therapies being used are Capmatinib, cabozantinib, and crizotinib. Their effectiveness depends on the level of MET amplification in the patient. Cricotinib shows a 50% response rate in NSCLC patients with MET amplification. 30 Other drugs include Savolitinib, Glumetinib, SAR125844, and Glesatinib their combinations are being investigated to enhance their efficacy and to encounter Second-Site Mutations resistance (MET-D1228N and MET-Y1230C mutations) against MET TKIs in both exon-14 mutations and MET amplifications. Some combinations being used are EGFR-TKI (Osimertinib) with MET inhibitor (Savolitinib) and Gefitinib with Tepotinib.33,37
RET
Rearranged during transfection (RET) normally encodes a tyrosine kinase receptor (transmembrane receptor) that binds neurotrophic factors derived from glial cell lines; rearrangements of these genes produce aberrant receptors that promote oncogenic consequences that further deregulate the downstream pathways, accounting for cell proliferation, migration, and survival.
Drug therapies that could be used are cabozantinib, vandetanib, and lenvatinib, but these drugs work against MEK TKI, which is why they are not that effective (18-37%) against RET rearrangements; specific RET-selective inhibitors are required. For example, the drugs selpercatinib (ORR-64%) and pralsetinib (ORR-61%) minimize off-target effects but have several adverse effects that can be managed (neutropenia, anemia, hypertension, and pneumonia); they have improved efficacy during the treatment. Ongoing trials for next-generation drug development are going on that are focused against resistance mutation mechanisms (a gatekeeper mutation RET-V804L) acquired in the patients; for example, TPX-0046 (a third-generation RET/SRC kinase inhibitor) and BOS172738 (a RET kinase inhibitor) are being explored.33,38
NTRK1, NTRK2, NTRK3
These are Tropomyosin Receptor Kinases (NTK) family genes that responsibly encode for what is required to normally regulate the development and function of the nervous system and mediate their signaling pathways (in cell proliferation, differentiation, and survival), but when genetic fusion takes place in these genes, overexpression is found that also activates fusion proteins; these collectively contribute to tumorigenesis. NTK gene fusions have a 0.1% to 1% frequency of occurrence in NSCLC, while they are found to be mutually exclusive with other oncogenic drivers. These fusions and Solvent-Front Mutations (NTRK-G595R) can provide resistance against EGFR TKI.39,40
Targeted drug therapies being used are Larotrectinib (a selective pan-TRK inhibitor, ORR-79%), entrectinib (a multi-kinase inhibitor against TRK, ROS1, and ALK), and Taletrectinib (for ROS1/NTRK inhibitors). In contrast, repotrectinib and selirectinib are next-generation inhibitors to counter resistance against first-generation inhibitors. 33
HER2 Mutations
A Human epidermal growth factor receptor2 (HER2) is a tyrosine kinase receptor it is a part of the EGFR family also known as ERBB2, this normally regulates cell proliferation and its survival by forming heterodimers along with other family members but because of some genetic amplifications and dysregulation receptor overexpression arises that activates the kinase domain mutations ie, kinase-activating exon-20 insertions, these mutations arises in 1–3% of lung adenocarcinomas. 33 They may contribute to the EGFR TKI resistance mechanism. 41
Target drug therapies being used are Trastuzumab (HER2-targeted monoclonal antibody), and Afatinib (HER2 TKI), whereas to tackle resistance (glycine at position 770 mutation and HER2-C805S) against these inhibitors, we use trastuzumab with ADCs (T-DM1), novel TKIs (poziotinib, pyrotinib, and mobocertinib), and Chemotherapy (OSS-50.9%) as a combinational therapy. 33 Many ongoing trials are working on combining strategies using targeted therapies and ICIs (immune checkpoint inhibitors) to enhance the efficacy and fight resistance mechanisms.30,42
ROS1
ROS proto-oncogene 1 is a receptor tyrosine kinase that shares structural homology with ALK. ROS rearrangement is found in ∼1–2% of NSCLC patients, and it involves fusions of the kinase domain of ROS1 with various fusion partners (CD74) to perform ROS1 kinase activity. 43 Because there is structural homology, the resistance mechanism of one mutation driver affects the treatment of another mutation driver by causing secondary mutations (ROS1-G2032R) and influencing downstream signaling. The drugs approved for ALK are also being used for ROS1 for example Crizotinib (RR-72% and PFS-19.2M).44,45
SRC Activation
SRC (proto-oncogene) is a tyrosine-protein kinase that regulates cell survival, differentiation, and functioning downstream of RTKs, but this SRC activation exhibits EGFR TKI and ALK TKI (in vitro) resistance in NSCLC patients. Its inhibitor (dasatinib) is used along with EGFR TKI (erlotinib) and ALK TKIs and shows good efficacy in the treatment outcomes (Table 4).
Molecular Alteration Drivers Responsible for Causing Lung Cancer, with Their Alterations and Therapies.

Signaling Pathways
MAPK Pathway
MAPK pathway reactivation plays a vital role in the EGFR resistant mechanism by arising BRAF-G469A and BRAF-V600E mutations along with NF1 gene loss and mutations in NRAS/KRAS and increasing their copy number is responsible for 1% of EGFR TKI's patients tackling the situation. MEK inhibitors with EGFR TKIs are being suggested. Whereas in ALK and ROS1 rearrangement, this pathway inhibits the resistance mechanism by ERK phosphatase DUSP6 down-regulation, and the KRAS amplification mechanism inhibitors against both are required to overcome the resistance. 44
PI3K–AKT Pathway
In the PI3K–AKT–mTOR pathway, the resistance mechanism is influenced by mutations in PIK3CA (present in ∼4% of patients) and PTEN loss (a negative regulator of PI3K signaling) in EGFR and HER2. Inhibitors against PI3K, HER2 TK, and EGFR TK are required as a combinational therapy to increase the treatment efficacy. Overexpression of mTOR also affects the efficacy of EGFR TKI against which rapamycin is used, while PI3K–AKT pathways are not involved in inducing resistance mechanisms in ALK- and ROS1-rearrangements.
JAK-STAT Pathway
The JAK-STAT pathway is also involved in EGFR TKI resistance mechanisms through downstream activation of NF-κB, enhanced by autocrine IL-6 signaling. So, inhibitors against the JAK or STAT3 (INCB39110) pathway, along with EGFR TK (osimertinib), are combinatory and used for better treatment outcomes. while JAK-STAT pathways are not involved in inducing prominent resistance mechanisms in ALK- and ROS1 rearrangements.46,47
Parallel Bypass Signaling Pathways
Different receptor tyrosine kinases can activate parallel bypass signaling pathways, which can disrupt the targeted mutation drivers and make them resistant to TKI therapies. For example, positive MEK amplification patients show resistance against EGFR TKIs by the activation of other pathways (MAPK and PI3K–AKT–mTOR pathways). The same goes with AXL RTK and HER2/HER3 activation; they also utilize the same mechanism against EGFR TKIs. EGFR shows a bypass mechanism by activating ALK- and ROS1-rearrangement. Up-regulation of FGFR1/FGF2, EPHA1, IGF1R, and KIT pathways also makes a patient resistant to various TKI therapies.
Survival and Anti-Apoptotic Pathways
BIM protein induces apoptosis in response to the EGFR TKIs intake; deletion of BIM through polymorphism makes the person resistant to these drugs. To overcome this, vorinostat-like drugs are advised, which can enhance the expression of BIM. NF-κB is a transcription factor that normally mediates cell proliferation, apoptosis, and inflammation, but in cancer patients, it exhibits a resistance mechanism against EGFR TKIs by activating the JAK–STAT3 pathway through IL-6 overexpression, and YAP1 (Hippo pathway effector) also activates a resistance mechanism against EGFR TKI and BRAF in NSCLC treatments. An inhibitor against YAP1, verteporfin, can improve the treatment of EGFR, BRAF, and MEK mutations. The loss of p16, a CDK inhibitor, also results in providing a resistance mechanism against EGFR TKIs; the addition of its inhibitor can enhance the treatment efficacy in NSCLC patients.
Histological Transformation
Patients with assimilated resistance mechanisms against TKIs undergo histological transformation, which converts NSCLC into small cell lung cancer, with loss of RB protein as a result of this transformation and loss of EGFR expression in individual patients, and further treatment could be given using BCL-XL inhibitors, which shows comparatively improved responses. In patients with ALK rearrangement, sarcomatoid carcinoma transformation is observed. Epithelial-to-mesenchymal transition is also found in patients who exhibit acquired resistance to EGFR TKIs, which is promoted by a cytokine TGF-β (transforming growth factor-β), ZEB1, and HDAC-mediated suppression.
Epigenetics
HDAC (histone deacetylase) activity promotes epigenetic changes that facilitate drug tolerance against EGFR TKIs and make them resistant to those drugs. Inhibitors against HDAC (panobinostat) are required along with EGFR TKIs to enhance their therapeutic efficacy.
Tumor Microenvironment (TME)
The interaction of tumor cells with the TME stromal components plays a significant role in inducing resistance responses against TKIs. 48 Increased permeation of TAMs (tumor-associated macrophages) in TME results in poor PFS and OSS in patients. 49 Its derived factors activate multiple signaling pathways (MAPK, PI3K, YAP, NF-κB, WNT, and RAS) to help in promoting resistance mechanisms. Up-regulation of PDL1 makes TME less beneficial to T cell-mediated responses, which contribute only towards drug resistance. Even inhibitors do not work efficiently because of the low immunity (low level of CD8+ T cell infiltration) in NSCLC patients. Ongoing trials are working in combination on oncoprotein inhibitors and immunomodulatory agents for better outcomes in NSCLC treatments. 30
TMB
Tumor mutation burden is usually found in NSCLC patients as a biomarker for ICI responses. Immune-oncology agents show improved outcomes in ORR and OSS improvement compared to chemotherapy. An elevated level of TMB exhibits resistance to various therapies, whereas it could benefit from using adjuvant chemotherapy. High TMB may also facilitate worse DFS and OSS values, which makes it a potential biomarker for ongoing treatment trials with adjuvant ICI. 50
Circulating Tumor DNA (ctDNA)
ctDNA detection after the post-surgery is an efficient prognosis factor for assuming chances of reoccurrence of the disease. ctDNA is a promising marker for minimal residual disease (MRD) detection; however, it remains challenging because of its low sensitivity and low abundance in a patient's blood sample. Positive post-surgical results for ctDNA detection correlate with severe RFS, whereas negative results show low reoccurrence risks. These results guide the better use of ACT (adjuvant chemotherapy).51,52
Signatures
To describe molecular groups and tumor subtypes, transcriptomic and multi-omic signatures are being developed to stratify relapse risks. However, implementing these clinical practices is challenging due to excessive costs, extended time requirements, fluctuations in tumor heterogeneity, and overall complexity. Several trials are being evaluated against specific gene signatures to improve their clinical practice and benefits.5,51
Epigenetic Markers
Epigenetic changes like DNA methylation and microRNA expression are effective prognosis biomarkers for better insight into tumor behavior, their risk of recurrence, DFS, and OSS in the patient. 5 Epigenetics refers to heritable genetic changes, these changes are the epigenetic modification of the gene expression. These changes are mediated by DNA methylation (gene silencing and TSG inactivation), histone modifications, and non-coding RNAs (miRNAs and lncRNAs). These methods are being used as a diagnostic and prognostic tool. Histone modification includes acetylation, methylation, and phosphorylation along with ubiquitylation and sumoylation, which silence gene expression and promote transcription.
Epigenetic modifications play a crucial role in cancer development by regulating genetic expression and silencing tumor suppressor genes (TSGs), which trigger cancer hallmarks such as proliferation, invasion, metastasis, apoptosis, and regulation of signaling pathways like the ERK family, NF-kB signaling pathway, and Hedgehog signaling pathway, thereby facilitating cancer growth and survival. Other epigenetic modifiers also include ncRNAs, microRNAs, and long ncRNAs. These epigenetic markers allow early screening, monitoring, and designing therapies by identifying their potential cancer biomarkers. HDAC (Histone deacetylases) have emerged as a favorable therapeutic target, and their overexpression is frequently found in NSCLC patients, which results in TSG silencing and abnormal transcription due to mutations found in histone acetyltransferase (HAT) and contributes to carcinogenesis. HDAC inhibitors are used to reverse abnormal epigenetic modifications, restore normal gene expression, reactivate silenced TSGs, and inhibit cancer cell growth. 27
Dual Drug Nanoformulations
As discussed before, drug resistance (intrinsic/acquired) is a major challenge against drug effectiveness due to repeated drug exposure while chemotherapy is performed, which leads to relapse, systemic toxicity, and therapeutic failure in 90% of the patients. To overcome the situation, drug combinations are used that serve better efficacy to target specific sites/pathways that have better performance compared to stand-alone drugs with minimized side effects/toxicity. Major challenges faced during dual drug selections include considerations of drug kinetics, cellular distributions, and their cellular uptake. 49
In the past few years, dual drug nanoparticle formulation has been used in extensive research; suitable formulations depend on the route of administration of the nano-drug, its size, and the physical and chemical properties of the drug 1 to be encapsulated inside the nanoparticles, which enable the proper technique to be chosen for drug nano-formation, which helps in determining/modifying the structure, size, texture (surface), and morphology, which also affects drug packaging, drug efficacy for entrapment, and their release kinetics. Also, other factors that allow for obtaining the selection criteria for NP preparations are polymer selection, drug selection, solvent nature analysis, toxicity detection, purity, stability, and scalability. 53 Thus, the pharmacokinetic, pharmacodynamic, and bioavailability profiles of medicinal drugs encapsulated in nanoparticles are significantly improved by prolonging their half-life and circulation time in the bloodstream. Nanoparticles provide the drug sufficient duration to maintain proximity to the tumour microenvironment, facilitating effective penetration and accumulation within tumour tissues, and enabling the release of the therapeutic payload at the tumour site. The physicochemical features of nanoparticles, including surface charge, are crucial for the application of any engineered nanoparticles in drug delivery systems. In the course of development, attaining a negative zeta potential is crucial, as it mitigates the danger of systemic toxicity and the aggregation of nanoparticles while simultaneously enhancing their internalization within cancer cells. 54
To achieve the synergistic activity, dual-drug nano packaging is required for their better therapeutic effects. 10 This nanomedicine has gained wide attention in cancer therapeutics due to its improved drug bioavailability, solubility, site specificity, less toxicity, multi-functionality, drug availability, and much more. 22 Moreover, their therapeutic effects remain challenging due to the co-precipitation and crystalline nanorod formation; it is also found that their retention time could be decreased if the external layer is absent, due to the gradual aggregation of the nanoparticles. 55
Design of Nanocarriers for Dual Drug Nanoformulations
Lipid-based nanocarriers—These carriers are made from phospholipids and are classified into 2 main types, ie, liposomes and micelles. They have lipid bilayers and aqueous cores where both hydrophobic and hydrophilic drugs can be encapsulated. They have prolonged ability of blood circulation with reduced reticuloendothelial system (RES) clearance. Higher accumulation in tumors can be achieved by liposomes when they are conjugated with PEG, which enhances their permeability, biocompatibility, and retention. 56
Polymer-based nanocarriers—these carriers are constructed using polymers; they have various subtypes: polymeric micelles, polymersomes, polyplexes, polymer-lipid hybrid systems, and protein-based systems. These polymeric nanoparticles are formed through the self-assembly of multiple polymers; they also co-deliver both hydrophobic and hydrophilic drugs, and their biodegradability, biocompatibility, and ability to conjugate with drugs and release them in tumor environments enhance their drug penetration.56,57
Metal-based nanocarriers- silver and gold are primarily used for the composition of metal-based nanocarriers; these particles have significant biomedical applications in diagnostics for distinguishing normal and healthy cells. Magnetic nanoparticles belong to this category and typically exhibit promising features for cancer therapy. 54
Self-assembling Nanocarriers—Artificial nanoparticles have been employed in drug delivery for numerous years and have fulfilled their intended function. Numerous nano delivery systems are produced annually in laboratories; however, when subjected to more complex in vivo models for preclinical and clinical trials, they frequently face restrictions that result in clinical failure. These constraints pertain to stability in systemic circulation, immunological responses, and challenges related to compatibility and degradation. Consequently, there is a growing need to utilize more stable, biocompatible, and biodegradable materials for drug delivery systems, minimizing immunogenicity and toxicity concerns, to attain an enhanced method for encapsulating therapeutic pharmaceuticals. Consequently, protein-based self-assemblies (SAs) are employed in the construction of nanocarriers. Proteins are non-toxic and readily decompose into their constituent subunits (amino acids), which can then be reutilized by peripheral tissues. 58 These self-assembly nanoparticles are readily manufactured, and specialized design strategies can yield stable platforms. Furthermore, it is important to acknowledge that SAs can prolong a drug's half-life and circulation time in the bloodstream while also effectively utilizing passive targeting. They can also be adjusted for active targeting. SAs frequently establish covalent conjugations, augmenting the hydrophobicity of proteins and transforming SAs into more intricate nanoparticles. Collectively, SAs have been proposed as efficient and appropriate frameworks for nanoparticle design. Moreover, SAs consist of cellular bases, which enhance tolerability relative to chemically synthesised nanoparticles. 54
Other nanocarriers, like dendrimers, have diverse functional characteristics other than drug delivery, ie, they are used as nanodevices. In drug delivery, they entrap drugs through noncovalent interactions or can covalently attach as a nano-construct approach. As they have multiple branches, they offer enough sample space for drug conjugation. Platinum-based drugs are commonly delivered through these nanocarriers, as they have enhanced solubility, drug loading capacity, and reduced toxicity with improved efficacy (Figure 3). Also, mesoporous silica, another NP, is widely utilized in drug delivery, as it has high loading and drug release capacity; it tackles NSCLC cells through the mechanism of endocytosis, as they have multifunctional properties that make them suitable for intracellular labeling and magnetic resource imaging investigations. 54

Nanoparticles for targeted dual drug delivery in NSCLC.
Tumor-targeting agents like folic acid (FA), transferrin, and other monoclonal antibodies, inserted in nanodrugs, can further improve therapeutic efficiency through a co-delivery system. This approach limits the drug loading, as it only delivers a restricted amount of drug, as nanocarriers could allow limited drug loading. 55 To improve the performance of polymeric nano drug release novel nanoparticles are formulated that respond to the combinatory signal such as pH/temperature, pH/redox, pH/magnetic field, temperature/reduction, double pH, pH and diols, temperature/magnetic field, temperature/enzyme, temperature/pH/redox, temperature/pH/magnetic, pH/redox/magnetic, temperature/redox/guest molecules, and temperature/pH/guest molecules have recently been developed they have shown good efficacy when transported to target sites (cellular compartments) for the cancer treatment. 59
Zhao et al, 2023 used gefitinib and bevacizumab for drug loading in dual-responsive nanoparticles (BCGN); it efficiently inhibits EGFR and signaling pathways in NSCLC. In vivo studies indicate it has strong tumor suppression activity, low toxicity, and optimal biodistribution in mouse models. In vivo, the study also showed good antitumor results and the least toxicity, ie, it overall enhances the treatment of NSCLC with high efficiency, accuracy, and safety. 23
Chen et al, 2018 also worked on this approach using ELTN and Fedratinib for drug loading through optimization and encapsulation in PEG-PLA poly (lactic acid) NPs. It efficiently releases the drug in the acidic conditions of tumor cells, and it also down-regulates the p-EGFR, p-JAK2, p-STAT3, and Surviving pathways. In vitro studies demonstrated that the drug is released efficiently and exhibits strong anti-cancer efficacy through cytotoxic mechanisms targeting various signaling pathways and tumor sites. In vivo study proved it shows reduced systemic toxicity, facilitating good anti-tumor efficacy, and it also promotes resistance reversal to enhance the treatment results, which was observed through immune-staining. 18
Du et al, 2022 used cisplatin (CDDP) and etoposide (Etp) for drug delivery in nanostructured lipid carriers (NLCs); ie, EtpP–CDDP NLCs form a size of 176.8 ± 4.9 nm when they were tested on A549/DDP cells of mice. In vitro, they showed drug potential (31.9 ± 3.2 mV), efficacy (57.4%±3.9%), a synergistic effect (CI50 values <1), and good distribution with high tumor activation rates. Also, improved cellular uptake and cytotoxicity were found with this nano dual drug (combinational) therapy. In vivo studies say it exhibits prolonged (up to 48 h.) plasma retention and tumor accumulation, facilitating growth inhibition with no major side effects. 60
Fan et al, 2021 used cisplatin (DDP) and oridonin (ORI) for drug loading layer-by-layer in NPs (D/O-NPs) of size 139.6 ± 4.4 nm with a potential value of (+13.8 ± 1.6 mV). The In vitro study was evaluated on A549/DDP cells (human cell line). In vivo studies were performed on A549/DDP cell xenografts of mice; both studies show significant cell cytotoxicity and synergistic effects with high cellular uptake and tumor growth inhibition with no side effects. 61
Xiao et al, 2022 used DTX and siCCAT2 (si-colon cancer-associated transcript-2) for drug delivery in PLGA copolymers, forming a size of 87.26 nm; it was further modified by the addition of peptide (transferrin) on its surface. This drug conjugate shows adequate target reach and efficient anti-tumor responses. Lu et al, 2018 used paclitaxel (PTX) and triptolide (TL) for drug delivery in LPNs (lipid and polymeric nanoparticles), forming 160 nm size of particles with a zeta potential value −30 mV and an efficiency of 85%. Both the in vitro and in vivo test results approved the synergic effects of this nanodrug conjugate with the fewest side effects. Liang et al, 2017 used GEM (hydrophilic) and PTX (hydrophobic) for drug delivery in targeted nanostructured lipid carriers (NLCs), then receptors that target the ligands were added ie, N-acetyl-
Menon et al, 2017 used poly(N-isopropyl acrylamide)-carboxymethyl chitosan shell and poly(lactic-co-glycolic acid) (PLGA), a multifunctional dual drug-loaded nanoparticle (MDNP), for drug loading that specially targets folate receptors. The drug controls the release of some therapeutic compounds, such as NU7441 (radiosensitizer) and gemcitabine (pH-dependent release). In vitro study with alveolar Type I cells shows better stability, uptake, and cytocompatibility, whereas an in vivo study demonstrates their least toxicity and high efficiency in tumor reduction. 22
Li et al, 2020 used erlotinib, apatinib, and icotinib alternatively with doxorubicin (DOX) for dual drug nanoformulation, where icotinib showed the best results, which were then encapsulated with DOX in NPs, forming a 65.7 ± 6.2 nm size and having good loading and releasing capacity. In vitro study demonstrated its efficient uptake, good cytotoxicity, and anti-migration capabilities, whereas an in vivo study confirmed the hypotoxicity of the drug and its specificity towards tumor cells rather than normal cells. 11 Zhang et al, 2021 used cisplatin prodrug (CPP) and etoposide (ET) for drug loading in nanoparticle in vitro studies to confirm its cytotoxicity, synergistic effects, drug release, effective targeting of the tumor site, enhanced retention, and improved permeability effects, whereas an in vivo study on murine lung cancer models shows its enhanced efficacy in tumor reduction and leaving no extra side effects by side. 17
Abdulkareem et al (2024) used metformin and artemisinin (ART) for drug delivery in niosomal nanoparticles. This drug conjugate limits the lung cell proliferation and has enhanced anti-proliferative properties against A549 cell lineage by altering gene expression of hTERT, Cyclin D1, BAX, BCL-2, Caspase 3, and Caspase 7. 20 Elgohary et al, 2018 used etoposide (ETP) and berberine (BER) for drug delivery in hydrophilic albumin nanoparticles (HSA NPs) with mannose and phenyl-boronic acid modifiers to enhance the drug efficacy, forming a size of 216.2 nm, efficacy of 69.5 and 87.6%, and loading of 10.52 and 14.04% for both drugs. Studies on A549 lung cancer cells demonstrated that the drug has strong cytotoxicity and better cellular uptake. In vivo, studies on mice predict higher levels of caspase-3 and reduced levels of VEGF compared with normal mice. 19
Chen et al, 2020 used cisplatin and EGCG for drug delivery in GE-Pt NP, forming a 75 nm size and zeta potential of +19.83 ± 0.25 mV. An in vitro study on A549 shows its improved cytotoxicity for tumor cells with a good cellular intake through endocytosis, which serves it as a better alternative for cisplatin resistance in the lung cancer patient. 12 Han et al (2018) used CsA and Gef for drug delivery in polyethylene glycol-block-poly (D, L-lactic acid) (PEG-PLA), forming 50 nm-sized (spherical) particles. This dual drug improves the Gef efficacy in Gef-resistant cells. In vitro studies declare the effectiveness of this drug, as it inhibits proliferation and enhances apoptosis in tumor cells after the administration of the drug. In vivo, studies declare that it reduces tumor cell growth with minimum side effects and toxicity by reversing MDR mechanisms (STAT3 activation). 10
Kulkarni et al, 2021 used Erlotinib (Erlo) and Quinacrine (QA) for drug delivery in NPs; for nano-formulation in vitro studies showed their enhanced synergic effects with reduced therapeutic dose when compared with plain drugs.
4
Mogheri et al, 2021 used Metformin and Silibinin (Sil) (natural herbal compounds) for drug delivery in poly(D,
Zhou et al, 2021 used gemcitabine (GEM) and 5-fluorouracil (5FU) for drug delivery in PEG-PLGA nanoparticles, forming GEM-5FU NPs. In vitro, studies reveal their induced apoptosis activity, improved stability, better solubility, and drug cellular uptake. Synergistic cytotoxic effects impact target tumor cells, transforming their TME (tumor microenvironment). In vivo studies are still required for better validation of this drug in terms of its efficacy, biodistribution, least side effects, and pharmacokinetics. Alagheband et al, 2022 used metformin and curcumin (Cur) for drug delivery in PLGA-PEG nanoparticles, forming Met-Cur-PLGA/PEG NPs, which have inhibitory effects against leptin (LEP) (a mitogenic agent (cytokine derived from adipocytes) in lung carcinogenesis). In vitro A549 studies reveal their higher cytotoxic effects and down-regulatory effects of LEP and LEP receptor genes that restrict the tumor growth pathways, whereas in vivo validations are yet to be discovered. 21
Zhou et al, 2020 used gefitinib (GFT) and curcumin (CUR) for drug delivery in liposomes NP, forming a 130 nm size and −22.2 mV zeta potential. In vitro, studies reveal their optimal cellular uptake and higher synergistic cytotoxicity against resistant tumor cells. In vivo, studies reveal their significant tumor reduction rates via improved tumor accumulation and enhanced synergic cytotoxic effects without accumulating other side effects. 64 Wang et al, 2018 used AZD9291 and paclitaxel (PTX) for drug loading in disulfide cross-linking micelles (DCMs) forming small-size NPs; in vivo studies on cancer-bearing mice reveal that they exhibit strong synergic effects against IKB-α/NF-κB/Bcl-2 and EGFR/Akt pathways with no additional side effects, ie, offering better efficacy and less toxicity in a patient being treated with this nano-drug. 65
Ganthala et al, 2022 used Ertb and Quercetin (Quer) for drug delivery in solid lipid nanoparticles (EQNPs), forming 87.3 ± 0.78 nm in size with + 13.4 ± 1.12 mV zeta potential with good drug encapsulation efficacy (77% Ertb and 71.4% Quer). In vitro studies on A549/ER cells reveal their apoptotic and inhibitory effects in prior-exposed ERTB-resistant cells. Whereas, in vivo, studies on the C57BL/6 mouse model show efficient cellular uptake and optimal inhibitory effects on n-EGFR/PI3K/AKT expression and tumor progression with the least side effects 66 (Table 5).
Different Nano-Drug Formulations with Their Composition, Mode of Action, Validation, and Trial Phase.

Discussion
Cancer has remained a major problem globally, affecting individuals irrespective of country. Lung cancer has topped the list for incidence as well as mortality. NSCLC is the top contributor to lung cancer subtypes, having a prevalence of 80–85% among other lung cancers. The key contributors to this disease have been environmental factors such as pollution, smoking cigarettes (one of the leading causes of NSCLC), and molecular alterations in EGFR, BRAF, KRAS, MET, ROS, etc Apart from these, mutations in signaling pathways such as MAPK, PI3K-AKT, and JAK-STAT are crucial in promoting oncogenic drivers that lead to tumor development. Many therapeutic advancements have been brought up over the decades, like nitrogen mustard, an alkylating agent that kills cells by chemically modifying their DNA, and platinum-based chemotherapy. There have been certain milestones in cancer therapeutics, many of which have been approved by the US FDA. The drawbacks of these medications have also been the cause of the advent of new alternatives related to cancer therapeutics. Nano-based approaches have shown a promising candidate for a new alternative. In the twentieth century, Paul Ehrlich, inspired by “Der Freischütz,” coined the term “magic bullet” for the first time and later expanded on the concepts of nanoparticles and drug targeting in medicine. In recent years, drug nanotechnology advancements have effectively overcome the drawbacks of traditional chemotherapy drugs. The nano-based drug delivery provides many advantages over conventional therapeutics. They are being used to deliver drugs that show better efficacy in place of just giving the drugs. The biocompatibility of these nanoformulations has also been well-studied for targeted drug delivery using different nanoparticles. Many researchers have formulated nanoformulations with either single or dual drugs. The single drug nanoformulation is effective in various in vitro and in vivo studies conducted in mouse models. Their efficacy compared to drugs is much better and has been reported by many researchers. On the other hand, dual drug nanoformulation has certain advantages, improving the drugs’ efficacy, providing a synergistic effect, and reducing drug resistance. The data from numerous studies show good anticancer efficacy on different cell lines, reducing the expression of several oncogenes and inhibiting certain oncogenic molecular pathways. These nanoformulations have been synthesized by taking both herbal and synthetic drugs or one from each. The selection of nanoparticle systems is the key to formulating nanoformulation. The optimal size, distribution, and characteristics of the particles are essential for developing a successful nanoformulation. The mode of delivery can be via inhalation, intravenous, intramural, transfection, microinjection, electroporation, etc The use of single-drug nanoformulation over dual-drug has been carried out mostly by researchers all over the globe. The paradigm shift towards dual drug nanoformulation requires more study and awareness in the scientific community. With the integration of personalized medicine and immunotherapy, the future of NSCLC treatment could see marked improvements in patient outcomes. The question remains whether these nanoformulations can be drug alternatives. The most reliable evidence would be conducting human trials, which need rigorous preclinical trials to support the data. The safety and tolerance of these approaches need to be further investigated. A suitable FDA-approved nanoformulation could become an ideal candidate for the management of NSCLC and lead to a new pathway to cancer therapeutics.
Conclusion
Cancer, being one of the deadliest diseases around the globe, mounts a burden on global healthcare. NSCLC has the highest contribution to mortality and incidence compared to other lung cancer subtypes. The multifactorial occurrence of the disease remains a cause of concern for individuals suffering from lung cancer. Many researchers have paved the way for different therapeutics and success in managing the disease. Their research has made significant contributions to the field of cancer biology, specifically cancer management. The finding of several factors that lead to NSCLC has brought about tremendous advantages in clinical settings. However, the management of the disease remains troublesome for professional healthcare workers. The prognostic factor includes the staging of NSCLC, which uses the size of the tumor (T), the locoregional lymph node invasion (N), and the metastasis status (M). IASCL, along with Cancer Research and Biostatistics (CRAB), has brought the eighth edition of the TNM classification. Factors such as smoking cigarettes and molecular alteration contribute the most in terms of the occurrence of the disease. The alteration in the genes such as EGFR, BRAF, KRAS, MET, ROS, etc The up- or down-signaling of pathways such as JAK-STAT, PI3K-AKT, MAPK, and other parallel signaling pathways play a key role in the development of cancer. With the need for better management, contributions from nano-based therapeutics have emerged as a potential candidate for managing the same. Nanotechnology in the past two decades has made a significant contribution to the healthcare sector. Cancer, with its alterations in different molecular drivers, has been targeted via nano-based formulations. The nano-based formulations are made up of different nanoparticles, such as polymeric, lipid, and metal-based. These include polymerosomes, liposomes, micelles, gold, silver, etc These nanoparticles, serving as a nanocarrier, have been well utilized by researchers for their enhanced efficacy, biodistribution, and targeted delivery. The in vitro and in vivo studies have demonstrated them to be an effective solution to multiple issues, such as drug resistance. Researchers have used many dual drug nanoformulations that yield synergistic effects and better efficacy with respect to just drugs. The use of cisplatin and etoposide in nanostructured lipid carriers showed better efficacy and synergistic value (CI < 1). Gefitinib and bevacizumab in dual-responsive nanoparticles showed efficient inhibition of EGFR and signaling pathways. Paclitaxel (PTX) and triptolide (TL) in LPN had 85% efficiency in both in vitro and in vivo studies. The combination of hydrophobic and hydrophilic drugs for nano-based dual drug delivery yielded positive results in both in vitro and in vivo analyses. GEM (hydrophilic) and PTX (hydrophobic) targeted in NLCs with NAG as a receptor, forming NAG-NLCs complexes, showed good antitumor efficacy and advanced cellular uptake in comparison to normal drugs. These studies have paved the way for researchers to use nano-based formulations and combine drugs for better management of NSCLC.
Footnotes
Acknowledgments
Faheem Ahmad is highly thankful to the Indian Council for Medical Research for providing the Junior Research Fellowship (3/1/3 JRF-2023/HRD(LS)-11).
Author Contributions
Conceptualization, FA and Tanuja; methodology, FA; validation, FA and KR; formal analysis, FA; resources, Tanuja; data curation, FA; writing—original draft preparation, FA; writing—review and editing, Tanuja, KR; supervision, Tanuja, KR; All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data collected were from public repositories, and the keywords are provided in the manuscript.
