Abstract
This study aimed to evaluate the effect in female rats of 6 weeks of ginger consumption on gestational diabetes by assessing glucose and lipid metabolism, oxidative damage, inflammation, and the WNT/β-catenin/glycogen synthase kinase-3 beta (GSK3β) signaling pathway. In this study, 40 adult female rats were divided into 4 equal groups as follows: pregnant rats, pregnant rats with diabetes, pregnant rats consuming ginger, and pregnant rats with diabetes consuming ginger. Induction of diabetes on day 0 of pregnancy was induced by streptozotocin (STZ) injection. Insulin, glucose, and lipid profiles as metabolic factors were measured along with the hepatic expression of glucose transporter type 4 (GLUT4), mechanistic target of rapamycin complex 1 (mTORc1), sterol regulatory element-binding protein-1c (SREBP-1c), peroxisome proliferator-activated receptor α (PPAR-α), β-catenin, GSK3β, PPAR-g, nuclear factor-κB (NF-kB), and nuclear factor erythroid 2-related factor 2 (Nrf2). The levels of tumor necrosis factor α (TNF-α), interleukin 1β (IL-1β), and oxidative parameters were measured in the liver. The level of metabolic factors was significantly impaired in diabetic pregnant rats compared to the control group. However, ginger improved these parameters in the diabetic pregnant—ginger group compared to the diabetic pregnant group. The hepatic expression of genes involved in metabolism, oxidative stress, inflammation, and the wingless and int-1 (WNT) signaling pathway was significantly impaired in diabetic pregnant rats, while ginger improved them in the hepatic tissue of diabetic pregnant rats. It can be concluded that ginger could improve gestational diabetes by genes involved in metabolism, the WNT signaling pathway, oxidative stress, and inflammation. These results suggest that ginger can be an adjunct drug during pregnancy as a metabolic modulator.
Keywords
Introduction
Gestational diabetes mellitus (GDM) as a type of diabetes can present with varying degrees of hyperglycemia during pregnancy. Rates of GDM are on the rise worldwide and affecting 5% to 10% of pregnant women. It is estimated that more than 70% of women with GDM and approximately 30% of their born children are prone to many abnormalities and diseases later in life, so it has a health and economic burden on health systems. 1 Pregnancy hypertension, insulin resistance, and subclinical metabolic dysfunction are symptoms of gestational diabetes. This type of diabetes usually disappears after pregnancy. 2 However, it can be a risk factor for type 2 diabetes and coronary artery disease in affected mothers. The treatment of gestational diabetes is somewhat complex, and recommendations for treatment regimens and insulin use are usually a priority. However, determining the appropriate dose of insulin is very difficult and in addition to developing insulin resistance, its clinical use in mothers and infants is risky. The details of the causes of GDM are completely unknown. 3
Pregnancy is a unique physiological period in which the mother's body undergoes metabolic adaptation to meet the developmental needs of the fetus. During pregnancy, other physiological adaptations include respiratory, hematological, renal, and cardiovascular adaptations.2,4 As pregnancy progresses, the metabolic needs of the mother and fetus 5 increase, along with the release of placental and growth hormones including estrogen, progesterone, leptin, cortisol and placental lactogen. 6 The consequence of this condition is hyperglycemia and dyslipidemia, a condition similar to insulin resistance 7 or diabetes mellitus. 8 Evidence suggests that these changes are compensated for by hypertrophy and hyperplasia of pancreatic β-cells, as well as increased glucose-stimulated insulin secretion. 9 Metabolic changes that occur during pregnancy can be due to changes in the expression and function of genes and proteins such as sterol regulatory element-binding protein-1c (SREBP-1c), glucose transporter type 4 (GLUT4), mechanistic target of rapamycin complex 1 (mTORc1), peroxisome proliferator-activated receptor gamma (PPAR-g), and PPAR-α that play a role in regulating glucose and lipid metabolism. In this condition, regulation of the expression of these genes is also changed. In addition, hyperglycemia-induced lipid toxicity, inflammation, and oxidative stress, along with disruption of cellular signaling pathways in β-cells and metabolically active organs such as the liver, play a role in the pathophysiology of GDM.2,10 Moreover, impairments in the wingless and int-1 (WNT) signaling pathways, the toll-like receptor 4 (TLR-4)/ nuclear factor-κB (NF-κB) pathway, decreased tumor necrosis factor α (TNF-α)-stimulated adenosine monophosphate-activated protein kinase (AMPK) activity, and increased serum levels of interleukin (IL)-1β, IL-6, TNF-α, and NF-κB activity are also implicated in GDM and insulin resistance.11–13 The activation of WNT/β-catenin signaling can increase a cell’s insulin sensitivity by increasing glucose uptake from the bloodstream. 3 However, impairments in the WNT signaling pathways can evolve into the pathogenesis of type 2 diabetes and affect insulin signaling in its target cells, in part due to a decrease in Wnt10b protein. 14 Therefore, it was hypothesized that activation of the WNT signaling pathway and improving lipid and glucose metabolism could play an important role in insulin sensitivity and reduction of insulin resistance in mothers with gestational diabetes. 4 Studies in the last decade have shown that lifestyle changes and the use of complementary and alternative medicine are effective treatment strategies to improve the condition of metabolic patients, including those with gestational diabetes.
The use of medicinal plants and their products, including ginger, has also received much attention in recent years. 15 Zingiber officinale Roscoe is a perennial flowering plant whose rhizome and root, known as ginger, are widely used as a spice and folk remedy. Many studies have shown the antidiabetic, anti-inflammatory, antioxidant, antihyperlipidemic, antihyperglycemic, and antinausea effects of ginger.16,17 Studies have shown that ginger extract or gingerol, as its main active ingredient, can increase insulin sensitivity and glucose uptake. Ginger can also increase high-density lipoprotein (HDL) cholesterol and improve dyslipidemia in obese people. 18 Hence, it seems to be a good treatment option for improving the metabolic status in patients with gestational diabetes. In addition, ginger can help reduce nausea and vomiting caused by chemotherapy or pregnancy. 19 Therefore, the aim of this study was to answer the question of whether ginger can improve gestational diabetes by targeting genes involved in nutrient metabolism, oxidative stress, inflammation, and the WNT signaling pathway.
Materials and Methods
Material Plant
Fresh ginger (Zingiber officinale) rhizome, purchased from a local herbal market in Shiraz, Iran, was confirmed by a botanist, Sedigheh Khademian, in the Department of Phytopharmaceuticals, School of Pharmacy, Shiraz University of Medical Sciences, Shiraz, Iran (voucher number PM: 948). In addition, it was approved using www.efloras.org/Flora of China and http://www.theplantlist.org. The ginger was then chopped into small pieces, dried at laboratory temperature, and finally ground into a powder.
Total Phenolic Content and Total Antioxidant Capacity
Total phenolic content (TPC) was assessed by the Folin–Ciocalteu spectrophotometric method. 20 Its value was calculated by the equation of the calibration curve created by gallic acid, and expressed as μg of gallic acid equivalents (GAE)/mg of sample. The scavenging activity of the DPPH (1,1-diphenyl- 2-picrylhydrazyl) radical was used to measure total antioxidant capacity (TAC). 17 DPPH inhibition (%) was calculated from the following formula:
% Inhibition = ((A blank–A sample)/A blank) × 100.
Animals
Forty adult female Wistar rats (220 ± 20g, 6-8 weeks) were maintained at 22°C, at relative humidity 38%, and with a 12/12 h of light/dark cycle. Rats had free access to chow and water. All stages of this research were based on the instructions for keeping and observing the ethical principles of working with laboratory animals approved by The Second Affiliated Hospital of Nanchang University (jxfy-obs-2022-016).
Study Design
After a week of adaptation, adult female rats were divided into four equal groups as follows: Group 1: pregnant rats, Group 2: pregnant rats with diabetes, Group 3: pregnant rats consuming ginger (100 mg/kg, by gavage), and Group 4: pregnant rats with diabetes consuming ginger (100 mg/kg, by gavage). Diabetes was induced on day 0 of pregnancy by intraperitoneal injection of a single dose of streptozotocin (STZ, 45 mg/kg, Sigma-Aldrich Chemie GmbH, Germany). 21 The animals fasted for 16 h before injection. Glucose levels were measured by a digital glucometer (Accu check, 41110000, USA) 3 days after STZ injection and 1 night of fasting using a lateral tail vein blood sample. Glucose levels above 180 mg/dL were considered diabetic levels. 21
Mating and Fertilization
The estrous cycle of each rat was determined based on the protocol described by previous studies.21,22 Briefly, vaginal smears of each virgin female rat were prepared and evaluated daily. The ratios of leukocytes, epithelial, and cornified cells were used in unstained vaginal smear photomicrographs to determine the stages of the estrous cycle. Female rats in the proesterous stage were mated with healthy male rats (2 females with 1 male) overnight. Early the next morning, female rats were examined by checking the outer surface of the vagina and the presence of white vaginal plaque caused by semen coagulation. The presence of a grayish-white clot blocking the vaginal opening was considered the first day of pregnancy. Each pregnant female rat was then transferred to a separate cage and the pregnancy process was followed by monitoring weight gain. At the end of the second week after delivery, blood and tissue samples were taken from healthy and diabetic mothers.
Sampling, Tissue Preparation, and Measurement of Biochemical Parameters
After the last treatment session, after one night of fasting, the animals were anesthetized with a combination of ketamine and xylazine. Blood samples were collected through a cardiac puncture and centrifuged (3000 ×
Molecular Measurement
The expression of SREBP-1c, mTORc1, PPAR-g, PPAR-α, GLUT4, inducible nitric oxide synthase (iNOS), NF-κB, and Nrf2 genes were measured. Total ribonucleic acid (RNA) was extracted from the liver tissue using a total RNA Purification kit (Jena Bioscience, Germany). Complementary DNA (cDNA) was synthesized from RNA samples by Primer Script reverse transcriptase (TaKaRa, Japan). Primers of SREBP-1c, mTORc1, PPAR-g, PPAR-α, GLUT, iNOS, NF-κB, and Nrf2, along with β-actin as internal control gene, were selected based on previous studies17,29 (Table 1). Real-time polymerase chain reaction (PCR) was performed by Real Q Plus 2x Master Mix Green (Amplicon, Denmark) in a Real Time PCR device (Applied Biosystem, USA). The expression of the target genes was normalized against β-actin expression, and their relative expression was calculated using the method 2−ΔΔCT.
The Primer Sequences Used in This Study.

Abbreviations: GLUT4, glucose transporter type 4; GSK3β, glycogen synthase kinase-3 beta; iNOS, inducible nitric oxide synthase; mTORc1, mechanistic target of rapamycin complex 1; NF-κB, nuclear factor-κB; Nrf2, nuclear factor erythroid 2-related factor 2; PCR, polymerase chain reaction; PPAR, peroxisome proliferator-activated receptor; TLR-4, Toll-like receptor 4; SREBP-1c, sterol regulatory element-binding protein-1c;
Histological Examination
The median lobe of the liver was fixed in 10% formaldehyde solution. After 24 h, this solution was replaced with a 4% formaldehyde solution. After at least a week, the samples were embedded in paraffin and 5 μm thick sections were cut. At least four sections of each tissue sample were stained using hematoxylin and eosin (H&E). Finally, histological evaluation was performed using a light microscope (Olympus, Japan) at a magnification of 40x.
Statistical Analysis
The data were recorded in Statistical Package for the Social Sciences (SPSS) version 18 and were analyzed by one-way analysis of variance. Tukey post hoc tests were used to compare the differences between groups. The results are presented as the means ± standard deviation (SD). P < .05 was considered the minimum statistically significant level.
Results
Total Phenol Content and DPPH Results
Total phenol content and DPPH radical scavenging capacity of ginger powder were measured. The total phenol content was 7.52 ± 0.02 (μg of GAE/mg of sample) and the IC50 of ginger powder was 7.10 μg/mL (Figure 1).

DPPH radical scavenging activity of ginger powder. Abbreviation: DPPH, 1,1-diphenyl- 2-picrylhydrazyl.
Changes in Body Weight in the Control and Pretreated Groups
The results showed that the body weight significantly decreased in pregnant rats with diabetes compared to healthy pregnant rats. However, pretreatment with ginger powder (100 mg/kg) improved these changes in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes (P < .05). There was no significant difference regarding body weight between healthy pregnant rats and pregnant rats consuming ginger (Figure 2 and Table 2).

The mean ± SD of the mean body weight during the study period in different groups. *P ≤ .05 (vs control group) and #P ≤ .05 (vs diabetic group and Table 2).
Ginger Powder Could Improve Lipid Profile, Glucose, Insulin, and HOMA-IR in the Control and Pretreated Groups
The mean ± SD of the fasting blood glucose (mg/dL), insulin (μU/L), and homeostasis model of insulin resistance (HOMA-IR) levels are presented in Table 3. The serum level of fasting Blood glucose (FBG) was higher and insulin lower in pregnant rats with diabetes than in the control group. However, pretreatment with ginger powder could significantly modulate these parameters in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes. The HOMA-IR index was significantly increased in the pregnant rats with diabetes compared to pregnant rats, while pretreatment with ginger could improve the level in pregnant rats with diabetes consuming ginger compared to the pregnant rats with diabetes without any intervention (Table 3). Insulin sensitivity and β-cell function significantly decreased in the diabetic pregnant group compared to the pregnant group. However, these parameters were significantly improved in the diabetic pregnant rats pretreated with ginger powder compared to the diabetic pregnant group (Table 3).
The Mean ± SD of the Mean of Final Body Weight, Liver Weight, and Liver Index in Different Groups.

*P ≤ .05 (vs the control group), # P ≤ .05 (vs the diabetic group).
The Mean ± SD of Fasting Blood Glucose (mg/dL), Insulin (μU/L), HOMA-IR, and Insulin Sensitivity in Different Groups.
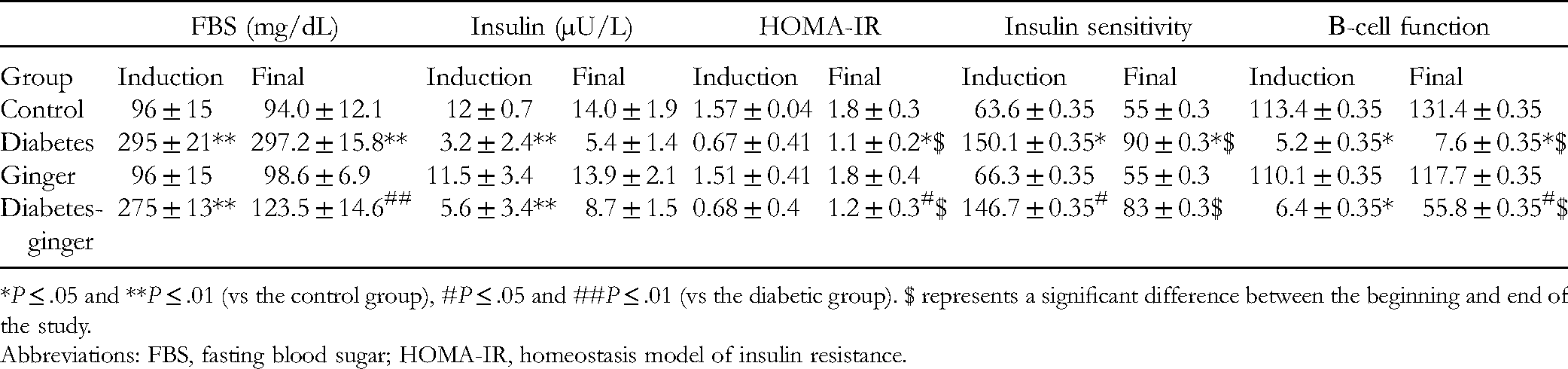
*P ≤ .05 and **P ≤ .01 (vs the control group), #P ≤ .05 and ##P ≤ .01 (vs the diabetic group). $ represents a significant difference between the beginning and end of the study.
Abbreviations: FBS, fasting blood sugar; HOMA-IR, homeostasis model of insulin resistance.
The mean ± SD of serum levels of lipid profiles including triglycerides (TG), total cholesterol (TC), low-density lipoprotein (LDL), and HDL are presented in Table 4. The serum levels of TG, TC, and LDL significantly increased and the HDL level decreased in diabetic pregnant rats compared to pregnant rats without diabetes. However, pretreatment with ginger improved these metabolic factors in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes (Table 4).
The Mean ± SD of Lipid Profile and Glucose Levels in Serum of Different Groups.

*P ≤ .05 (vs the control group), #P ≤ .05 (vs the diabetic pregnant group).
Abbreviations: HDL-c, high-density lipoprotein cholesterol; LDL-c, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglycerides.
Ginger Powder Could Improve Hepatic Oxidative Damage in the Control and Pretreated Groups
The liver activity of SOD, glutathione peroxidase (GPx), and CAT enzymes was measured along with MDA level to assess the oxidative status. The levels of these enzymes significantly decreased and the MDA level increased in diabetic pregnant rats. Pretreatment with ginger improved these parameters in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes (Figure 3).

Mean ± SD of MDA level (A), Nrf2 expression (B), and antioxidant enzyme activity (C) in different groups. *P < .05 versus the pregnant rats without diabetes (the control group). #P < .05 versus pregnant rats with diabetes (the diabetic group). Abbreviations: MDA, malondialdehyde; Nrf2, nuclear factor erythroid 2-related factor 2.
The Nrf2 gene was evaluated as a gene controlling the state of oxidative damage in this study. Nrf2 expression in the liver tissue was significantly reduced in the diabetic pregnant group compared to the control group, whereas pretreatment with ginger improved Nrf2 expression in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes without any intervention (Figure 3).
Ginger Powder Could Inhibit Inflammation by Decreasing IL-1β and TNF-α Activity and Expression of iNOS, TLR-4, and NF-κB
Diabetes increased the levels of IL-1β and TNF-α and the expression of NF-κB, TLR-4, and iNOS in the liver tissue of the diabetic pregnant group (Figure 4). However, pretreatment with ginger ameliorated the hepatic levels of IL-1β and TNF-α and expression of these genes in pregnant rats with diabetes consuming ginger compared to pregnant rats with diabetes without any intervention (Figure 4).

Mean ± SD of tissue levels of IL-1β and TNF-α (A) and the expression of iNOS, TLR-4, and NF-ĸB (B) in different groups. *P < .05 versus the control group. #P < .05 versus the diabetic group (pregnant rats with diabetes). Abbreviations: IL, interleukin; iNOS, inducible nitric oxide synthase; NF-κB, nuclear factor-κB; TLR-4, toll-like receptor 4; TNF-α, tumor necrosis factor α.
Ginger Powder Could Ameliorate the Gene Expressions Involved in Lipid and Glucose Metabolism
In this study, the hepatic expressions of mTORc1, SREBP-1c, PPAR-α, PPAR-g, and GLUT4 were measured to evaluate the status of lipid and glucose metabolism. The expression of PPAR-α significantly increased in the diabetic pregnant group compared to the pregnant group, while pretreating with ginger powder could significantly ameliorate its expression in the pregnant rats with diabetes consuming ginger compared to the diabetic pregnant group. The hepatic expression of PPAR-g, GLUT4, mTORc1, and SREBP-1c decreased in the pregnant rats with diabetes compared to the pregnant group. However, pretreating diabetic pregnant rats with ginger improved their expression compared to the diabetic pregnant group (Figure 5).
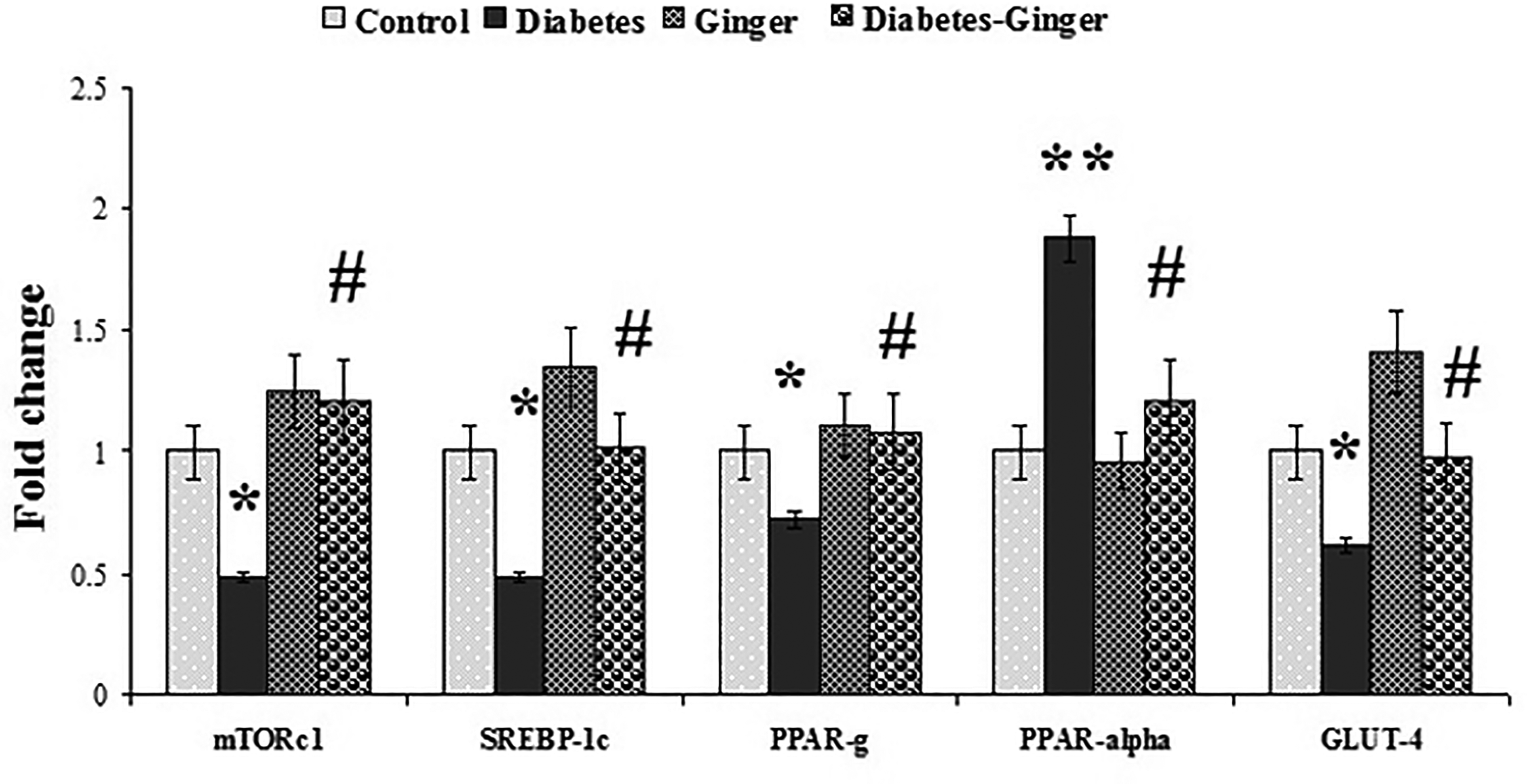
The mean ± SD of mTORc1, SREBP-1c, PPAR-g, PPAR-α, and GLUT4 expression in liver of the control and pretreated groups. * P < .05 and **P < .01 versus the control group. #P < .05 versus the diabetic pregnant group. Abbreviations: mTORc1, mechanistic target of rapamycin complex 1; PPAR, peroxisome proliferator-activated receptor; SREBP-1c, sterol regulatory element-binding protein-1c; GLUT4, glucose transporter type 4
Ginger Powder Could Ameliorate the WNT/β-Catenin/GSK3β Pathway in Liver of Diabetic Pregnant Rats
In this study, the hepatic expression of β-catenin and glycogen synthase kinase-3 beta (GSK3β) was measured to evaluate the status of the WNT/β-catenin/GSK3β pathway. The expression of β-catenin and GSK3β significantly increased in the diabetic pregnant group compared to the control group. Pretreating with ginger powder could significantly ameliorate the expression of β-catenin and GSK3β in the pregnant rats with diabetes consuming ginger (the diabetic-ginger group) compared to the diabetic pregnant group. There was no significant difference between the control and ginger groups regarding expression of these genes (Figure 6).

The mean ± SD of β-catenin and GSK3β expression in liver of control and pretreated rats. *P < .05 versus the control group (pregnant rats without diabetes). #P < .05 versus The diabetic group (pregnant rats with diabetes). Abbreviation: GSK3β, glycogen synthase kinase-3 beta.
Histological Finding
Liver tissue from all groups was examined with a light microscope (Olympus, Japan) at 40 × magnification (Figure 7). Fatty degeneration and diffused steatosis were observed in the diabetic pregnant rats compared to the normal structure of hepatocytes in the control rats. However, pretreatment with ginger improved these features in the ginger-diabetic pregnant group (Figure 7).

Light microscopic examination of liver sections in different groups was performed with × 40 magnification. Histological findings of the control and ginger (100 mg/kg) groups showed normal arrangement and structure of hepatocytes and proper central vein. The presence of fat droplets and hepatic steatosis is well established in hepatocytes of the diabetic pregnant group, while pretreatment with 100 mg/kg body weight (BW)/day of ginger significantly improved these features in the liver of diabetic pregnant rats.
Discussion
During pregnancy, the mother’s body undergoes a series of physiological changes to support the needs of the developing fetus. The most important of these changes include the adaptation of the cardiovascular, renal, respiratory, metabolic, and hormonal systems. Changes in the serum levels of insulin-resistant hormones such as estrogen, progesterone, leptin, cortisol, lactogen, and growth hormone are essential, in addition to meeting the metabolic needs for fetal growth and development. As the pregnancy progresses, the function of these hormones increases the concentration of blood glucose and free fatty acids. In this condition, the mother’s body increases glucose-stimulated insulin secretion to maintain glucose homeostasis through hypertrophy and hyperplasia of pancreatic β-cells. This condition leads to insulin resistance. However, the level of insulin secreted is not enough to maintain glucose and leads to hyperglycemia during pregnancy, which is known as gestational diabetes. Our results showed that STZ injection caused gestational diabetes through inflammation, oxidative damage, changes in glucose and lipid metabolism, and the WNT/β-catenin/GSK3β pathway. In this study, we showed that serum levels of insulin, glucose, and lipid profiles, along with insulin resistance and insulin sensitivity indices were significantly altered in diabetic rats. Abnormally high levels in the plasma lipid profile and glucose, known in diabetes as dyslipidemia and hyperglycemia, respectively, are due to lack of insulin. The metabolic pathways which are either inhibited or activated by insulin are disrupted in these conditions. mTORc1, SREBP-1c, PPAR-α, PPAR-g, and GLUT4 genes are the major genes in these metabolic pathways that are affected by insulin. Our results showed that the expression of GLUT4, mTORc1, SREBP-1c, PPAR-α, and PPAR-g genes involved in glucose and lipid metabolism were significantly altered in hepatic tissue of diabetic rats. At the molecular level, insulin resistance due to a defect in insulin signaling leads to the accumulation of glucose (hyperglycemia) and fatty acids and their storage form, triglycerides, and contribute to the development of GDM. 30 Dysfunction in insulin signaling leads to inadequate expression and insertion of GLUT4 which is responsible for the transport of glucose into the cell and can be one of the causes of hyperglycemia. It has been well shown that insulin-stimulated glucose uptake decreases in GDM. 31 In addition, changes in expression of insulin receptor substrate 1 (IRS-1), phosphoinositide 3-kinases (PI3K), and GLUT4 contribute to GDM. 30 In addition, proinflammatory cytokines, reactive oxygen species (ROS), and free saturated fatty acids increase intracellular concentrations of diacylglycerol in insulin target cells and inhibit intracellular insulin signaling (IRS-1 and PI3K) by activating protein kinase C (PKC) via IκB kinase (IKK).32,33 mTORc1 is activated by insulin and is involved in modulating the expression of genes involved in glucose and lipid metabolism. SBREP-1c, which plays a key role in hepatic lipogenesis and facilitates the storage of fatty acids as triglycerides, is regulated by insulin via mTORc1. It regulates genes involved in lipogenesis. The WNT pathway, TNF-α, energy status, and receptor tyrosine kinases, including the Akt/protein kinase B (PKB), mitogen-activated protein kinases (MAPK)/extracellular signal-regulated kinase (ERK), and Jun kinase (JNK) pathways, are the upstream signaling pathways of mTORc1 and may play an important role in the metabolic adaptation during pregnancy. PPAR-α is a major regulatory factor in lipid metabolism and is involved in the adaptive response to long-term fasting in conditions of energy deprivation by activating ketogenesis. PPAR-α increases the uptake, utilization, and oxidation of fatty acids by increasing the transport of fatty acids, the binding and activation of fatty acids, and the oxidation of peroxisomal and mitochondrial fatty acids. PPAR-g regulates fatty acid storage and glucose metabolism. PPAR-g also increases insulin sensitivity by stimulating lipid uptake and adipogenesis by adipocytes, reducing lipotoxicity, inducing fibroblast growth factor 21 (FGF21), and increasing nicotinamide adenine dinucleotide phosphate (NADPH) production. PPAR-g also inhibits inflammation by promoting anti-inflammatory M2 macrophage activation. Therefore, in the absence of insulin lipolysis increases and lipogenesis decreases which can be a reason for dyslipidemia or increased serum lipid profile and hyperglycemia. The results of this study showed that one of the mechanisms of physiological metabolic adaptations during pregnancy is a change in the expression of mTORc1, SREBP-1c, PPAR-α, and PPAR-g genes. Disruption of their expression can lead to metabolic disorders and hyperglycemia and even in advanced cases cause gestational diabetes. However, the results in the groups receiving ginger showed that ginger can significantly improve the metabolic status by modulating the expression of these genes. Pretreating with ginger could improve gestational diabetes by inhibiting inflammation, oxidative damage, and the WNT/β-catenin/ GSK3β pathway. In agreement with our findings, Hajimoosayi et al 34 showed that ginger could improve fasting blood sugar (FBS), insulin, and HOMA-IR in women with GDM. Adefegha et al 35 showed that ginger could improve postprandial hyperglycemia associated with T2MD by inhibiting α-amylase and α-glucosidase activities. Akhani et al 36 reported that ginger increases insulin levels and decreases serum glucose, cholesterol and triglyceride levels in mice with type 1 diabetes. Bahramian et al 37 showed that ginger capsules could improve postprandial hyperglycemia and insulin, and reduce the frequency of visits to the gynecologist in women with GDM. It seems that ginger can improve the health of mothers with gestational diabetes and their infants. Akbari et al 16 also showed that ginger can improve homocysteine metabolism and inhibit the oxidative damage caused by its increase. The results of the evaluation of metabolic factors and genes involved in glucose and lipid metabolism showed that ginger could significantly improve metabolic disorders in gestational diabetes. In this study, we showed that ginger could decrease insulin resistance and increase insulin sensitivity. These effects may be due to the widespread role of ginger and its effective compounds in improving β-cell function and increasing glucose uptake by target or nontarget insulin tissues. 38 Moreover, it was shown that the gene expression of GLUT4 was improved following the consumption of ginger.38,39 In line with our results, ginger exerts hypoglycemic effects by improving pancreatic β-cell function, increasing peripheral glucose utilization, increasing hepatic glycogen synthesis, and inhibiting hepatic glucose production.39,40 In this study, the results also showed that the serum lipid profile could be significantly improved by ginger. Studies that have been conducted since 2000 on the hypoglycemic, hypolipidemic, anti-inflammatory and antioxidant functions of ginger have shown that ginger, and its extract or active compounds can reduce hyperlipidemia and hyperglycemia, and inhibit inflammation and oxidative damage by modulating the expression genes involved in these conditions.41,42 Our results showed that β-catenin and GSK3β expression were significantly altered in liver tissue in diabetic rats. However, the expression of these genes showed a significant improvement in diabetic rats receiving ginger. It is possible that one of the cellular mechanisms involved in physiological adaptation during pregnancy is a change in signaling of the WNT/β-catenin/GSK3β signaling pathway.
Our results also showed that ginger improved oxidative damage and inflammation in the liver tissue of rats with gestational diabetes by increasing Nrf2 expression and antioxidant enzyme activity (SOD, GPx, and CAT), but decreasing TLR-4, NF-κB, and iNOS expression and IL-1β, TNF-α, and MDA levels. It is well known that inflammation and oxidative damage are involved in the pathogenesis of gestational diabetes and their inhibition has a significant role in improving the status of diabetes. On the other hand, the anti-inflammatory and antioxidant role of ginger and its products has been well demonstrated in previous studies. Inhibition of inflammation and oxidative damage by ginger and its products such as gingerol, shogaol, and other structurally related substances is through suppression of 5-lipoxygenase or prostaglandin synthetase43,44 and inhibiting the synthesis of proinflammatory cytokines such as IL-1, TNF-α, and IL-6 and IL-8.41,45 Ginger extract and 6-shogaol inhibit expression of iNOS, NF-κB, and cyclooxygenase 2 (COX-2) and the production of nitric oxide (NO), prostaglandin E (PGE), TNF-α, and IL-1β. 46 Ginger enhances free radical scavenging and strengthens the antioxidant system by increasing the expression of genes that control the activity of enzymatic and non-enzymatic systems.41,43,44,47 As previously described, ROS and TNF-α can induce insulin resistance, which is inhibited by ginger. 48 Lee et al 44 also showed that ginger can inhibit inflammation and oxidative stress by inhibiting the expression of NF-κB and iNOS, reducing the levels of TNF-α, IL-1β, and MDA, and enhancing the expression of Nrf2 and antioxidant enzymes. These results well suggest that ginger can act as an antioxidant and anti-inflammatory. Evidence has also shown that homocysteine, a nonprotein amino acid made from the demethylation of methionine, can lead to GDM through dysfunction of pancreatic β-cells and oxidative damage. 3 Interestingly, Akbari et al 16 reported that ginger can inhibit ethanol-induced hyperhomocysteinemia. On the other hand, paying attention to appropriate amounts of micronutrients such as sufficient levels of iron, zinc, and vitamin D during this period can also play a significant role in maternal and fetal health during and after pregnancy. 49 Interestingly, Ahmad Mustafa et al 17 showed that ginger and ginger + zinc could improve oxidative damage, inflammation and autophagy due to fructose, and regulate glucose and lipid metabolism and zinc homeostasis in rats with metabolic syndrome. Therefore, it is safe to say that ginger can reduce inflammation and oxidative stress by improving the expression of genes and enzymes involved in these processes and can be one of the main mechanisms for improving metabolic disorders caused by diabetes. Evidence has also shown that impaired neurohormonal levels, such as those of adiponectin and leptin, are involved in the pathogenesis of GDM 3 and may explain what we have not been able to fully achieve through ginger treatment. Adiponectin, resistin, and leptin are also involved in insulin signaling and the metabolism of fatty acids and glucose, and can enhance processes such as β-oxidation and inhibit gluconeogenesis. 50 Therefore, further studies are necessary to discover other mechanisms involved in these complications and the pathways that lead to GDM improvement.
Conclusion
It can be concluded that ginger can improve gestational diabetes by modulating genes involved in glucose and lipid metabolism, inflammation and oxidative damage, and the WNT pathway. These results show that ginger can be used as an adjuvant drug during pregnancy as a metabolism modifier. However, clinical studies are needed to evaluate this suggestion.
Footnotes
Acknowledgement
We would like to thank the scientific and research group affiliated with Dr Abolfazl Akbari for their guidance.
Authors’ Contributions
LL was involved in methodology, investigation, and writing original draft. SZ was involved in methodology, writing original draft, and resources. AA was involved in conceptualization, supervision, project administration, analysis, and writing review and editing. BT was involved in conceptualization, supervision, project administration, writing review and editing, and resources.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support forthe research, authorship, and/or publication of this article: The authors state that the intellectual property of this research belongs to the scientific and research group affiliated to Dr. Abolfazl Akbari.
Ethical Approval /Patient Consent
All stages of this research were based on the instructions for keeping and observing the ethical principles of working with laboratory animals approved by The Second Affiliated Hospital of Nanchang University (jxfy-obs-2022-016).
Statement of Human and Animal Rights
All stages of this research were based on the instructions for keeping and observing the ethical principles of working with laboratory animals approved by The Second Affiliated Hospital of Nanchang University (jxfy-obs-2022-016).
