Abstract
Osteoporosis is a metabolic skeletal disorder characterized by increased fragility and fracture risk as s result of reduced bone mineral density and microstructural destruction and caused a heavy burden on families and society. Current medicines, on the other hand, have some limitations, with side effects and doubts regarding long-term efficacy being highlighted. Studies seeking for natural constituents as potential treatment options therefore come into focus. Icariin is a phytochemical derived from a traditional Chinese medicine, Herba epimedium, that has been used to treat orthopedic disorders in ancient China for thousands of years, including osteoporosis, osteoarthritis, and fracture. Icariin belongs to a category of prenylated flavonoids and has been shown to help reduce osteoporosis bone loss while having relatively low side effects. Icariin's anti-osteoporosis properties manifest in a variety of ways, like promoting osteogenesis, suppressing osteoclastogenesis and bone resorption, regulating migration, proliferation, and differentiation of mesenchymal stem cells, enhancing angiogenesis, anti-inflammation, and antioxidation. These procedures entail a slew of critical signaling pathways, such as PPARγ, ERα/AKT/β-catenin, and MAPK. Therefore, icariin can be an applicable alternative to improve osteoporosis although the underlying mechanisms have yet to be fully understood. In this study, we searched using the terms “icariin” and “osteoporosis,” and included 64 articles meeting the inclusion criteria and reviewed the research of icariin in anti-osteoporosis over the last 10 years, and discussed new prospects for future study. Therefore, this review may provide some references for further studies.
Introduction
Osteoporosis is defined as a systemic skeletal disease characterized by low bone mass and microarchitectural deterioration of bone tissue, with a consequent increase in bone fragility and susceptibility to fracture. 1 Osteoporosis affects roughly 200 million people globally, with one in every three women and one in every five males suffering from osteoporotic fractures. 2 With the aging of the population, the cost of osteoporosis rapidly grew, placing a significant burden on families and society. WHO announced diagnostic criteria using SD scores of bone mineral density (BMD) with osteoporosis defined as a BMD T-score of −2.5 or less and osteopenia or low bone mass defined as a BMD T-score of −1 and −2.5. 3 The pathogenesis of osteoporosis involves numerous interactions of hazards, including mechanical loading, microdamage, genetics, estrogen insufficiency, aging, and oxidative stress. Anti-resorptive, anabolic, and mixed-effect medications for osteoporosis prevention or treatment include estrogen, raloxifene, alendronate, denosumab, teriparatide, and romosumab. 4 The most significant shortcomings of existing medications are their adverse effects and questions regarding their long-term usefulness. Therefore, it is crucial to investigate better alternatives.
Because kidney deficiency was thought to be a common cause of orthopedic ailments in ancient China, the Herba epimedium known as Yin Yang Huo (Chinese Pinyin) was utilized as a tonifying-kidney agent to cure orthopedic-related diseases. It is the dried leaf of the Chinese medicinal plant Epimedium brevicornum Maxim, which is primarily produced in the provinces of Gansu and Sichuan. Epimedium koreanum Nakai, Epimedium brevicornum Maximowicz, Epimedium pubescens Maximowicz, and Epimedium sagittatum Maximowicz are among the various species. 5 Herba epimedium is now used in China as an adjuvant therapy combined with modern medicine to achieve better clinical efficacy. Interestingly, Herba epimedium doesn't appear to produce any noticeable adverse effects. 6 Additionally, Herba epimedium has been shown to be effective in reducing osteoporosis bone loss in clinical investigations using formulae including the herb. 7 Herba epimedium treats orthopedic illnesses by acting on a variety of targets thanks to the presence of different phytochemicals such as flavonoids, phenolic glycosides, polysaccharides, lignans, alkaloids, organic acids, trace elements, etc.7‐9 Epimedium-derived phytoestrogen flavonoids were successful in reducing bone loss in late postmenopausal women in a 24-month randomized double-blind placebo-controlled clinical experiment. 10 The primary active flavonoid glucoside identified from Herba epimedium, icariin (ICA, C33H40O15, 676.67 g/mol), is thought to be essential for Herba epimedium's ability to treat osteoporosis. 11 Previous studies have already proved its positive effect in bone metabolism, such as anti-osteoporosis, anti-osteoclastogenesis, osteogenesis, chondrogenesis, boosting osteoblastogenesis, and so on.12‐14 A lot of star molecules like ALP, OCN, Runx-2, Osterix, BMP-2 MAPKs, and NF-κB are involved in ICA regulation of bone metabolism.15‐21 Most notably, ICA is also thought to as the chemical marker for Herba epimedium quality control. To further evaluate the efficacy of ICA in anti-osteoporosis, a systematic review and meta-analysis were conducted, demonstrating that ICA is effective in reversing osteoporosis in ovariectomized rats. 22 In addition, ICA may exhibit similar efficacy to bisphosphonates like alendronate and etidronate in increasing bone mass.23,24 ICA improves bone mass in multiple manners (Figure 1). Firstly, ICA promotes osteogenesis via targeting osteoblasts which are in charge of the synthesis and mineralization of bone organic matrix. Secondly, ICA prevents osteoclast differentiation from the monocyte/macrophage lineage and osteoclast bone resorption, which both contribute to bone loss. Then, ICA controlled mesenchymal stem cell migration, proliferation, and differentiation in order to stimulate osteoblastogenesis and inhibit adipogenesis. ICA also promotes angiogenesis while reducing inflammation and oxidative stress. As a result of its many uses, ICA may be a viable alternative in the treatment of osteoporosis.

Schematic presentation of the role of icariin in anti-osteoporosis. Icariin exerts anti-osteoporosis effects via promoting osteogenesis, suppressing osteoclastogenesis and bone resorption, regulating migration, proliferation, and differentiation of mesenchymal stem cells, enhancing angiogenesis, anti-inflammation, and antioxidation.
Method
Three databases (PubMed, MEDLINE, and Web of Science) were used for preliminary literature search from 2011 to 2021. Icariin, osteoporosis are used as keywords. The title and abstract of the articles were screened according to the following inclusion criteria: (i) Original research articles, but not reviews, were included. (ii) Studies focused on that icariin exerted its efficacy against osteoporosis, including primary osteoporosis, secondary osteoporosis, osteoporotic bone loss, osteoporotic fractures, and osteoporotic bone defects, were included. The search process was presented in Figure 2.
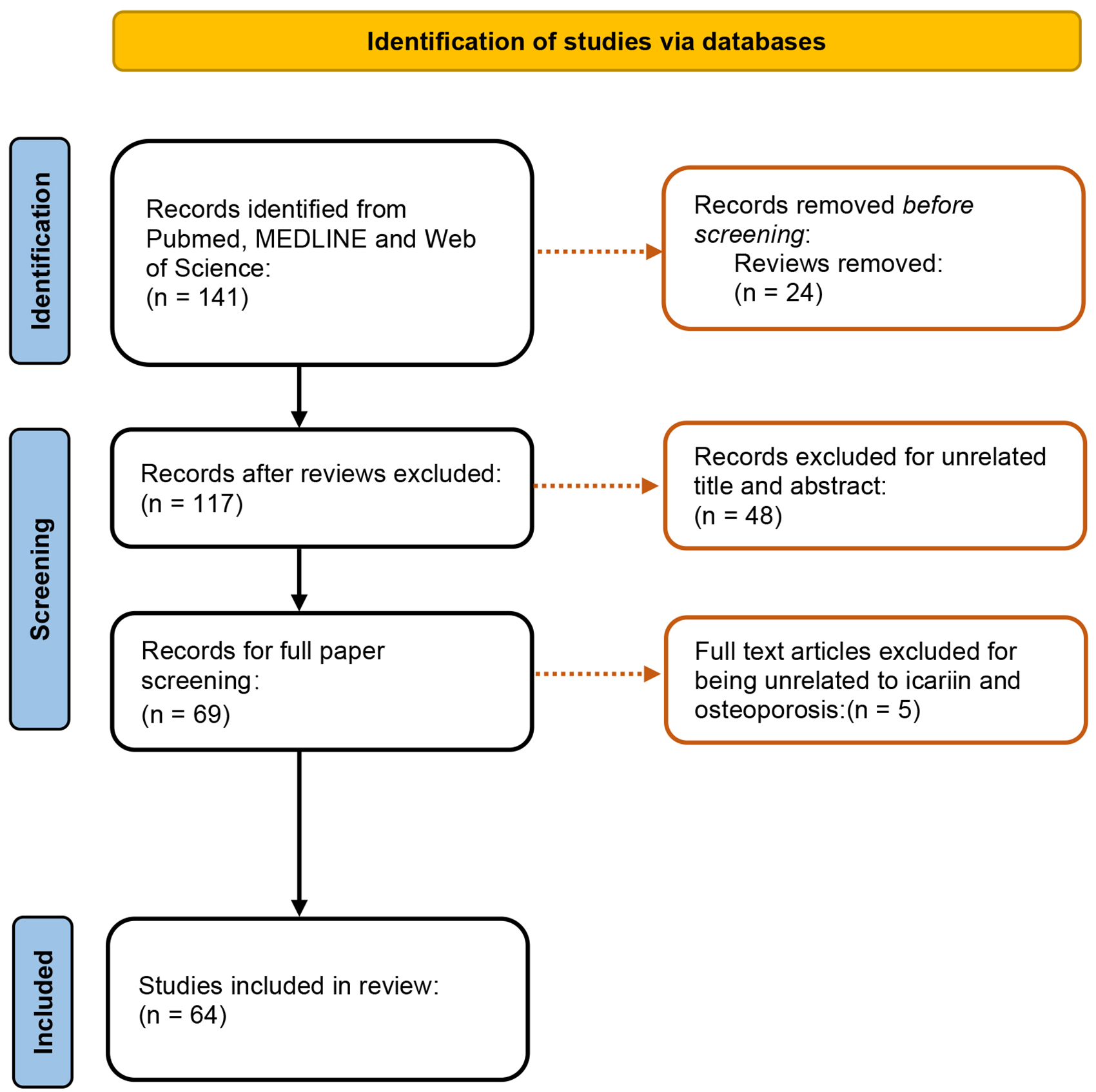
Flow diagram of the process of inclusion and exclusion.
Pharmacokinetics of Icariin
In ancient China, the Herba epimedium, the source of ICA, was used to cure osteoporosis. The substitution group at the C-8 location (Figure 3) of the prenylated flavonoid ICA is thought to be primarily responsible for the pharmacological activity. This substitution group may be the cause of ICA's greater ability than genistein to stimulate osteogenic differentiation. 25 The pharmacokinetics of ICA are associated with its administration. Following oral administration, ICA is digested in the small intestine by bacterial community β-glucosidase and lactase phlorizin hydrolase and absorbed in the small intestine.26‐28 91.2% of ICA is transformed into icariside II after oral delivery, compared to 0.4% after intravenous administration, which was considered to be the predominant form in rats’ plasma. After oral administration of ICA, LC-MS/MS method was applied to determine the pharmacokinetic parameters, which revealed that the peak time (Tmax) of ICA was 15.0 ± 0.0 min and elimination half-life (t1/2) was 73.9 ± 107.4, 29 while other studies have reported different values.30,31 Because most pharmacokinetic investigations used flavonoid extract from Herba epimedium rather than pure ICA and a different solvent to dissolve ICA, these heterogeneity may be the result of these factors. 29 There were 19 different metabolites altogether that were discovered in the plasma of rats, including icaritin, icariside I, icaritin-3-O-gluA, desmethylicaritin and its three isomers, icariside II and its one isomer, icaritin-7-O-gluA, icaritin-3,7-di-O-gluA, 1,3-isoprene alcohol icariside II, 1,3-isopren alcohol icaritin and its two isomer, allylic alcohol icaritin and its one isomer, icaritin-3-O-rha-7-O-gluA and 1,3-isoprene icariside II. According to the plasma levels, it was believed that icaritin was the main metabolite of ICA. 28 But according to a different study, icariside II makes up 91.2% of ICA's metabolites. 29 Since ICA was metabolized by humans into icaritin, icariside II, and desmethylicaritin,32,33 this suggests that ICA metabolism varies by species. According to all evidence, it is certain that icaritin, icariside II, and desmethylicaritin were commonly mentioned as being active components. After absorption, ICA was dispersed to a variety of tissues, including the testicles, uterus, ovary, liver, lung, kidney, heart, and brain. The distribution varied depending on the tissue and the gender. 31 According to the results, ICA was more widely distributed in the lungs, liver, and reproductive systems than in other tissues, and its concentration in all tissues was higher in male rats overall—with the exception of the genital organs, where it was higher in female rats. 31 However, due to the chemical structure of ICA as a diglycoside, oral administration restricted the pharmacokinetic features of ICA and had some issues with intestinal tract uptake. 27 This issue may be resolved by some pharmacological techniques, such as the use of β-cyclodextrin and propyleneglycol-liposomes, which will increase its bioavailability.34,35

Chemical structure of icariin.
Toxicity of Icariin
Because of its structural resemblance to 17-estradiol (E2), ICA is a flavonoid phytoestrogen that can bind to estrogen receptors and have estrogenic effects. 36 As a result, it may have side effects similar to those of estrogen, including uterine hemorrhage, endometrial cancer, breast cancer, and venous thrombosis.37,38 On the other hand, research on the negative impacts of ICA is scarce. A study using animals revealed that the formula Er-Xian decoction, which contains the herb Herba epimedium as a key ingredient, had no overt adverse effects on the uterus that resembled those of estrogen. 39 Through tissue-specific estrogen-like effects without any uterotrophic effects, administration of ICA reduced bone loss in long bones and the lumbar spine, 36 indicating that ICA might not promote uterine-related disorders such as endometrial cancer. Rats given ICA orally at doses up to 120 mg/kg showed minimal toxicity but no overt damage. 16 According to a randomized, double-blind, placebo-controlled clinical trial involving 58 postmenopausal women, oral administration of a purified extract of Epimedium spp. containing 6.9% ICA and 48.2% prenylflavonoids for 6 weeks did not result in any overt adverse events, and the influence of ICA on hematological, hepatic, and renal parameters was rated as CTCAE Grade 1 (asymptomatic; clinical or diagnostic observations only; intervention not indicated). 40 Even very high doses of ICA are still highly tolerated although gastrointestinal symptoms may occur at 1680 mg. 41 At the cellular level, ICA equal to or less than 10−5M is unlikely to possess numerous toxicity. 42 In general, administration of ICA is safe. Besides, we can expect a systematic review and meta-analysis of ICA safety data registered in INPLASY. 43
Preclinical Studies of Icariin
Numerous studies have shown that ICA is capable of improving the bone loss and microarchitectural deterioration of osteoporosis, and we summarized the preclinical studies of ICA for osteoporosis (Table 1).
Preclinical Studies of Icariin Against Osteoporosis.

The authors did not state the specific number of rats included in the study and only indicated that 12 rats were selected randomly from all rats to be measured by DXA.
We are not clear about the specific style of injection as the authors of this mentioned article did not state it.
Mechanical characteristics of femur include peak load, ultimate stiffness, ultimate strength, and Young's modulus.
Biomechanical parameters include three-point bending tibia, compression of vertebra, load testing of femoral head.
Mechanisms of Icariin in Anti-Osteoporosis
Icariin Promotes Osteogenesis
Following childbirth, bone undergoes a dynamic remodeling process that involves both bone resorption by osteoclasts and bone formation by osteoblasts. The synthesis and mineralization of bone organic matrix, such as collagen, osteopontin, and osteocalcin, is carried out by osteoblasts, which are mesenchymal stem cells’ offspring. Bone loss is thought to be primarily caused by osteoblast dysfunction. 63 Numerous in vitro and in vivo investigations suggested that ICA can promote osteogenesis by specifically targeting osteoblasts. By measuring bone mineral density, a meta-analysis of seven fully randomized studies revealed that ICA considerably increased bone mass, and no discernible difference was identified between ICA and estrogen. 64
By increasing the expression of the genes Runt-related transcription factor 2 (RUNX2) and Bone Morphogenetic Protein 2, ICA increases the formation of extracellular matrix and mineralization in osteoblasts (BMP2). Furthermore, ICI182780, a high-affinity estrogen receptor (ER) antagonist, prevented these effects, indicating that ICA may act via the ER pathways. 65 Bone adaptation is sort of dependent on the mechanical environment and exercise has a beneficial impact on osteogenesis. Exercise-induced skeletal adaptation to ICA therapy was enhanced in part by the ER/AKT/β-catenin pathway. 66 ICA promotes bone formation in osteoblasts in rats by activating the cAMP pathway, which is localized to osteoblast primary cilia, as demonstrated by siRNA suppression of a protein involved in cilium assembly. 54 This research strengthened the link between cAMP-mediated signaling and osteogenesis and provided fresh insights into the mechanism and function of ICA. When ICA regulates osteogenic development and mineralization, PKA and CREB are found downstream of cAMP. The cAMP/PKA/CREB pathway was discovered to be time-dependently activated by ICA therapy, which was reported to induce BMP2-mediated osteogenesis. 67 In hFOB 1.19 cells, an in vitro experiment showed that ICA increased the number of calcified nodules, cell viability, and ALP activity. It also upregulated the expression of Smad4, BMP-2, OPG, RANKL, Cbfa1/RUNX2, and the OPG/RANKL ratio. The results suggested that the BMP-2/Smad4 pathway is involved in the process of regulating bone formation by ICA in hFOB 1.19 cells. 68 By boosting the expression of DEC1, a bone enhancer implicated in the PI3K/Akt/GSK3/β-catenin integrated pathway, ICA increased osteogenic activity. 59 The expression of PEDF, PDIA3, NPAT, and HSP70 is regulated by ICA therapy through pathways like Fas and p53, which results in increased osteogenesis, according to bioinformatics and western blot analysis. 69 Furthermore, ICA at a dose of 5 mg/kg per day significantly corrected OPG-deficient-induced bone loss and bone strength reduction. 70 Results from RT-PCR demonstrated that ICA clearly increased the expression of BMP2, BMP4, Runx2, OC, Wnt1, and Wnt3a in OPG KO mice. 70 It's interesting to note that ICA may promote osteogenesis by suppressing the adipocytic transdifferentiation of osteoblasts by downregulating the expression of peroxisome proliferator-activated receptor gamma (PPARγ) and CCAAT/enhancer binding protein alpha (C/EBPα) through interaction with ERs. 65
Icariin Suppresses Osteoclastogenesis and Bone Resorption
Osteoclasts are multinucleated cells produced from the monocyte/macrophage lineage that are stimulated by two main factors: monocyte/macrophage colony-stimulating factor (M-CSF) and receptor activation of NF-B ligand (RANKL). M-CSF extends survival and encourages proliferative growth. Osteoblasts and stromal cells of the bone marrow are the main surface cells that express RANKL. The commitment of monocyte/macrophage cells to the osteoclast lineage and the activation of mature osteoblasts begin after the binding of RANKL and RANK, a transmembrane molecule on the surface of osteoclasts. 71 Proteolytic enzymes and hydrochloric acid are secreted by osteoclasts during the bone resorption, mineral dissolution, and bone degradation processes. Previous research has demonstrated that Herba epimedium flavonoids (HEF), which contain ICA, can decrease osteoclast proliferation and differentiation, induce apoptosis and cell cycle arrest, and inhibit bone resorption in order to modify the development and activity of osteoclasts. 72 To prevent osteopenia, ICA reduced the disordered metabolism of bone minerals and improved the stability of bone apatite. 52 Additionally, ICA reduced mature osteoclast function in a concentration-dependent manner, in part by preventing the development of the crucial F-actin ring for osteoclast function. 73 The results of an in vitro investigation utilizing osteoclast precursors, which showed a reduction in the number of TRAP-positive multinucleated cells after ICA treatment, validated the effects on suppressing osteoclastogenesis.36,73
By interacting with its cognate receptor RANK, RANKL controls the formation and activity of osteoclasts, and OPG blocks RANKL's effects by out-competing RANK. 71 As a result, OPG/RANKL ratios mean a lot in osteoclast differentiation regulation. Osteoclast differentiation is suppressed and vice versa when OPG/RANKL ratios are upregulated. OPG/RANKL ratios, which are lower in osteoporosis rats and are reversed by ICA treatment, have been shown to govern the differentiation of osteoclasts. 52 The TRAF6 signaling cascade, which is a crucial signaling adaptor molecule found in the progenitors of osteoclasts, also plays a significant role during the interaction of RANK and RANKL. 74 ICA downregulated RANKL-simulated TRAF6 expression and inhibited ERK and then decreased the expression of c-Fos and NFATc1, resulting in osteoclastogenesis and bone resorption quelled. 75 ICA was reported to prevent RANKL-induced osteoclastogenesis by suppressing nuclear factor-κB (NF-κB) and mitogen-activated protein kinase (MAPK). 73 What's more, the Wnt/β-catenin pathway is also implicated in how ICA prevents osteoclastogenesis. The Runx2 gene is activated by ICA through an upregulation of the Wnt/β-catenin pathway, preventing osteoclast differentiation. A treatment with ICA significantly raised the levels of Lrp6, β-catenin, and Runx2 expression while downregulating the levels of GSK-3 expression, according to Western blot and RT-qPCR results. GSK-3 phosphorylation is increased and β-catenin phosphorylation is inhibited when Wnt receptors Lrp5/6 are active. The Wnt/β-catenin pathway is then activated when the accumulating β-catenin is transported to the nucleus. As a result, Runx2 is turned on, enhancing osteoblast differentiation and proliferation while suppressing osteoclast differentiation and proliferation. 76 Most notably, some researchers contend that blocking bone resorption may be the primary reason for ICA to generate bone-protective benefits, 77 while most researchers believe that ICA can be exploited for an anabolic agent.
Glucocorticoid-induced osteoporosis (GIOP) is a prevalent type of secondary osteoporosis brought on by prolonged systemic glucocorticoid use. In addition to increasing morbidity and mortality, glucocorticoids also impair bone production, speed up bone loss, and promote bone resorption. 78 The previous work demonstrated that ICA preserved osteocyte viability to control bone remodeling and offered preliminary evidence that ICA could treat osteoporosis brought on by glucocorticoids. 23 But more recently, it was discovered that administration of ICA treated the imbalance in calcium homeostasis and bone resorption brought on by dexamethasone (DXM), increased serum alkaline phosphatase, tartrate-resistant acid phosphatase, osteocalcin, and deoxypyridinoline, suppressed the expression of cathepsin K, and prevented the downregulation of microRNA (miR)-186 brought on by DXM. Additionally, miR-186 inhibits osteoclasts’ production of cathepsin K, a key collagenase involved in the destruction of bone matrix. Together, ICA and GIOP prevent bone loss through cathepsin K inhibition and miR-186 activation. 53
Icariin Regulates Migration, Proliferation, and Differentiation of Mesenchymal Stem Cells
From a variety of tissues, including bone marrow, adipose tissue, umbilical cord, peripheral blood, and placenta, mesenchymal stem cells (MSCs) can be isolated.79‐83 MSCs are thought of as multi-lineage differentiation potential cells with the ability to differentiate into a variety of cells, such as adipocytes, osteoblasts, and chondrocytes, among others. Cytokines such as peroxisome proliferator-activated receptor gamma (PPARγ), CCAAT/enhancer binding protein alpha (C/EBPα), Runt-related transcription factor 2 (Runx2), and Osterix influence the way MSCs differentiate into osteoblasts or adipocytes. The promotion of adipogenic differentiation is carried out by PPARγ and C/EBPα, whereas the promotion of osteogenic differentiation is carried out by Runx2 and Osterix. 84 Numerous disorders stem from MSCs’ disordered differentiation, and a lack of osteoblastogenesis or an excess of adipogenesis from MSCs results in a number of bone ailments like osteoporosis. 85 These suggest that MSCs are a potentially effective osteoporosis treatment. Naturally, it is also the focus of contemporary study. The following are three ways that ICA can act as a regulator for mesenchymal stem cells throughout their migration, proliferation, and differentiation (Figure 4).

Schematic presentation of the mechanisms of icariin regulating migration, proliferation, and differentiation of mesenchymal stem cells (MSCs). Created with BioRender.com.
MSCs initially migrate to the surface of the bone, then take part in bone formation to become osteogenic cells. Effective homing makes it possible for stem cell therapy to deliver desired capabilities. 86 Similar to this, encouraging MSC migration is advantageous for using stem cells to treat osteoporosis. While studies on the processes by which ICA enhances MSCs migration are scarce, ICA showed a good result in encouraging MSCs migration. Nevertheless, some progress has been made. An ideal concentration of 1 mol/L ICA increased MSCs’ ability to migrate and encouraged the development of actin stress fibers, which is necessary for whole-cell migration. Additionally, ICA-induced MSC migration involves MAPK signaling. 87 Stem cell homing depends on the stromal cell-derived factor-1α (SDF-1α)/C-X-C chemokine receptor type 4 (CXCR4) axis. ICA improved the chemotactic capacity of SDF-1α, which consequently boosted MSCs’ migration. In addition, ICA increased the amount of HIF-1α in MSCs, which is expected to activate the CXCR4 luciferase promoter. Additional research revealed that HIF-1α encouraged MSCs to migrate. Therefore, the SDF-1α/HIF-1α/CXCR4 pathway was a consistent promoter of MSCs migration when using ICA. 88
Classic ERs include ERα and ERβ which all matter in regulating osteogenic and adipogenic differentiation. ICA activated the ER signaling pathway to promote osteoblast differentiation and suppressed adipogenic differentiation of MSCs by greatly upregulating the expression of both ERα and ERβ. 89 Exercise enhanced this process via upregulating the expression of ERα and then strengthening the Akt/β-catenin pathway. 66 Specific MAPK inhibitors, including the JNK inhibitor SP600125 and the ERK inhibitor U0126, reduced the effects of ICA on osteoblast differentiation. ICI182780, an ER antagonist, decreased ICA-mediated effects and prevented ERK and JNK phosphorylation, indicating that ICA's activation of the ER signaling pathway also activates the ERK and JNK pathways. 90
STAT3 is a member of the signal transducer and activator of transcription (STAT) protein family, involved in several cellular processes like cell growth and differentiation. 91 STAT3 gene dominant-negative mutations is related to osteoporosis and pathologic fractures. ICA treatment increased the levels of STAT3 phosphorylation, and restraining STAT3 activation blocked ICA-induced osteoblast differentiation, indicating that STAT3 is incorporated in ICA-induced osteoblast differentiation. 47
Transmembrane receptors like Notch are important for cell differentiation and function. Four Notch receptors (Notch-1, −2, −3, and −4), five classical ligands (Jagged (Jag) 1, Jag2, Delta-like (Dll) 1, Dll3, and Dll4), and two non-classical ligands (delta-like homolog (Dlk) 1 and Dlk2) make up the Notch signaling system. The hairy and enhancer of split (HES) and HES with a YRPW motif (HEY) families of target genes are examples of those whose transcription is aided by the activation of the classical Notch signaling pathway. Notch activation prevents cell maturation during the early stages of osteoblast development, and it also prevents further cell differentiation and increases the number of defective osteoblasts.92‐96 The expression of Notch3, Jag1, Heys, and other Notch signaling molecules was downregulated after treatment with ICA, which also improved osteogenic differentiation. In line with this, Notch signaling activation downregulated the mRNA expression of osteogenic markers such as osteocalcin, ALP, collagen-1α1, and bone sialoprotein (BSP), as well as the protein expression of Runx2 and Osx and the osteogenic differentiation produced by ICA. 97
Previous studies reported that RhoA and TAZ activation causes MSCs to differentiate into osteoblasts, but the precise link between RhoA and TAZ was unknown.98,99 The RhoA-TAZ signaling pathway is involved in the process of ICA boosting proliferation and osteogenic differentiation of rat adipose-derived stem cells with the optimal concentration of 10−7 M, as shown in a study by blocking RhoA with C3 (RhoA inhibitor) and transfecting TAZ with siRNA. 100
Through the canonical Wnt signaling pathway, ICA enhanced osteoblast differentiation and inhibited adipocyte differentiation of rats’ bone marrow mesenchymal stem cells (BMSCs). 101 It's interesting to note that ICA controlled the Wnt signaling pathway to control the bidirectional differentiation of human bone marrow mesenchymal stem cells (hBMSCs). 102 In addition, more research confirmed the role of sclerostin in the Wnt/β-catenin system, indicating that ICA promoted BMSC osteogenic differentiation via upregulating the Wnt/β-catenin pathway by blocking sclerostin's activity. 103 BMPs are participated in the downstream of canonical Wnt signaling pathway to boost osteogenic differentiation. 70 BMP2 is frequently used in clinical settings to enhance bone growth because it plays a crucial part in causing osteoblast differentiation. Due to its low cost and ability to activate the cAMP/PKA/CREB pathway and increase BMP-2 protein production, ICA may provide a promising alternative for the treatment of osteopenia. 67 A study used senescence-accelerated mouse SAMP6 to further elucidate the underlying mechanism of ICA enhancing BMP-2 induced differentiation. Connective tissue growth factor (CTGF) was also found to be involved, they discovered. Osteoblast differentiation brought on by BMP-2 is negatively regulated by CTGF. Phosphate-Smad1/5/8 levels were raised by ICA via suppressing CTGF expression.104,105 To our excitement, besides, microRNA also plays a crucial role in mediating the promotion of ICA to osteoblast differentiation. 106 An in vitro investigation shown that ICA had a potent influence on osteoblast development in MC3T3-E1 cells via the miR-153/Runx2 pathway. 107 Even though MC3T3-E1 cells are not MSCs, this result gave rise to the possibility that ICA might improve MSCs’ ability to differentiate into osteoblasts through a microRNA-mediated mechanism. It should be examined whether ICA might control MSCs’ differentiation into osteoblasts through microRNA-mediated pathways and whether microRNAs are involved in the process.
Autophagy is crucial to the development of bone MSCs and is linked to the beginning of osteoporosis. In general, a decline in autophagy appears to be a factor in osteoporosis and bone loss. 108 Intriguingly, ICA increased the BMSCs’ levels of autophagy in vivo, and in vitro, ICA-induced osteogenic differentiation was blocked by chloroquine (an autophagy inhibitor), suggesting that ICA increases the BMSCs’ osteogenic differentiation through increasing autophagic activity. 49
Icariin Enhances Angiogenesis
For bone formation, angiogenesis and vascularization are essential processes. It has been discovered that signaling molecules that promote blood vessel growth and the pro-angiogenic process improve the osseointegration of biomaterials and promote bone formation. 109 The presence of vessels in specific locations mediates the growth of the bone vascular system, generates a unique metabolic and molecular microenvironment, maintains osteoprogenitor cells around the vessels, and combines angiogenesis with osteogenesis, and reversal of these vessels decline by pharmacological treatment is allowed to restore bone mass. 110 Angiogenesis-osteogenesis coupling is a term used to describe the link between the development of bone and the vascularization of ossifying tissue in both space and time. An important trend for the prevention of osteoporosis can be the activation of angiogenesis. 111 Studies conducted both in vivo and in vitro have demonstrated that ICA can encourage angiogenesis. The capacity of bone microvascular endothelial cells to migrate and undergo angioplasty as well as their survival rate were both dramatically improved by ICA. 112 Following ICA therapy, a considerably higher density of newly created blood vessels was found using histomorphometric analysis, and vascular endothelial growth factors were elevated as well. 113 Mechanistically, ICA modulated endothelial cells function by upregulating the eNOS expression via activating the EGF-EGFR signaling pathway. 114 ICA dramatically upregulated the level of VEGF and TGF-β1, angiogenesis-promoting molecules, in secreted extracellular vesicles and promoted the proliferation of microvascular endothelial cells. 115 AKT signaling pathway appears to be involved in the upregulation of angiogenic genes by ICA, including angiotensin 1 and vascular endothelial growth factor. 116 Moreover, ICA promoted angiogenesis via MEK/ERK- and PI3K/Akt/eNOS-dependent pathways.117,118
The Anti-Inflammatory and Antioxidative Effects of Icariin
Pro-inflammatory cytokines also play a role in the way estrogen causes bone loss. Reduced inflammation slows osteoporosis bone loss. 16 Interleukin-1, tumor necrosis factor-, and interleukin-6 are pro-inflammatory cytokines that have been shown in both human and animal trials to activate osteoclasts, which may explain why bone loss at menopause is accelerated by these cytokines.119‐122 By suppressing JNKs, p38, and NK-B signaling pathways and downregulating the expression of miR-34c, ICA reduced bone loss brought on by LPS. 123 According to a study on ICA's ability to prevent cartilage deterioration, it reduces inflammation by blocking the NF-κB/HIF-2α signaling pathway. 124 Additionally, ICA controlled inflammation by signaling through the HO-1/Nrf2 and NF-κB pathways. HO-1 and Nrf2 expression rose with ICA, while NF-κB expression decreased. The antioxidant enzyme HO-1's gene is activated as a result of accumulated Nrf2. Furthermore, COX-2 may also contribute to the anti-inflammatory effects as evidenced by the fact that ICA more potently inhibits COX-2 expression than indomethacin. 125 Meanwhile, ICA was also controlling the cutaneous hypothalamus-pituitary-adrenal axis to reduce inflammation. 126
In mitochondria, ROS are produced, and this ROS production has an effect on harming the cell membrane, cytoplasm, and even DNA. A surplus of ROS encourages osteoclastogenesis and speeds up bone resorption. 127 Numerous studies have demonstrated that ROS are important in osteoporosis and bone loss. Osteoporosis brought on by high ROS can be significantly improved by reducing oxidative stress. 128 More studies have confirmed ICA's capacity to reduce oxidative stress. 129 Therefore, it was thought that ICA prevented bone loss by reducing oxidative stress, however, the molecular processes are yet unknown. Both in vitro and in vivo investigations were carried out to examine the effectiveness of ICA in correcting iron overload-induced bone loss. ICA prevented bone loss and resorption caused by iron excess as well as the differentiation, activity, and differentiation of osteoclasts. 51 However, only a small number of studies have explicitly shown how ICA stops bone loss by reducing oxidative stress. In a study on Alzheimer's disease, it was discovered that ICA treatment decreased iron levels and lipid peroxidation while increasing the activity of glutathione peroxidase and superoxide dismutase. 130 Aβ25-35 increased the levels of both intracellular and mitochondrial ROS in PC12 cells, while Icariside II, a metabolite of ICA, reduced this. 131 There should be more research showing how ICA directly stops bone loss by reducing oxidative stress and examining the underlying mechanisms.
Prospects and Challenges
In this review, we have outlined the updated data on ICA's anti-osteoporosis effects over the last 10 years. It has been shown that the effectiveness of bone protection induced by ICA is mediated through tremendous acknowledged pathways, making it a potential candidate for osteoporosis. Pharmacological research was conducted to look into the pharmacological properties of ICA in order to increase its effectiveness in combating osteoporosis. Intestinal hydrolyzed metabolites such as icaritin, baohuoside I, and icariside I were discovered to be better absorbed and to have an improved anti-osteoporosis activity. Additionally, enteric-coated capsules containing both snailase and ICA were used to boost intestine hydrolysis and ICA absorption. 132 The first-pass effect after oral administration, lack of targeting ability, poor water solubility, and low bioavailability of ICA were all considered drawbacks. A targeting drug delivery system of biomineral-binding liposomes loaded with ICA was developed to address these issues and provide greater therapeutic effects than ICA itself. 50 Calcium phosphate cement scaffold, another ICA delivery device, was created and then placed to a calvarial defect to show how the combination of a local sustained-release system with systemic administration could increase the efficacy of osteogenesis and angiogenesis. 55
Numerous studies have demonstrated the effectiveness of ICA in combating osteoporosis both in vitro and in vivo, yet some researchers have expressed different views. A comparison research revealed that icariside II, an ICA metabolite, had greater osteogenic potential than ICA. 133 Another comparison study found that human embryonic stem cell-derived mesenchymal progenitor cells were little affected by ICA, and they stressed that this may be because ICA cannot be digested in a single cell culture medium. 134 On the other hand, recent research has focused on the cellular or molecular level. Further research is desperately needed in this regard to assess its clinical efficacy against osteoporosis. Additionally, more research stressing the negative impacts of ICA on humans needs to be done.
MSCs, which are readily available, immunosuppressive, multipotent stem cells with the capacity to develop into a range of cells, are the focus of osteoporosis research. It is undeniable that ICA can help MSCs differentiate into osteoblasts. The issue is that its metabolites, not ICA, may be the active form. Given that ICA can be applied locally and escape from intestinal microbiome metabolism and first-pass effects, it is necessary to clarify the actual or more effective active form of ICA.
Conclusion
ICA has the potential to be developed as a contender for the treatment of osteoporosis due to its osteogenic activity. There are obstacles in this process, though. Studies on the function and mechanism of ICA in reducing bone loss and the development of pharmacological techniques to improve its absorption and safety are still needed. However, a clinical trial needs to be scheduled as soon as possible.
Footnotes
Acknowledgments
The authors sincerely appreciate the Key Research and Development Program of Luzhou (2020-SYF-31); Natural Science Foundation of Hospital (T.C.M) Affiliated to Southwest Medical University (2021ZKMS050); The Union Project of Luzhou City and Southwest Medical University under Grants (2021); University-level Fund Innovation Team Project of Southwest Medical University in 2017 (Bone Pathology Innovation Team of Integrated Traditional Chinese and Western Medicine to SL); Southwest Medical University High-Level Talents (Zhong Shizhen Team) Special Support Program for their support.
Availability of Data and Materials
The datasets generated during and/or analyzed during the current study are available in the [Pubmed] repository, [https://pubmed.ncbi.nlm.nih.gov/]; [Web of Science] repository, [![]() ].
].
Authors’ Contributions
All authors contributed to the study conception and design. Long L, Wang X, and Lei Y designed the present manuscript. Long L completed the manuscript. Guo S, Wang C, Dai W, Lin B, and Xie M performed a literature search and selected this study to be performed. Long L, Xu H, and Li S revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
