Abstract
Preclinical literature suggests that icariin, a flavonoid found in Epimedium, may have potential for medical and psychiatric conditions. The objective of this study was to examine the safety, tolerability, and pharmacokinetics of orally administered icariin at doses of 100 to 1,680 mg/day in 24 healthy adult participants. Cognition, mood, and side effects were assessed over 5 days. Multiple blood samples were obtained over 24 hours to assess bioavailability and pharmacokinetics. Data were analyzed using a Wilcoxon signed rank test and Mann-Whitney U test. At all doses, either very low or undetectable blood levels of icariin were observed, demonstrating the low bioavailability of the oral formulation and preventing a determination of pharmacokinetic properties. No significant between-group differences were observed on side effect scales, either by self-report, or on cognitive assessments. A statistically significant, but not clinically significant, increase in self-reported depressive symptom severity was observed with icariin relative to placebo. Tolerability of icariin was good except at the highest dose. Two participants receiving 1,680 mg of icariin discontinued the study drug due to gastrointestinal symptoms. Bioavailability of oral icariin appears to be low at all doses tested. Although icariin appears generally to have a favorable tolerability profile, the highest doses may be associated with gastrointestinal distress. Different drug formulation and delivery method may be needed to assess the pharmacokinetic profile of icariin adequately.
Icariin is a flavonoid derived from Epimedium (ie, Horny Goat Weed or Yin Yang Huo). Like sildenafil, icariin is a phosphodiesterase type-5 inhibitor. 1 Additionally, preclinical studies suggest that icariin may have a variety of other important properties and potential in treating illnesses in humans. Icariin appears to regulate osteoblast anabolism and prevents bone loss in animal models. 2 -6 Preclinical data suggest that icariin has a variety of central nervous system (CNS) effects. Prior animal studies demonstrated that icariin blocked glucocorticoid receptor downregulation in a mouse model of social defeat 7 and had antidepressant properties in an animal model of depression and antidepressant response. 8 Icariin decreased β-amyloid deposition, 9,10 reduced infarct size and edema following stroke, 11 and improved memory following hypoperfusion 7 as well as in an animal model of Alzheimer’s disease. 12 Animal studies with icariin demonstrated that it is poorly absorbed orally, eliminated quickly, and there is a variety of metabolites. 13 Studies of icariin metabolism by human intestinal microflora also demonstrate a complex pattern of metabolism. 14 In fact, these microflora studies indicate that icariin may be rapidly metabolized in the gut to icariside II prior to absorption; therefore, when studying orally administered icariin, the circulating plasma levels of the compound may be quite low.
Human data on icariin are limited; to our knowledge, only 1 placebo-controlled trial has been reported. A randomized, double-blind, placebo-controlled trial of Epimedium-derived flavonoids that included icariin (60 mg/day) in 100 postmenopausal women found significantly greater bone mineral density in the icariin group, compared with the placebo group, after 24 months. 15 The authors did not report safety and tolerability data. Other reports in humans suggest improvement in immune functioning and a reduction in lipid levels in patients with end-stage renal disease given Epimedium. 16 -19 An open-label study of icariin (300 mg/day) demonstrated a decrease in depression and anxiety scores in psychiatric patients with bipolar disorder and alcohol use following 8 weeks of administration. 20 The pharmacokinetics of icariin in healthy volunteers following oral administration was studied using a 50% extract of Epimedium. In this study, a maximal plasma concentration (C max) of 7.89 ng/mL was observed following an 80 mg dose of icariin. 21
Given the potential of icariin in the treatment of a variety of diseases and the limited available human data, its tolerability, safety, bioavailability, and pharmacokinetics were assessed in the current study using a commercially available icariin preparation. The primary aim of the study was to determine the pharmacokinetic profile of oral icariin in humans using serial blood draws over 24 hours following a single dose of the drug. The secondary aims focused on the effect of icariin on cognition, mood, and associated side effects, as measured by the validated research assessments. Demographic characteristics of participants randomized to icariin vs. placebo groups are provided in Table 1. At baseline, icariin and placebo groups did not differ significantly in sex and ethnicity; however, the participants in the icariin group were significantly younger than those in the placebo group, t(22)=2.14, P = 0.044.
Demographic and Baseline Characteristics by Treatment Group.
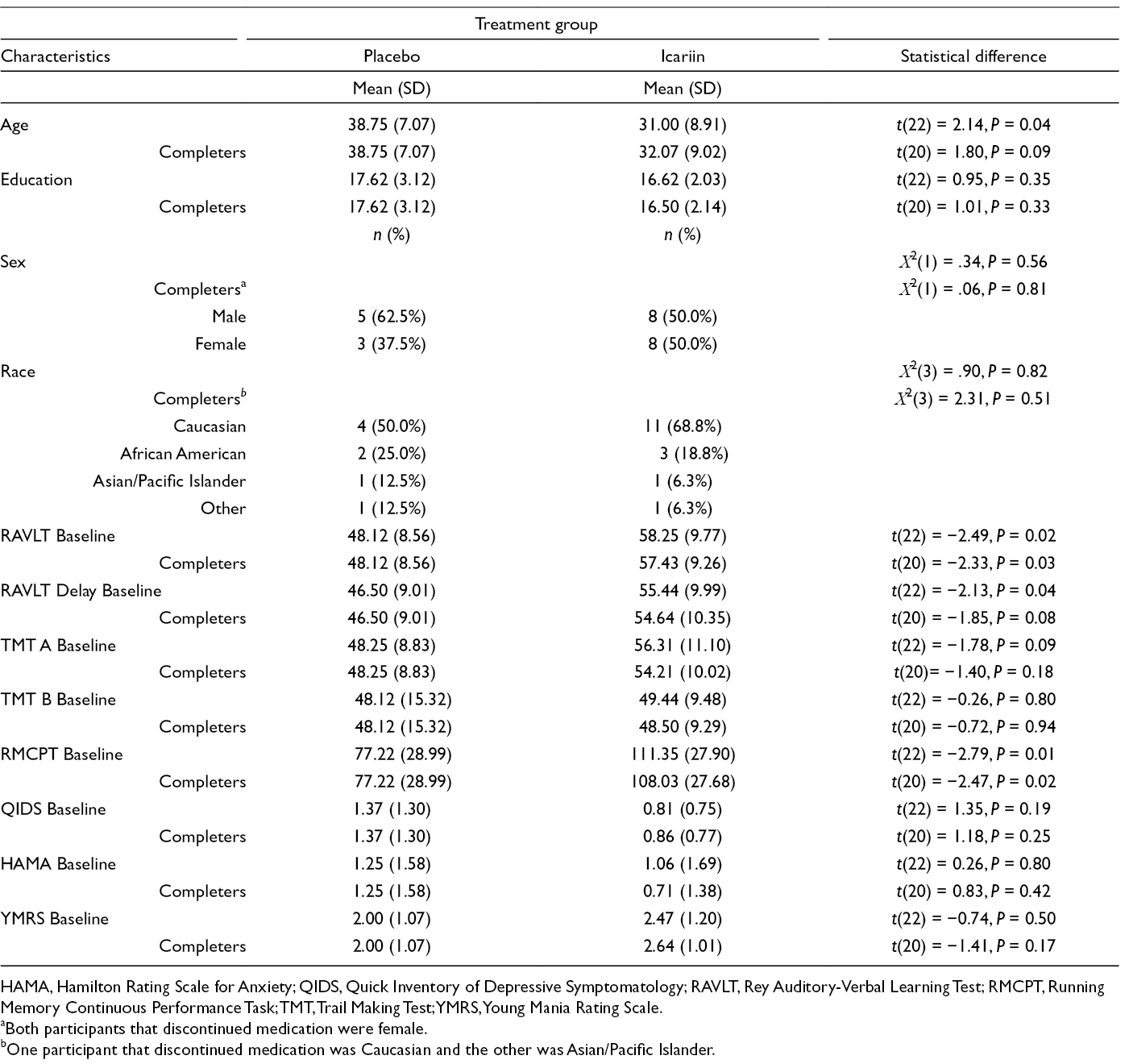
HAMA, Hamilton Rating Scale for Anxiety; QIDS, Quick Inventory of Depressive Symptomatology; RAVLT, Rey Auditory-Verbal Learning Test; RMCPT, Running Memory Continuous Performance Task; TMT, Trail Making Test; YMRS, Young Mania Rating Scale.
aBoth participants that discontinued medication were female.
bOne participant that discontinued medication was Caucasian and the other was Asian/Pacific Islander.
Few side effects were observed in the icariin group except at the highest dose (1,680 mg). There were no statistically significant differences between placebo and icariin groups on either Systematic Assessment for Treatment Emergent Effects (SAFTEE) (P = 0.184) or Arizona Sexual Experience Scale (A-SEX) (P = 0.663) side effect scales. There were two potentially icariin-related discontinuations during the study, with both participants reporting gastrointestinal (GI) distress on the SAFTEE scale 12 hours after receiving their first dose of 1,680 mg/day of icariin. Both participants discontinued study medication after day 2 of the study. In general, GI discomfort was the most frequently reported side effect (21.4%) in the study, followed by drowsiness (17.8%), appetite changes (10.7%), and muscle twitching (10.7%).
There were no significant differences between treatment groups on cognitive change (Supplemental Table S1), including Rey Auditory-Verbal Learning Test (RAVLT), Trail Making Test (TMT), and Running Memory Continuous Performance Task (RMCPT) scores. One significant between-group difference on change in mood scores was observed in the total sample and in study completers. Based on the Mann-Whitney U test, the placebo group showed a decrease in Quick Inventory of Depressive Symptomatology Clinician-Rated (QIDS-C) change score, while the icariin group showed a slight increase with a significant between-group difference (P = 0.012, without correction for the 8 comparisons). While statistically significant, this finding may, however, not be clinically significant, because the highest QIDS-C score observed was 5, which is within the normal range. When looking only at study completers, the effect held (P = 0.017; Supplemental Table S2).
The primary aim of this pilot study was to determine the pharmacokinetic properties of orally administered icariin in healthy adults. To our knowledge, this is the first study to examine both icariin plasma levels and brain effects. However, the analysis of plasma samples revealed low bioavailability of icariin, such that the pharmacokinetic profile of the drug could not be determined. Low observed bioavailability of icariin is consistent with limited changes on mood and cognitive assessments. However, the lack of changes may also be attributable to a lack of brain effects with icariin, or the use of healthy controls, with minimal baseline mood symptoms and normal cognition. Limitations associated with the trial included the small sample size and baseline differences on some measures between groups, as well as a brief observation period, absence of analysis of icariin metabolites, and lack of concentration analysis beyond 1 day. It is possible that longer trial duration is needed in order to observe significant changes on the assessments.
Icariin appeared to be well tolerated, except at the highest dose tested. However, poor bioavailability precluded a determination of the pharmacokinetic properties. It cannot be completely ruled out that the poor bioavailability of icariin may be associated with the specific formulation of the product. Other methods of product delivery, such as intravenous formulations, may need to be explored in future studies.
Experimental
Trial Description
This pilot study was a randomized, double-blind, placebo-controlled, parallel-group clinical trial (NCT02112123) to evaluate the pharmacokinetics and safety of icariin, an over-the-counter supplement, in healthy individuals (N = 24) at UT Southwestern Medical Center, Dallas, TX. The trial was conducted between 7/2/2014 and 9/6/2016, with all participants recruited from the community using free and paid advertising. The study protocol was reviewed and approved by the UT Southwestern Institutional Review Board, and all participants signed an informed consent form undergoing any study procedures.
Sample Size and Randomization
The study used a sample size of 24 (N = 24), a group of 6 participants per icariin dose. The Wilcoxon-Mann-Whitney power analysis was conducted using median difference between icariin and placebo groups with Poisson distribution (mean of 3) of scores of cognition, mood, and side effects. A power of 76.2% was obtained from the proposed sample size of 24 (2:1 ratio icariin and placebo groups) at α = 0.05. Randomization was performed by a statistician using a computer-generated random number sequence, with participants randomized in a 2:1 icariin-to-placebo ratio in each dose group of 6 participants.
Inclusion and Exclusion Criteria
The included participants were healthy men and women (18 - 50 years old) with ≥12 years of education (or equivalent), a body mass index between 18.5 and 30, no medication changes within 30 days of baseline, and baseline RAVLT T-score ≥35 on trials A1 to A5 indicating normal baseline memory performance. The exclusion criteria were as follows: history of any major psychiatric illness, as assessed by Structured Clinical Interview for DSM (SCID-IV); current tobacco use; history of neurological disorders (eg, recurrent seizures or any seizure in the past 12 months, stroke, brain surgery, traumatic brain injury, and loss of consciousness >1 minute, multiple sclerosis, Parkinson’s disease); use of CNS-acting medications; hypertension (ie, systolic blood pressure >140 or diastolic blood pressure >90); medication therapy for erectile dysfunction or opioid therapy within 30 days of the baseline or initiation of new medications within 30 days of baseline; clinically significant vital signs values (body temperature ≥100.5°F [38°C], baseline heart rate <50 bpm or >100 bpm); clinically significant laboratory values (eg, hypokalemia, hypernatremia) or ECG results at baseline; medical contraindications to icariin; history of myocardial infarction, cancer, or diabetes; vulnerable populations (ie, pregnant or nursing women, incarcerated, those with severe cognitive disorders); baseline QIDS-C score >7; history of suicide attempt or current suicidal ideation; history of special education or intellectual disability; current participation in any other pharmacotherapy studies.
Intervention Description
Following the baseline visit, participants completed a 24-hour stay at the UT Southwestern Clinical Research Unit (CRU, an inpatient research facility), where the study intervention was initiated and blood samples drawn. Five dose levels were evaluated (100, 200, 400, 840, and 1,680 mg or placebo) over a 5-day administration period. The administration was in blocks of 6 participants who received either icariin or identical placebo and only moved to a higher dose if the previous dose was well tolerated. For example, if 100 mg was well tolerated, based on side effect and adverse event assessment in 6 participants, then the next 6 participants received 200 mg doses. Different healthy controls were used for each dose level, with the total treatment duration for each participant lasting 5 days. The first dose was administered orally by a nurse at the UT Southwestern CRU on an empty stomach (i.e. not within 1 hour prior or within 2 hours following meals). The second dose was also administered by a nurse following the last blood drawn at 24 hours. The participants were supplied with icariin doses for days 3–5 at the time of discharge.
Icariin, as a 20% icariin concentration in a 500 mg Horny Goat Weed capsule (Barlowe’s Herbal Elixirs, West Palm Beach, FL), was used in the study (Lot #C20121115). This product was selected because it was commercially available and Barlowe’s Herbal Elixirs provided a Certificate of Analysis (CoA). The CoA was a document issued by the manufacturer that confirmed that the product met product specifications, and included results from quality control testing of an individual batch of icariin. The preparation was changed to 600 mg (Lot #CYYH-A-01948) during the last 2 dose phases of the study due to commercial availability of the product; however, the dosing was adjusted to deliver the correct amount of icariin. Each of the Barlowe’s Herbal Elixirs icariin capsules had a label claim of 100 mg of icariin, and this concentration was confirmed through liquid chromatography mass spectrometry (LC-MS)/MS determination. The matching placebo, identical in appearance to the active drug, was prepared at Abrams Royal Pharmacy (Dallas, TX), and was encapsulated in clear and tasteless gelatin capsules. The placebo capsules contained the following ingredients: food color powder blue, food color powder yellow, food color powder brown, microcrystalline cellulose, silicon dioxide, and calcium carbonate. Both participants and research personnel remained blinded to the treatment assignments.
Pharmacokinetic Profile Determination
Icariin (98+%), purchased from INDOFINE Chemical Company (Hillsborough, NJ), was used as the analytical standard as received. Biochanin A, purchased from Sigma-Aldrich (St Louis, MO), was used as the internal standard and used as received. All other reagents were purchased and used as received from Fisher Scientific (Hampton, NH).
Bioanalytical Sample Preparation
The bioavailability and pharmacokinetics of orally administered icariin were determined by plasma analysis following the initial oral dose of icariin. After dosing, 15 blood samples (approximately 2 mL each) were obtained over 24 hours at 0, 30, 60, 90, 120, 150, 180, 210, 240, 300, 360, 420, 480, 1080 and 1440 minutes. These samples were turned into plasma and stored at −80 ± 10°C until analysis.
Bioanalytical Method
An LC-MS/MS method for the determination of icariin in human plasma was developed and qualified with a lower limit of quantitation of 1 ng/mL. The plasma from subjects receiving doses of 100 mg (n = 6), 200 mg (n = 5), 400 mg (n = 6), 840 mg (n = 3), and 1,680 mg (n = 3) was analyzed. Essentially all plasma samples contained levels of icariin below the lower limit of quantitation with the exception of a few samples.
Specifically:
One subject in the 100 mg dose group had a plasma concentration of 2.8 ng/mL at the 1 hour time point
One subject in the 200 mg dose group had consistent blood levels between 1 and 2 ng/mL at all post dose time points
One subject in the 1,680 mg dose group had plasma concentrations of 2.1 and 1.6 ng/mL at the 6 and 8 hours time points, respectively.
LC-MS/MS analysis of the capsules demonstrated that the capsules contained 82% of the label claim; therefore, the lack of bioavailability was not due to improperly formulated capsules. The bioanalytical method was qualified through analysis of 3 calibration curves prepared on 3 separate days. The calibration curve was determined to be linear (r > 0.99) over the concentration range of 1.0 to 1000 ng/mL on all 3 days. Accuracy and precision were assessed by determination of the percent relative error (accuracy) and percent standard deviation (precision) and the results of this determination are presented in Supplemental Table S2. The accuracy varied between 93.9% and 114%. The precision varied between 2.87% and 10.2%.
Bioanalytical Conditions
The icariin plasma concentrations were determined using an in-house developed liquid chromatography tandem mass spectrometry method. Standards and samples (100 µL) were prepared by protein precipitation after addition of the internal standard biochanin A. Chromatographic separation was achieved using gradient elution with a Kinetex XB C18 30 × 2.1 mm, 2.6 µm, column, which was maintained at 60°C, and an injection volume of 1 µL. Mass spectrometric quantitation of icariin and biochanin A was carried out in multiple-reaction monitoring mode with positive polarity, by monitoring the precursor >product transitions of m/z 677.4 > 531.2 for icariin and m/z 284.9 > 152.0 for IS. Unit resolution was used for both precursor and product ions. The analytical data obtained were processed by Analyst software (version 1.6.2). The same methodology was utilized for analysis of the commercially available icariin capsules.
Study Assessments
Various assessments were administered to participants to examine safety, tolerability, and effect of icariin on cognition and mood. The SCID-CV 22 was used to rule out exclusionary psychiatric illness, while mood was assessed using clinician-rated instruments, such as Young Mania Rating Scale, 23 Hamilton Rating Scale for Anxiety, 24 and QIDS-C. 25 Different cognitive domains were evaluated using RAVLT (verbal declarative memory), 26 TMT (executive function, attention, sequencing, visual search, motor function), 27 and the RMCPT (working memory). 28 To monitor icariin safety and tolerability, vital signs were collected at each visit, an ECG was obtained at baseline, 24 hours after the first dose of study medication, and at the final study visit, and a physical examination was performed at baseline and exit. The side effects were monitored using the SAFTEE 29 and A-SEX, 30 as well as via a psychiatric evaluation at each study visit. Data were analyzed using SPSS 24.0, and Wilcoxon signed rank test and Mann-Whitney U-test nonparametric tests were utilized in the analysis.
Supplemental Material
Supplemental Materials - Supplemental material for Human Safety and Pharmacokinetics Study of Orally Administered Icariin: Randomized, Double-Blind, Placebo-Controlled Trial
Supplemental material, Supplemental materials, for Human Safety and Pharmacokinetics Study of Orally Administered Icariin: Randomized, Double-Blind, Placebo-Controlled Trial by E. Sherwood Brown, Collette Bice, William C. Putnam, Richard Leff, Alexandra Kulikova, Alyson Nakamura, Elena I. Ivleva, Erin Van Enkevort, Traci Holmes, and Nyokabi Miingi in Natural Product Communications
Footnotes
Acknowledgments
Dr Brown has research grants from NHLBI, NIAAA, NIA, the Stanley Medical Research Institute, and Otsuka, and serves on an advisory board for Allergan. Dr Ivleva has research grants from NIH. Other authors have no interests to declare.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by the National Center of Complementary and Integrative Health (R21 AT007869-01).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
