Abstract
The solubility of icariin in acetone, acetoacetate, chloroform, and light petroleum in the 283.2‐318.2 K range was measured by ultraviolet-visible spectrophotometry. As the temperature increased, the solubility of icariin in the 4 solvents gradually increased. The solubility data correlated with the modified Apelblat equation. The dissolution enthalpy and entropy of icariin were determined using van’t Hoff plots. The dissolution enthalpy and entropy of icariin in the 4 solvents increases as acetone >acetoacetate >chloroform >light petroleum, which can be explained by the difference of the polarity indices between them. The polarity indices of the solvents affect the solubility behavior.
Introduction
Icariin {3-[(6-deoxy-α-L-mannopyranosyl)oxy]−7-(β-D-glucopyranosyloxy)−5-hydroxy-2-(4-methoxyphenyl)−8-(3-methyl-2-buten-1-yl)−4H-1-benzopyran-4-one; Figure 1}, a natural flavonoid isolated from Epimedium Herba (Chinese herbal name of Yinyanghuo), is considered to be the major bioactive substance of E. Herba, which is widely applied in the treatment for urinary, nervous system, cardiovascular, and endocrine diseases. 1,2

Molecular structure of icariin.
Icariin has been reported to have a broad range of therapeutic applications such as inhibiting osteoclastogenesis, antifibrotic, and anti-inflammatory effects, improving endothelial function and inhibiting atherosclerosis progress. 3 -6 For pharmaceutical use, icariin is mainly extracted from the powdered caudex and frond of Epimedium grandiflorum Morr using solvents such as water, alcohols, or mixtures of these, then purified and crystallized from the solution. Therefore, it is important to have thermodynamic data for the solubility of icariin in different solvents. However, the solubility of icariin in different organic solvents at different temperatures is not complete. 7,8 So, it is necessary to determine systematically the solubility of icariin in other organic solvents.
In this study, the solubility of icariin in acetone, acetoacetate, chloroform, and light petroleum from 283.2 to 318.2 K was measured by ultraviolet-visible spectrophotometry (UV-Vis). By utilizing the van’t Hoff equation, the dissolution enthalpy and entropy of icariin were estimated based on the regression of the solubility data.
Experimental
Materials and Apparatus
The icariin (purity higher than 0.990 in mass fraction) sample was supplied by Shanghai Tauto Biotech Co., Ltd (Shanghai, China). The acetone, acetoacetate, chloroform, and light petroleum (analytical purity grade, boiling point range 60‐90 ℃) were provided by Tianjin Kermel Chemical Reagent Co., Ltd (Tianjin, China). The physical properties of the 4 organic solvents are listed in Table 1. All the solvents were used without further purification.
Purity Levels, Densities (ρ), and Refractive Indexes (n D) of Acetone, Acetoacetate, Chloroform, and Petroleum Ether (T = 293.2 K).
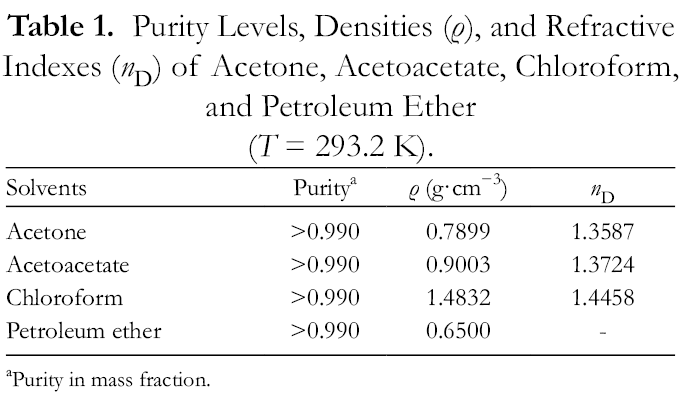
aPurity in mass fraction.
X-ray diffraction (XRD) was performed using an XRD-6100 X-ray diffractometer (SHIMADZU, Japan) with Cu κα radiation.
The absorbance measurements of icariin samples were obtained with an SP-752 UV-Vis spectrophotometer (Shanghai Spectrum Instrument Co., Ltd., Shanghai, China).
Sample Preparation
An excess amount of icariin was added to the solvents (acetone, acetoacetate, chloroform, and light petroleum) in a specially designed, sealed 10-mL dual-wall flask. The flask was kept at a constant temperature by circulating water (±0.1 K) controlled by a thermostat between the outer and inner walls of the flask. The solution in the flask was constantly mixed with a magnetic stirrer for 3 hours. After the equilibrium was reached, the stirring was stopped, and the solution was allowed to stand in a water bath maintained at an appropriate temperature for 2 hours.
An aliquot of each solution was evaporated in vacuo and the obtained residue dissolved in ethanol. The resulting solution was diluted to the appropriate concentration with ethanol, and then the absorbance of the solution measured, and the solubility of icariin in the different solvents calculated. The wavelength used for the measurements in ethanol was 270 nm. All experimental measurements were carried out under atmospheric pressure.
The mole fraction solubility of the solute (x 1) in different pure solvents was calculated by the following equation (1) 7 -9 :

where m 1 (g) represents the mass of the solute (icariin) and m 2 (g) represents the mass of the solvent (acetone, acetoacetate, chloroform, and light petroleum). M 1 (g·mol-1) and M 2 (g·mol-1) are the molar masses of the solute and solvent, respectively.
Sample Analysis
To investigate possible polymorphisms of icariin during the solution process, the XRD spectrum of raw icariin was compared with that of icariin equilibrated with different solvents. In this regard, the icariin equilibrated with different solvents was precipitated, separated from the solvents, and dried at room temperature. Then, tests were carried out using an XRD-6100 X-ray diffractometer with Cu κα radiation. The instrument was operated at 2000 W (X-ray tube), with a fixed tube current of 30 mA and a voltage of 40 kV. Samples were measured by a fixed time step scanning method in the range of 2.5°‐30° (2θ) at 0.02° intervals. Data were analyzed using the software Jade (Version 6.5).
The concentrations of icariin were determined by measuring UV absorbance. We measured the absorbance of the standard solution and the samples of icariin at the maximum absorption wavelength. The wavelength used in the measurements in ethanol was 270 nm. 10,11 The icariin concentrations were calculated from the calibration curve. The calibration curve (Figure 2, A = 0.0395C + 0.0029, R 2 = 0.9992) for estimating icariin concentrations was prepared using standard solutions in the appropriate concentration range (5-20 μg mL-1).

The calibration curve of icariin.
The saturated solubility of icariin (x 1) in the 4 solvents was measured 3 times. The uncertainty of the experimental solubility values x 1 is u r (x 1) = 1.36 × 10-2, resulting from the uncertainties in temperature measurements, dilution of the solution, and instrument error. 12,13
Results and Discussion
The XRD patterns of icariin in each solvent are shown in Figure 3. The results indicated that icariin has polymorphs in different solvents. These problems can be solved in the following ways: evaporate the solvent under vacuum, then dissolve the obtained residue in ethanol. Dilute the solution to the appropriate concentration with ethanol, and then measure the absorbance.

The X-ray diffraction patterns of icariin in different solvents.
Solubility data for icariin in acetone, acetoacetate, chloroform, and light petroleum at different temperatures are presented in Table 2. The temperature dependence of icariin solubility in various solvents can be described by the modified semi-empirical Apelblat equation (2). 14,15
Solubility (x1) of Icariin in Acetone, Acetoacetate, Chloroform, and Petroleum Ether in Different Temperatures.a

aStandard uncertainties u are u (T) = 0.1 K, u r (x 1) = 1.36 × 10-2.
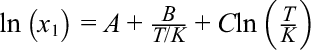
where x 1 is the molar solubility of icariin, T is the absolute temperature, and A, B, and C are the parameters determined by least-squares analysis. Values of the calculated solubility for icariin are also shown in Table 2. Table 3 lists the parameters of A, B, and C, along with the root-mean-square deviation (rmsd):

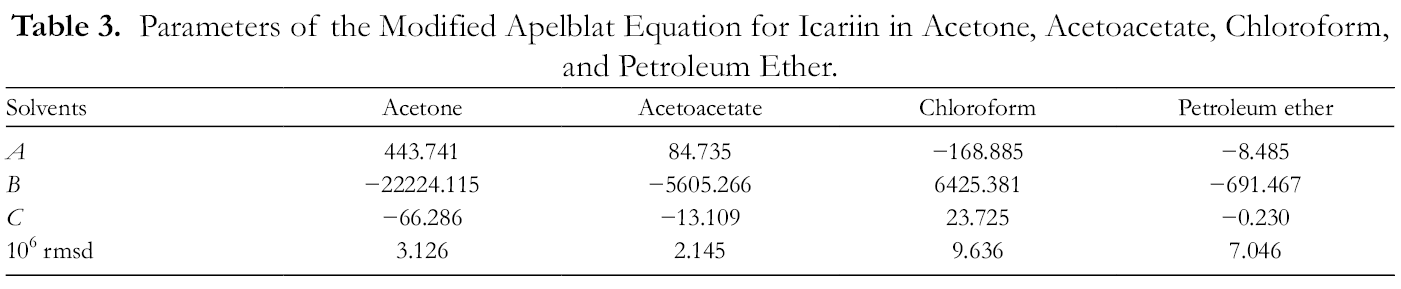
Parameters of the Modified Apelblat Equation for Icariin in Acetone, Acetoacetate, Chloroform, and Petroleum Ether.
where n is the number of experimental points, xi calc represents the calculated solubility, and xi represents the experimental solubility values. The solubility data of icariin in acetone, acetoacetate, chloroform, and light petroleum at different temperatures are plotted in Figure 4. The van’t Hoff plots obtained from the linear fit of ln x 1 versus 1/T are shown in Figure 5.

Solubility of icariin in acetone, acetoacetate, chloroform, and light petroleum. (A) Solubility of icariin in acetone and acetoacetate. (B) Solubility of icariin in chloroform and light petroleum.  , acetone (experimental values),
, acetone (experimental values),  , acetone (calculated values).
, acetone (calculated values).  , acetoacetate (experimental values),
, acetoacetate (experimental values),  , acetoacetate (calculated values).
, acetoacetate (calculated values).  , light petroleum (experimental values),
, light petroleum (experimental values),  .
.  , light petroleum (calculated values)
, light petroleum (calculated values)  , chloroform (experimental values), chloroform (calculated values).
, chloroform (experimental values), chloroform (calculated values).

The van’t Hoff plots of ln x 1 versus 1/T in different solvents. ◆, acetone; ■, acetoacetate; □, chloroform; △, light petroleum.
It is indicated that the solubility of icariin in acetone, acetoacetate, chloroform, and light petroleum increases with increasing temperature based on the experimental data in Table 2. The solubility of icariin in acetone and acetoacetate is significantly higher, compared with that in chloroform and light petroleum, with the solubility in acetone being highest. The calculated solubilities are highly consistent with the experimental data, demonstrating that the solubility data of icariin in varied organic solvents can be correlated with the modified Apelblat equation.
The solubility of icariin in the 4 solvents increases as light petroleum ≈chloroform <acetoacetate <acetone from 283.2 to 318.2 K (Table 2). This phenomenon results from the polarity indices of the solvents, which are the key factors that influence the solubility behavior of icariin. The solvent polarity is the most defining factor playing a role in the material solubility. However, in the case of some solutes, solvent polarity was not the dominant factor for the degree of solubility. The intermolecular interactions between solvent molecules and solute determine the solubility. 16 Different from the other 3 organic solvents, the higher polarity index of acetone induces a higher solubility of icariin. The dielectric constants of the 4 organic solvents were as follows: acetone (20.7) >acetoacetate (6.03) >chloroform (4.7) >light petroleum (1.80). The dipole moments of the 4 organic solvents were as follows: acetone (2.88) >acetoacetate (1.78) >chloroform (1.20) >light petroleum (1.80).
The logarithm 1of the mole fraction of a solute can be related by the van’t Hoff equation as a linear function of the reciprocal of the absolute temperature in the solution shown below 17,18 :

where x 1 is the mole fraction solubility, ΔH d and ΔS d are the dissolution enthalpy and entropy, respectively, T is the absolute temperature, and R is the gas constant.
The van t Hoff plots can be obtained from the linear fit of loge(x 1) versus 1/T in Figure 5. The dissolution enthalpy and entropy of icariin can be calculated from the slope and intersect of these plots in Table 4. The Gibbs energy of the solution can be calculated by the following equation 19,20 :
Dissolution Enthalpy, Entropy of Icariin in Different Solvents and the Gibbs Energy of Solution at 298.2 K.

Abbreviations: ΔG d, Gibbs energy of solution; ΔH d, disslution enthalpy; ΔS d, dissolution entropy.

Table 4 shows that the process of icariin solution in the 4 solvents over the experimental temperature range is endothermic (ΔH d > 0), which explains the enhanced solubility of icariin in the 4 organic solvents with the rise of temperature. The heat of solution of icariin in acetone is larger than that found in the other 3 solvents. The heat of solution in light petroleum is the lowest. Therefore, the solubility of icariin in acetone is most affected by temperature, while the solubility in light petroleum is minimally affected.
Since the interaction between the icariin molecule and the solvent molecule is stronger than the interaction between the solvent molecules, the dissolution process is endothermic. The course of icariin dissolving in acetone, acetoacetate, chloroform, and light petroleum was not spontaneous at any temperature and had no driving force (ΔH d > 0, ΔS d < 0).
In this study, the solubility of icariin in acetone, acetoacetate, chloroform and light petroleum in the range of 283.2‐318.2 K was determined by UV-Vis spectrophotometry. The solubility of icariin in the 4 solvents was a function of temperature, with solubility increasing with temperature. The dissolution enthalpy and entropy of icariin were evaluated by the van’t Hoff equation, using the solubility data obtained from the experiment. The Gibbs energy of the solution was calculated from the enthalpy and entropy. The dissolution enthalpy and entropy of icariin in the 4 solvents increased as acetone >acetoacetate >chloroform >light petroleum ether, which can be explained by the difference in polarity indices between them.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Na Wang would like to recognize funding from the Key Research and Development Program of Shaanxi Province (2017SF-302). Jiao Xie acknowledges the Natural Science Foundation of Shaanxi Province (2019JQ-388) and the clinical research award of the Second Affiliated Hospital of Xi’an Jiaotong University (XJEFY-2018094) for this work.
