Abstract
Introduction
Inflammatory Bowel Disease (IBD) is globally widespread, with higher incidence in North America, Europe, and Scandinavia compared to other regions, indicating regional differences in prevalence. IBD affects all genders but is common in ages 20–40 or 25% have a family history. Ulcerative Colitis (UC) and Crohn's disease share traits but differ in location, causing lifelong morbidity.1–3 UC is a persistent inflammatory factor affecting the colon and rectum, marked by inflammation and the development of ulcers in their inner linings. It significantly impacts the quality of life for those affected, causing signs and symptoms such as abdominal pain, diarrhea, rectal bleeding, and weight loss. 4 While the exact cause of UC remains elusive, it is believed to outcome from an unfortunate immune defence mechanism to gut microorganisms in genotypically susceptible subjects. However, it has been claimed that immunological dysregulation is a key factor in the emergence of UC (1). Proinflammatory cytokines like tumor necrosis factor, interferon, interleukins IL-1, IL-6, and IL-12, also the response of inflammatory facilitate or inducible nitric oxide synthase and cyclooxygenase-2, are believed to be key mediator that alters the inflammation process. 5 Conventional treatment approaches include anti-inflammatory drugs, immunosuppressants, and biological therapies. Nevertheless, not every patient exhibits a positive response adequately to these treatments, and many suffer adverse drug reactions that can limit their efficacy and long-term use. Despite the fact that these therapies work, they are linked to serious side effects, including high blood pressure, diarrhoea, cramps, and abdominal pain that is accompanied by a fever.4,6 The management of UC poses a complex challenge for healthcare professionals and researchers alike. In recent years, there has been a growing interest in exploring alternative or adjunct therapies to enhance the management of UC. One such avenue of investigation is the use of combination therapy involving existing medications with diverse mechanisms of action. In this context, the current study seeks to investigate the potential synergistic effects of combining Papaverine and Sulfasalazine in a rat model of UC. Papaverine, an opium alkaloid with antispasmodic properties, has been traditionally used to treat various spasms, particularly those involving the gastrointestinal tract. Its mechanism of action involves the inhibition of phosphodiesterase enzymes, leading to increased levels of cyclic AMP and cyclic GMP.7,8 Sulfasalazine, the most ancient medication within this category, is composed of a combination of 5-aminosalicylic acid (5-ASA) and sulfapyridine. When ingested, colonic bacteria break down Sulfasalazine into its constituent parts: sulfapyridine and 5-ASA. While 5-ASA is believed to be the active ingredient that effectively treats UC, Sulfapyridine is thought to be responsible for some of the medication's unwanted side effects. The precise way of Sulfasalazine's action is not entirely clear, and it remains uncertain whether Sulfasalazine itself or its metabolites, like sulfapyridine and 5-aminosalicylic acid, are key mediators of its anti-inflammatory effects. 9 In the management of mild-to-moderate UC, topical and oral 5-ASA compounds have continued to play a crucial role, whether for initiating therapy or maintaining it. On the other hand, aminosalicylates are not recommended for patients with mild to chronic UC, for whom systemic corticosteroids are the preferred initial therapy option. 10 Additionally, Papaverine has demonstrated vasodilator effects and has found applications beyond its traditional use, such as in the treatment of acute mesenteric ischemia. 11 With antispasmodic and vasodilator properties, it shows promise in managing UC symptoms. Its ability to relieve involuntary muscle contractions might alleviate abdominal pain, while vasodilatation could improve blood flow to inflamed tissues, potentially aiding healing. 12 Inducing UC involves administering a 5% solution of Dextran sulfate sodium which triggers an inflammatory response in the gastrointestinal tract, mimicking the pathophysiological processes seen in UC patients. 13 The similarity between the induced model of UC and the normal disease condition in humans lies in the inflammatory response within the gastrointestinal tract. 14 The induced model offers insights into disease mechanisms and treatment efficacy, advancing our understanding of UC and guiding novel therapeutic approaches. The rationale behind combining Papaverine and Sulfasalazine lies in their distinct mechanisms of action. By targeting different aspects of the inflammatory process, this combination therapy aims to provide a more comprehensive approach to managing UC, potentially enhancing efficacy and minimizing adverse effects associated with higher doses of individual medications. The findings could offer valuable information for both clinical practice and further research in the quest for more effective and safer treatments for UC.
Materials and Methods
Experimental Animals
Thirty adult (6-week-old) Wistar rats of both sexes, weighing 180 ± 20 g, were obtained from a breeder registered with the Committee for Control and Supervision of Experiments on Animals (CCSEA). The animal study protocol, numbered ASCB/IAEC/16/22/161, was received from the Institutional Animal Ethical Committee. The rats were housed in a standard laboratory conditions with 23 ± 2 °C as temperature, 40 ± 10% humidity, and a natural light-dark cycle (12 h each), following CCSEA guidelines in New Delhi, India. They had free access to water and a standard chow pellet diet (Ashirwad Industries, Mohali, Punjab). The rats underwent a one-week acclimatization period before the commencement of as experiments. Then animals were randomly allocated into the different study groups.
Induction of Ulcerative Colitis (UC)
Experimental were separated into five groups consisting of six rats in each group, Group 1: Normal control; Group 2: DSS control; Group 3: Sulfasalazine control; Group 4: Papaverine Control; Group 5: Combination of Sulfasalazine + Papaverine with 6 animals in each group. In order to induce UC, 5% Dextran sulphate sodium solution was prepared with distilled water and given orally (1 ml/kg),15–17 with a total duration of 21 days. On the other hand, standard medication (Sulfasalazine 200 mg/kg) and test medication (Papaverine 200 mg/kg)18–20 were given throughout the study. The animal was fasted overnight (for about 16 h) on the last day of the experimental model before the blood sampling, then blood samples were preserved for biochemical analysis, further euthanized the animals and the colon was isolated for histopathological purposes.
On a weekly basis closely tracked the animal's water, food consumption and changes in body weight and also observed their daily stool characteristics, including the presence of visible blood, and stool consistency. On the basis of these details the Disease Activity Index (DAI) were calculated by using the following formula: DAI = (body weight decrease + stool consistency + rectal bleeding total score) divided by 3.
Evaluation of Lactate Dehydrogenase (LDH)
The blood was centrifuged to separate the plasma, and the LDH levels in the plasma was then measured using ERBA commercial kits, following the standard protocol. The spectrophotometer measured the LDH concentration by observing the rate at which lactate is converted to pyruvate, a reaction catalyzed by the LDH enzyme.
Evaluation of Nitric Oxide (NO)
Generation of NO was assessed by quantifying nitrite levels using the Griess method. To summarize the procedure, samples of the colon were homogenized with buffer. The resulting supernatant was mixed with a solution containing 1% sulphanilamide and 0.1% N-(1-naphthyl) ethylenediamine dihydrochloride, and further mixed within 5% phosphoric acid. The fractions of nitrite were resolved into micromolar (μM) units by measuring the absorbance at wavelength 492 nm.
Evaluation of Myeloperoxidase (MPO) Activity
Colon samples (10 mg) were weighed and then treated in a chilled potassium phosphate buffer (50 mM, pH 6.0) with the addition of Cetyltrimethylammonium bromide (0.5%). Following this, a mixture of diluted H2O2, O-dianisidine, after some time reported absorbance at 450 nm immediately using a spectrophotometer. MPO expression was determined and expressed as units U/mg. 21
Evaluation of Lipid Peroxidation Status
The assessment of Malondialdehyde (MDA), which is a resultant product when polyunsaturated fatty acids in cell membranes degrade, is a meaningful test for lipid oxidation. In this process, colon samples were homogenized in 2 mL of chilled 1.15 M KCl buffer pH 6.0. To determine lipid peroxidation, a modified version of the method by Ohkawa
Evaluation of Superoxide Dismutase (SOD)
The colon samples SOD activity measured by Kono
Evaluation of Catalase (CAT)
The CAT procedure involves measuring the enzymatic expression of CAT, an enzyme that disintegrates hydrogen peroxide into water and oxygen. Collect fresh Blend the tissue with ice-cold phosphate buffer in a blender and subsequently centrifuge the resulting homogenate and collect the supernatant. Add 1 mL of 30 mM hydrogen peroxide and sample into the cuvette. Instantly mix the contents with a glass rod with a flattened tip, and place the cuvette in the spectrophotometer. The initial absorbance ought to be approximately between 0.500 and 0.520. Adjust the absorbance if necessary by adding more phosphate buffer to decrease it or more hydrogen peroxide to increase it. In a separate cuvette, take 1.9 mL of phosphate buffer and add 1 mL of 30 mM hydrogen peroxide. Then, add 0.1 mL of CAT solution (approximately 10 units) to the cuvette. 24
Histopathology of Colon
To identify the tissue damage caused by the inducing agent in animals from the illness group and the healing effect of the therapeutic medicine in the treated group, histopathology of the organs was performed. The animals were sacrificed, and the colon was extracted before being fixed in a 10% neutral buffered formalin solution for at least 6 h. The Kharar-based Dr Sandhu Diagnostic Lab performed the histopathology. Haematoxylin and eosin staining was used to prepare colon tissue on slides. Images were taken from the ready-made slides using a photomicroscope for analysis.
Statistical Analysis
The data were all given as Mean ± SEM. GraphPad Prism Stat (GraphPad Software, version 8.0.1 Inc., USA) was used to analyse the data from all groups using a One-way ANOVA followed by Tukey's test and Two-way ANOVA followed by Bonferroni post hock -tests. A p-value of 0.05 was deemed significant.
Results
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Disease Activity Index
The DSS exhibits clinical symptoms such as diarrhoea, faeces with blood, and a decrease in body weight. These particular attributes are assessed when calculating the DAI, which also takes into account the extent of inflammation. From the first week to the last week variation was observed in the DAI score in the DSS-induced group maximum incline upto 9 compared to the control. The DAI score decline in the case of alone Papaverine (200 mg/kg) with 3.33 and the Sulfasalazine (200 mg/kg) with 3.3 and similarly in the combination group Papaverine and Sulfasalazine the decreased DAI was observed closer to normal value ie 2.8, respectively as shown in Figure 1.

Histogram represents the effect of Papaverine alone and combined use with Sulfasalazine on Disease Activity Index. All the values are expressed as mean
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Body Weight of Rats
The weekly body weight in DSS solution-treated animals showed a decline throughout the treatment period, significant (p˂0.05) alterations were observed on days 14th and 21st as in comparison to the control group. However, the Sulfasalazine treatment group showed a rise in body weight; whereas treatment with Papaverine showed a modest incline in body weight the significant (p˂0.05) changes were observed on days 14th and 21st in the correlation with the disease control group. However, treatment with a combination of Papaverine and Sulfasalazine revert the significant (p˂0.05) on days 14th and 21st as to the normal body weight of the animal in comparison to the disease control group as shown in Figure 2.
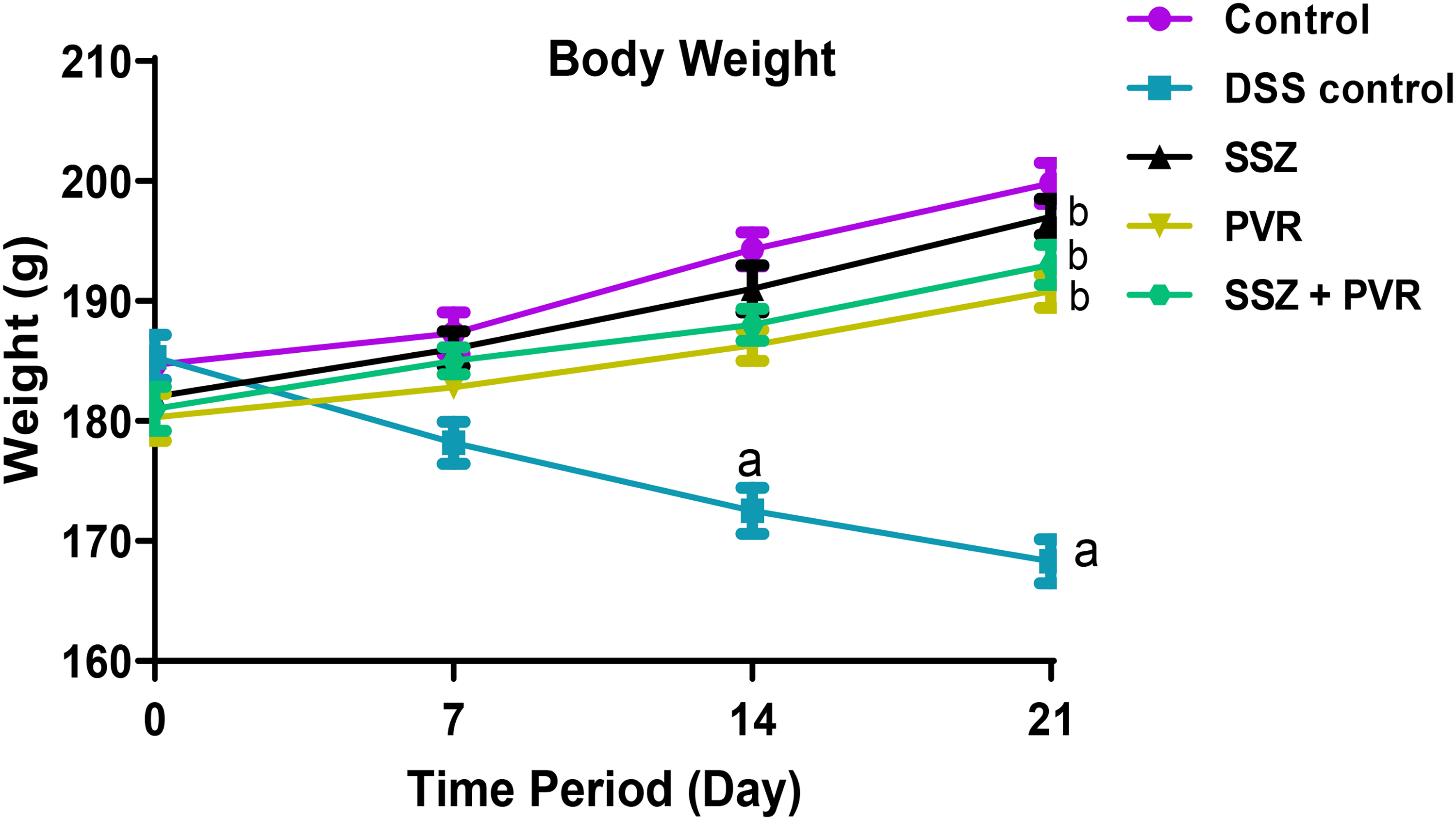
Histogram represents the effect of Papaverine alone and combined use with Sulfasalazine on Body Weight. All the values are expressed as mean
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Feed Intake of Rats
The weekly feed intake in treatment with DSS solution resulted in a significant (p˂0.05) decline in feed consumption on days 14th and 21st as compared to the normal control group. However, treatment with Sulfasalazine on days 14th and 21st expressed a significant (p˂0.05) increase in feed intake, whereas treatment with Papaverine represented a slight increase in feed intake as compared to the disease control group. However, treatment with a combination of Papaverine and Sulfasalazine also exhibited a significant (p˂0.05) increase in feed intake in comparison to disease control and as represented in Figure 3.

Histogram represents effect of Papaverine alone and combined use with Sulfasalazine on Feed Intake. All the values are expressed as mean
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Water Intake of Rats
The weekly water intake in treatment with DSS solution resulted in a decrease in water intake significant (p˂0.05) on days 14th and 21st in contrast to the normal control group. However, treatment with Sulfasalazine exhibited a significant increase (p˂0.05) on days 14th and 21st in water intake, whereas treatment with Papaverine showed a slight incline in water intake in comparison to the DSS-treated group. However, treatment with a combination of Papaverine and Sulfasalazine also exhibited a significant increase (p˂0.05) on days 14th and 21st in water intake in comparison to the disease control group as represented in Figure 4.

Histogram represents effect of Papaverine alone and combined use with Sulfasalazine on Water Intake. All the values are expressed as mean
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Antioxidant Enzymes
The levels of antioxidant CAT and SOD in DSS solution treatment exhibited a significant decline (p˂0.05) as compared to the normal control group and, Sulfasalazine group presented a significant (p˂0.05) increase in the levels of CAT and SOD, whereas treatment with Papaverine showed a slight (p˂0.05) increase in CAT and SOD levels in comparison to the disease control group. However, treatment with a combination of Papaverine and Sulfasalazine represented a highly (p˂0.05) significant increase in the levels of CAT and SOD near their normal levels as compared to the disease control group as illustrated in (Table 1).
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Antioxidant Enzymes and Oxidative Stress Markers Level.

Values are expressed as mean
Where a p˂0.05 as compared to the normal Control group, and b p˂0.05 as compared to the DSS, and c p˂0.05 as compare to SSZ (One-way ANOVA followed by Tukey's test) DSS: Dextran Sulphate Sodium, PVR: Papaverine, SSZ: Sulfasalazine.
Effect of Papaverine Alone and Combined Use with Sulfasalazine on Oxidative Stress Markers Levels Abbreviations
In the case of the DSS solution treatment showed a significant increase(p˂0.05) in values of NO, MPO, LDH and MDA in comparison to the normal control group. However, Sulfasalazine group exhibit a significant (p˂0.05) lower in the values of NO, MPO, LDH and MDA, whereas in Papaverine treatment group showed a slight decrease in the levels (p˂0.05) of NO, MPO, LDH and MDA in comparison of the disease control group. However, treatment with a combination of Papaverine and Sulfasalazine showed a highly (p˂0.05) significant decrease in the levels of NO, MPO, LDH and MDA near their normal levels in contrast to the disease control group, as illustrated in (Table 1).
Effect of Papaverine Alone and Combined Use with Sulfasalazine Histopathology of Colon
In the context of an UC rat model, the study assessed the impact of combining Papaverine and Sulfasalazine. The results showed that this combination therapy led to moderate improvements in mucosal damage, accompanied by a reduction In the invasion of inflammatory cells, notably macrophages, monocytes, and neutrophils, in the submucosal and lamina propria. These findings provide valuable insights into the potential therapeutic benefits of this combination for tissue healing and the mitigation of free radical burden in UC as presented in (Figure 5).

i).Pictorial representation demonstrates effect of Papaverine alone and combined use with Sulfasalazine on histopathology and ii). histogram represents the histological score in Colon (a) Control group (b) DSS treated group induces an influx of macrophages monocytes and neutrophils into the submucosa and lamina propria and cause mucosal damage categorized by loss of crypt glands and epithelium destruction (c) SSZ treated group reduced mucosal damage as compare to DSS group (d) PVR treated group showing mild change in mucosal damage and (e) (PVR + SSZ) Papaverine, particularly in combination with the standard drug shows moderate change in mucosal damage and reduces the influx of macrophages monocytes and neutrophils into the submucosa and lamina propria. All the values are expressed as mean ± SEM (n = 6). Where, ap˂0.05 versus Control and bp˂0.05 versus DSS. The statistical data analysis was presented by one-way ANOVA analysis using the Turkey's test. DSS: Dextran Sulphate Sodium, PVR: Papaverine, SSZ: Sulfasalazine.
Discussion
UC is a persistent inflammatory condition of the colon and rectum, associated with the category of chronic IBD. This ailment is identified by inflammation and the formation of ulcers in the epithelial lining of the colon, resulting in a variety of incapacitating as symptoms.13,25 The precise origin of UC remains unidentified, though there is a consensus that it results from an abnormal immune response in individuals with a genetic predisposition. 26 This study delved into the potential of utilizing Papaverine, alone and in conjunction with a standard drug, as a treatment strategy for UC. The research utilized a rat model induced by a 5% DSS solution to simulate UC conditions and assessed the disease's severity through various biochemical markers and histological evaluations.27,28 In rats exposed to DSS, body weight, feed and water consumption decreased significantly (p < 0.05). Sulfasalazine increased these parameters (p < 0.05), while Papaverine slightly elevated them (p < 0.05). Combining Papaverine and Sulfasalazine yielded similar effects (p < 0.05). The relationship between DSS and DAI alterations is that DSS is responsible for inducing colitis, while DAI is used to quantify and monitor the resulting changes in the disease activity. As the colitis progresses or responds to various treatments, the DAI may be altered or adjusted to reflect the severity of the condition at different points in time. Researchers use these alterations in the DAI to assess the effectiveness of interventions and to gain insights into the progression of the disease in the animal model. DSS-induced poor stool consistency and increased DAI score (p < 0.05). Sulfasalazine, Papaverine, combo partially relieved as symptoms (p < 0.05). Papaverine showed potential in enhancing body weight, feed intake and countering colitis-like symptoms. The findings underscored the potential role by regulating the oxidative stress in the progression of UC. Through the assessment of biomarkers such as CAT, SOD, MDA, and nitrate levels, it was observed that the UC-induced rats exhibited increased oxidative stress, marked by reduced antioxidant enzyme levels and elevated lipid peroxidation indicators (p < 0.05). 28 This oxidative damage was further implicated in DNA and protein oxidation and the breakdown of erythrocyte membranes. 29 Notably, Papaverine treatment exhibited antioxidant and anti-inflammatory potential by effectively reducing MDA and nitrate levels (p < 0.05) while increasing CAT and SOD levels (p < 0.05). Papaverine demonstrates treats, vasospasms; vasodilator, targets heart, intestines and blood vessels. 11 Remarkably, Papaverine was the inaugural Phosphodiesterase (PDE) inhibitor for diverse ailments. It blocks PDE-4 and PDE-5, elevating cAMP and cGMP levels, respectively. Its cAMP and cGMP elevation diminishes inflammatory markers (NO, COX-2, TNF-α, IL), and enhances antioxidant enzymes (CAT, SOD), mitigating oxidative stress (p < 0.05). 30 Consequently, Papaverine curbs apoptosis and ameliorates UC symptoms, presenting a potential therapeutic as avenue. This suggests a promising mechanism through which Papaverine can counteract the oxidative stress associated with UC. Papaverine shows protective effects on spinal cord injury (SCI) in rat model; arise from its free radical scavenging and anti-inflammatory, involving the suppression of the Receptor for Advanced Glycation End-products (RAGE)-High Mobility Group Box 1 (HMGB1) axis. 31 Inflammatory indicators, including MPO and NO, were also significantly (p < 0.05) elevated in the UC-induced rats. 32 The study highlighted the potential of Papaverine, particularly in combination with a standard drug, to attenuate these elevated levels (p < 0.05), indicating it's anti-inflammatory properties. Nitric oxide, generated mainly through iNOS under inflammatory conditions, can contribute to the exacerbation of inflammation and tissue damage. 13 Papaverine's ability to suppress NO levels (p < 0.05) could provide a mechanism for reducing inflammation and subsequently ameliorating UC symptoms. Histopathological changes refer to alterations in the microscopic structure. They provide valuable insights into the state of the tissues and organs at a cellular level. 33 Histological analysis provided further insights into the effects of Papaverine treatment. UC-induced rats displayed evident mucosal damage characterized by epithelial erosion, ulceration, and depletion of mucin-producing goblet cells. The influx of immune cells into the submucosal layers further underscored the extent of inflammation. 13 Importantly, the treatment with Papaverine, particularly in combination with the standard drug, demonstrated improvements in these histological features (p < 0.05), suggesting its potential to mitigate UC-associated tissue damage. By targeting oxidative stress and inflammation, Papaverine appears to address key factors driving the disease's progression. 34 While these findings suggest the potential therapeutic value of Papaverine in UC treatment, it's crucial to acknowledge the need for further research. The study serves as a preliminary as investigation, promising outcomes in a controlled rat model. For clinical translation, rigorous safety and efficacy assessments, ideally involving human subjects, are imperative to establish. In conclusion, this study illuminates Papaverine's potential as a curative intervention for UC. By addressing free radical stress and inflammation, Papaverine, especially in conjunction with a standard drug, demonstrates promising effects in ameliorating the disease-associated markers and histopathological changes.
The limitation of this study is the inadequacy of well-equipped laboratory and research facilities, which constrained the range of parameters that could be investigated. Despite demonstrating promising improvements in body weight, biochemical markers, and histopathological outcomes, the precise mechanisms by which Papaverine and Sulfasalazine exert their effects in treating UC remain unresolved. Further research is necessary to elucidate these mechanisms in order to refine treatment strategies and develop novel therapeutic approaches for UC colitis.
Summary and Conclusion
In the DSS 5% group, the rats showed significantly lower body weight, food, and water consumption, also decreased levels of the free radical scavengers such as CAT and SOD. Conversely, NO and lipid peroxidation levels were significantly elevated in this as group, indicating the induction of the disease and its associated abnormalities both biochemically and histologically. The combination of Papaverine with Sulfasalazine demonstrated a significantly beneficial and promising effect in DSS-induced UC.
On the basis of these outcomes the present investgation, summarized that Papaverine, particularly in combination with the standard drug, shows promising potential for treating DSS-induced UC. This conclusion is supported by significant improvements in body weight, biochemical and histopathological parameters, and enhanced antioxidant defence. These findings are of vital clinical importance and warrant further extensive research and studies.
The findings presented above make it clear that Papaverine, particularly in combination with the standard drug might be a useful remedy in the management of UC.
Footnotes
Acknowledgements
The authors are gratefull to the Director of institute and management committee member of Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, BELA (Ropar) for granting the necessary learning access.
Abbreviations
UC: Ulcerative Colitis;DSS: Dextran Sulphate Sodium ; IBD: Inflammatory bowel disease; DAI: Disease activity index; ROS: Reactive Oxygen species; MPO: Myeloperoxidase; NO: Nitric oxide; MDA: Malondialdehyde; SOD: Superoxide dismutase; CAT: Catalase; LDH: Lactate dehydrogenase SD: Standard deviation; ANOVA: Analysis of variance; 5-ASA: 5-aminosalicylic acid.
Author Contribution
All authors contributed equally to this work. Bei Huang: Conceived the animal experiments, Yanbang Niu: Curated and arranged the data, Haili Zhang: Revised the article and provided supportive work, Shengdong Zhang: Corresponding author; designed the experiments, Abhishek Gautam: Conducted the experiments. Gurpreet Kaur: Provided additional support and contributions, Ajay Singh Kushwah: Conceived the ideas and implemented the animal experiments.
Availability of Data and Material Statement
All relevant data are contained within the article. The original contributions presented in the study are included in the article and Supplemental material. Further data can be provided upon request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The animal study protocol (ASCB/IAEC/16/22/161) was approved by the Institutional Animal Ethical Committee Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, Bela, Ropar, Punjab-140111, and all experiments were conducted in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CCSEA), New Delhi, India.
Funding
Xi’an Science and Technology Program Project Grant Number:24YXYJ0079
Statement of Human Rights and Informed Consent
This article does not include any studies involving human subjects; therefore, informed consent is not applicable.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
