Abstract
Cryptotanshinone is a quinone diterpene extracted from the traditional Chinese medicine Salvia miltiorrhiza root that shows obvious anticancer activity. The aim of this study was to investigate the mechanism of action of cryptotanshinone as an antigastric cancer agent, as well as a chemotherapy potentiator. A gastric cancer model was established by tumor transplantation, and mice were treated with either 5-fluorouracil or cryptotanshinone, or both drugs. The tumor mass was recorded, and the tumor suppression rate was calculated. Pathological changes were observed by hematoxylin and eosin staining, gene transcription was detected by quantitative polymerase chain reaction, and protein expression by Western blotting. The results showed that cryptotanshinone could reduce the tumor mass, increase the tumor suppression rate, and enhance the chemotherapeutic effect of 5-fluorouracil by a mechanism related to inhibition of the Janus kinase 2/signal transducer and activator of transcription 3 signaling pathway.
Introduction
Gastric cancer (GC) is one of the most common types of cancer in the world. Its pathogenesis is related to many factors, such as chronic lack of fresh vegetables and fruits, long-term consumption of smoked and salted foods, Helicobacter pylori infection, genetic factors and gene expression. Studies predicted that in 2020, the incidence of stomach cancer would rank fifth worldwide, and its fatality rate would rank third. More than 60% of GC cases occur in East Asia. 1 In China, GC ranks third in incidence after lung cancer and colorectal cancer, and its mortality rate is second to only lung cancer and liver cancer; approximately 374 000 GC-related deaths were projected in China in 2020. 2
Early GC usually has no obvious clinical symptoms, and patients are typically in the progressive stage of disease at diagnosis and thus during the course of treatment. In China, the 5-year survival rate is less than 30% due to the extensive metastasis of progressive GC with high drug resistance. 3 The failure of chemotherapy is an important cause of death for GC patients, and the sensitivity of tumors to chemotherapy drugs is gradually decreasing due to the development of multidrug resistance (MDR).
Traditional Chinese medicine has gained more attention globally in recent years because of its therapeutic efficacy and low incidence of side effects. It now also plays a larger role in illness prevention and treatment. Salvia miltiorrhiza, a traditional Chinese medicinal plant, is commonly utilized as an adjuvant treatment for postoperative chemotherapy. According to network pharmacological studies, Salvia miltiorrhiza is anticipated to be considered as a potential component of traditional Chinese medicine for the treatment of GC. 4 The tanshinones in Salvia miltiorrhiza are mostly fat-soluble diterpenes and are mainly synthesized and accumulate in the periderm of the roots. Modern medical research has identified 15 active tanshinone-like compounds in Salvia divinorum, including tanshinone I, tanshinone IIA, tanshinone IIB, cryptotanshinone (CPT) and isocryptotanshinone (Figure 1). The biosynthesis of tanshinones occurs in three stages: geranylgeranyl pyrophosphate synthesis, cyclization, and hypotanshinone diene modification. From the tanshinone biogenesis pathway, it is clear that hypotanshinone diene is the first product in the formation of the tanshinone skeleton, and through a series of biosynthetic pathways, active ingredients such as tanshinone IIA and CPT are finally formed in reactions catalyzed by the cytochrome P450 enzyme oxidases, decarboxylase, dehydrogenase, and reductase. 5 Although there have been some reports on the anticancer benefits of CPT on several malignancies, such as lung, 6 colorectal, 7 breast, 8 and bladder 9 cancer, and chronic myeloid leukemia, 10 it is unclear exactly how CPT exerts its anticancer activity.
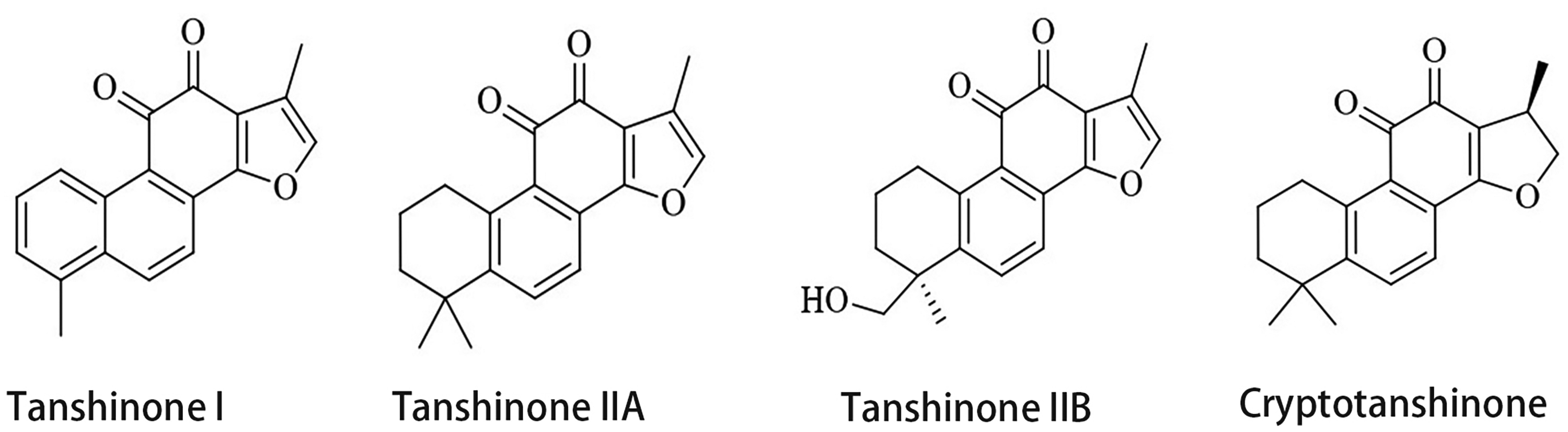
Chemical formula of the main active ingredients of tanshinone in Salvia divinorum.
The latest studies have shown that cell proliferation and cell survival can be controlled through the regulation of Pim-1 signaling by Janus kinase (JAK)/signal transducer and activator of transcription (STAT). 11 JAK2 is a member of the tyrosine kinase family, and STAT3 is a cytoplasmic transcription factor in the STAT family. JAK2/STAT3 is most closely related to cancer; these proteins can mediate signaling between the plasma membrane and the nucleus to impact a variety of cellular activities. When various carcinogens, radiation, viruses, growth factors, proto-oncogenes, and cytokines connect with the appropriate cell-membrane receptors, they activate intracellular JAK and induce the phosphorylation of STAT3 at tyrosine 705 [phosphorylated STAT3: p-STAT3 (Tyr705)]. The phosphorylated tyrosine binds reversibly to the SH2 region of another STAT3 monomer to form the STAT3 homodimer, which can translocate to the nucleus, where it functions as a transcription factor and promotes gene transcription, regulates tumor cell cycle progression and inhibits apoptosis.12,13 In addition, 5-FU resistance was shown to be reduced by inhibiting the JAK2/STAT3 pathway. 14 There is no such report on the interaction between CPT and JAK2/STAT3 in GC, despite an increasing number of reports implicating JAK2/STAT3 and CPT in GC proliferation. Previous studies on GC cytology have confirmed that CPT can induce apoptosis and amplify the effects of chemotherapy agents on GC cells, so this study was conducted in nude mice to further investigate the mechanism of action of CPT.15–17
Tumor cell growth and proliferation are closely related to the imbalance in pro- and antiapoptotic signaling and abnormal regulation of the cell cycle. Programed cell death is mainly dependent on the mitochondrial pathway, and stress signals lead to altered mitochondrial membrane permeability and cytochrome C release from the mitochondrial membrane gap, followed by activation of a protein hydrolysis cascade of cysteine caspases and, ultimately, apoptosis. 18 The B-cell lymphocytic-leukemia pro-oncogene-2 family can be divided into two major groups: the antiapoptotic factors [mainly B-cell lymphoma-2 (Bcl-2), B-cell lymphoma-XL (Bcl-XL), myeloid cell leukemia-1 (Mcl-1)] and the proapoptotic factors [mainly B-cell lymphoma-2-associated X (Bax), Bak, Bcl-XS, Bad, Bik and Bid]. Among them, Mcl-1, Bcl-XL, and Bcl-2 are cell death regulators that oppose apoptosis and shield cells from this form of death in response to external stimuli. 19 The dynamic changes between proapoptotic and antiapoptotic proteins have a crucial effect on the mitochondrial pathway.
Materials and Methods
Materials
TRIzol lysis solution was purchased from Life Technologies Corporation, NovoStart SYBR quantitative polymerase chain reaction (qPCR) SuperMix Plus from Novoprotein, PrimeScript™ RT Reagent Kit with gDNA Eraser and LA Taq from Takara, and a BCA kit from Sigma (St. Louis). Goat antirabbit IgG was obtained from Beijing Zhongshan Biological Technology Co., rabbit antibodies against mouse JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, Bax, survivin, phosphorylated JAK2 (p-JAK2), phosphorylated STAT3 (p-STAT3), P-glycoprotein (P-gp) and GAPDH from Abcam, CPT (purity > 98%) from Shanghai Yuanye Bio-Technology Co., Ltd, electrochemiluminescence (ECL) Western blot detection reagents from Beyotime Biotechnology, and dimethyl sulfoxide from Fisher Scientific.
Cell Culture
The 5-FU-resistant GC cell line SGC-7901/5-FU was kindly provided by Fudan University. The MDR subline SGC-7901/5-FU was developed by exposing parental cells to increasing concentrations of the anticancer drug 5-FU. All cells were cultivated in RPMI-1640 supplemented with 10% heat-inactivated fetal bovine serum at 37 °C in a 5% CO2 atmosphere.
Animal Experimental Design
Male 5-week-old BALB/c-nu mice were purchased from Beijing Huafukang Biotechnology Co., Ltd. All mice were bred and maintained in a specific pathogen-free (SPF) clean laboratory with adequate food and water. Approximately 0.2 mL of 5-FU-SGC-7901-resistant tumor cell suspension (containing 1 × 107 cells) was inoculated subcutaneously into the right forelimb of 10 mice under aseptic conditions, and solid subcutaneous tumors reached 0.5–1.0 cm in diameter in approximately 2 weeks. These tumors were excised under aseptic conditions, and the hard fish-like tissue was cut into pieces of approximately 0.5 mm × 0.1 mm and stored in cold saline solution.
We purchased 30 more of the same type of mice. After the mice were anesthetized, an incision (approximately 1.0–1.5 cm) was made in the middle of the upper abdomen, and the stomach was lifted through the incision. The visceral peritoneum was punctured with a 1.0–1.5 mm glass elbow probe along the longitudinal axis on the anterior side of the gastric sinus near the lesser curvature. A fragment of tumor tissue of the appropriate size was selected and inserted into the space under the plasma layer of the gastric wall, and the abdomen was closed layer by layer after confirming the absence of obvious blood leakage.
Through a literature review,20,21 we found that CPT is typically administered by intraperitoneal injection at a concentration of 10–40 mg/kg. However, the antitumor effect of CPT was not very obvious at a concentration of 15 mg/kg. When CPT was administered at 20 mg/kg, the tumor mass was obviously reduced, and the mice did not have any adverse effects. However, when the concentration of CPT was 25 mg/kg, some mice showed low spirits and reduced activity, and some even died. Therefore, we decided to use CPT at a concentration of 20 mg/kg.
After 14 days of feeding, the mice were randomly divided into six groups (6 mice per group). The mice in the model group were not treated; those in the control group received an intraperitoneal injection of 10 mL/kg of 0.9% sodium chloride solution (NS); those in the 5-FU group received an intraperitoneal injection of 5-FU (20 mg/kg) dissolved in 0.9% NS; those in the CPT group received an intraperitoneal injection of CPT (purity > 98%, 20 mg/kg) dissolved in 10 mL/kg of 0.9% NS; and those in the CPT + 5-FU group received equal amounts of 5-FU and CPT (20 mg/kg each) dissolved in 10 mL/kg of 0.9% NS through intraperitoneal injection. Treatments were administered twice a week for 6 consecutive weeks. At the end of the intervention period, the mice were sacrificed, and the tumor specimens inoculated into the stomach were dissected intact.
The study was reviewed and approved by the Animal Ethics Committee of Anhui University of Traditional Chinese Medicine (Ethical approval number: AHUCM-mouse-2022019). All experimental procedures were performed under the guidance of the ethics committee to minimize animal suffering.
Quantitative Real-Time PCR (qRT-PCR) Assay
Total RNA was extracted from mouse tumor tissue using TRIzolTM according to the manufacturer's instructions. cDNA was then synthesized using 15 ng of isolated RNA using a relevant synthesis kit. Afterwards, the mRNA levels of JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, Bax, survivin, and MDR1 were quantified using SYBR Green qPCR Master Mix with an Applied Biosystems 7900HT Fast qPCR System based on specific primers for JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, Bax, Survivin, P-gp, and GAPDH. The primer sequences are listed in Table 1. The qPCR assay was performed in two steps, with 3 replicate wells for each target gene. In the first stage, the samples were denatured at 95 °C for 1 min. In the second stage, the samples were denatured at 95 °C for 5 s and annealed at 60 °C for 15 s in a total of 40 cycles. The third stage was 15 s at 95 °C, 1 min at 55 °C, and 30 s at 95 °C.
Primers for qRT-PCR Analysis.

Abbreviations: Bcl-2, B-cell lymphoma-2; Bcl-XL, B-cell lymphoma-XL; Mcl-1, myeloid cell leukemia-1; Bax, B-cell lymphoma-2-associated X; P-gp, P-glycoprotein; qRT-PCR, quantitative real-time polymerase chain reaction.
Western Blot Analysis
Proteins from tumor tissue were extracted with RIPA lysis buffer in the presence of PMSF and a protein phosphatase inhibitor. Then, the protein concentration in the supernatant was quantified by a BCA kit. Equal amounts of isolated protein (30 µg/lane) were separated by SDS–PAGE and transferred onto polyvinylidene difluoride membranes, which were blocked in Tris-buffered saline at pH 7.6 and 0.05% Tween 20 and then incubated with specific monoclonal primary antibodies. The primary antibodies were diluted as follows: JAK2 (1:5000), STAT3 (1:1000), Bcl-2 (1:2000), Bcl-XL (1:2000), Mcl-1 (1:2000), Bax (1:2000), survivin (1:1000), p-JAK2 (1:4000), p-STAT3 (1:1000), P-gp (1:2000), and GAPDH (1:2000). After washing, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies (1:5000) at room temperature for 50 min. Finally, the protein levels were quantified using ECL Western blot detection reagents.
Statistical Analysis
All analyses were conducted using SPSS 23.0 software (SPSS Inc.). Data are presented as the `x ± s. If the variance was homogeneous, single-factor analysis of variance and the t test were used to compare sample means. A nonparametric test was used if the variance was not uniform. P values <.05 were considered indicative of a significant difference.
Results
CPT not Only Inhibits GC Growth but Also Potentiates the Anti-GC Effect of 5-FU
In the clean SPF laboratory, mice that had been successfully transplanted with tumors were given no treatment, 0.9% NS, CPT, 5-FU, or CPT + 5-FU for 6 weeks. After complete tumor debridement, the tumors from each group were weighed using electronic scales, and the tumor suppression rate was calculated. Compared with the model group, the control group showed no significant difference in the tumor suppression rate (P > .05), while the CPT group had a significant difference in tumor suppression rate at 76.3% (P < .05). The tumor suppression rate in the CPT + 5-FU group was 80.9%, which was significantly different from that in the 5-FU group (P < .05). The results showed that CPT not only inhibits GC growth but also potentiates the anti-GC effect of 5-FU (Table 2). Pathological sections were observed after hematoxylin and eosin staining, and the results indicated a lower tumor cell density in the CPT, 5-FU, and CPT + 5-FU groups, with obvious foci of tumor necrosis and inflammatory cell infiltration. In the CPT + 5-FU group, the tumor cell density was decreased, and numerous necrotic foci were observed (Figure 2).

Histopathological section of tumor, stained by HE. (A)The tumor cells in the model group were dense and uniformly arranged, and no obvious foci of tumor necrosis were seen. (B)The tumor cells in the control group were denser, arranged in a nested pattern, with abundant cytoplasm and red staining. (C)Tumor cell density was reduced in the 5-FU group, and some foci of tumor necrosis and inflammatory cell infiltration were seen. (D)Tumor cell density was reduced in the CPT group, and tumor necrosis foci and inflammatory cell infiltration were seen. (E) The tumor cell density decreased in the CPT + 5-FU group with a large number of necrotic foci. Abbreviations: HE, hematoxylin and eosin; CPT, cryptotanshinone
Tumor Tissue Weight(`x ± s) and Tumor Inhibition Rate of Each Group.

Compared with the model group, the difference was not statistically significant (P > .05).
Compared with the model group, the difference was statistically significant (P < .01).
Compared with the 5-FU group, the difference was statistically significant (P < .01).
CPT can Inhibit the Expression of JAK2/STAT3 Signaling Pathway Components
The inhibition of tumor growth by CPT was further verified by qPCR and WB to determine whether this inhibitory effect was related to the JAK2/STAT3 pathway. This study involved blank, saline, and CPT intervention for two weeks. qPCR assays demonstrated that CPT could inhibit the transcription of JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, and survivin mRNA and enhance Bax mRNA transcription (Figure 3). The difference between the model group and the control group was not statistically significant (P > .05), but the difference between the model group and the CPT group was statistically significant (P < .05). Furthermore, WB experiments demonstrated that CPT decreased the levels of p-JAK2, p-STAT3, Bcl-2, Bcl-XL, Mcl-1, and survivin and increased Bax levels (Figure 4). The difference between the control group and the model group was not statistically significant (P > .05), but the difference between the control group and the CPT group was significant (P < .05). WB and qPCR assays showed that the inhibition of tumor growth by CPT was related to the JAK2/STAT3 signaling pathway.

CPT inhibits the transcription of genes related to the JAK2/STAT3 signaling pathway. Expression of JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, Bax, Survivin mRNA in gastric cancer tissues, respectively. Values are mean ± SD from triplicate experiments. * P < .05 versus control group, **P < .01 versus control group, #P < .05 versus 5-FU group, ## P < .01 versus 5-FU group. Abbreviations: JAK2, Janus kinase 2; STAT3, signal transducer and activator of transcription 3; CPT, cryptotanshinone; Bcl-XL, B-cell lymphoma-XL; Bcl-2, B-cell lymphoma-2; Mcl-1, myeloid cell leukemia-1; Bax, B-cell lymphoma-2-associated X.

CPT inhibits the expression of the JAK2/STAT3 signaling pathway-related proteins. Protein expression of JAK2, p-JAK2, STAT3, p-STAT3, Bcl-2, Bcl-XL, Mcl-1, Bax, Survivin in gastric cancer tissue. * P < .05 versus control group, ** P < .01 versus control group, # P < .05 versus 5-FU group, ## P < .01 versus 5-FU group. Abbreviations: JAK2, Janus kinase 2; STAT3, signal transducer and activator of transcription 3; CPT, cryptotanshinone; Bcl-XL, B-cell lymphoma-XL; Mcl-1, myeloid cell leukemia-1; Bax, B-cell lymphoma-2-associated X; p-JAK2, phosphorylated Janus kinase 2; p-STAT3, phosphorylated signal transducer and activator of transcription 3.
The Mechanism of Action by Which CPT Enhances the Anti-GC Effect of 5-FU is Also Related to the JAK2/STAT3 Signaling Pathway
In this study, we performed qPCR and WB assays to further verify the ability of CPT to reverse tumor resistance. CPT, 5-FU, and CPT + 5-FU were administered for two weeks. The experimental results showed that compared with the control group, the 5-FU group had significantly higher MDR1 and P-gp levels. In contrast, MDR1 and P-gp levels were lower in the CPT + 5-FU group (Figure 5), and the difference was statistically significant. In addition, compared to the 5-FU group, the CPT + 5-FU group had significantly lower mRNA levels of JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, and survivin; significantly lower protein levels of p-JAK2, p-STAT3, Bcl-XL, and survivin; and significantly higher Bax mRNA and protein levels (Figures 3 and 4). Therefore, CPT can inhibit the transcription of the drug resistance gene MDR1 and reduce P-gp expression. In addition, the mechanism of action also impacts the JAK2/STAT3 signaling pathway.

CPT can reverse the transcription of the drug resistance gene MDR1 and reduce the expression of P-gp. (A) The expression of MDR1 in gastric cancer tissues. (B) Protein expression of P-gp in gastric cancer tissue. Values are mean ± SD from triplicate experiments. * P < .05 versus control group, **P < .01 versus control group, # P < .05 versus 5-FU group, ## P < .01 versus 5-FU group. Abbreviations: MDR1, multidrug resistant 1; CPT, cryptotanshinone; P-gp, P-glycoprotein.
Discussion
As an incredibly important active ingredient, CPT has been widely studied in recent years. Studies have revealed that CPT may be utilized in conventional medicine for both cancer prevention and treatment. 22 Additionally, CPT has been demonstrated to be an active secondary metabolite that improves the effectiveness of anticancer medications, restores sensitivity to several chemical drugs, and reduces the adverse effects of anticancer drugs on normal cells. 23 The anticancer effects of CPT on other cancer cells have been well investigated,24,25 but its impact on the apoptosis of human GC cells and its mechanism of action have not been thoroughly explored. Therefore, we used SGC-7901 GC cells to construct a mouse transplantation tumor model to examine the inhibitory effect of CPT on GC cells. According to the findings, CPT inhibited the growth and proliferation of GC cells by promoting the expression of antiapoptotic genes in GC cells.
In the present work, we evaluated the ability of CPT to prevent GC in an experimental mouse model. The main findings were as follows: (a) CPT can inhibit GC growth by suppressing JAK2/STAT3 signaling and (b) CPT potentiates the anti-GC effect of 5-FU by reversing the transcription of the drug resistance gene MDR1 and decreasing P-gp expression.
As reported in the literature, CPT can induce the apoptosis of mouse liver cancer Hepa1-6 cells through the JAK2/STAT3 signaling pathway, thereby suppressing cell proliferation. 26 Moreover, the JAK2/STAT3 signaling pathway contributes to GC development, and its downregulation inhibits GC growth. We first investigated whether CPT has an inhibitory effect on GC. The results showed that the tumor masses in mice treated with CPT were significantly smaller, and molecular analyses of the tumors revealed that CPT inhibited tumor growth by reducing the mRNA levels of JAK2, STAT3, Bcl-2, Bcl-XL, Mcl-1, and survivin; decreasing p-JAK2, p-STAT3, Bcl-2, Bcl-XL, Mcl-1, and survivin protein levels; and increasing Bax mRNA and protein levels. The survival of cancer cells is usually dependent on elevated levels of Bcl-2 family members, which bind to and inactivate proapoptotic proteins containing the BH3 structural domain. 27 Bcl-XL is a mitochondrial membrane protein that regulates the stability of the mitochondrial internal environment in terms of voltage and permeability in response to different stimuli, maintains the integrity of the outer mitochondrial membrane, and thus promotes cell survival 28 ; its overexpression leads to a notable rise in the population of surviving cells. 29 Bax proteins interact with members of the Bcl-2 protein family in a physiological environment to activate apoptosis in response to diverse stimuli. By interacting with BH3-only activators, cytoplasmic Bax monomers are produced during apoptosis, leading to mitochondrial outer membrane permeabilization and allowing apoptotic agents to enter the cytoplasm, which leads to cell death. Since the formation of membrane complexes involving Bax is irreversible, Bax activation represents a decisive and irreversible point in the process of cell death. 30 Survivin antagonizes apoptosis and promotes mitosis, and STAT3 can contribute to the upregulation of survivin expression through IL-1. 31
In addition, we investigated whether CPT can reverse 5-FU resistance in GC. P-gp, the most important protein associated with drug resistance, is encoded by MDR1, and P-gp is commonly overexpressed in drug-resistant cancer cell lines. P-gp binds to drugs and pumps them out of the cell when the ATP supply is sufficient, leading to an increased extracellular drug concentration and a decreased intracellular drug accumulation; specifically, cells become drug-resistant, triggering MDR and chemotherapy failure. In a study of colon cancer, 17 CPT was found to effectively prevent P-gp-mediated drug resistance and to help colon cancer cells that overexpress P-gp overcome MDR by reducing P-gp expression and lowering ATPase activity, thus enhancing the intracellular accumulation of the chemotherapeutic drug, but no research has been undertaken to determine the underlying mechanism. An experimental drug-resistant cell line was used, and by comparing the CPT + 5-FU group with the 5-FU group, we found that CPT reversed the transcription of MDR1 and reduced the expression of P-gp, thus reversing tumor drug resistance; we also found the signaling pathway that plays a role in the underlying mechanism. This result suggests that CPT is a potential chemotherapy potentiator, and the next step for our group will be to detect the binding of P-gp to the abovementioned proteins using protein chip technology to further explore the mechanism of its potentiating effect.
In summary, our findings demonstrate that CPT inhibits GC growth and potentiates the effect of 5-FU chemotherapy in experimental mice, and its mechanism of action is connected to the JAK2/STAT3 signaling pathway. These findings provide a rationale for the anticancer properties of CPT.
Supplemental Material
sj-tif-1-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-1-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-2-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-2-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-3-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-3-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-4-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-4-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-5-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-5-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-6-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-6-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-7-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-7-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-8-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-8-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-9-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-9-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-10-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-10-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-11-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-11-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-tif-12-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-tif-12-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-jpg-13-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-jpg-13-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-jpg-14-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-jpg-14-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-jpg-15-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-jpg-15-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Supplemental Material
sj-jpg-16-npx-10.1177_1934578X221130874 - Supplemental material for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model
Supplemental material, sj-jpg-16-npx-10.1177_1934578X221130874 for Cryptotanshinone From Salvia miltiorrhiza Inhibits the Growth of Tumors and Enhances the Efficacy of Chemotherapy in a Gastric Cancer Mouse Model by Wenkai Wu, Yezhi Cao, Ling Cheng, Linghu Wang, Qingsheng Yu, Hui Peng, Fuhai Zhou, Haiwei Liu and Qi Zhang in Natural Product Communications
Footnotes
Authors’ Note
Data are available from the authors upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was reviewed and approved by the Animal Ethics Committee of Anhui University of Traditional Chinese Medicine (Ethical approval number: AHUCM-mouse-2022019). All experimental procedures were performed under the guidance of the ethics committee to minimize animal suffering.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the General Project of Anhui Natural Science Foundation (2008085MH266), the Natural Science Research Project of Anhui Universities (KJ2020A0448), Clinical Scientific Research Project of the First Affiliated Hospital of Anhui University of Chinese Medicine (2020yfyzc02), Anhui Province University Outstanding Young Talents Support Program Project (gxyq2019035 and gxyq2021184), and Key Research and Development Plan of Anhui Province (202004j07020047).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
