Abstract
Objective
Based on this premise, the present study aimed to evaluate the antibacterial activity of ethanolic extracts of wax (EECMr) and fermented pollen (EEPMr), as well as pure honey (HMr) from Melipona rufiventris, against multi-resistant strains of Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa.
Methods
Microbiological assays were performed using the broth microdilution method to evaluate the intrinsic activity of the bioproducts, their antibiotic combination effects, and beta-lactamase inhibition by determining Minimum Inhibitory Concentration (MIC).
Results
The products tested did not show clinically relevant intrinsic activity against the strains tested, exhibiting MIC ≥ 1024 µg/mL. However, when combined with the evaluated antibiotics, they reduced the MIC, indicating a potentiation effect of the antibiotic activity. For S. aureus, a reduction in the MIC of gentamicin from 64 to 32 µg/mL and norfloxacin from 128 to 64 µg/mL was observed when combined with EEWMr and EEPMr, respectively. Results for E. coli demonstrated a reduction in the MIC of gentamicin from 64 to 16 µg/mL for HMr and EEPMr. Additionally, the combination of ampicillin with EEWMr resulted in a threefold MIC reduction, from 64 to 8 µg/mL, attributed to its action on the beta-lactamase enzymatic mechanism in E. coli. For P. aeruginosa, all three bioproducts showed a reduction in the MIC for gentamicin when combined with EEWMr and EEPMr, decreasing the MIC from 8 to 4 µg/mL.
Conclusion
The bioproducts of M. rufiventris show a promising future as potential beta-lactamase inhibitors.
Introduction
Infections caused by multidrug-resistant microorganisms represent one of the major public health problems, particularly those caused by pathogens resistant to most of the antibiotics available on the market.1,2 Antibiotic and drug resistance has become a global concern, with the indiscriminate use of antibiotics and self-medication as the main contributors, being key factors in the emergence and spread of resistance. Furthermore, the excessive use of antibiotics to treat secondary infections caused during the Coronavirus Disease (Covid-19) pandemic has contributed to the development of resistant strains.3,4
Among the resistance mechanisms acquired by bacteria, the active efflux mechanism stands out, mediated by efflux pumps present in the bacterial membrane that expel drugs from the interior of the bacterial cell5,6; membrane impermeability, which alters the membrane structure and reduces the size of the entry channels 7 ; modification of the antibiotic target or model, altering the binding site 8 ; and enzymatic inactivation, where enzymes like beta-lactamase neutralize the action of antibiotics by degrading the beta-lactam ring present in their structure. 9
Staphylococcus aureus is a commensal bacterium that resides in the mucosal and skin folds of healthy individuals, but when present in the bloodstream, it can cause issues ranging from chronic infections, endocarditis, osteomyelitis, to pneumonia.1,6,10 S. aureus has a variety of antibiotic-resistant strains and is therefore classified as one of the main bacteria in the ESKAPE-E group.10,11
This group includes the most pathogenic bacteria responsible for hospital-acquired infections due to high antibiotic resistance rates, also including Pseudomonas aeruginosa and Escherichia coli.10,12 P. aeruginosa is associated with urinary and respiratory infections in humans, especially those with cystic fibrosis.13,14 E. coli is a Gram-negative pathogen primarily associated with intestinal infections or urinary tract infections, causing hemolytic uremic syndrome.15,16
With the rise of bacterial resistance, the search for new alternatives to control these infections has become essential, with natural products standing out as promising options. 17 The exploration of natural substances synthesized by animals, such as stingless bees, has been studied as a therapeutic strategy since the earliest civilizations.18,19
Stingless bees (tribe Meliponini) constitute a group of hymenopterans widely distributed around the globe, with the highest concentration in the Neotropical region, where nearly 300 species are found in Brazil alone. 20 Some of these species are particularly notable for the commercial value of their products, such as honey, propolis, pollen, and wax. 21 The bioproducts of bees, especially stingless bees, have been shown to exhibit various bioactivities, including antioxidant, 22 antibacterial, 23 antifungal, and anti-inflammatory properties, all associated with the chemical structures of the compounds present in these products.22,24 In addition to their commercial and biological value, stingless bees play a crucial ecological role as the main pollinators of most plants, making them essential for maintaining native flora and for agriculture.25,26
Melipona rufiventris (Lepeletier, 1836), popularly known as “Uruçu-amarela,” is a species of stingless bee native to the biomes of the Cerrado, Caatinga, and Atlantic Forest in Brazil.20,23 This species of stingless bee has been little studied, which justifies the investigation of the antimicrobial potential of its products. It is also worth mentioning that the natural products of M. rufiventris have promising potential for inhibiting the beta-lactam resistance mechanism present in multi-resistant strains of E. coli. 27 This study evaluates the intrinsic antibacterial activity and antibiotic potentiation effects of ethanolic extracts of wax (EEWMr), fermented pollen (EEPMr), and pure honey (HMr) from Melipona rufiventris against multidrug-resistant Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa.
Methodology
Collection and Preparation of Extracts from M. rufiventris Products
Samples of honey, wax, and fermented pollen (“saburá”) from Melipona rufiventris were collected at Recanto das Abelhas, located in the municipality of Crato, Ceará, Brazil, with geographic coordinates 7°27’50” S and 39°28’13” W. The samples were stored in plastic jars in a standard refrigerator until the extracts were prepared.
27
Collection was conducted with authorization from the System of Authorization for Biodiversity Information (SISBIO), under number 85035-1, granted by the Chico Mendes Institute for Biodiversity Conservation (ICMBio). For the preparation of the extracts, 170.8 g of fermented pollen (“saburá”) and 83.6 g of wax were used. The material was crushed and placed into glass jars. Subsequently, 500 mL of 99.5% ethanol was added to each extract and kept in contact with the product for 72 h to allow the solvent to extract the compounds. After this period, the solution was filtered and subjected to rotary evaporation under vacuum (Fisatom Scientific Equipment Ltd, Brazil), where the obtained crude extract was placed in a water bath (Quimis Scientific Equipment Ltd, Brazil) to evaporate the excess solvent. At the end of the process, the ethanolic extract of fermented pollen from M. rufiventris (EEPMr) and the ethanolic extract of M. rufiventris wax (EECMr) were obtained. For the honey from M. rufiventris (HMr), no extract was prepared, and it was used in its pure form. The yield calculation was made using the following formula (Calculation 1), where the product yield in percentage is the ratio of the final product weight (extract (pf)) multiplied by 100 and divided by the initial product weight (pi).
27
Microbiological Assays
Substances
For the dilution of the substances, 10 μL of the antibiotics gentamicin, norfloxacin and ampicillin (Sigma Co., St. Louis, USA) and the EEPMr, EEWMr and HMr were weighed, then diluted in dimethylsulfoxide (DMSO) (500 μL, at a concentration of less than 5%) and sterile water (9265 μL). All substances were diluted to a standard concentration of 1024 μg/mL.
Culture Media
Two culture media were used for the assays: Mueller Hinton Agar (Sigma-Aldrich Brazil Ltda), prepared according to the manufacturer's recommendations, was used as the solid medium for bacterial strain cultivation; and Brain Heart Infusion (Acumedia Manufacturers Inc.), prepared at a 10% concentration, was used as the liquid medium for the microdilution plates.
Bacterial Strains
Standard strains S. aureus ATCC LB25923, E. coli ATCC LB25922, and P. aeruginosa ATCC 29336. And multiresistant strains of S. aureus 10, E. coli 06, and P. aeruginosa 24, 28 were used throughout this study (Table 1).
Amc—Amoxicillin + Clavulanic Acid, Ami—Amikacin, Amox—Amoxicillin, Amp—Ampicillin, Asb—Ampicillin + Sulbactam, Azi—Azithromycin, Ca—Cefadroxil, Cef—Cephalexin, Cfo—Cefoxitin, Cip—Ciprofloxacin, Cla—Clarithromycin, Clin—Clindamycin, Cmp—Cefepime, Cro—Ceftriaxone, Ctz—Ceftazidime, Eri—Erythromycin, Imi—Imipenem, Lev—Levofloxacin, Mer—Meropenem, Mox—Moxifloxacin, Oxa—Oxacillin, Pen—Penicillin, Ptz—Piperacillin.

Minimum Inhibitory Concentration (MIC) Assay
The Minimum Inhibitory Concentration (MIC) of EEPMr, EEWMr, and HMr was determined according to the broth microdilution method proposed by Javadpour et al, 29 with adaptations. The strains were inoculated onto solid media 24 h before the assays and maintained in a microbiological incubator. After this period, the bacterial inoculum was suspended in saline solution to a McFarland scale of 0.5, approximately 1.5 × 108 CFU/mL. Microtubes were filled with 900 μL of liquid medium and 100 μL of the bacterial inoculum, then plates were filled with 100 μL of the final solution from the microtubes. Microdilution was performed by adding 100 μL of the extracts and honey to the plates, with serial dilutions up to the second-to-last well of the plate (1:1), which was used as the growth control. Dilutions ranged from 512 μg/mL to 8 μg/mL, Table 2). Plates were maintained in a microbiological incubator for 24 h, and after this period, readings were performed by adding 20 μL of resazurin (7-hydroxy-3H-phenoxazine-3-one 10-oxide). Resazurin oxidizes in the presence of an acidic medium produced by bacterial growth, leading to a color change from blue to pink. 30 The MIC was defined as the lowest concentration where no growth was observed, and the assays were carried out in triplicate. 31
This Table Shows a Sequence of Decreasing Concentrations, Typically Used to Determine the MIC According to the Methodology Described in the Methods Section.

Antibiotic Potentiation Assay
To assess whether EEPMr, EEWMr, and HMr have the ability to modify the action of the antibiotics norfloxacin, gentamicin, and ampicillin, a comparative study was conducted between the effects of the standard antibiotics, evaluating their ability to reduce the MIC when combined with the extracts and honey. A sub-inhibitory concentration (MIC/8) of 128 μg/mL of the inhibitors and compounds was used. In the assays, the bacterial inoculum was prepared by suspending it in a saline solution corresponding to the McFarland scale of 0.5, approximately 1.5 × 108 CFU/mL. 150 μL of the inoculum, along with 188 μL of the extracts and honey, were transferred into a microtube and completed with 1162 μL of liquid medium. 100 μL of the microtube contents were transferred to the microdilution plate, to which 100 μL of the antibiotic was added, followed by serial dilution (1:1) ranging from 512 to 0.5 μg/mL (Table 2).
The sub-inhibitory concentration (MIC/8) was calculated based on the lowest concentration that did not show direct antibacterial activity, according to the study by Coutinho et al. 32
Beta-Lactamase Presence Verification
To verify the presence of the beta-lactamase enzymatic mechanism in the evaluated strains, a comparative assay was performed using the standard inhibitors for this mechanism (Sulbactam) and ampicillin. The MIC of ampicillin alone was compared with the MIC of ampicillin combined with sulbactam (50%) (Amp + Sulbactam). The presence of beta-lactamase is evidenced when there is a reduction of at least threefold in the MIC value compared to ampicillin alone. 33
Statistical Analysis
Central data and standard deviations were obtained according to the methodology of Freitas et al 34 for microbiological analysis. Data were analyzed using the GraphPad Prism 6.01 statistical software, and the ANOVA test was applied. A post hoc Bonferroni test was performed (P < .05 was considered significant, and P > .05 was considered non-significant).
Results
Yield of M. rufiventris Wax and Saburá Extracts
The EEWMr achieved a yield of 19.47%, while the EEPMr achieved a yield of 11.77%, as shown in Calculations 2 and 3.

Minimum Inhibitory Concentration (MIC)
When evaluating the minimum inhibitory concentrations of bee products, it was observed that the MIC values for pollen extract (EEPMr), wax extract (EEWMr) and honey (HMr) were equal to or greater than 1024 µg/mL against the multidrug-resistant strains S. aureus 10, E. coli 06 and P. aeruginosa 24, for both the standard and multidrug-resistant strains. These results indicate that the products did not show direct antibacterial activity at the concentrations tested, making it difficult to determine an exact MIC for them.
Modification of Standard Antibiotic Action
The assays on the modification of antibiotic action by combining Melipona rufiventris products with the multidrug-resistant strain Staphylococcus aureus 10 showed a reduction in the minimum inhibitory concentration (MIC) of the antibiotic norfloxacin. When combined with the ethanolic extracts of wax and pollen, the MIC was reduced from 128 µg/mL to 64 µg/mL. However, no change in the MIC was observed when combined with honey. For gentamicin, the results indicated that all three products (wax, pollen, and honey) were able to reduce the MIC from 64 µg/mL to 32 µg/mL. These data are presented in Table 3 and Figure 1.
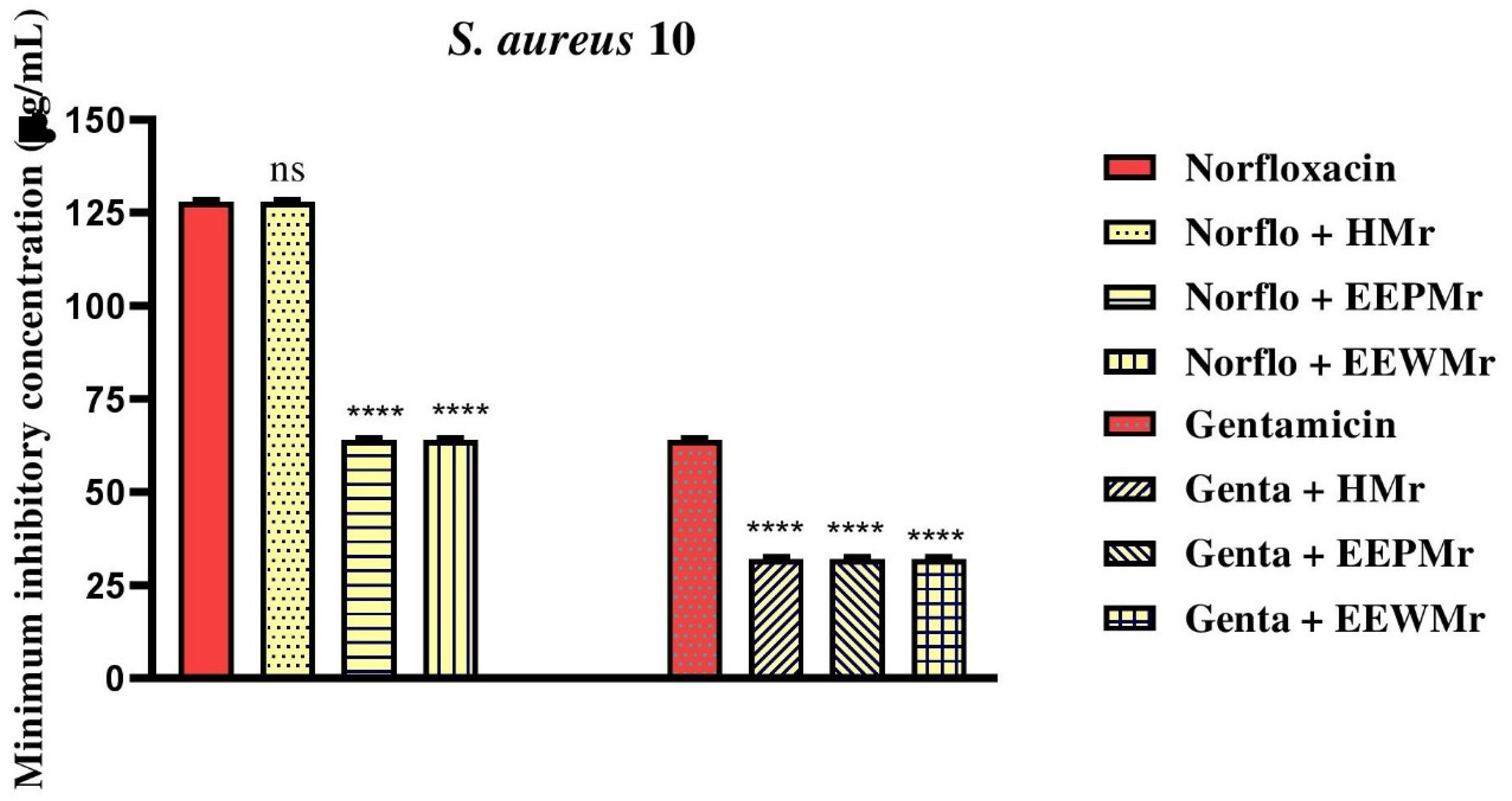
Evaluation of Antibiotic Potentiation by Melipona rufiventris Honey (HMr), Melipona rufiventris Ethanol Extract of Pollen (EEPMr), and Melipona rufiventris Ethanol Extract of Bee Wax (EEWMr) in the Bacterial Strain S. aureus 10. Genta: Gentamicin; Norflo: Norfloxacin; ****: P < .0001 versus Antibiotic Control; ns: Not Significant.
Minimum Inhibitory Concentration (MIC) Values (Expressed in µg/mL) of the Combinations of Melipona rufiventris Honey (HMr), Melipona rufiventris Ethanol Extract of Pollen (EEPMr), and Melipona rufiventris Ethanol Extract of Bee Wax (EEWMr) with Standard Antibiotics (Norfloxacin, Gentamicin, and Ampicillin) for the Multi-Resistant Strains S. aureus 10, E. coli 06, and P. aeruginosa 24.

****: P < .0001 versus antibiotic control; ns: not significant.
The association of Melipona rufiventris products with the standard antibiotic norfloxacin did not show any potentiation of antibacterial activity against the Escherichia coli 06 strain, as presented in Table 3 and Figure 2. However, when associated with gentamicin, the MIC was reduced from 16 µg/mL to 8 µg/mL with honey, and to 2 µg/mL with the ethanol extracts of bee wax (EEWMr) and pollen (EEPMr), indicating a threefold reduction in the MIC of gentamicin. These results are illustrated in Table 3 and Figure 2.

Evaluation of Antibiotic Potentiation by Melipona rufiventris honey (HMr), Melipona rufiventris Ethanol Extract Of Pollen (EEPMr), and Melipona rufiventris Ethanol Extract of Bee Wax (EEWMr) in the Bacterial Strain E. coli 06. Genta: Gentamicin; Norflo: Norfloxacin; ****: P < .0001 versus Antibiotic Control; ns: Not Significant.
The association of norfloxacin with the products evaluated in this study for the Pseudomonas aeruginosa 24 strain did not result in statistically significant changes. However, for gentamicin, a reduction in the MIC from 8 to 4 µg/mL was observed with the ethanol extracts of bee wax (EEWMr) and pollen (EEPMr), while honey (Melipona rufiventris) did not show any reduction in the MIC. These results are presented in Table 3 and Figure 3.

Evaluation of Antibiotic Potentiation by Melipona rufiventris Honey (HMr), Melipona rufiventris Ethanol Extract of Pollen (EEPMr), and Melipona rufiventris Ethanol Extract of Bee Wax (EEWMr) in the Bacterial Strain P. aeruginosa 24. Genta: Gentamicin; Norflo: Norfloxacin; ****: P < .0001 Versus Antibiotic Control; ns: Not Significant.
Beta-Lactamase Assays
The presence of the beta-lactamase enzyme was investigated by comparing the MIC of ampicillin alone and the MIC of ampicillin combined with sulbactam. Significant reductions in the MICs were observed: from 1024 to 64 µg/mL for S. aureus 10, from 64 to 4 µg/mL for E. coli 06, and from 512 to 2 µg/mL for P. aeruginosa 24, as shown in Table 3. These results indicate the presence of the beta-lactamase enzyme in the analyzed strains.
Regarding the combination of Melipona rufiventris extracts (EEPMr, EEWMr, and HMr) with ampicillin, no reduction in the MIC was observed for S. aureus 10, with all three products showing an MIC of 1024 µg/mL, as shown in Table 3 and Figure 4. However, for E. coli 06, a significant reduction was observed: the MIC of ampicillin was reduced from 64 µg/mL to 16 µg/mL with HMr and EEPMr, and to 8 µg/mL with EEWMr, as shown in Figure 5. These data suggest that the combination of M. rufiventris products with ampicillin may inhibit beta-lactamase activity.

Evaluation of Antibiotic Potentiation by M. rufiventris Honey (HMr), M. rufiventris Pollen Ethanol Extract (EEPMr), and M. rufiventris Bee Wax Ethanol Extract (EEWMr) in the Bacterial Strain S. aureus 10. Amp: Ampicillin; ****: P < .0001 versus Antibiotic Control; ns: Not Significant.
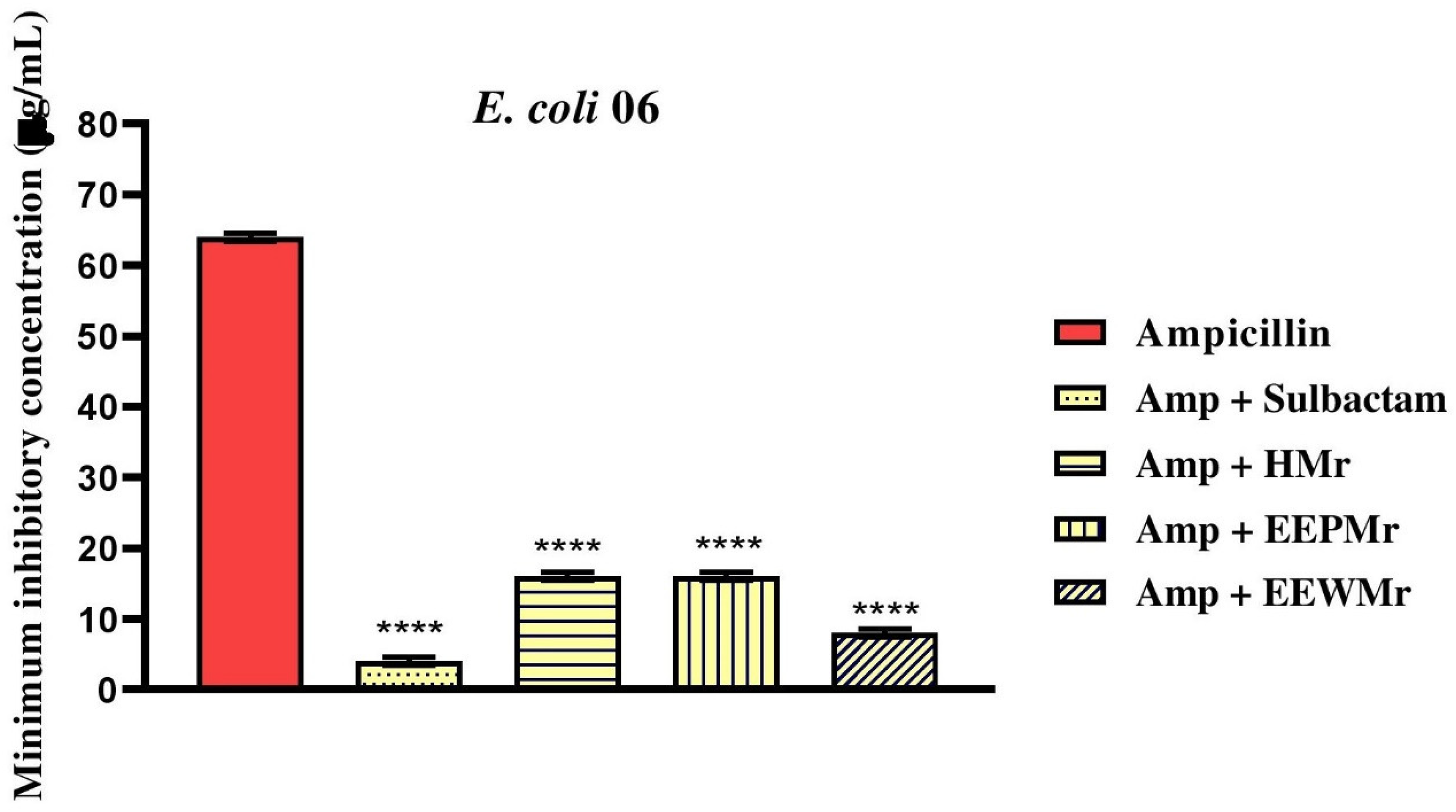
Evaluation of Antibiotic Potentiation by M. rufiventris Honey (HMr), M. rufiventris Pollen Ethanol Extract (EEPMr), and M. rufiventris Bee Wax Ethanol Extract (EEWMr) in the Bacterial Strain E. coli 06. Amp: Ampicillin; ****: P < .0001 versus Antibiotic Control; ns: Not Significant.
On the other hand, for P. aeruginosa 24, no change was observed in the MIC of the products HMr, EEWMr, and EEPMr when combined with ampicillin, remaining at 512 µg/mL for all cases, as shown in Figure 6 and Table 3.
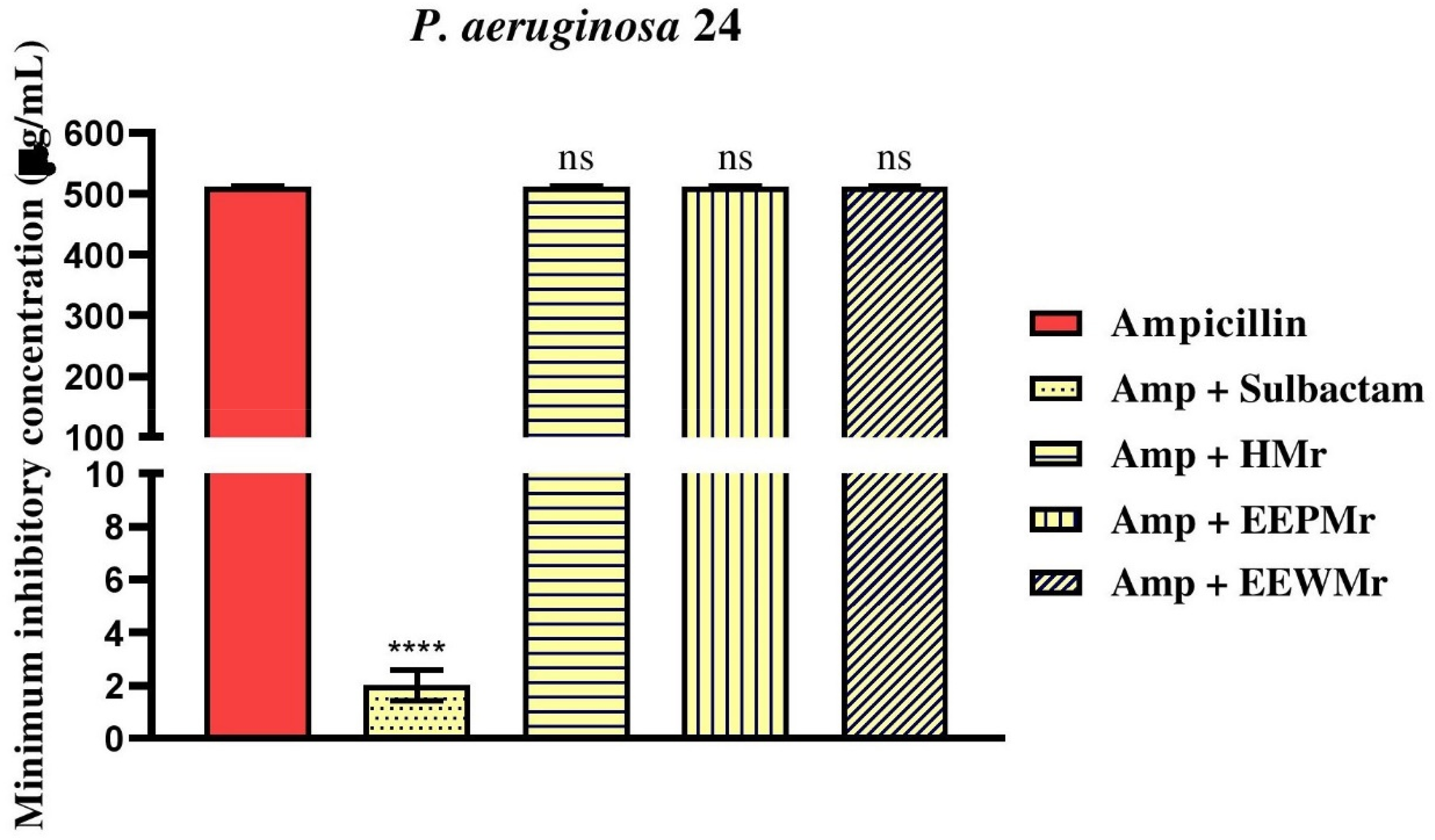
Evaluation of Antibiotic Potentiation by M. rufiventris Honey (HMr), M. rufiventris Pollen Ethanol Extract (EEPMr), and M. rufiventris Bee Wax Ethanol Extract (EEWMr) in the Bacterial Strain P. aeruginosa 24. Amp: Ampicillin; ****: P < .0001 versus Antibiotic Control; ns: Not Significant.
Discussion
The antibacterial activity of natural or synthetic compounds is commonly evaluated through minimum inhibitory concentration (MIC) assays, which determine the lowest concentration required to inhibit bacterial growth. 35 In the present study, the MIC results showed that the wax, fermented pollen, and honey extracts from Melipona rufiventris did not exhibit clinically relevant antibacterial activity against the tested strains. None of the products were effective in inhibiting the growth of the multi-resistant strains S. aureus 10, E. coli 06, and P. aeruginosa 24, nor the standard strains S. aureus ATCC LB25923, E. coli ATCC LB25922, and P. aeruginosa ATCC 29336. 36
Honey is a viscous liquid synthesized by bees, obtained through the practice of meliponiculture since the early civilizations, for both food and medicinal purposes. In addition to honey, other products such as propolis and beeswax hold commercial and medicinal value due to the presence of compounds with bioactive properties, such as phenolic acids and flavonoids.37–39 Fermented pollen is characterized by a mixture of floral pollens collected by bees, combined with salivary enzymes and nectar. This material is stored in the hive in pollen pots coated with wax and can be used as a dietary supplement. 40
This is the first study to evaluate both the direct action and the antibiotic combination of Melipona rufiventris wax, fermented pollen, and honey extracts against the multi-resistant strains S. aureus 10, P. aeruginosa 24, and E. coli 06, as well as the standard strains S. aureus ATCC LB25923, E. coli ATCC LB25922, and P. aeruginosa ATCC 29336. Literature reports the action of the ethanolic extract of Melipona rufiventris geopropolis against the S. aureus ATCC 12692 strain. The study demonstrated a bacterial growth inhibition capacity of 70.9%, and this inhibition was likely related to the presence of compounds such as para-hydroxybenzoic acid and gallic acid, with the latter being capable of interfering with the bacterial cell membrane, making it more susceptible to antibiotics. 23 Dos Santos et al, 41 evaluated the ethyl acetate extract of M. subnitida geopropolis against S. aureus 10, E. coli 06, and P. aeruginosa 26, reporting a MIC of 128, 85, and 512 µg/mL for S. aureus 10, E. coli 06, and P. aeruginosa 24, respectively.
Santini et al, 27 characterized M. rufiventris propolis using Ultra-High Performance Liquid Chromatography – High-Resolution Mass Spectrometry (UHPLC-HRMS), revealing the presence of gallic acid, ellagic acid, p-coumaric acid, and naringenin. The acids, gallic and ellagic, do not exhibit direct antibacterial activity against S. aureus IS-58 and K2068 strains (MIC ≥ 1024 µg/mL), but they reduce the MICs of tetracycline and ciprofloxacin for the tested strains. Reductions of 256 to 128 µg/mL were observed for tetracycline associations, and for ciprofloxacin, the reductions were from 161.27 to 128 µg/mL. 6
Santini et al, 27 also evaluated the ethanolic extract of M. rufiventris propolis against Gram-positive and Gram-negative bacterial strains. The results showed that the extract had significant activity against Gram-positive bacteria, which may be associated with the presence of an additional membrane in Gram-negative bacteria, making it more difficult for the compounds to enter the bacterial cell. The extract showed significant activity (<100 µg/mL) against the standard strains Bacillus cereus (ATCC 11778) and S. aureus (ATCC 6538), and moderate activity (500 > 100 µg/mL) against S. epidermidis (ATCC 12228), Listeria monocytogenes, and Streptococcus mutans (ATCC 25175). However, these results were not replicated for the standard strains S. aureus ATCC LB25923, E. coli ATCC LB25922, and P. aeruginosa ATCC 29336, which were studied in the present work.
Combinatory compound assays are used to evaluate the capacity of these compounds to potentiate or not the action of standard antibiotics by modifying their MIC at a sub-inhibitory concentration (MIC/8) of the compound. In some cases, such combinations can be beneficial, lowering the antibiotic concentration required to inhibit bacterial growth; in other cases, the combination becomes harmful, as the MIC increases and a higher concentration is needed to inhibit growth due to the association with the evaluated compound.32,42,43
Thus, the antibiotic combination with the M. rufiventris products EEWMr and EEPMr for S. aureus 10 showed a positive MIC modification of norfloxacin by both extracts, whereas pure honey was unable to modify the MIC of the evaluated antibiotic, reducing it by half. For gentamicin, a reduction from 64 to 32 µg/mL was observed for EEWMr, EEPMr, and HMr. Regarding the ampicillin combination with M. rufiventris products, no statistically significant differences were observed from the antibiotic control, suggesting that these products do not act on the beta-lactamase resistance mechanism present in S. aureus 10.
Bacterial strains that carry the enzymatic resistance mechanism and produce the beta-lactamase enzyme can resist beta-lactam antibiotics such as penicillins, cephalosporins, cefamycins, and carbapenems. This enzyme degrades the beta-lactam ring found in this class of antibiotics, which is responsible for the drug's effectiveness. To reverse this resistance, inhibitors of this enzyme, such as Sulbactam (a standard beta-lactamase inhibitor), are used.9,44
The combinations for E. coli 06 showed that the combination of norfloxacin with EEWMr, EEPMr, and HMr did not result in a reduction in the MIC of this antibiotic, indicating that the extracts were unable to modify its action against the tested strain. In contrast, combinations with gentamicin resulted in a MIC reduction by half for MMr and a threefold reduction for EECMr and EEPMr. These data suggest that the products have a good potential to enhance the action of gentamicin against the tested strain.
Other positive modifications for E. coli were also observed with the combination of ampicillin with the products. The MIC was reduced twofold when combined with MMr and EEPMr, and threefold in combination with EECMr. These results were close to the control, where ampicillin was combined with the beta-lactamase inhibitor Sulbactam, which showed a MIC of 4 µg/mL (a fourfold reduction). The reduction observed in the ampicillin-Sulbactam combination highlights the presence of beta-lactamase, while the data from the combinations with the extracts and honey suggest a possible action of stingless bee products on the beta-lactamase enzymatic mechanism present in the strain.
The findings of Santini et al, 27 as well as the results obtained in our study for the E. coli 06 strain, suggest a promising scenario for the use of M. rufiventris products as inhibitors of the beta-lactamase enzymatic resistance mechanism, present in both Gram-negative and Gram-positive strains. This becomes even more evident when these products are combined with beta-lactam antibiotics. The same study observed a significant reduction in the MIC of amoxicillin (a beta-lactam antibiotic) when combined with propolis extract, reducing from 0.3125 to 0.078 µg/mL against the S. aureus (ATCC 6538) strain. 27
For P. aeruginosa 24, only the fermented pollen and wax extracts were able to modify the MIC of gentamicin, reducing it by half. In contrast, honey, when combined with gentamicin, did not show a MIC different from the positive control. None of the products (HMr, EEPMr, and EEWMr) modified the MIC when combined with norfloxacin and ampicillin. These results suggest that the products did not exhibit activity against P. aeruginosa 24, except for the extracts in combination with gentamicin. Additionally, no action was observed on the beta-lactamase enzymatic mechanism.
The ability to modify the MIC of norfloxacin, gentamicin, and ampicillin by the ethyl acetate extract of M. subnitida geopropolis was presented in the work by Dos Santos et al, 41 where the authors reported a reduction in the MIC of ampicillin from 256 to 192 µg/mL and of gentamicin from 64 to 16 µg/mL, and for norfloxacin, the MIC increased from 170 to 512 µg/mL against S. aureus 10. For P. aeruginosa 24, reductions were observed only for gentamicin (from 32 to 16 µg/mL) and norfloxacin (from 64 to 8 µg/mL), while in E. coli 06, reductions were observed for all three antibiotics, from 84 to 16 µg/mL for the ampicillin combination, from 13 to 1 µg/mL for gentamicin, and from 426 to 64 µg/mL for norfloxacin. The data obtained for the ampicillin antibiotic were similar to our results for the E. coli strain.
This is a preliminary study investigating the direct activity against standard and multi-resistant strains of S. aureus, E. coli and P. aeruginosa, as well as the association with standard antibiotics. It therefore has some limitations that should be addressed in future research. Studies using bacterial strains that carry other resistance mechanisms are needed to elucidate the possible adjuvant potential. In addition to chemical prospecting aimed at identifying the composition of the extracts and honey and their possible bioactive compounds, in vivo and in silico tests or toxicological tests. The lack of direct action shown by the extracts and honey of M. rufiventris should not be considered a limitation, since compounds that do not show direct activity are excellent candidates for adjuvants in the inhibition of efflux pumps, a potential only shown in associations with antibiotics. 45 Therefore, these results are in line with the aim of the study to investigate the action of natural products in the treatment of infections through antibiotic association, without interfering with their direct action.
Conclusion
This study represents a novel approach in evaluating the antibacterial activity of honey, fermented pollen, and beeswax from Melipona rufiventris against multiresistant and standard strains. Although these products did not exhibit clinically relevant antibacterial activity when tested in isolation, M. rufiventris products demonstrated significant potential in modulating the activity of specific antibiotics. The fermented pollen and beeswax extracts consistently reduced the minimum inhibitory concentration (MIC) of gentamicin for the three multiresistant strains tested, while honey displayed more limited, but still relevant, activity, especially in combination with ampicillin against E. coli. These results suggest that M. rufiventris products may function as modulators of antibiotic activity, with particular emphasis on their potential interference with the beta-lactamase enzymatic mechanism. The reduction in MIC observed indicates a potentiating effect of these products when combined with beta-lactam antibiotics, aligning with previous findings that point to the presence of bioactive compounds, such as phenolic acids and flavonoids, in stingless bee products. However, the lack of significant activity against P. aeruginosa and the limitations in modifying the MIC of other antibiotics, such as norfloxacin, highlight the need for further studies to elucidate the molecular mechanisms at play.
These findings are preliminary and, therefore, require additional validation. Further research should include the chemical characterization of the active compounds, toxicity studies, and in vivo assays. Additionally, expanding the study to assess the potential of these products against other bacterial strains and resistance mechanisms is essential. These efforts will be crucial for the future development of M. rufiventris products as adjuvants in antibiotic formulations, supporting the fight against antimicrobial resistance.
Footnotes
Acknowledgements
We would like to express our gratitude to Mr Chagas and his wife Celene, as well as to Valmir, the owners of Recanto Das Abelhas, Crato – Ceará, for providing the bioproducts.
Ethics Considerations
No human or animal subjects were involved in this study.
Funding
We also thank the Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (FUNCAP) and BPI BP5-0197-00174.01.00/22 Edital/Chamada: Bolsa de Produtividade em Pesquisa, Estímulo à Interiorização e Inovação Tecnológica – BPI 04/2022. We appreciate the support provided to the PDJ 2023 project – Chamada CNPq N° 32/2023 - Pós-Doutorado Júnior, process 172358/2023-0, and the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). Hélcio Silva dos Santos acknowledges financial support from the PQ/CNPq (Grant#: 306008/2022-0), FUNCAP (Grants#: ITR-0214-00060.01.00/23, UNI-0210-00337.01.00/23, FPD-0213-00088.01.00/23).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All datasets generated for this study are included in the manuscript.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This research did not involve human participants, human tissue, or any procedures requiring human subjects. Additionally, animal experiments were not conducted in this study.
Use of AI Tools Declaration
The authors declare they have not used Artificial Intelligence (AI) tools in the creation of this article.

