Abstract
The present study aimed to explore the therapeutic effects of carvacrol on the ulcerative colitis (UC) inflammation model induced by DSS (dextran sulfate sodium) in rats. Forty rats were randomly divided into the blank, model, low-, middle-, and high-dose carvacrol groups. The rats drank 4.5% DSS for 5 consecutive days to induce the UC model, except for the blank group. Different concentrations of either carvacrol or purified water were supplied by intragastric administration for 7 consecutive days to assess the protective effect of carvacrol on inflammation in UC rats involving the TLR4/NF-κB signaling pathway. In vivo administration of carvacrol was found to ameliorate the symptoms of colitis in rats. Carvacrol significantly improved the pathological symptoms of colitis in the model group and reduced the DAI and pathological scores of rats. At the same time, carvacrol can reduce the expression of IL-6 and TNF-α induced by DSS, inhibit the activation of TLR4, hinder the nuclear translocation of NF-κB (P65), reduce the cleavage of the apoptosis-related protein cleaved-caspase 3, and protect colon cell apoptosis. The results of in vitro experiments showed that carvacrol has a significant inhibitory effect on cell apoptosis caused by the activation of TLR4 induced by LPS and pcDNA3.1tlr4. Research results revealed that carvacrol has a certain protective effect against inflammation-induced injury in ulcerative colitis rats via inhibiting the activation of the TLR4/NF-κB signaling pathway.
Introduction
Ulcerative colitis (UC) is a nontransmural, recurrent inflammatory disease (IBD) confined to the colon, and is associated with abdominal pain, diarrhea, mucopurulent stools, and postacute symptoms.1,2 In the past 20 years, the incidence and prevalence of UC in Asian countries have been increasing year by year, and UC is a high-risk factor for colorectal cancer.3,4 The disease is difficult to be cured, easy to recur, and tends to become cancerous, which seriously affects the quality of life and has been classified by WHO as one of the modern intractable diseases. Finding new effective colitis treatment drugs and methods is a thorny problem that needs to be solved urgently.
Chinese herbal medicines and their monomeric active substances have significant curative effects in the treatment of ulcerative colitis, with a low recurrence rate, good long-term curative effect, few side effects, and broad research prospects. 5 Carvacrol has shown good anti-inflammatory effects. For example, in interleukin 1β (IL-1β)-induced osteoarthritis, carvacrol can inhibit nitric oxide (NO) and prostaglandin E2 (PGE2). 6 Carvacrol can improve the memory impairment of Parkinson's rats through its antioxidant effect. 7 Carvacrol also significantly downregulated TNF-α, IL-1β, and PGE2 and significantly increased TGF-1β, thereby inhibiting the inflammatory response and improving gouty arthritis. 8 Because of the good anti-inflammatory activity of carvacrol, we have reason to speculate that it may have a therapeutic effect on UC.
The TLR4/NF-κB pathway has been widely reported in the research of ulcerative colitis. Toll-like receptors (TLRs) are a type of pattern recognition receptors widely distributed on the surface of immune cells.9,10 Ten TLRs have been reported on mammalian cell membranes, among which TLR4 is considered to be most closely related to immune and inflammatory functions. 11 Activated TLR4 can activate the transcription of NF-κB (p65) to the nucleus, promote the transcription of varieties of inflammatory factors, and cause colonic mucosal damage. 12 TLR4 and NF-κB are lowly expressed in the normal colonic mucosa, while they are overexpressed in UC colonic mucosa, suggesting that TLR4 receptors and their signaling pathways may be an important link in the pathogenesis of UC. 13 At the same time, LPS induces the onset of UC and triggers the secretion of cytokines IL-1β, IL-6, and TNF-α, which leads to the destruction of the intestinal mucosal barrier. 14 These factors further induce cellular immunity, leading to the continuous development of inflammation. However, the effect of carvacrol on ulcerative colitis is still unclear. This study intends to explore the influence and mechanism of carvacrol on ulcerative colitis based on the classic inflammation pathway TLR4/NF-κB.
Experimental
Animals and Grouping
Forty SD rats were purchased from the Chengdu Dashuo Experimental Animal Co. Ltd (certificate No. SCXK201302, Chengdu, Sichuan, China). The animal experiments were approved by the Ethics Committee of the Chengdu University of TCM. Rats were housed in polycarbonate cages in an animal laboratory with a 12 h light/12 h dark cycle under conditions of controlled temperature (25 ± 1 °C) and in a humidified atmosphere (50 ± 10%). Following a week of adaptation, rats were randomly divided into blank, model (4.5% DSS), low- (25 mg/kg), middle- (50 mg/kg), and high-dose carvacrol group (100 mg/kg). The rats in the blank group received a basic diet and ready access to clean drinking water. The rats in other groups were given 4.5% DSS to drink freely for 5 consecutive days. After the successful model building, the rats in blank and model groups received 0.5 mL saline by intragastric administration, and the rats in the carvacrol groups received an equal volume of 25, 50, and 100 mg/kg carvacrol intragastrically once a day for 7 consecutive days. After completing the experiment, the animals were injected with 1% sodium pentobarbital (50 mg/kg) and sacrificed by carbon dioxide inhalation to obtain tissue and blood.
Hematoxylin/Eosin (H&E) Assays
In a parallel animal assay (6 rats per group), the colitis model establishment and drug treatment were the same as described above. On the 12th day, rats were euthanized to collect colon tissue, which was fixed with 4% formaldehyde, and embedded in paraffin. Tissue sections (5 μm in thickness) were prepared according to standard protocols for hematoxylin/eosin (H&E) staining.
TUNEL Alexa Fluor 488 Staining
The section of colon tissue was fixed with 4% paraformaldehyde for 30 min. Then, slips were treated, respectively, with 0.1% Triton X-100 to permeabilize for 20 min and 5% goat serum to block for 30 min. Tissue sections were stained with Alexa Fluor 488 at 37 °C for 30 min in the dark. Then, the tissue was washed with PBS. Cell nuclei were counterstained with DAPI staining solution. After mounting, the slides were observed and pictured through an Olympus BX51 fluorescence microscope (Olympus, Japan).
Western Blot
Proteins were extracted using RIPA Lysis Buffer (Shanghai Weiao Biotechnology Co., Ltd, China), and their concentrations were measured using a BCA protein reagent kit (Beijing Solarbio Science & Technology Co., Ltd, China). The p65 protein in the nucleus was extracted according to the instructions with the Nuclear Protein Extraction Kit (R0050, Solarbio), with protein H3 as an internal reference for nuclear proteins. Proteins (30 μg) were separated on SDS-PAGE gels (8%), transferred onto a polyvinylidene difluoride (PVDF) membrane, and blocked with 5% skim milk for 1 h at room temperature. The PVDF membranes were incubated with anti-NF-κB p65 (AN365, Beyotime) (1:400), anti-cleaved-caspase3 (ab32042, Abcam) (1:400), anti-H3 (ZIKER-0349R, ZIKER BIOTECHNOLOGY) (1:400), anti-TLR4 (ab22048, Abcam) (1:400) and anti-β-actin (K200058M, Solarbio) (1:1000) primary antibody solution (BOSTER Biological Technology Co., Ltd, China) overnight at 4 °C. After incubating with horseradish peroxidase-conjugated goat anti-rabbit secondary antibody or goat anti-mouse secondary antibody (BOSTER Biological Technology Co., Ltd, China) at room temperature for 1 h, the membranes were reacted with an ECL hypersensitive chemiluminescence kit (Shanghai Weimao Biotechnology Co., Ltd, China) according to the manufacturer's protocol.
Cell Proliferation Assay
Rat colonic mucosal epithelial cells (RCME), purchased from YIPU BIOLOGY (CP-R170, Wuhan, China), were cultured in DMEM/high glucose medium (Hyclone, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, USA) and 1% glutamine (Sigma, USA), and incubated at 37 °C. Cell proliferation was measured using a Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan). Cells were seeded in a 96-well plate at a density of 6 × 104/mL. When the cells reached 60% to 70% confluence, they were incubated with either carvacrol (10 μM) or pcDNA3.1tlr4 plasmid (100 μg/mL) for 48 h. After the treatment, the original medium was aspirated and discarded, and fresh medium and CCK-8 at a ratio of 10:1 (100 µL) were added to each well, and incubation was continued for 1 h. The absorbance (
Detection of Apoptosis by Flow Cytometry
RCME cells were evenly spread at the bottom of each well of a 6-well plate at a density of 5 × 104/well and cultured for 24 h with DM/F12. When cell confluence reached 70%, the cultures were treated with different drugs for 48 h. After treatment, the cells were digested with trypsin, washed twice with pre-cooled PBS, and centrifuged at 4 °C for 5 min. Then, 5 μL of annexin V-FITC and 5 μL of propidium iodide (PI) solutions were added to each tube. After mixing, the cells were incubated in the dark at room temperature for 15 min. Apoptosis was determined with a flow cytometer and calculated after analysis of the data (Becton Dickinson).
Statistical Analysis
All data were analyzed by one-way analysis of variance and Duncan's test using the SPSS 19.0 package (SPSS Inc. Chicago, IL, USA). All results are shown as the means ± standard deviation, and
Results
Protective Effect of Carvacrol on DSS-Induced Colitis Rats
As shown in Figure 1A, compared with the blank group, the colon length of the model group was significantly shortened; however, treatment with 50 and 100 mg/kg carvacrol significantly reduced the shortening of the colon length. The disease activity index (DAI) score was determined by weight loss, stool consistency, and stool bleeding. As shown in Figure 1B, the DAI score of rats treated with 50 and 100 mg/kg carvacrol was significantly lower than that of the DSS alone group, and the DAI score of the blank group was zero. As shown in Figure 1C, there was no significant difference in the body weight of rats before the experiment. After finishing the experiment, compared with the model group, the weight loss of the rats treated with 100 mg/kg of carvacrol was significantly reduced.

Evaluation of enteritis in rats. (A) A schematic presentation of experimental design, structure of carvacrol, and colon length of rats. (B) Disease activity index scores in each group. (C) Effects of carvacrol on body weight. *
Effect of Carvacrol on Pathological Changes and Inflammatory Factors in Colon Tissue of Rats With Ulcerative Colitis
The colon tissue of rats in each group was stained with HE, and the intestinal mucosal injury and inflammation were observed under a microscope. As shown in Figure 1A, the colon mucosal epithelial tissue of the blank group is complete, the structure is clear, the glands are intact, and there is no disease. In the model group, the colon mucosa was extensively absent. Most of the glands were incomplete, and inflammatory cells infiltrated extensively into the muscle layer, showing typical inflammatory changes, and the degree of inflammation is very serious. In the carvacrol intervention group, mucosal damage of colon tissue was mild, inflammatory cell infiltration and diffusion were rare, the muscle layer was slightly thickened, and the degree of inflammation was mild. Tunel staining results showed that there was obvious apoptosis in the colon tissue of the model group. In the carvacrol high-dose treatment group, the apoptosis of colon cells caused by DSS almost disappeared, indicating that carvacrol can attenuate DSS-induced cell apoptosis. As for inflammatory factors, there was no significant difference in TNF-α and IL-6 between the blank group and the carvacrol high-dose group. However, the model group and the carvacrol high-dose group were significantly different (

Effects of carvacrol on histochemistry and inflammatory factors in rats with colitis. (A) Images of colon tissue following hematoxylin-eosin (H&E) staining. (B) Images of colon tissue following TUNEL staining. (C) The relative levels of TNF-α and IL-6 in serum. *
Effect of Carvacrol on Protein Expression of TLR4/NF-κB In Vivo
In this study, Western blotting was used to detect TLR4, NF-κB (p65), and cleaved-caspase1 protein expression in rat colon tissues. As shown in Figure 3A to D, compared to the blank group, TLR4, P65 in the nucleus, and cleaved-caspase1 protein expression levels significantly increased in rat colon tissues in the model group. However, the expression of these proteins activated by DSS had been significantly inhibited in the carvacrol high and medium-dose treatment groups.
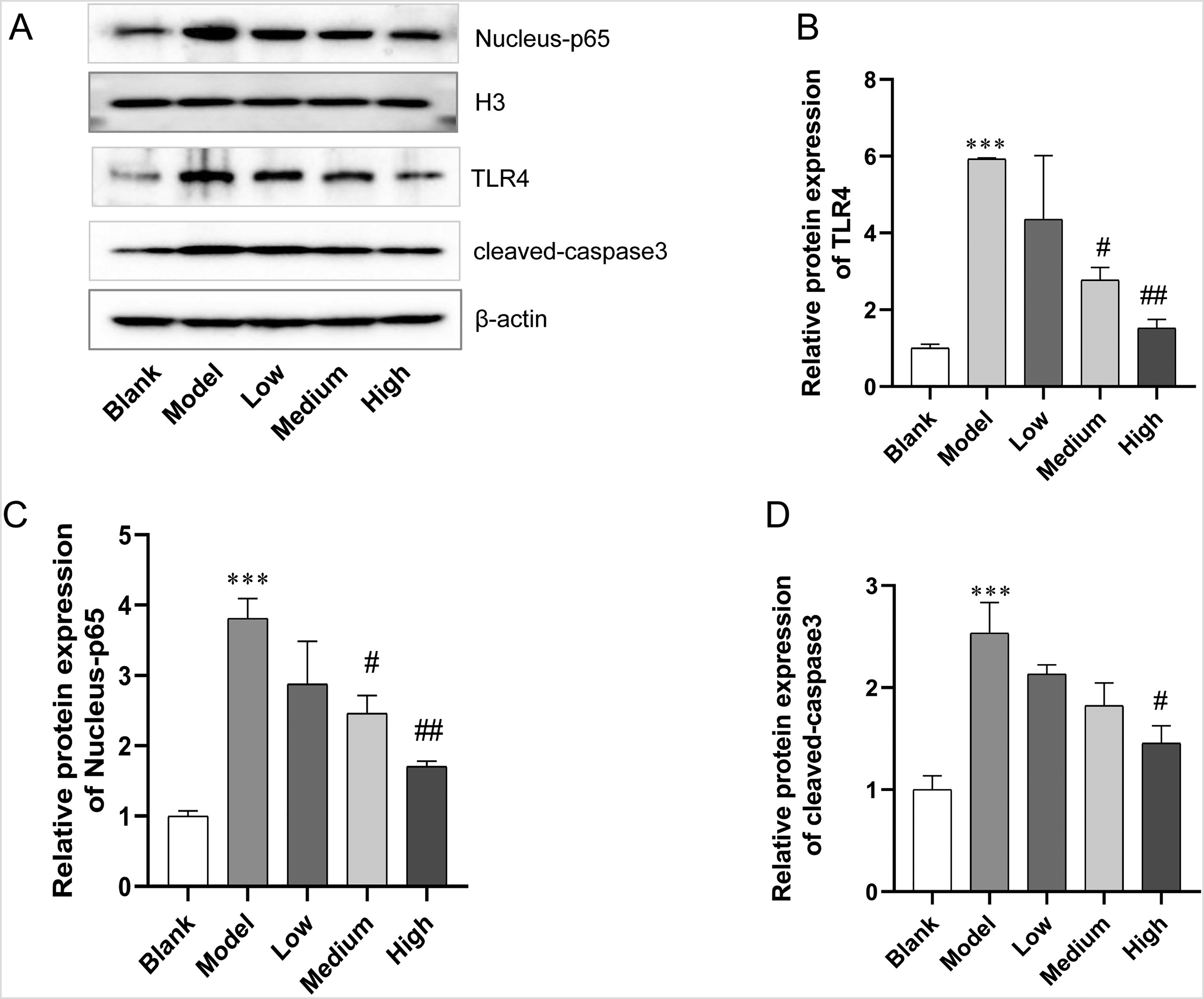
Effect of carvacrol on protein expression of TLR4/NF-κB in vivo. (A) The images of TLR4/NF-κB pathway-related proteins TLR4, Nucleus-p65, and cleaved-caspase3 in colon tissue by Western blot. (B) Relative expression of TLR4. (C) Relative expression of Nucleus-p65. (D) Relative expression of cleaved-caspase3. ***
The Biological Effects of Carvacrol and TLR4 on Colonic Mucosal Epithelial Cells (RCME)
LPS can induce UC pathogenesis and trigger cells to secrete cytokines IL-1β, IL-6, and TNF-α leading to intestinal mucosal damage. Therefore, to clarify this speculation, we investigated whether LPS is involved in the TLR4-mediated cell biological behavior of colon cancer cells. To verify further the role of TLR4 in the colon, after transfection with pcDNA3.1tlr4 plasmid, compared with the blank group cells, the level of TLR4 was significantly increased (Figure 4A). Colon cancer cell proliferation, apoptosis, and expression of the inflammatory factors IL-6 and TNF-α were examined in each group. MTT results showed no significant toxicity of carvacrol on RCME cells. MTT and flow data analysis showed that transfection of pcDNA3.1tlr4 and LPS-treated cells decreased viability and increased apoptotic cells compared to the blank group and that carvacrol treatment effectively reversed these responses. As for inflammatory factors, there was no significant difference in TNF-α and IL-6 between the LPS group and the pcDNA3.1tlr4 group, which were significantly higher than the levels in the blank group (Figure 4G). However, the carvacrol treatment group showed significantly reduced levels (

The biological effects of carvacrol and TLR4 on colonic mucosal epithelial cells (RCME). (A) The images of proteins TLR4 were activated by pcDNA3.1tlr4. (B) Relative expression of TLR4. (C) Cytotoxicity of carvacrol to RCME. (D) The effect of TLR4 and carvacrol on the survival rate of RCME cells. (E) Apoptotic rate of RCME cells. (F) The result of apoptotic rate. (G) The relative levels of TNF-α and IL-6 of RCME cells. *
Effect of Carvacrol on Protein Expression of TLR4/NF-κB In Vitro
To verify further the effect of the TLR4/BNFKB pathway on the colon, the effects of carvacrol on the protein of TLR4 and NF-κB in vitro were carried out. As shown in Figure 5, LPS and pcDNA3.1tlr4 can significantly activate the expression of TLR4, and NFκB/p65 moves to the nucleus, which in turn leads to capase1 cleavage activation, and this reaction can be offset by carvacrol. This observation indicates that carvacrol inhibits the apoptosis of colon cancer cells through TLR4/BNFKB inhibition (Figure 5).
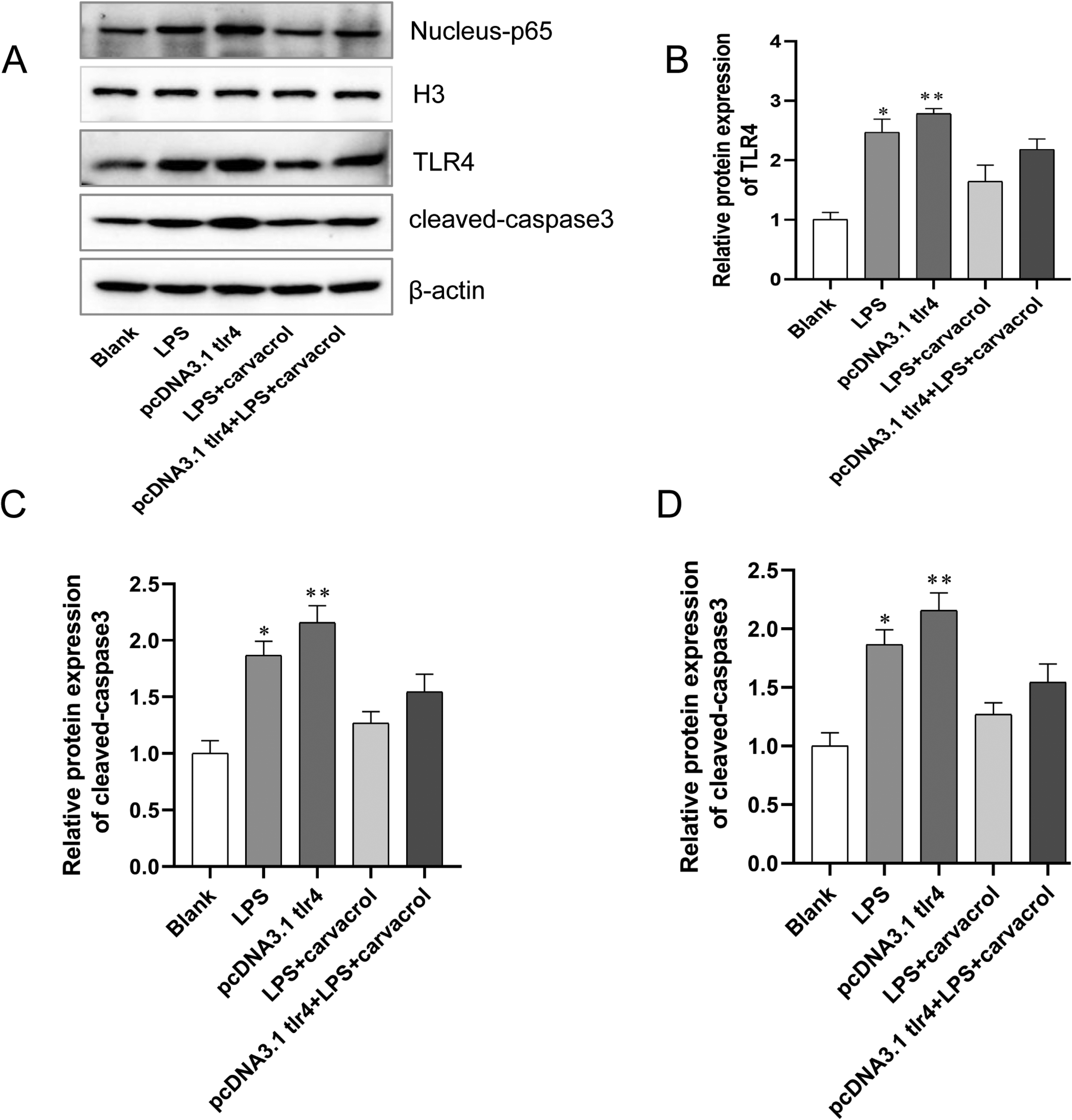
Effect of carvacrol on protein expression of TLR4/NF-κB in vitro. (A) The images of TLR4/NF-κB pathway-related proteins TLR4, Nucleus-p65, and cleaved-caspase3 in colon tissue by Western blot. (B) Relative expression of TLR4. (C) Relative expression of Nucleus-p65. (D) Relative expression of cleaved-caspase3. *
Discussion
The onset and progression of UC are related to a variety of risk factors, among which inflammation plays an important role in the progression of UC. Carvacrol plays an active role in the treatment of arthritis diseases. 5 Therefore, this study hypothesizes that carvacrol can reduce the inflammatory response in the intestine of rats with DSS-induced colitis and colon cell apoptosis, thereby controlling the progression of colitis. In the end, the present study revealed that carvacrol can protect against inflammation-induced injury by inhibiting the activation of the NF-κB signaling pathway in UC rats.
UC is a chronic disease that is currently being studied. The establishment of the UC model is the first step in the study of physiology and pathology. At present, chemical induction is a commonly used modeling method in the research of basic drugs for UC, and the DSS-induced model is the most widely used.15,16 Therefore, the DSS-induced UC model was chosen for research to evaluate the therapeutic effect of carvacrol on UC. The change in animal behavior is a sign to evaluate whether the model is a success, and it is also a standard to evaluate whether the drug is effective. In this experiment, the DAI score of rats decreased after DSS induction. It manifested as decreased voluntary activity, loss of appetite, loose stools, and mucous blood in the stool, and weight loss. Carvacrol can significantly restore the colon length in colitis rats. Histopathological observation showed that the degree of colonic necrosis was reduced, the infiltration of inflammatory cells was reduced, and the area of ulcers was significantly reduced. At the same time, carvacrol also decreased the colon cell apoptosis induced by DSS. These studies indicated that carvacrol can significantly improve experimental ulcerative colitis.
Inflammation is closely related to the occurrence and development of colitis. 17 Previous research reports showed that carvacrol could ameliorate thioacetamide-induced hepatotoxicity by abrogation of oxidative stress, inflammation, and apoptosis in the liver of Wistar rats. 18 IL-6 is an important factor in the inflammatory response, with the ability to activate and induce T and B cell differentiation, enhance NK cells, and promote phagocytosis. 19 As an acute-phase response factor, it participates in colon tissue immunity, pathological response, and inflammatory process of colonic tissues, leading to damage of colonic tissues. 20 In this study, the level of TNF-α and IL-6 in the colon of UC model rats increased significantly. Both showed similar trends, but carvacrol treatment could reduce their content. Inflammatory stimuli induce macrophages to secrete TNF-α, which further activates macrophages and nuclear transcription factors such as NF-κB, thereby promoting the release of other inflammatory factors. 21
Apoptosis plays an important role in the late stage of inflammatory reaction, and TLR4/NF-κB is also a key signal to inhibit intestinal epithelial cell apoptosis.22,23 Studies have shown that TLR4 is involved in the signal pathway of NF-κB nuclear translocation induced by LPS stimulation. 24 Therefore, blocking the TLR4/NF-κB signaling pathway to down-regulate the levels of inflammatory factors and pro-apoptotic factors has become one of the ways to treat UC and prevent carcinogenesis. The green fluorescence of colon cells in the model group was significantly stronger than that in the blank group and carvacrol high-dose treatment group, which indicated that carvacrol can protect colon cells from apoptosis. LPS can activate the expression of TLR4 and increase the expression of cleaved-caspase3, a protein that promotes apoptosis. Flow cytometry results showed that compared with the carvacrol combination treatment group, the LPS group and pcDNA3.1tlr4 cell apoptosis increased. Western blot results showed that TLR4 protein and NF-κB in the nucleus were highly expressed in UC rats of the model group, which shows that the high expression of TLR4 and NF-κB is closely related to the pathogenesis of UC. The expression of TLR4 and NF-κB in the carvacrol high and medium dose groups was significantly lower than that in the model group, indicating that carvacrol has a significant effect on blocking the TLR4/NF-κB signaling pathway. Research reports have shown that the production of UC inflammatory factors is closely related to the TLRs/NF-κB pathway. 25 In inflammation and immune response, this pathway mainly plays a pivotal role in these reactions by regulating the expression of immune and inflammation-related factors and inflammatory transmitters. 26 NF-κB presents an over-activated state in UC. It can lead to the over-expression of cytokines and adhesion molecules, amplifying the inflammatory response. 27 Studies have found that NF-κB is a marker of intestinal mucosal infection. The content of NF-κB can reflect the severity of the disease and can also evaluate the treatment effect. 25
Conclusion
In summary, the present study revealed that carvacrol plays a protective role against inflammation-related injury in UC rat and colonic mucosal epithelial cells via inhibiting the activation of the TLR4/NF-κB signaling pathway to reduce inflammation, enhance the viability, and attenuate apoptosis. This study may provide a potential therapeutic strategy for future treatment of colitis, and carvacrol may improve the outcome of colitis. However, the mechanism of carvacrol in the treatment of ulcerative colitis in this article is not deep enough, and further experiments are needed to prove it.
Footnotes
Authors’ Note
Study on the curative effect and social benefit of acupoint catgut embedding therapy on ulcerative colitis based on the theory of “Shu Mu points matching.”
Author Contributions
Du Qiang conceived and the study, Liu Miao, Mao gang, Zhou Xuelei, Wan Xuemei, Zhang Fan, Dai Li, Chen Ya, Dai Ning, Zhang Yan conducted the experiments and analyzed the data, Du Qiang wrote the manuscript, all authors read and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of the Chengdu University of TCM.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
