Abstract
Introduction
The gastrointestinal tract (GIT) is constantly at risk because of exposure to numerous pathogens and toxins of food and environmental origins.1,2 Ulcerative colitis (UC) is one of the most common GIT disorders. 3 UC is an inflammatory bowel disease (IBD) characterized by weight loss, severe abdominal pain, dehydration, anemia, fever, and colon ulceration. This ulceration makes the GIT unable to absorb nutrients and finally leads to bloody diarrhea and mucus. 4 UC is life-threatening because once ulceration becomes severe, it might develop into colorectal cancer. 5
The pathogenesis of UC involves several pro-inflammatory mediators and cytokines, for instance, tumor necrosis factor-α (TNF-α), interleukin (IL)-1β, IL-6, and nitric oxide (NO).6,7 NO plays an important role in antibacterial action and tumor removal in the body, but NO produced by inducible nitric oxide synthase (iNOS) during inflammation promotes inflammatory reactions such as vascular permeability, edema, and promotes biosynthesis of inflammatory mediators to worsen inflammation.8,9 It has been reported that iNOS is involved mainly in the inflammatory response and is expressed when stimulated with lipopolysaccharide (LPS). 10 LPS stimulates innate immune cells such as macrophages and neutrophils, which produce proinflammatory molecules such as IL-1 and TNF, as well as free radicals, resulting in significant sequel inflammation. These disorders result in the destruction of the ultrastructure of colon tissue, manifested by swelling of the mucosa and eruption of the epithelium.11,12 Pro-inflammatory cytokines mentioned above, and NO are important indicators of colitis, which themselves cause oxidative stress. Hence, reducing these inflammatory mediators is the role of antioxidation. RAW 264.7 macrophage cells have been proven to be activated by LPS and release NO, making them the best model for studying inflammation. 13
Polyphenols are known to relieve colitis and prevent colon cancer caused by colitis.14,15 Polyphenols inhibit macrophages and reduce the expression of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. 16 Flavonoids are polyphenolic compounds that have therapeutic effects on IBDs. 17 Chlorogenic acids, which can be measured using high-performance liquid chromatography (HPLC), are plant polyphenols that inhibit the synthesis of pro-inflammatory cytokines such as IL-1β. 18 Chlorogenic acid functions as an antioxidant by lowering IL-1β.
Methods
Preparation of A. elata Extract
AE was purchased from a local market Sunchang-gun, Jeollabuk-do, Republic of Korea, in May 2020, and the voucher specimen (No. 14100691) was deposited at the Department of Biotechnology, Jeonbuk National University, Iksan, Republic of Korea. The plant was washed with tap water to remove foreign substances, sliced into 2 mm thick pieces, and dried at 60 °C. The dried material was pulverized with a grinder (Philips, HR3752/00), passed through a 20-mesh sieve (0.841 mm), and stored at 4 °C for further use. The dried powder (1 kg) was extracted with distilled water at 80 °C for 5 h. The extract was centrifuged for 10 min (3000 ×
Total Polyphenol Content
To estimate the total polyphenol content in AE extract, we adopted the method of Folin-Denis. 25 In brief, 0.5 mL of 1 N Folin-Ciocalteu's reagent (Cat No. F9252, Sigma) was added to 1 mL of a diluted sample (Conc. 10 mg/mL) and mixed thoroughly. Next, 1 mL of 5% Na2CO3 (Cat. No. 222321, Sigma) was added to the mixture before incubation in the dark for 1 h, and the absorbance at 725 nm was measured using a spectrophotometer (Agilent, Agilent 8453). The total polyphenol content of the extract was expressed as mg gallic acid (Sigma, St. Louis) equivalents (GAE)/g.
Total Flavonoid Content
Moreno's technique was used to determine the total flavonoid content of AE. 26 Simply, a mixture was made of 0.5 mL of the extract (10 mg/mL), 0.1 mL of 10% aluminum nitrate, 0.1 mL of 1 M potassium acetate, and 4.3 mL of ethanol (Daejung Chem. and Metal Co. Ltd). The reaction mixture was incubated at room temperature for 40 min, and the absorbance measured at 415 nm. The flavonoid content was determined using the standard curve of quercetin (Sigma) (25-200 μL/mL). The extract's total flavonoid content was reported as mg quercetin equivalents (QE)/g.
Analysis of Chlorogenic Acid in AE Extract
Chlorogenic acid in the AE extract was measured using an Agilent 1200 series system (Agilent Technologies) consisting of a vacuum degasser, quaternary pump, autosampler, DAD (diode array detector), and Agilent ZORBAX Eclipse Plus C18 column (5 μm, 250 × 4.6 mm). Samples and standard solutions were filtered through a 0.2 μm filter using HPLC-grade solvents prior to analysis. Gradient elution was conducted with solvent A; 1% formic acid (Cat. No. F0507, Sigma) in water and solvent B; methanol (Daejung Chem. and Metal Co. Ltd), as follows: (0 min) 75% A:25% B, (20 min), 50% A:50% B, (25 min), and 20% A:80% B. The HPLC injection volume was 20 µL (20 °C), and the flow rate was 1.0 mL/min. The absorption spectrum of chlorogenic acid was recorded at 325 nm.
Animal Study Design
Sixty male C57BL6/J mice (20-22 g) were obtained from Hanil Laboratory Animal Center. The animals were kept at 23 ± 1 °C, humidity 50 ± 10%, and with a 12-h light/dark cycle; feed and water were provided
For sacrifice, the mice were anaesthetized with urethane (700 mg/kg) to minimize suffering. Then the blood was collected from the left ventricle. Serum was separated after centrifugation at 3000 rpm for 10 min and stored at −80 °C for further analysis. The liver, spleen, and colon were collected immediately after sacrifice. The length of the colon and the weight of the spleen were measured. Disease activity index (DAI) was evaluated based on the clinical progression of colitis, calculated by combining the degree of bloody stool, stool softness, and weight loss, according to Park's method. 25
Histopathology
The liver and colon were fixed in 4% paraformaldehyde. The samples were dehydrated, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). The slides were then examined under a light microscope (100×).
Immunohistochemistry
The colons were fixed in 4% paraformaldehyde. Dehydrated samples were embedded in paraffin, sectioned at 5 µm, and incubated for 1 h at 60 °C. Afterwards, the samples were deparaffinized with xylene, rehydrated with ethanol, the antigen retrieved with citrate buffer, quenched with H2O2 and methanol, and a blocking solution was applied. The primary antibodies were then applied and incubated overnight at 4 °C (TNF-α and IL-1β diluted in PBS 1:500). Labelling was removed under a high-power field microscope (400×) after the application of the secondary antibody. A microscope was used to examine the slides. The free software ImageJ v.1.80 and the Color Deconvolution2 plug-in were obtained from the NIH website (https://imagej.nih.gov/ij) for immunoreactivity analysis.
Cell Line Preparation
The RAW 264.7 cells were taken from the Korean Cell Line Bank (KCLB). The cells were grown in Dulbecco's Modified Eagle's Medium (Cat. No. 0030034DJ) with 10% fetal bovine serum (Cat. No.10082147) and 1% penicillin/streptomycin (Cat. No. 15070063) at 37 °C with 5% CO2. The cells that were cultured in trypsin (0.025%, Cat. No. R001100) and PBS (phosphate-buffered saline, Cat. No. 28348) were isolated and used throughout the experiment. All cell culture reagents were obtained from Gibco. Cell preparation protocols were performed as reported in a previous study. 12
Cytotoxicity
To assess the cytotoxicity, MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, Cat. No. M2128, Sigma) was used.
19
Briefly, cells were seeded into a 96-well plate (1 × 105 cells/well) and treated with 100 μL of diluted
Nitric Oxide Assay
Cells were treated with AE extract (25, 50, 75, 100, and 150 µg/mL) for 2 h and then LPS (1 μg/mL) was added and incubated for 24 h. Afterwards, 100 µL of the supernatant was mixed with 100 µL of Griess reagent, Cat No. MAK367-1KT (0.1% N-(1-naphthyl) ethylenediamine and 1% sulfanilamide 1:1) in a new 96-well plate and permitted to react for 10 min. Absorbance was measured at 570 nm using a microplate reader (Tecan infinite M200Pro, Männedorf).
Measurement of Inflammatory Mediators
The cells were treated with AE extract (25, 50, 75, 100 µg/mL) for 1 h and then exposed to LPS (1 μg/mL) for 18 h. Commercial ELISA kits were used to quantify the concentrations of tumor necrosis factor-alpha (TNF-α), Cat No. RAB0477; interleukin-1 β (IL-1β), Cat. No. RAB0275-1KT; and interleukin-6 (IL-6), Cat No. RAB0308-1KT, according to the manufacturer's (Sigma) instructions. Additionally, the morphology of cells treated with LPS (1 μg/mL) and AE extract (25, 50, 100, and 150 μg/mL) was examined under an optical microscope (400×).
Statistical Analysis
The results are stated as mean ± standard error of the mean (SEM). Statistical analysis was performed using Prism 5.0 (Graphpad Software Inc.). The Bonferroni
Results
Analysis of AE Extract
To explore the active components of the AE extract, polyphenols, flavonoids, and chlorogenic acid were measured. The regression equations obtained indicate slight differences between the data, y = 0.0055x − 0.0318 (R2 = 0.9969) and y = 0.0014x + 0.0011 (R2 = 0.9989 for polyphenols and flavonoids, respectively. AE extract at concentrations of 10,000, 5,000, 2,500, 1,250, 625, and 312.5 mg/mL revealed 186.8, 105.4, 62.7, 38.7, 21.4, and 1.9 mg GAE/g polyphenols, respectively, and 81.9, 40.2, 23.9, 12.2, 6.8, and 3.5 mg QE/g flavonoid, correspondingly. The result signifies that polyphenols and flavonoid content are related proportionally to the concentration of AE extract.
Chlorogenic acid is one of the polyphenols abundant in vegetables and fruits and has antioxidant and cytoprotective effects.
26
Our HPLC analysis showed that a solid peak of chlorogenic acid was observed at a retention time of 7.921 min, consistent with the standard peak. The chlorogenic acid content in the
Disease Activity index
Table 1 demonstrates that AE extract ingestion at concentrations of 250, 500, 750, and 1000 mg/kg significantly reduced the DAI compared to that in the DSS group. This result indicates the beneficial effects of AE extract when given to mice with DSS-induced colitis.
Disease Activity Index.

The data are reported as means ± SD (n = 6).
*:
AE Extract Protects Against Histological Changes in DSS-Induced Colitis
To prove whether TE extract possesses hepatotoxic effects, the liver tissues of all animals were examined. Normal liver structures are composed of small lobules of a roughly hexagonal shape arranged around the portal vein (Figure 1A, B, C, D, and F). This result proves that the AE extract is harmless to the liver.
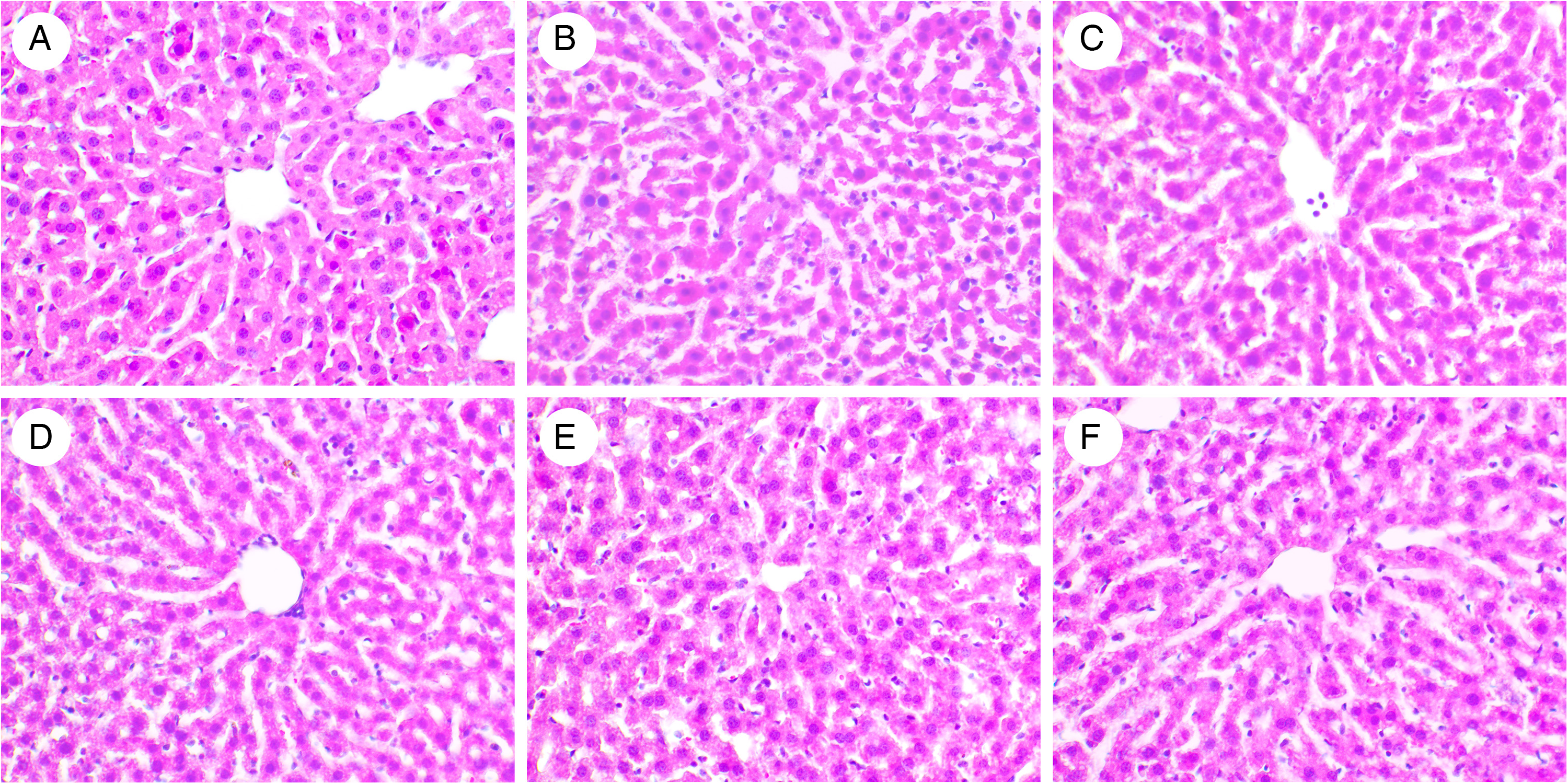
Effects of
To examine the protective effects of AE extract on DSS-induced colitis, histological analysis was performed. Cont (Figure 2A) and AE-1000 (Figure 2B) revealed normal tissue architecture (different layers clearly appear), while DSS induced inflammation in the colon (Figure 2C) was shown by immune cells infiltration in the mucosa and submucosa layers with hypertrophy of the muscle layer and loss of the crypt. However, AE-treated groups exhibited less damage (relative appearance of crypt cells, Figure 2D; relative reduction in immune cell infiltration, Figure 2E; and no hypertrophy, Figure 2F) compared with those in the DDS group. The result indicates a dose-dependent effect of the AE extract.

Effects of
To confirm further the protective effects of AE extract, pro-inflammatory cytokines were quantified in the colon using immunohistochemistry. The results demonstrated that there were no substantial differences in TNF-α and IL-1β levels between the control and AE-1000 groups. TNF-α and IL-1β levels, on the other hand, were noticeably higher in the DSS group compared to the control group (

Immunoreactivity tumor necrosis factor-α (TNF-α) at 200× magnification. Cont (A), dextran sulfate sodium (DSS) (B),

Immunoreactivity of interleukin 1 beta (IL-1β) at 200× magnification. Cont (A), dextran sulfate sodium (DSS) (B),
AE Extract Protects Against Cell Death and NO Overproduction
MTT was performed, in RAW 264.7 cells, to evaluate the viability of cells treated with 25, 50, 75, 100, and 150 µg/mL of AE extract. The result showed that cells treated with AE concentrations of 25 to 100 µg/mL caused no significant changes in cell viability. However, cells treated with 150 µg/mL considerably reduced cell viability compared with that of the Cont cells (
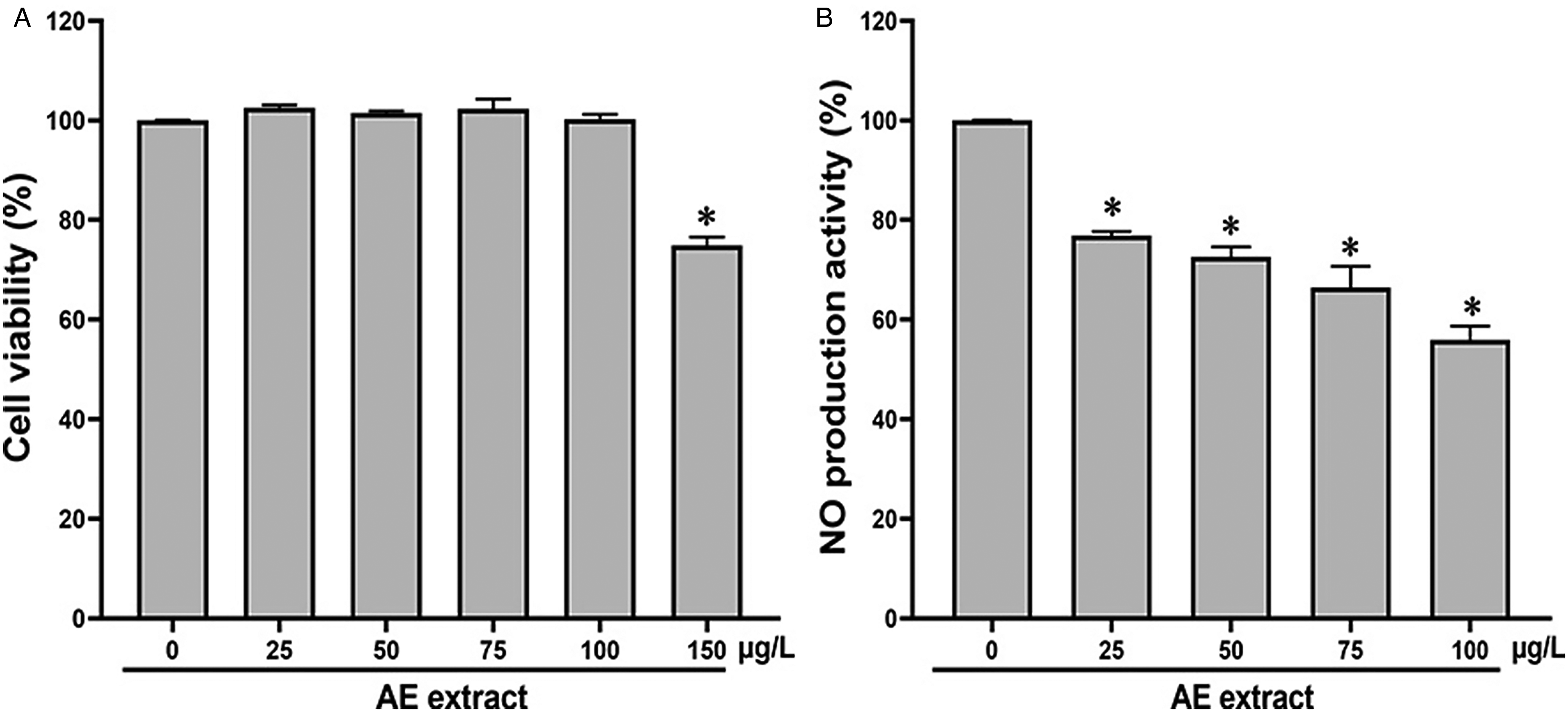
Cell viability assay on RAW 264.7 cells culture (A) and the nitric oxide (NO) production (B), and NO production inhibitory activity % (C) on LPS-treated RAW 264.7 cell culture incubated for 24 h with
AE Extract Curbs Pro-Inflammatory Cytokines Secretion
To figure out whether AE extract reduces the pro-inflammatory markers, RAW 264.7 cells were pretreated with LPS and then treated with AE extracts (25, 50, 75, 100 µg/mL). Cytokines were measured using commercially available ELISA kits. The levels of TNF-α, IL-1β, and IL-6 significantly increased in LPS-treated cells compared to those in the Cont cells (
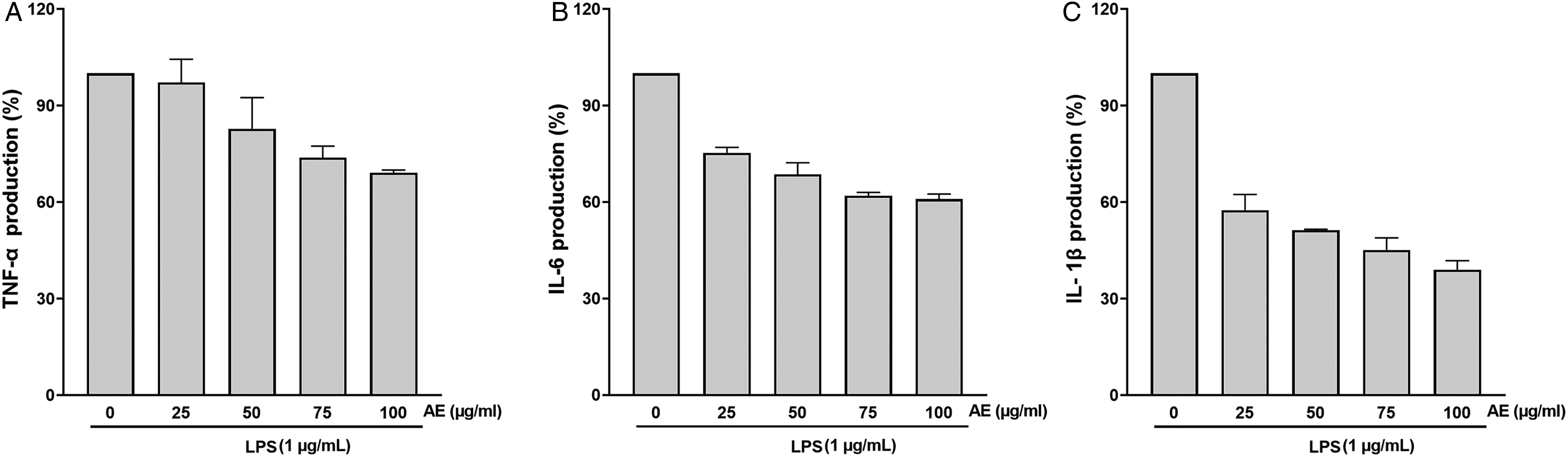
Effects of
AE Extract Improves Cell Morphology Deteriorated by LPS Exposure
As demonstrated in Figure 7, control cells showed a round, regular shape while LPS-treated cells were elongated, with granulated and separated cytoplasm and bigger nuclei. However, cells treated with AE extract showed substantial improvements in morphology and size, in a dose-dependent manner.
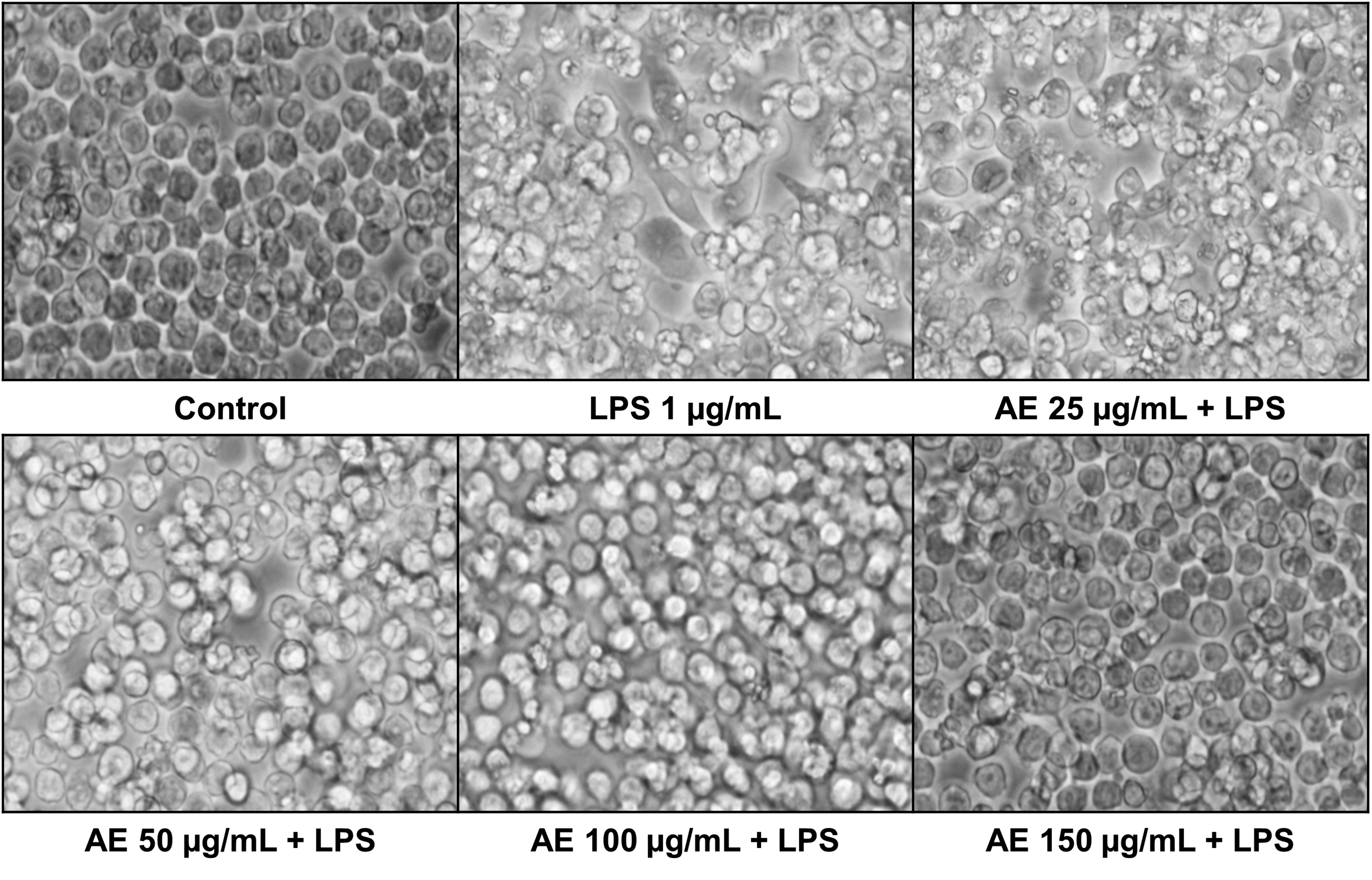
Optical microscopy (×400) images of RAW264.7 macrophage cells subjected to lipopolysaccharide (LPS) at a dose of 1 µg/ mL alone or with
Discussion
UC is a global disease that threatens the lives of people worldwide. 28 To slow the progression of the disease, doctors recommend a variety of treatments, including antibiotics, immunosuppressive drugs, and corticosteroids. However, there are concerns about their potential side effects. 29 Polyphenols and flavonoids contribute to a wide range of biological activities, including anti-inflammatory, immune regulation, antioxidant, cardiovascular protection, and anticancer. 30 The current study sought to identify the anti-inflammatory properties of AE extract in mice and RAW 264.7 cells exposed to DSS and LPS, respectively.
In this study, polyphenol and polyphenol-based substances (flavonoid and chlorogenic acid) were detected as key components of the AE extract. Perhaps DSS is the most used substance to induce colitis in animals because of its simplicity and effectiveness. 16 The extract is safe in the animal experiment using C57BL/6 mice as no hepatotoxicity was observed. When AE extract was applied to a DSS-induced colitis mouse model, the DAI was decreased, damage to the colon tissue was suppressed, and expression of pro-inflammatory cytokines (TNF-α, IL-1β) was increased compared to the DSS group, confirming that the AE extract has an attenuating effect on colitis. In the cell experiment using RAW 264.7 cells, concentrations with low cytotoxicity were selected. The inflammation was modulated and the antioxidant effect through the NO inhibitory activity was confirmed.
The polyphenol and flavonoid contents of the AE extract (10 000 µg/mL) were found to be 186.8 mg GAE/g at and 81.9 QE/mg, respectively. According to our results in Table 1, the result showed that the AE extract polyphenol contest is comparable with previous studies by Nikolovska et al 31 and Aboelmaati et al 32 investigating natural products with antioxidant activity. Moreover, polyphenols are substances with antioxidant activity 33 and, hence, the protective effects can be attributed to this property. All treated groups showed an index compared with the DSS group. Besides, the length of the colon decreased less than that of the DSS group, and the weight of the spleen tended to increase less than that of the DSS group, confirming the colitis attenuating effect of AE extract.
As shown in Figure 2, H & E staining of the colon slides demonstrated the inflammatory cells and damage to the epithelial tissue in the colitis groups. However, in the AE extract groups, fewer inflammatory cells and less damage were observed in a dose-dependent manner, showing that the AE extract appears to have colitis mitigating effects. These effects are attributed to the polyphenol- and flavonoid-rich AE extracts.32,33 It has been reported that
LPS causes toxicity in macrophage cells.38,39 We confirmed that LPS reduced cell viability and improved morphology after 18 h. Nonetheless, AE-pretreated cells improved the cell viability in a dose-dependent manner. Furthermore, NO production, overproduced by LPS, was reduced in cells pretreated with AE extract. Lee and Kang confirmed that AE extract exhibited a sufficient inhibitory effect on NO production and suppresses inflammatory reactions by regulation of NF-kappa B signaling in macrophage cells.
37
The findings of this experiment are compatible and directly compared with those of a recent report, which, similar to our protocol, assessed AE against LPS-induced inflammation in RAW 264.7 cells and DSS-induced colitis in mice, in which oxidative stress, TNFa, IL-6, IL-1b, and NO production were substantially reduced.
40
Moreover, we confirmed that a single plant extract of AE extracts, not a combined treatment of
Limitations of This Research
There are several limitations to this study, including the absence of endoscopy imaging to trace the progression of the treatment, which is due to the unavailability of endoscopy in our unit. Although AE treatment involves controlling the inflammatory mediators, the exact mechanism of action remains undetermined.
Conclusion
To sum up, we confirmed the polyphenol and flavonoid content of AE extract and found that it had antioxidant and anti-inflammatory properties by suppressing inflammatory mediators in RAW 264.7 cells and mice. Hence, UC was significantly reduced. These findings suggest that AE extract could be an effective phytomedicine with clinical applications in the treatment of colitis. More research is needed to determine the underlying molecular mechanism.
Footnotes
Authors’ Contributions
EL, SWL, GOA, and HJT conceived and wrote the manuscript; YJY, HYS, DA, and THJ performed the experiments; BTO, BYP, ISK, SHL, and JHL analyzed the data; HJY and JHL supported the study. SHL, CE, and HGO revised the manuscript. All authors have agreed to be held accountable for all aspects of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sunchang Research Institute of Health and Longevity; National Research Foundation of Korea (NRF) grant funded by the Korean government (NRF-2019R1A6A1A03033084); Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry and Fisheries (IPET) through Agriculture, Food and Rural Affairs Convergence Technologies Program for Educating Creative Global Leader, funded by Ministry of Agriculture, Food and Rural Affairs (MAFRA)(grant number:320005-4).
