Abstract
Context and objective
In the context of valorization of wood waste from the timber industry, this work aims to study the chemical composition and to evaluate the biological capacities of coproducts from Pinus caribaea Morelet.
Methods
With this mind, sawdust, bark, and knot-wood from P caribaea were analyzed by high-performance liquid chromatography-mass spectrometry. The antioxidant activities of extracts and reference compounds were assessed in vitro using 2 methods: 2.2-diphenyl-1-picrylhydrazyl and 2.2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid). For antimicrobial evaluation, coproduct extracts were assayed against Staphylococcus aureus, Staphylococcus epidermidis, Escherichia coli, and Candida albicans.
Results
Despite the variable composition of sawdust, bark, and knot-wood, the composition of P caribaea is typical of the Pinus genus, with the presence of flavonoids, stilbenes, and lignans. Seven compounds were elucidated: 2 lignans (nortrachelogenin and matairesinol), 3 flavonoids (pinocembrin, pinobanksin, pinobanksin 3-acetate), 2 stilbenes (pinosylvin, pinosylvin monomethyl ether), and 1 phenolic acid (caffeic acid).
Conclusion
The ethanolic extracts of bark and knots showed significant antioxidant activity. The knots showed significant antibacterial properties against S epidermidis.
Introduction
Undervalued, coproduct from the timber industry (bark, sawdust, and knot) possesses important potential due to its high quantity, low cost, availability, and richness in natural bioactive components. Specialized metabolites ensure the durability of wood through the resistance against biological degradation. The molecules display various activities, such as antioxidant, antifungal, or insecticide activities. 1 In particular, phenolic compounds such as phenolic acids, stilbenes, and tannins play a key role in plant development, mainly for their protection against damage and infections.2,3 These natural polyphenols have attracted attention for a wide range of applications: fine chemicals, dietary supplements, food additives, and cosmetic or pharmaceutical products.
Generally, geographical location, season, age, wood parts, and even harvesting conditions influence the quantity and originality of natural molecules.4-6 Tropical woods are a promising resource because of their adaptation to severe conditions such as strong ultraviolet rays and high temperatures. Indeed, high levels of phenolic compounds have been found in tropical heartwood. 7 High quantities of polyphenols, especially flavonoids, stilbenes, and lignans have been found in the Pinus genus.5,8-10 Flavonoids such as pinocembrin and pinobanksin in P pinaster have demonstrated antioxidant, anti-inflammatory, anticancer, and neuroprotective activities.11-13 Stilbenes are known for their antifungal properties, which promoting the wood durability. 14 Thus, pinosylvin is the main stilbene found in Pinus genus, especially in P strobus and P resinosa, 15 while pinosylvin monomethyl ether (PMME) is widely present in P sylvestris. 16 As reported by Umezawa, lignans are beneficial to human health, with antitumor, antioxidant, and antiviral potentials. 17 Nortrachelogenin is the major lignan in P sylvestris,8,18 P pinaster, 10 and P resinosa. 19
Native from Central America, Pinus caribaea Morelet is located throughout the tropics for timber production and is highly adaptable to different environments. Despite its widespread use, few studies have been carried out on the coproducts of this species. The first chemical studies of this species revealed a high proportion of α-pinene on the resin. 20 Further studies of the essential oil in the needles revealed a high terpene content.21,22 Pine bark has been studied for the antiradical and anticancer activity of condensed tannins. 23 However, the chemical composition of this pine has not been precisely described, and there are no reports on the biological activities of its extractives. Considering its potential, the aim of this study is to investigate the polyphenols content of P caribaea coproducts (barks, sawdust, and knot) and to evaluate the antioxidant and antibacterial activities of this species.
Results and Discussion
Mass Yield of Pine Extracts and Characterization
Essential oils
Essential oils of P caribaea sawdust were obtained with a mass yield of 0.01% (w/w). The main compounds detected by gas chromatography GC-FID-MS were longifolene (16%), longiborneol (9%), and hexadecanoic acid (9%) (Figure 1). Longifolene was found in high proportions in P densiflora and P thunbergii from Japan and has shown antitermite activities. 24 Hexadecanoic acid, present at 18% in P massoniana needles, had significant antibacterial activity against pathogenic bacteria 25 and anti-inflammatory activity. 26 The essential oils from P caribaea bark had a mass yield of 0.003% (w/w), and the main volatile compound was α-terpineol (54%). This is a tertiary monoterpenoid alcohol, commonly found in industry for its fragrant properties as an essential oil. Overall, yields of essential oils from sawdust and bark are modest, and this may act as an obstacle for future uses of these oils.

Structures of the main compounds found in essential oils of Pinus caribaea coproducts (sawdust and bark).
Extractives
The extractions of the different parts of P caribaea by ethanol and cyclohexane had shown that knot-wood contained the highest proportion of extractives compared to other coproducts (Table 1). Knots are located on areas of mechanical tension perpetually exposed to pathogen aggression (bacteria, fungi) or air oxidation where natural compounds are exceptionally concentrated.27,28 In particular, a study revealed the high concentration of extractives in P radiata knot-wood from New Zealand, specifically a large amount of resinous material. 29 The Gabaston's study indicated a mass yield of 7.5% of knot extractives from P pinaster using ethanol for extraction. 30 Despite high extraction rates, pine knots have a minor biomass compared to sawdust and bark, and their separation from the rest of the wood requires an additional step.
Mass Yield (in %) of Pinus caribaea Extractives.

Results in deviations according to starting material of 47.44 g and 100 g.
Comparison of sawdust from P caribaea and P pinaster
Sawdusts of P caribaea and P pinaster were analyzed by high-performance liquid chromatography–ultraviolet–mass spectrometry (HPLC–UV–MS) and displayed a similar composition (Figure 2).

High-performance liquid chromatography–ultraviolet (HPLC-UV) chromatographic fingerprint of ethanolic sawdust extracts of Pinus pinaster and Pinus caribaea compared to the ethanolic knots extract of P caribaea registered at 254 nm (a) and 280 nm (b).
Chemical composition of ethanolic extracts of P caribaea biomass (sawdust, bark, and knot)
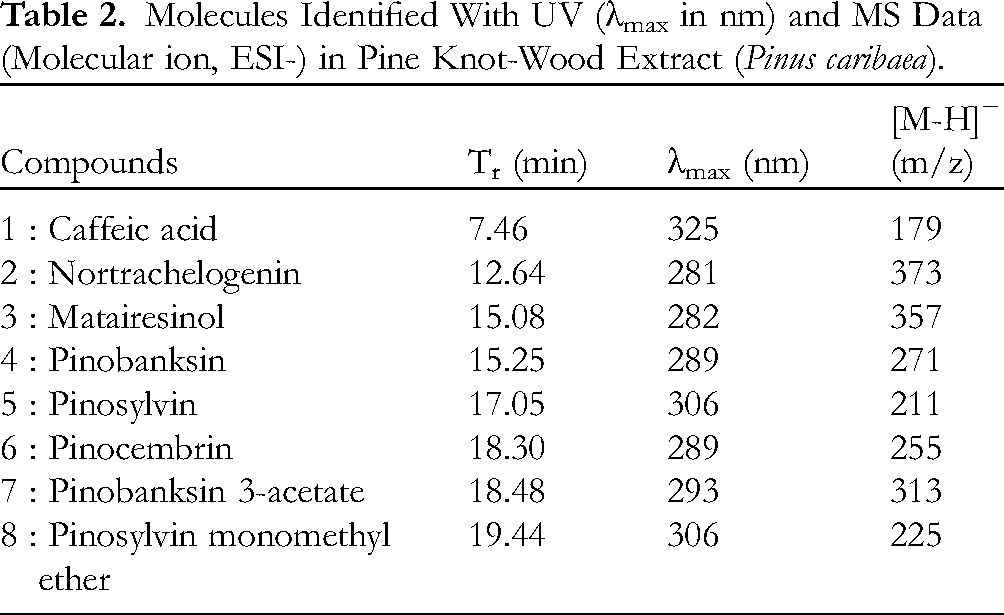
Pinus caribaea coproducts were analyzed by HPLC–UV–MS in order to determine the polyphenols composition. Based on a comparison of retention times, UV data (λmax) and mass spectrometry data. 30 Pinus caribaea knots’ extract contained 7 polyphenols and one phenolic acid (Figure 3A).

High-performance liquid chromatography-diode array detector (HPLC-DAD) chromatograms of Pinus caribaea biomass (a: knots, b: sawdust, c: bark) at 280 nm. 1: caffeic acid; 2: nortrachelogenin; 3: matairesinol; 4: pinobanksin; 5: pinosylvin; 6: pinocembrin; 7: pinobanksin 3-acetate; 8: pinosylvin monomethyl ether.
The identified compounds were 2 lignans (nortrachelogenin, matairesinol), 3 flavanones (pinocembrin, pinobanksin, pinobanksin 3-acetate), 2 stilbenes (pinosylvin, PMME), and 1 phenolic acid (caffeic acid) (Figure 4, Table 2). Pinosylvin, PMME, and the phenolic acid showed λmax at 306 and 325 nm. These absorption bands are linked to the presence of a double bond in E configuration. Nortrachelogenin, matairesinol, pinocembrin, pinobanksin, and pinobanksin 3-acetate showed λmax between 281 and 290 nm according to the lack of conjugation between aromatic rings in their chemical structure. 31
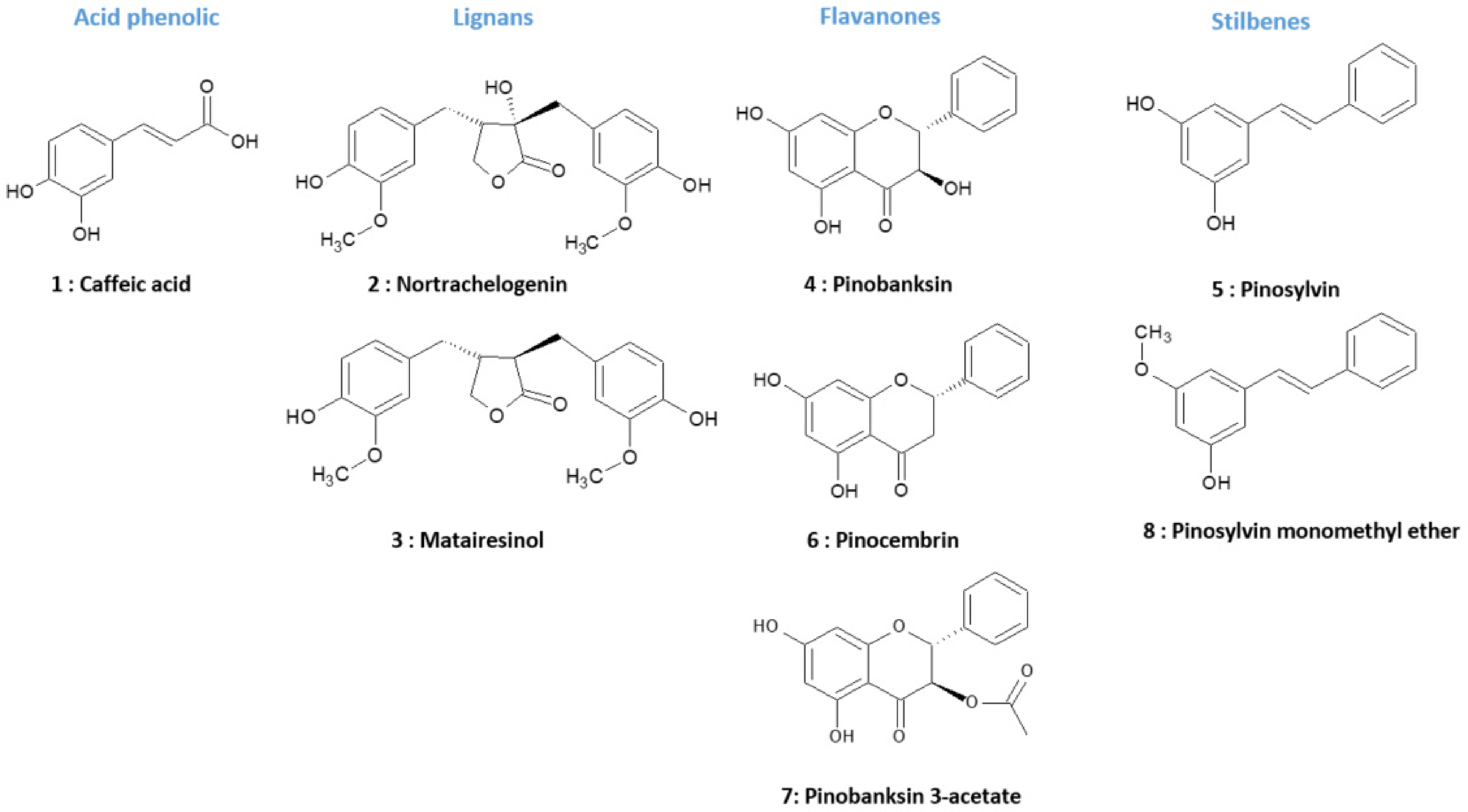
Structures of molecules 1: caffeic acid; 2: nortrachelogenin; 3: matairesinol; 4: pinobanksin; 5: pinosylvin; 6: pinocembrin; 7: pinobanksin 3-acetate; 8: pinosylvin monomethyl ether (PMME).
Molecules Identified With UV (λmax in nm) and MS Data (Molecular ion, ESI-) in Pine Knot-Wood Extract (Pinus caribaea).
The polyphenolic composition of P caribaea was closed to that of other species of the Pinus genus. Indeed, lignans, flavonoids, and pinosylvins were mainly found in P banksiana, P contorta, P resinosa, P strobus, P sibirica, and P sylvestris.14,15 Nortrachelogenin, pinocembrin and pinosylvins were found in P pinaster.
30
The flavonoids composition in P caribaea sawdust, barks, and knots have been determined. Compounds
Quantification of pinocembrin in pine knot extract
Pinocembrin is a natural flavonoid with numerous biological activities, including antioxidant, antimicrobial properties, and beneficial effects on vasodilation, anti-inflammation, and neuroprotective.13,32 To determine the concentration of this flavanone in P caribaea knot-wood, calibration curves were plotted with a pinocembrin standard. For ethanolic extract of P caribaea knots, pinocembrin was present at 9.2% (w/w). In comparison with the literature, Gabaston et al 30 have shown that the concentration of pinocembrin in P pinaster knots was 3.17% (w/w). Thus, our results unambiguously confirmed the presence of this bioactive molecule in this tropical wood and specifically in knot wood. This experiment confirmed the presence of this bioactive molecule in this tropical wood, specifically in the knot wood. In the ethanolic extract of sawdust, pinocembrin was present at 0.67% (w/w). In comparison, Hillis's study reported a pinocembrin content of 0.037% in heartwood and 0.115% in knot-wood of P radiata from New Zealand. 29
Antioxidant Assays
To investigate antioxidant activity of extracts and reference molecules, 2 radical-scavenging assays were used: DPPH (2,2-diphenyl-1-picrylhydrazyl radical) and ABTS (2,2′-azinobis[3-ethylbenzothiazoline-6-sulfonic acid]). The DPPH assay measures the ability of an extract to quench DPPH radicals by electron donation, while the ABTS test assesses the ability to scavenge cations from ABTS radical by reduction via electron donation or hydrogen atom transfer. 33 Results were expressed as the concentration required to scavenge 50% of the radicals (IC50) and compared to references. Ascorbic acid is the reference for the DPPH test. Trolox (6-hydroxy-2,5,7,8 tetramethylchroman-2-carboxilic acid) and ascorbic acid are references for the ABTS assay.
For the DPPH assay, the best IC50 values were obtained with pinosylvin and the ethanolic extract of bark and knot compared with ascorbic acid. This experiment showed that all ethanolic extracts (sawdust, bark, and knot) of P caribaea had a high capacity to scavenge DPPH radicals (Table 3). In contrast, apolar extracts from the cyclohexane extraction showed no significant effects at a concentration of 100 µg/mL.
Antiradical Activities of Pinus caribaea Extracts.

Data represent IC50 values in µg/mL (ie, the concentration of extract that scavenge 50% of radicals) mean ± SD of 2 independent experiments, NA, no activity found; ND, not determined.
For the ABTS assay, under the same conditions, the ethanolic extract of knot showed the best IC50 value (17.25 ± 0.78 µg/mL) compared with other biomass. Ethanolic extracts of P caribaea (sawdust, bark, and knot) were most active than cyclohexane extracts but had lower values than antiradical references AA and Trolox (Table 3). These results suggested that all polar extracts had an interesting antiradical activity against DPPH and ABTS radicals compared with reference compounds, especially knots. On the other hand, ethanolic sawdust from P pinaster was evaluated under the same conditions but showed a low capacity to quench DPPH radicals (72% for P pinaster vs 92% for P caribaea, Supplemental Data).
This experiment confirmed the role of polar molecules as polyphenols containing free radical scavenging functional groups. These compounds have one (or more) aromatic rings bearing one or more hydroxyl(s) potentially able to quench and stabilize free radicals. 34 Indeed, polyphenols present in wood are important for their antioxidant14,15 and antimicrobial properties, which are necessary against microorganisms attack when the branch breaks. 35 Barks were more active against DPPH radicals. A study had demonstrated the high content of extractives in bark, 36 in particular the richness of phenolic compounds in pine bark compared to epicea. 37 According to the same extraction procedure and at the same concentration, the ethanolic extract of P caribaea sawdust showed more significant anti-radical activity than P pinaster sawdust.
In parallel, 3 reference molecules (pinocembrin, pinobanksin, and pinosylvin) were evaluated for their ability to scavenge radicals. Pinocembrin and pinobanksin showed no antioxidant effect compared to pinosylvin. Figure 3 shows that pinosylvin and its derivatives were present in sawdust and knots, which could potentially explain their antiradical capacities.
Antibiotic Assays
Pinus caribaea extracts and pure compounds were evaluated for their antibiotic capacities against Staphylococcus epidermidis, Staphylococcus aureus, Escherichia coli, and Candida albicans. The concentration of the extract that inhibited 50% of the cell growth (IC50) was determined by measuring OD turbidimetry in a spectrometer at 590 nm.
Depending on the microorganisms tested, P caribaea extracts showed significant activity against S epidermidis (Table 4). The best IC50 values were obtained with knot extracts (ethanol and cyclohexane) (Table 4). Pinus caribaea extracts showed no antibiotic activities against E coli and S aureus. Compared with the literature, knot extracts of P sylvestris, P resinosa and pure compounds such as pinocembrin and lignans were inactive against E coli at 100 µg/mL. Stilbene such as pinosylvin and PMME had a moderate effect against E coli. 38 The presence of pinosylvin and in general trans-stilbenes in P sylvestris (38%)5,14 and P sibirica (28%) 15 plays an important role in wood durability, with significant fungicidal capacities against wood-destroying fungi. 38 These compounds and PMME showed the highest antimicrobial activities when tested separately as pure compounds. Accordingly, our experiment had shown that pinosylvin, pinocembrin, pinobanksin, and acetate-3-pinobanksin at 100 μg/mL were not active against E coli, S epidermidis, S aureus, and C albicans. In general, our study showed low antimicrobial activities of the extracts. Moreover, the isolated products did not show any effect. It would, therefore, be interesting to study the chemical composition of the cyclohexane extract in order to assess the contribution of least polar molecules in biological activities.
Antibiotic Activities of Pinus caribaea Extracts (Sawdust, Bark, and Knot Wood) and Reference Compounds.

Data represent IC50 values in µg/mL (ie, the concentration of the extract that inhibits 50% of the bacteria growth) mean ± SD of 2 independent experiments; NA, no activity found; ND, not determined.
An experiment with P caribaea extracts was carried out on fungi (C albicans), but no significant antifungal effect was observed. In spite of this, the literature had described several antifungal effects of pine knot, such as a growth inhibition of 80% for P resinosa and 85% for P strobus, P sibirica, and P sylvestris at 100 µg/mL against C albicans. 38 The experiment was also carried out on 3 commercial polyphenols (pinosylvin, pinobanksin and pinocembrin) and a polyphenol from the F5 fraction (pinobanksin 3-acetate) but the results did not reveal any inhibition of microorganism growth.
Conclusion
The phytochemical analysis of P caribaea coproducts demonstrated the presence of 7 compounds already identified in the Pinus genus. Pinocembrin was quantified showing a concentration 3 times higher in the knots of this tropical wood than in P pinaster. Extraction experiments showed that extractives are more concentrated in knots than in sawdust and bark, this tissue having a strategic position justifying a high content of defense molecules. Biological activities revealed variable results depending on coproducts, with polar extracts showing the highest bioactivity. In our experiment, ethanolic extracts of knots were the most antioxidant, followed by bark and sawdust. Polar sawdust containing natural free radical scavengers demonstrated DPPH and ABTS radical inhibition capacities. The extracts of knots showed significant inhibition against S epidermidis compared to sawdust and bark. Our present study could contribute to the biological valorization of pine residues.
Experimental
Chemicals and Reagents
Pinus caribaea sawdust, bark, and knots were obtained from a local garden center. Materials were air-dried at 35 °C, ground into fine particle size in a blender (Knife Mill GM 300) and ground in a second crusher (Polymix PX-MFC 90 D) to the required particle size (1 or 0.5 mm). Pinus pinaster sawdust was collected in Bordeaux and was ground under the same conditions as described above. The solvents used for extraction were HPLC-grade solvents (cyclohexane, ethanol, methanol, and acetone) from Fischer Chemicals. Formic acid and standards for pinocembrin, pinobanksin, and pinosylvin were purchased from Sigma-Aldrich.
For rapid detection, preparative thin-layer chromatography was performed on precoated silica gel (60 F254, Merck) with different solvent systems (MeOH, C6H12, CH2Cl2, EtOH, and AcOEt). Spots were visualized under UV (365 and 254 nm) and vanillin sulfuric.
Column chromatography was performed using silica gel 60 (0.015-0.040 mm, Merck) as the stationary phase. Various solvent systems were used (MeOH, C6H12, CH2Cl2, EtOH, and AcOEt). Lipophilic Sephadex was used with MeOH (100%) as solvent. Preparative plates were employed with glass plates (20 cm × 20 cm) coated with 2 mm of silica gel (Kieselgel 60 PF254, Merck). After air-drying for 24 h, plates were activated at 90 °C for 3 h.
Extractions
To obtain the essential oils, pine sawdust (127.66 g) or bark (405.05 g) was distilled with a Clevenger apparatus. To obtain extractives, each coproduct powder (sawdust, bark, or knots, 100 g) was macerated in 1L of cyclohexane for 6 h under ultrasonic agitation. After filtration and evaporation of the solvent, C6H12 extracts were obtained. Residues were dried in an oven (Memmert Universal Oven UF1060) at 40 °C for 24 h. The second extraction was carried out under the same conditions with 1L of EtOH/H2O (70/30). After filtration and evaporation of solvent, ethanolic extracts were obtained. A slightly higher proportion (85:15 of EtOH/H2O) was used to obtain the ethanolic extract for pinocembrin quantification. The mass yields of products were given in %.
Purifications
The ethanolic extract of sawdust (3.13 g) was purified on a chromatography column eluted with a gradient of C6H12/AcOEt/MeOH (C6H12/AcOEt [100/70 to 100/0]; AcOEt/MeOH [90/10, 50/50 to 0/100]). The 15 fractions resulting from this separation were analyzed by HPLC–UV–MS. Fraction F6 from ethanolic pine sawdust (252.31 mg) underwent a second purification through lipophilic sephadex with MeOH (100%).
Characterizations by GC-FID-MS Chromatography
Essential oils were analyzed by gas chromatography coupled to a mass detector (Clarus® 580, Perkin Elmer Inc) and a flame ionization detector (Clarus® 580, Perkin Elmer Inc). Helium was used at 1 mL/min in EI mode. Capillary column was an elite-5MS (30 m × 0.25 mm, 0.25 μm) (Perkin Elmer Inc). The injection temperature was set at 250 °C. Analyses were performed using a temperature program, with an initial 3 min hold, starting from 50 °C to 250 °C with a 5 °C/min heating ramp. The final temperature was stable for 15 min. Mass range was set at 40 to 500 m/z. The peaks were identified both by comparison of mass spectra from libraries and by the retention indices. This parameter was calculated for all volatile components using a homologous series of n-alkanes C8-C27. Retention indices were compared with available literature data. 39 For the mass spectra comparison and identification, we used Mass Finder 2.3 library, Wiley, and NIST library. Percentages of constituents were calculated with the area from the FID-GC chromatogram corrected with the number of carbon atoms of the corresponding compound.
Characterizations by HPLC–MS Chromatography
The HPLC–UV–ESI–MS system consisted of a Shimadzu LC-20AB pump, a SIL-20AC autosampler, a CTO-10AS oven column and, for detection, the Shimadzu SPD-M20A photodiode array detection connected in series with a Shimadzu LCMS-2020 ESI mass spectrometer. The LC-MS instrument was regulated by Shimadzu LCMS LabSolution software. The HPLC analytical column used was a Shimadzu Shim-pack GISS-HP reversed phase C18 column (particle size 3.0 mm, 100 mm × 3.0 mm I.D.) preceded by a guard Shim-pack GISS C18 column (particle size 3.0 mm 10 mm × 1.5 mm I.D.,). The mobile phase consisted of ultrapure H2O with 0.1% formic acid (A) and ACN with 0.1% acid formic (B). The gradient was as follows: 0 to 3 min, 10% B; 3 to 6 min, linear gradient from 10%B to 20% B; 6 to 10 min, linear gradient from 20%B to 30% B; 10 to 12 min, linear gradient from 30%B to 35% B; 12 to 22 min, linear gradient from 35%B to 60% B; 22 to 29 min, linear gradient from 60%B to 100% B and 29 to 35 min, linear gradient from 100%B to 10% B and then 10% B was maintained for 2 min for column re-equilibrium. The gradient was delivered at a flow rate of 0.4 mL/min, and the injection volume was 1 or 10 μL. The temperature of the column oven was 40 °C. For UV-Visible detection, the wavelengths scan were set at 200 to 400 nm. The electrospray ionization (ESI) source was operated in negative ionization mode (scan range m/z 300-500). The mass detector parameters were as follows: nebulizing gas flow (nitrogen), 1.5 L/min; drying gas flow, 10 L/min; desolvation line temperature, 250 °C; and heat block temperature, 300 °C. The ion spray voltage was set at −4.5 kV.
Extracts for analysis were dissolved in methanol grade HPLC at 1 mg/mL filtered on filters (PTFE, 0.45 μm). This protocol has been adapted from the Gabaston's study on P pinaster. 30 For the quantification of pinocembrin, standards have been injected independently in triplicate at different concentrations (5, 10, 100, 300, and 500 μg/mL). A calibration curve line was established between concentration of the standards and the peak's area. Pinocembrin was quantified at its maximum wavelength (280 nm) by UV/vis detector (Supplemental Data, Figure A-G).
Characterization NMR of Pinobanksin 3-Acetate (Compound 7 )
Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker DPX 500 NMR spectrometer (1H NMR: 500 MHz, 13C NMR: 125 MHz). The solvents used were CDCl3 or dimethyl sulfoxide (DMSO)-d6. Chemical shifts (δ) were recorded in parts per million (ppm) relative to residual solvent peaks (CDCl3: 1H NMR: δ = 7. 26 ppm and 13C NMR: δ = 77.00 ppm; DMSO-d6: 1H NMR: δ = 2.50 ppm and 13C NMR: δ = 39.52 ppm). Coupling constants were reported in hertz (Hz). Multiplicities were reported as s (singlet), br s (broad singlet), d (doublet), t (triplet), br (broad), or m (multiplet).
1H NMR were consistent with literature data 40 : 7.47 (2H, d, J = 8.0 Hz, H-2′, H-6′), 7.42 (3H, m, H-3′, H-4′, H-5′), 6.04 (1H, d, J = 2.0 Hz, H-8), 6.00 (1H, d, J = 2.0 Hz, H-6), 5.80 (1H, d, J = 11.7 Hz, H-3), 5.36 (1H, d, J = 11.7 Hz, H-2), 2.022 (3H, s, CH3CO) (Supplemental Data Figure H-L).
Antiradical Evaluation (DPPH and TEAC)
2.2-diphenyl-1-picrylhydrazyl assays
Radical scavenging activity was observed by the discoloration of purple DPPH radical to pale color measured at 515 nm, in the presence of radical scavenging molecule. The positive control was a 12 mg/mL AA (ascorbic acid) solution, and extracts diluted in ethanol had the same concentration. Aliquots of 20 μL of the extract, the positive control (AA) and the negative control (EtOH) were loaded in 96-well microtiter plates (polystyrene cell culture plate, corning incorporated costar) and supplemented with 180 µL of DPPH solution (100 µM). Absorbance at 515 nm was measured at 0 and 30 min after incubation in the dark at room temperature using a microplate reader (EPOCH 2-microplate reader; BioTek Instruments). The experiment was repeated 3 times. For the data, an average of all repetitions was calculated. Percentage inhibition was calculated (1):
TEAC tests
The antioxidant scavenging capacity will be noticed by the discoloration of the blue chromophore, characteristic of ABTS•+. 33 The ABTS•+ solution was prepared by mixing 7 mM ABTS with 2.45 mM potassium persulfate (1:1 vol/vol) and leaving in the dark, for 19 h, until the absorbance was stable. The standard ranges were performed in ethanol (2.5, 0.5, 0.25, 0.1, 0.05, and 0.025 mg/mL). The positive control was Trolox solution at 5 mg/mL (1-day storage). Samples to screen were diluted in ethanol at 5 mg/mL. The ABTS•+ solution was diluted with ethanol to obtain an absorbance at 734 nm of 0.700 ± 0.02 at 30 °C. 41 For screening, 4 µL of extract (5 mg/mL), positive (trolox dilutions) or negative control (ethanol) were loaded into 96-well microtiter plates (cell culture plate polystyrene, corning incorporated costar) and supplemented with 196 µL of radical ABTS•+ to give a final concentration of 100 µg/mL in the well. Absorbance at 734 nm was measured at 0 min and after 6 min of incubation at 30 °C using a microplate reader (EPOCH 2-microplate reader; BioTek Instruments). For the data, an average of all repetitions was calculated. As for DPPH assay, the percentage of inhibition was calculated according to equation (1) where Ao is the absorbance of ABTS•+ in ethanol and A1 is the absorbance of the sample with ABTS•+ minus the absorbance of the sample alone at 734 nm. For extracts with high percentage of inhibition, IC50 were calculated as previously described (Supplemental Data, Table A).
Antibiotic Assays
Bacterial and yeast strains used were E coli (ATCC 25922, CIP 7624), S epidermidis (ATCC 12228, CIP 1200), and C albicans (ATCC 10231, CIP 4872). The organisms have been streaked on LB agar (Lysogenic Broth Lennox medium [LB, tryptone: 10 g.L−1, yeast extract: 5 g.L−1, NaCl: 5 g.L−1]; CONDA) and incubated at 37 °C for 12 h for the bacteria or 48 h for the fungi. The positive control was gentamycin (0.01 mg/mL) for S epidermidis and E coli, while amphotericin B (0.1 mg/mL) was the antifungal reference for C albicans. The extracts were prepared in DMSO at 10 mg/mL and then diluted in LB until a concentration of 1 mg/mL. Antibiotic assays were performed under aerobic conditions on microplates (sterile transparent 96 wells PS flat bottom, Greiner) in LB medium. An overnight culture (one colony added to 5 mL of LB) was adjusted to an OD600nm between 0.1 and 0.2. Each well was filled with 20 μL of diluted culture, 20 µL of extract or molecule or antibiotic reference, and 160 μL of LB. For the OD600nm of extracts in the environment, 20 µL of extract or molecule and 180 µL of LB were put in the well. For the cell growth curve reference, 20 µL of the diluted culture and 180 µL of LB. The plate was incubated at 37 °C with orbital shaking at 125 r.min−1 in a microplate incubator reader (EPOCH 2 microplate reader, BioTek Instruments). Cell growth was monitored by measuring the OD600 every 30 min for 24 or 48 h. Data were calculated by averaging all replicates compared to control data. For screening, bioassays for antibacterial and anti-yeast activities were carried out for all extracts at 100 µg/mL. IC50 were calculated for compounds or extracts with the highest activities. The strain growth rates (µ) were determined for each concentration. A sigmoid dose–response curve was obtained by plotting growth rate against concentrations of extracts or tested molecules to determine IC50 values.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231211958 - Supplemental material for Polyphenol Content, Antioxidant, and Antibiotic Activities of Pinus Caribaea Morelet Forestry Coproducts
Supplemental material, sj-docx-1-npx-10.1177_1934578X231211958 for Polyphenol Content, Antioxidant, and Antibiotic Activities of Pinus Caribaea Morelet Forestry Coproducts by Cynthia Sinyeue, Louisa Maerker, Linda Guentas, Valérie Medevielle, Frédérique Bregier, Vincent Chaleix, Vincent Sol and Nicolas Lebouvier in Natural Product Communications
Footnotes
Acknowledgements
The authors are grateful to the Government of New Caledonia for the PhD thesis grant of Cynthia Sinyeue and to the University of New Caledonia Foundation (Project NORAS). The authors are would like to thank Kristina O'Shea for proofreading the manuscript.
Author Contributions
Conceptualization: CS, VS, NL. Methodology: CS, LG, VM, VS, NL, VC, FB. Validation: LG, VS, NL. Formal analysis: CS, LG, NL, VS. Investigation: CS, LM, VM, NL. Resources: VS, NL. Writing - Original Draft: CS. Writing - Review & Editing: LG, VS, NL. Visualization: CS. Supervision: VS, NL.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Government of New Caledonia for the PhD thesis grant of Cynthia Sinyeue and by the University of New Caledonia Foundation (Project NORAS). University of New Caledonia Foundation, Government of New Caledonia, (grant number Projet NORAS, PhD thesis grant of Cynthia SINYEUE).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
