Abstract
Zhuang Medicine Jin-mu Granules (ZMJG) are prescriptions derived from the Zhuang nationality, which is the largest minority among 56 ethnic groups in China. They have been widely used in the treatment of chronic pelvic inflammatory disease (CPID) in Guangxi Zhuang Autonomous Region for clearing away heat and toxins, removing dampness and poisoning. CPID is a common gynecological disease of female reproductive organs and surrounding tissues and is characterized by persistent and recurrent symptoms, causing serious physical and psychological damage to the patient. Preliminary research found that ZMJG have beneficial effects on CPID model rats, but the metabolic mechanism underlying their protective effects is unclear. In this study, we used the strategy of combining network pharmacology, pharmacodynamic, and metabolomic approaches to investigate the molecular mechanisms and potential targets of ZMJG for the treatment of CPID. First, a network diagram of “medicinal materials-components-targets-pathways” based on network pharmacology was constructed to obtain a preliminary understanding of the biologically active compounds and related targets of ZMJG and clarify their molecular mechanism in CPID. Subsequently, the in vivo efficacy of ZMJG was verified in a rat model. Furthermore, we analyzed the corresponding metabolomics profile to explore the differentially induced metabolic markers and elucidate the metabolic mechanism by which ZMJG treat CPID. The results show that the therapeutic effect of ZMJG on CPID is mediated through multiple pathways, metabolic pathways, and multi-component multi-target modes, providing a detailed theoretical basis for the development and clinical application of ZMJG and a new research idea for the treatment of CPID in Chinese medicine.
Keywords
Introduction
Chronic pelvic inflammatory disease (CPID) has been attributed to the inadequate and untimely diagnosis and treatment of pelvic inflammatory disease. Under normal circumstances, the female reproductive tract exerts a natural defense against pelvic inflammatory disease. However, compromised in this natural defense, low body immunity or exogenous pathogen invasion can cause pelvic inflammatory disease. Traditional Chinese medicine classifies pelvic inflammatory disease as “menstrual disorder,” “dysmenorrhea,” “infertility,” and so on,1–4 with symptoms similar to those of “leucorrhoea” due to damp heat toxin in Zhuang medicine. At present, Western medicine has no specific drugs for CPID disease; the primary treatment is comprised of antibiotics that can easily lead to a series of adverse reactions. Nevertheless, studies have shown that traditional Chinese medicine plays a significant role in the treatment of CPID. 5 Therefore, it is particularly important to clarify the pathogenesis of CPID and identify relevant drug targets. As a promising treatment strategy, multi-target therapy emphasizes the idea of “network targets and the overall theory in multi-component therapy,” which is consistent with the theory of traditional Chinese medicine. Traditional Chinese medicine formulations have broad prospects in the prevention and treatment of complex metabolic diseases, which are attributed to the targets of multiple components that produce combined or synergistic effects. 6
Network pharmacology is methodologically holistic, systematic, and dynamic, consistent with the characteristics of the holistic view of Chinese medicine and evidence-based treatment and prescription dispensing. Thus, network pharmacology provides a bridge to study the relationship between traditional Chinese medicine and modern pharmacology and offers a new research direction for the modernization and development of Chinese medicine. Zhuang Medicine Jin-mu Granules (ZMJG) are a traditional Zhuang prescription for the clinical treatment of CPID and are widely used in Guangxi Zhuang Autonomous Region. ZMJG contain 9 traditional Chinese medicine: (1) Jin Gangci (Smilax scobinicaulis C. H. Wright, family Liliaceae). The medicinal material is liana from sarsaparilla, which grows in low mountain shrubs or valley banks and is mainly distributed in Shandong, Jiangsu, Zhejiang, Fujian, Taiwan, Jiangxi, Henan, Hubei, Yunnan, Guizhou, Hunan, Guangxi, and Guangdong in China. The active ingredients include amino acids, flavonoids and their glycosides, sterols, saponins, and ester compounds. Studies have found that the plant has several pharmacological effects, such as bacteriostasis, immunosuppression, neuroprotection, anti-inflammatory, analgesic, anti-oxidation, and antitumor. 7 (2) Huo Tanmu (Polygonum perfoliatum L. [family Polygonaceae]) is produced in Guangxi, Southern Shaanxi, Southern Gansu, East China, Central China, South China, and Southwest China. The active ingredients in Huo Tanmu include flavonoids, steroids, and phenolic acids. Modern pharmacological research has shown that the plant has anti-oxidation, anti-viral, anti-inflammatory, analgesic, and anti-tumor effects. 8 (3) Tu Fuling, the dried rhizome of Smilax glabra Roxb., is produced in Gansu, Guangxi, Yunnan, and the provinces of the Yangtze River Basin in China. The active ingredients of Tu Fuling are mainly flavonoids and their glycosides, sterols, phenylpropanoids, sugars, organic acids, and volatile oils. Modern pharmacological studies have shown that the plant has anti-inflammatory, analgesic, cellular immune suppression, antibacterial, cardiovascular system protection, diuretic, and anti-tumor effects.9,10 (4) Hu Zhang (Polygonum cuspidatum Sieb.et Zucc., family Polygonaceae) is produced in Southern Shanxi, Southern Gansu, Guangxi, Sichuan, Yunnan, and Guizhou. The active ingredients include flavonoids, anthraquinones, coumarins, stilbene, and some fatty acids. It has anti-inflammatory, myocardium protection, anti-tumor, metabolic regulation, anti-infection, and anti-oxidation effects.11–13 (5) Huang Bo is the dried bark of the Rutaceae plant Phellodendron chinense Schneid. It is mainly produced in Henan, Anhui, Guangxi, Ningxia, and other areas of China. The active ingredients include alkaloids and flavonoids. Alkaloids are the most important active ingredients with relatively high contents, including berberine, phellodendron, jatrorrhizine, tetrandrine, and magnolin. Huang Bo has various pharmacological effects, such as lowering blood pressure, immunosuppression, lowering blood sugar, antibacterial, and antioxidant. 14 (6) Ku Shen is the dried root of the legume Sophora flavescens Ait. It is produced in Hebei, Guangxi, Shanxi, Shaanxi, Gansu, Henan, Hubei, Shandong, Jiangsu, and other areas of China. Ku Shen mainly contains alkaloids and flavonoids and has anti-inflammatory, anti-tumor, antibacterial, analgesic, and other pharmacological activities.15,16 (7) Da Xueteng is the dried cane of Sargentodoxa cuneata (Oliv.) Rehd. et Wils, family Lardizabalaceae, and is produced in Sichuan, Guizhou, Hubei, Hunan, Yunnan, Guangxi, Guangdong, and other areas of China. The active ingredients of Da Xueteng include phenols, saponins, flavonoids, and anthraquinones. The plant has anti-bacterial, anti-inflammatory, immunosuppressive, anti-tumor, and anti-viral effects.17,18 (8) Li Toucao is the whole plant or root of Viola japonica Langsd., family Violaceae. The plant is produced in Hubei, Sichuan, Guangxi, Guizhou, Shaanxi, and other areas of China. Li Toucao contains coumarins, organic acids, and flavonoids and is effective in treating refractory wounds, septic arthritis, liver damage, and chronic osteomyelitis. 19 (9) Bai Beiyegen is the dried root of Mallotus apelta (Lour.) Muell. Arg., family Euphorbiaceae. It is produced in Guangxi, Guangdong, Hunan, Jiangxi, Yunnan, Hainan, and Fujian in China. The active ingredients include flavonoids, sterols, organic acids, and terpenoids, which have antiviral, antitumor, liver protection, anti-inflammatory, and other pharmacological effects. 20 ZMJG have been found to eliminate dampness toxin, and stop bleeding and can be used for leucorrhea, abdominal pain, itching, dysmenorrhea, and other diseases. To meet the clinical needs, a research team evaluated the pharmacology, acute toxicity, pharmacodynamics, and possible anti-inflammatory mechanism of ZMJG prepared from the original prescription decoction in the early stage. They demonstrated that ZMJG are effective in the treatment of CPID. However, the multi-component multi-target mechanism remains unclear. With the development of bioinformatics, systems biology provides a more comprehensive method to integrate compound-target interactions from the molecular to the system level. One of the most important applications of systems biology is network pharmacology, which can be used to understand the complex action mechanism of traditional Chinese medicine formulations, propose a complete drug-target interaction network, and determine drug interactions more efficiently. 21 Metabolomics is an important part of systems biology that systematically studies the changes in metabolites during the dynamic process of metabolism and reveals the nature of metabolism in the body.22–24 Metabolomics examines changes in the entire organism, thereby overcoming the shortcomings of a single index or few indicators. Furthermore, it has temporal, dynamic, holistic, and systematic characteristics. Metabolomics emphasizes both commonality and individuality, which is consistent with the holistic view and syndrome differentiation of Chinese medicine. Metabolomics can be used to analyze serum samples through liquid chromatography with tandem mass spectrometry (LC-MS/MS) combined with multivariate statistical methods, compare changes in different metabolites of various groups, identify metabolic markers, and obtain the corresponding metabolic pathways. These clarify the relevant metabolic mechanisms and provide a foundation for future research.25–27
This study used a combination of network pharmacology, pharmacodynamics, and serum metabolomics methods with LC-MS/MS technology to study the molecular mechanism and potential targets of ZMJG in the treatment of CPID. First, a network diagram of “medicinal materials-components-targets-pathways” related to CPID was established through network pharmacology to identify potential biologically active compounds and related targets of ZMJG. Subsequently, a CPID rat model was established with a mixed bacterial solution of Staphylococcus aureus and Escherichia coli together with mechanical damage, and related pharmacodynamic experiments were performed to demonstrate the efficacy of ZMJG in vivo. LC-MS/MS technology was used to analyze the blank, model, and drug administration groups. The serum samples of animals in the drug group were analyzed, the potential endogenous metabolites were screened through multivariate data analysis, and the corresponding metabolic pathways were determined. Finally, the results of network pharmacology and metabolomics were combined for systematic analysis. This study emphasized the reliability and effectiveness of methods based on network pharmacology and metabolomics, which identified and verified the complexity of natural compounds in ZMJG, and combined the analyzed metabolic pathways to illustrate the mechanism of action for ZMJG in the treatment of CPID.28,29 The overall process of this study is shown in Figure 1.
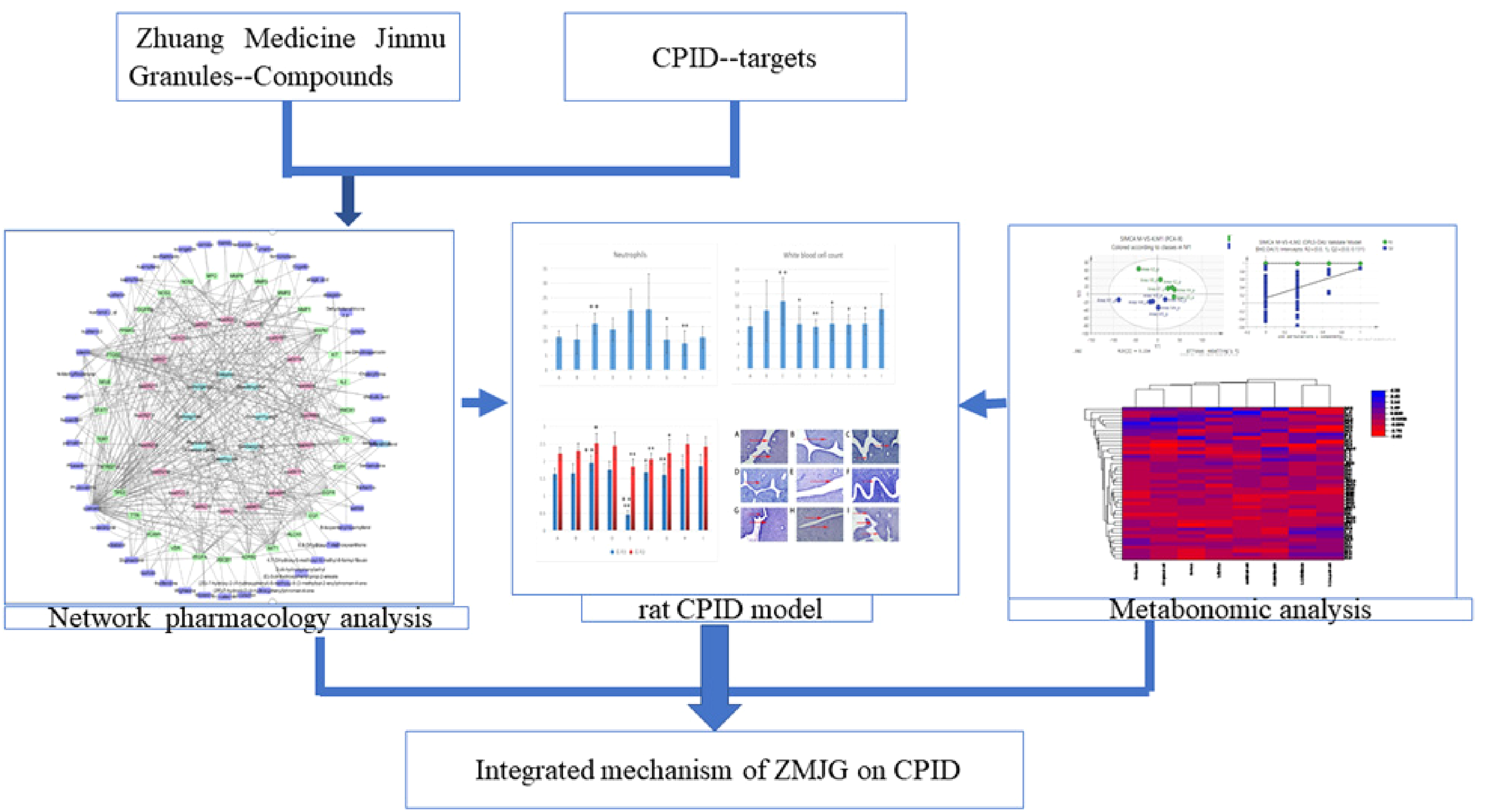
Comprehensive flow chart for the treatment of CPID with ZMJG. First, a network diagram of “medicinal materials-components-targets-pathways” based on network pharmacology was constructed to predict the molecular mechanism of ZMJG in the treatment of CPID. Subsequently, the in vivo efficacy of ZMJG was verified in a rat model. Furthermore, the corresponding metabolomics profile was used to explore the differentially induced metabolic markers and provide the metabolic mechanism of ZMJG in the treatment of CPID. These results indicate the mechanism of action of ZMJG in the treatment of CPID.
Materials and Methods
Network Pharmacology Analysis
Collection and Screening of the Active Components of ZMJG
Using the pharmacology database and analysis platform of traditional Chinese medicine system pharmacology (TCMSP; http://tcmspw.com/tcmsp.php), 9 traditional Chinese medicines, such as “Tu Fuling,” “Hu zhang,” “Huang bo,” and “Ku Shen” were imported to determine the corresponding chemical components. Given that ethnic drugs were present in the compound, we reviewed relevant literature to determine the corresponding chemical components. When oral bioavailability (OB) ≥ 30% and drug-likeness (DL) ≥ 0.18 are selected as the conditions for screening chemical components and target proteins, the drug targets retrieved may present with some concerns, such as nonstandard naming. Therefore, UniProt (https://www.uniprot.org/) can be used to retrieve the current protein names by inputting the target protein name and limiting the species to “human.”30–32
Collection of CPID-Related Targets
We searched the GeneCards database (https://www.genecards. org/) for the related target information of CPID using the keyword “chronic pelvic inflammatory disease” in Venny 2.1.0, created the Venny with the related target protein of ZMJG, mapped and selected the common target and then searched for the corresponding chemical components and herbs of the common target. Thereafter, the common targets and corresponding components were introduced into Cytoscape 3.6.0,33–35 the relevant topological data degree value was derived and nodes larger than the average degree value were screened to identify the primary chemical components of ZMJG used to treat CPID.
Construction of the Target Interaction Network (Protein-Protein Interaction)
To clarify the interaction between ZMJG targets in the treatment of CPID, selected targets were introduced into the STRING network platform (https://string-db.org/), the protein type was set as “homo sapiens,” and the minimum interaction threshold was set as “medium confidence” (0.4) to determine the protein interaction relationship and construct a protein-protein interaction (PPI) network. Cytoscape 3.6.0 was used to visualize the PPI network,36,37 and the “degree” and “betweenness centrality” of nodes were analyzed to determine the core target of ZMJG in the treatment of CPID.
Gene Ontology Function Enrichment and KEGG Pathway Analysis
Using gene ontology (GO) analysis through the DAVID 6.8 database (https://david.ncifcrf.gov/), the key targets were analyzed for biological process, molecular function, and cellular component. Results with a P of <0.01 and false discovery rate (FDR) of <0.05 were selected and processed visually.38–40 Based on the KEGG data, the key target pathway was analyzed to determine the key target enrichment pathway.
Construction of the CPID “Medicinal Materials-Components-Targets-Pathways” Network
According to the chemical composition prediction results, the target and pathway of ZMJG, corresponding relationships between medicinal materials and components, components and targets and targets and pathways were established with Excel. A network diagram was constructed with Cytoscape 3.6.0. The nodes in the network represented medicinal materials, components, targets and pathways, whereas the edges represented medicinal material–component, component–target, and target–pathway interactions.41–43
Preliminary Verification of the Mechanism of Action
Based on Pymol and Autodock software, the screened active ingredients were molecularly docked with the core targets. The traditional Chinese medicine systems pharmacology database and analysis platform (TCMSP) were used to download the mol2 format file of the active ingredients for the core formula as the ligand and download the PDB format file of the core target from the PDB database(http://www.uniprot.org/) as the receptor. The core target protein was imported into Pymol software to remove water molecules and ligands and then imported into Autodock Tool for molecular docking. The docking effect was evaluated according to the binding energy.
Experimental Validation of ZMJG on CPID in Vivo
Animal Experiment
Ninety specific-pathogen free Sprague–Dawley (SD) female rats of SPF grade 7-8 weeks without drug intervention, weighing 200 ± 20 g, were purchased from Hunan Slike Jingda Laboratory Animal Co., Ltd and kept in the animal room of Guangxi University of Traditional Chinese Medicine (Certificate No.: 2021SCXK Xiang [2019-0002]). Rearing conditions were as follows: room temperature of 20 °C ± 5 °C, relative humidity of 50% ± 20%, 12-h light/dark cycle, and adaptive feeding was provided for 7 days before experiments; rats were fed in a cage with ad libitum access to food and water. During the animal experiments, all ethical issues of cooperation with animals were approved by the Laboratory Animal Ethics Committee of Guangxi University of Traditional Chinese Medicine (approval number: DW20200415-53).
Reagents and Reagents
ZMJG was developed by our research group (batch number: G20190601). Huahong granules were purchased from Honghong Pharmaceutical Co., Ltd (batch number: 20180403), dexamethasone acetate tablets from Nanguo Pharmaceutical Co., Ltd (batch number: 171202), acetonitrile, methanol and ultrapure water from Merck Group Co., Ltd, formic acid from Shanghai CNW Co., Ltd, and 2-chlorophenylalanine from Shanghai GL Biochem Co., Ltd.
The ZMJG test solution was prepared by obtaining the appropriate amount of ZMJG and converting the prescription dose into rat dose; the test solution was prepared for the high, medium and low-dose groups according to its concentration. The prepared solution was stored in a refrigerator at 4 °C for subsequent experimentation. The Huahong granule test solution was prepared by obtaining the appropriate amount of Huahong granules and adding pure water; the prepared solution was stored in a refrigerator at 4 °C. The dexamethasone acetate test solution was prepared by obtaining the appropriate amount of dexamethasone acetate tablets and adding pure water; the solution was stored in a refrigerator at 4 °C. For the excipient group, the excipient of ZMJG was prepared using distilled water and stored in a refrigerator at 4 °C.
The bacterial solution was prepared using a 1:1 ratio of Escherichia coli and Staphylococcus aureus diluted with normal saline in a sterile environment to prepare 1.5 × 108 bacterial cells/mL. E. coli and S. aureus were provided by the Department of Microbiology and Immunology, Guangxi University of Traditional Chinese Medicine, China.
Instruments
Instruments used in this study included a low temperature, high-speed centrifuge; biological spectrophotometer (Eppendorf); ultra-low temperature refrigerator (Sanyo Electric Co., Ltd); vortex oscillator (Hailmen Qilinbeier Instrument Manufacturing Co., Ltd); electronic analytical balance (Cydolis Scientific Instruments Beijing Co., Ltd); analytical instruments (Tanon); automatic rapid hemorheology tester (Chongqing Weiduo Technology Co., Ltd); constant temperature incubator (Shanghai Yuejin Medical Instrument No. 1 Factory); and fluorescence digital imaging microscope (Olympus).
Methods
Model Preparation, Grouping, and Administration
After 1 week of adaptive feeding, 90 female SD rats were randomly divided into 9 groups: blank group; sham surgery group; model group; dexamethasone acetate group (Western medicine positive control); Huahong granule group (traditional Chinese medicine positive control); low-, medium- and high-dose ZMJG groups; and excipient group. Each group comprised 10 rats. Except for the blank and sham surgery groups, all groups were treated as study models. After fasting for 1 day with access to water, the modelling surgery was imitated. Accordingly, 10% chloral hydrate was injected intraperitoneally at 0.35 mL/100 g. The middle and lower portions of the rat abdomen were disinfected with iodophor, and then, the surgical area was disinfected with ethanol. A small incision was made on both sides of the uterus using surgical scissors. Using a 1-mL syringe, the tip of the needle was buried deep into the uterine cavity tissue, after which 0.05 mL of bacterial solution was injected into both sides of the uterus, followed by suturing of the incision. The blank group received no treatment, whereas the sham surgery group underwent only opening and closing of the abdomen. After modelling, all abdomens were disinfected daily to prevent infection, and rats were weighed every other day. After 14 days of successful modelling, the drug was administered once a day via gavage at a rate of 10 mL/kg for 14 days. The excipient group received administered excipients via gavage, whereas the blank and sham surgery groups received distilled water via gavage.
Observation of Uterine Anatomy and Morphology
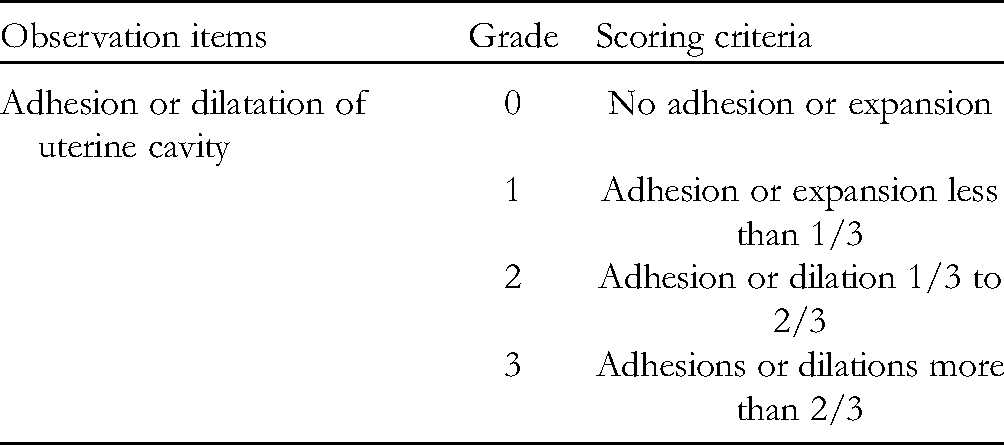
A day after the last administration, 10% chloral hydrate was intraperitoneally injected into the rats at a rate of 0.35 mL/100 g. After achieving anesthesia, blood was drawn from the rat abdominal aorta to dissect and expose the uterus. The uterus was observed using double-blind experiments and scored according to the scoring standard of uterine anatomical morphology (detail in Table 1).
Scoring Standard for the Observation of Uterine Anatomy and Morphology.

Blood Routine Test (Leukocyte and Neutrophil)
After intraperitoneal injection of 10% chloral hydrate at a dose of 0.35 mL/100 g, 1.5 mL of blood was drawn from the abdominal aorta and added to the blood collection vessel treated with EDTA-K2. The blood was mixed with an anticoagulant by gentle shaking. Thereafter, routine blood tests were performed, with subsequent data being recorded.
Coefficient of Spleen and thymus in Rats
After removing the spleen and thymus of rats, the bloodstains were washed out with normal saline, and the organ coefficients were calculated.
Pathological Examination of the Uterine Tissue
A 2-cm section of the rat uterus was cut and fixed with 4% paraformaldehyde solution. After 24 h, the solution was changed again; the specimen was dehydrated, permeabilized and embedded in paraffin to prepare 2- to 3-μm thick tissue slices. After hematoxylin and eosin (HE) staining, the slides were observed under a light microscope at 100× magnification; the pathological changes in the uterus were scored according to the scoring index detailed in Table 2.
Grading Criteria of Uterine Lesions.
Collection and Pre-treatment of Serum Samples
After the last administration, the rats were anesthetized via an intraperitoneal injection of 10% chloral hydrate at 0.35 mL/100 g. Blood was collected through the abdominal aorta and placed into the blood collection vessels without an anticoagulant. After maintaining at room temperature for 2 h, the serum was separated through centrifugation at 3000 rev. min−1 for 10 min and stored at −80 °C. Serum was collected, but urine and saliva were not. The sample was thawed at room temperature before detection. Pipette 100 µL of serum samples into a 1.5-mL EP tube, add 300 μL of methanol, and 10 μL internal standard (3.0 mg/mL, 2-chlorophenyl alanine), vortex mixed for 30 s and centrifuged at 12000 rev. min−1 at 4 °C for 15 min. Subsequently, 200 μL of supernatant was filtered into a new EP tube through a 0.22-μm filter. The filtrate was then transferred into the injection bottle for LC-MS/MS detection.
Rat Serum Sample Processing for Metabolomic Analysis
LC-MS/MS was used for serum metabolomic analysis. Chromatographic conditions were as follows: the chromatographic column was Hyper gold C18(100 mm × 2.1 mm, 1.9 μm), column temperature was 30 °C, current speed was 0.30 mL/min, injection volume was 10 μL and the mobile phases were water containing 0.1% formic acid (A) and methanol containing 0.1% formic acid (B). The gradient elution condition was 0-2 min, 5%-20%B; 2-17 min, 20%-95%B; and 17-20 min, 95%-5%B.
Mass spectrometry conditions were as follows: the ion source was electrospray ionization, the temperature was 300 °C, the sheath gas flow rate was 45 arb, the auxiliary gas flow rate was 15 arb, flow rate of the tail gas was 1 arb, the capillary temperature was 350 °C, the electrospray voltage under positive ion mode was 3.0 kV, and electrospray voltage under negative ion mode was 3.2 kV.
Screening and Identification of Different Metabolites
Based on the screening criteria of VIP > 1 and P < 0.05, potential metabolites were identified using the MetaboAnalyst5.0 platform.
Metabolic Pathway Analysis
MetaboAnalyst database is used to analyze the metabolic pathways. This database is mainly based on the Kyoto Encyclopedia of Genes and Genomes (KEGG) metabolic pathway and Human Metabolome Database and can be combined with the results of pathway enrichment analyzes to identify the most relevant metabolic pathways.44,45
Statistical Analysis
Data were expressed as means ± standard deviations (x ± s). SPSS 20.0 statistical was used for data analysis, and GraphPad Prism 7.0 was used for mapping. One-way analysis of variance was used for comparison between-groups comparisons, and Kruskal–Wallis test was used for grade data. Differences were considered statistically significant at P < 0.05.
Compound Discoverer software (Thermo Fisher Scientific) was used to extract and pre-process LC-MS/MS data, which were normalized and post-edited in Excel 2010. Finally, data were sorted into a two-dimensional data matrix, which included retention time, molecular weight, observation volume (sample name), and sample number. Data such as the number of substances (ID) and peak intensity can also be extracted. A total of 2836 and 872 features were obtained in the positive and negative modes, respectively. The edited data matrix was imported into the SIMCA-P14.1 (UmetricsAB, Umea, Sweden) software for multivariate statistical analysis.
Results
ZMJG Chinese Herbal Medicine: Composition and Target Prediction
By searching the TCMSP research platform and relevant literature, 137 chemical constituents with non-repetitive and effective targets were screened, including 14 constituents of Tu Fuling, 10 of Hu Zhang, 38 of Huang Bo, 45 of Ku Shen, 5 of Da Xueteng, 13 of Jin Gangci, 10 of Huo Tanmu and 2 of Bai Beiyegen. Overall, 514 targets were identified from the UniProt database via de-duplication.
Screening of CPID-Related Targets
From the GeneCards database, 3503 disease targets associated with CPID were identified. The top 200 targets with a large “score” value were selected, and 31 common targets were screened by mapping with ZMJG targets in Venny 2.1.0, as shown in Figure 2. The common target and corresponding components were imported into Cytoscape 3.6.0, after which the chemical components of ZMJG that are involved in the treatment of CPID were identified, namely, quercetin, β2 adrenergic receptors, cyclooxygenase 2, matrix metalloproteinase, isorhamnetin, berberine, stigmasterol, sitosterol, kaempferol, luteolin, neoastilbin, formononetin, lycopodine, and naringenin.
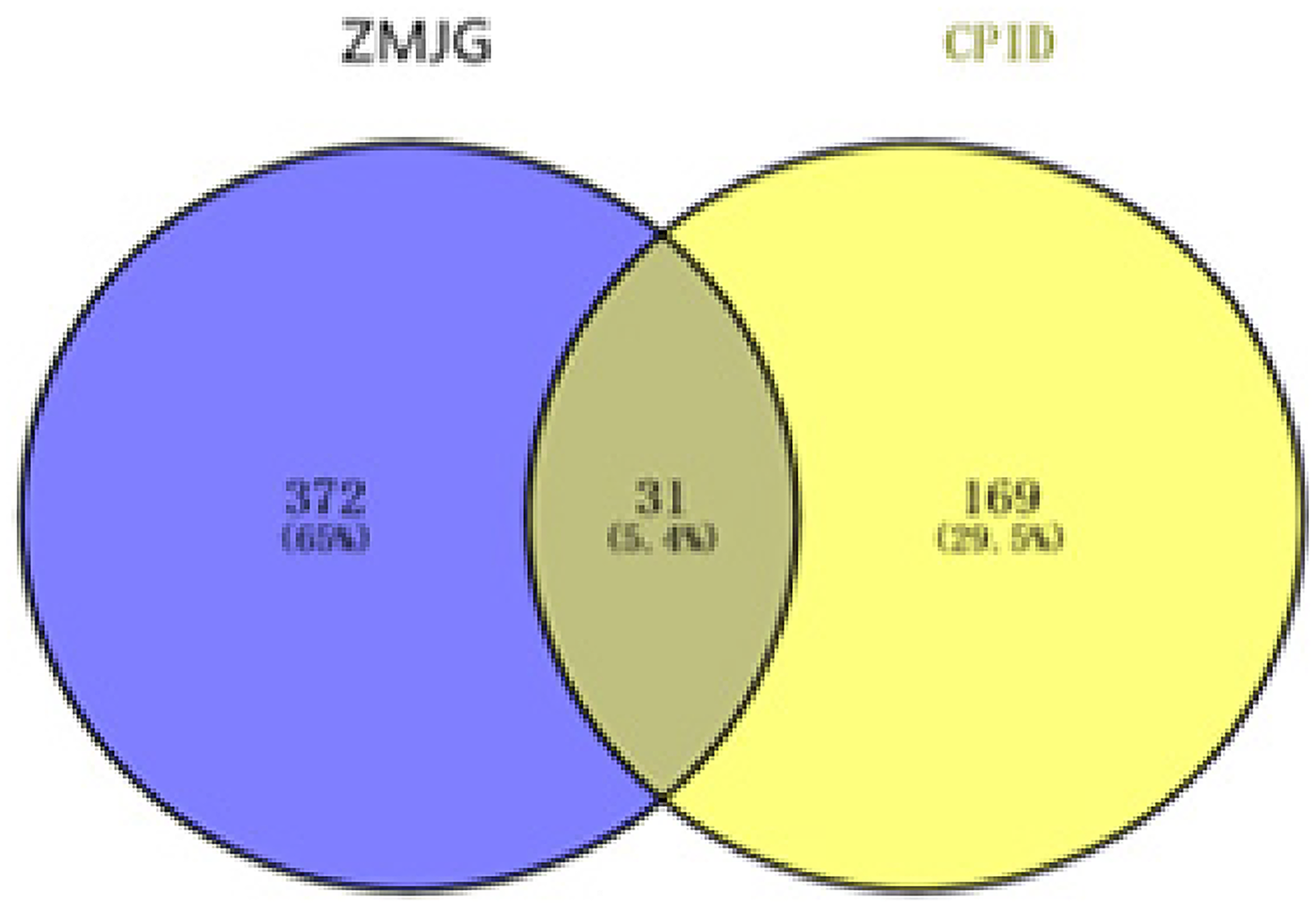
Intersection Venn graph of ZMJG and CPID.
PPI Analysis of ZMJG Target
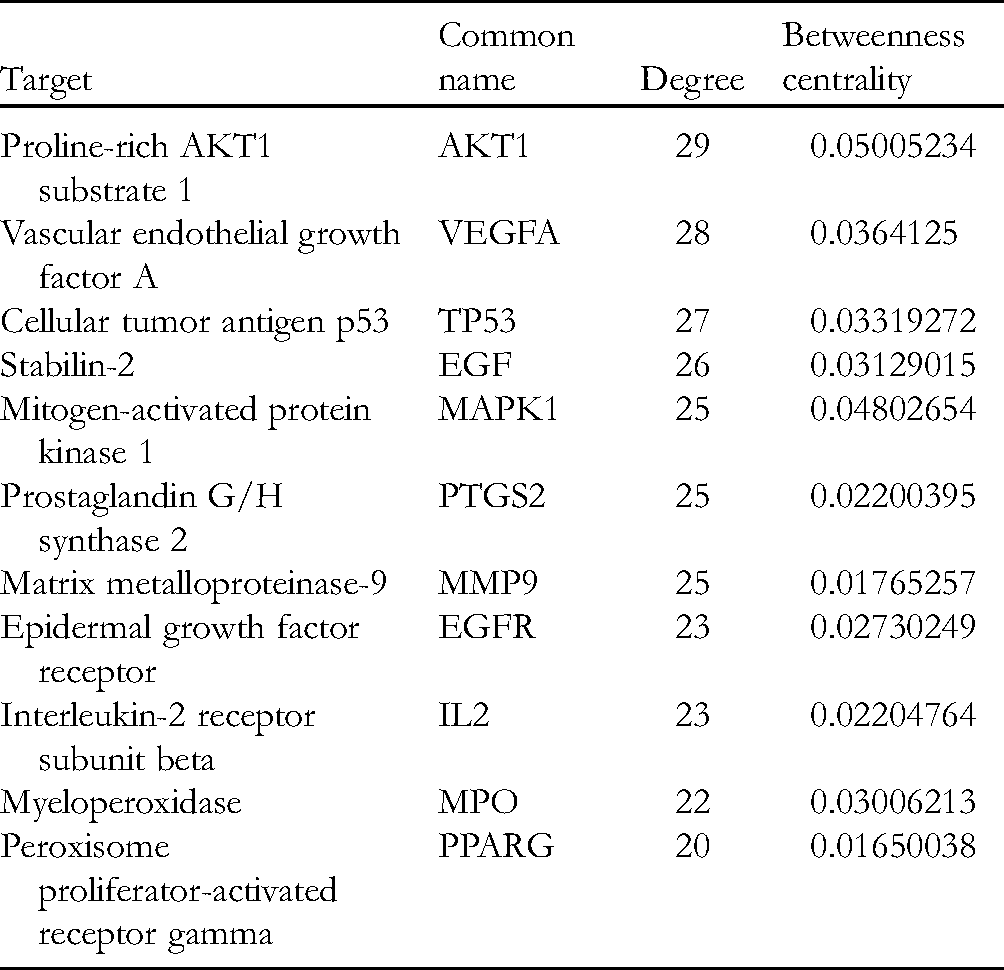
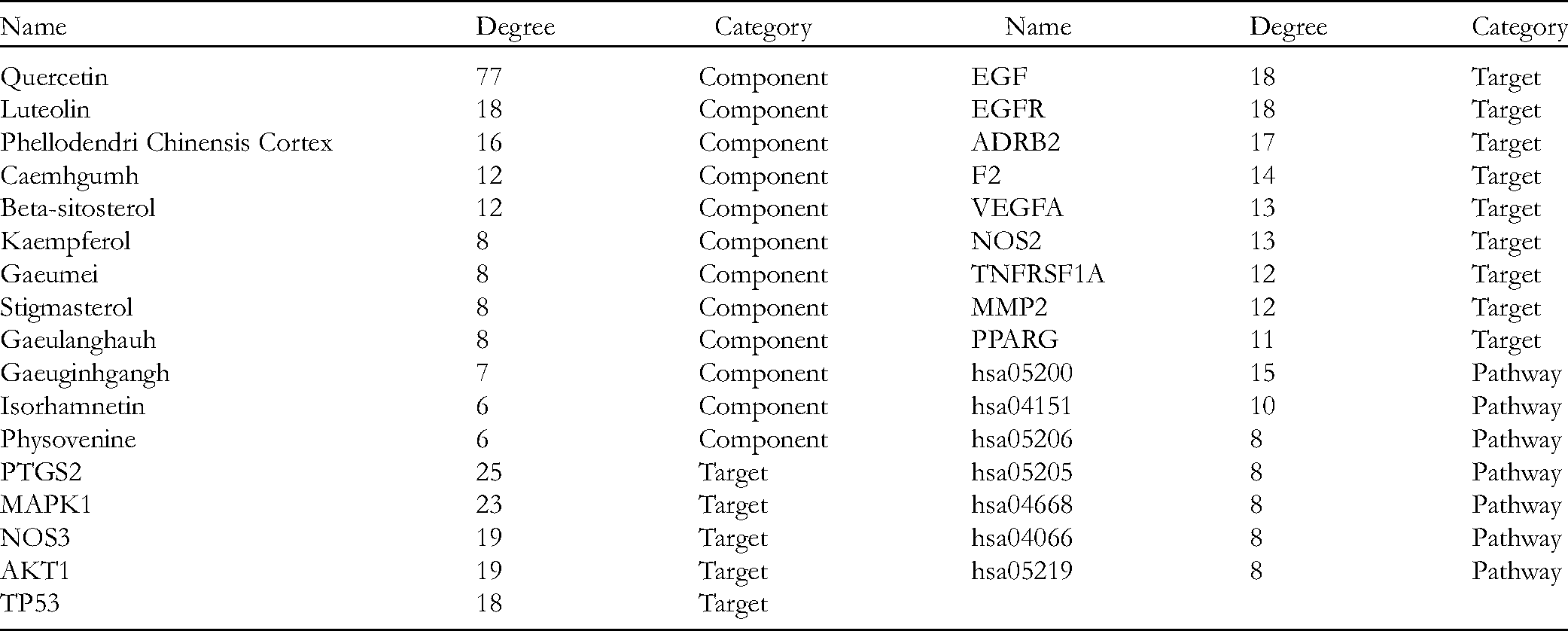
Data of 31 target proteins of ZMJG involved in the treatment of CPID were introduced into the STRING database to construct the PPI network of target protein interaction. As shown in Figure 2, 31 nodes and 280 interactions were identified, with an average degree of 18.1. The network data built on the STRING platform were imported into Cytoscape 3.6.0; the average degree value of the nodes was determined to be 18.06451613. Notably, 14 nodes had a greater than average degree value. The average median centrality of the nodes was 0.013867261, with 12 nodes having larger than average median centrality. As shown in Table 3, 11 node and median centrality values were higher than the average values for AKT1, VEGFA, TP53, EGF, MAPK1, PTGS2, MMP9, EGFR, IL2, MPO and PPARG.
Core Protein Targets of ZMJG.
GO Enrichment Analysis Results
Using the DAVID database, the GO function of 31 key targets was enriched and 77 enriched items (P < 0.01, FDR < 0.05) were identified. Among them, 48 enriched items were identified following biological process analysis, including the positive regulation of cell proliferation, regulation of signal transduction, activity of a positively regulated kinase and hormone response. Cellular component analysis showed that nine items were enriched, including plasma membrane and extracellular space. Molecular function analysis identified 20 items, including enzyme binding, protein binding and enzyme activity regulation. ZMJG reportedly plays a role in the treatment of CPID by participating in the regulation of a variety of various biological processes. Thus, we selected the top 10 channels according to the FDR value, as shown in Figure 2.
Results of KEGG Pathway Analysis
Through pathway enrichment analysis, 31 key targets were found to be significantly enriched in 32 pathways (P < 0.01, FDR < 0.01). The top 20 pathways were selected, as shown in Table 4 and Figure 2. We confirmed the involvement of the TNF signaling pathway, HIF-1 signaling pathway, estrogen signaling pathway, hepatitis C, PI3K-Akt signaling pathway, VEGF signaling pathway, and others; the findings suggested that the action mechanism of ZMJG in the treatment of CPID involves multiple pathways.
Key Targets of Zhuang Medicine Jin-mu Granules in the Treatment of CPID Participate in Pathway Enrichment Information.

Abbreviation: FDR, false discovery rate.
Construction of the “Medicinal Materials-Components-Targets-Pathways” Network
Data regarding the medicinal materials, compounds, key targets and pathways were imported into Cytoscape 3.6.0 to construct the “medicinal materials-components-targets-pathways” network. As shown in Figure 3. The network comprised 8 medicinal materials, 49 compounds, 31 targets, and 20 pathways, with a total of 108 nodes. The network topology data showed that the average degree of compounds was 5.375, with 12 compounds exhibiting a higher than average degree. The average target degree was 10.5161, with 14 targets showing a higher than average degree. The average pathway degree was 7.25, with 7 targets showing a higher than average degree. These results are summarized in Table 5. This network involved not only one molecule interacting with multiple targets but also different molecules interacting with the same target protein; this finding indicates that ZMJG exhibits multicomponent, multitarget and multichannel characteristics, preliminarily explaining the material basis for ZMJG and the related mechanism through which it alleviates inflammation.

Network diagram of “medicinal materials-components-targets-pathways.” From inside to outside, the concentric circles in the figure are herbs, pathways, targets, and components. There are 8 kinds of herbs, 49 kinds of compounds, 31 kinds of predicted targets, and 20 signal pathways in the network. The green rectangle in the inner circle represents the 8 herbs in ZMJG, the pink rectangle represents the compound, the light green rectangle represents the potential prediction target, and the outermost blue rectangle represents the predicted signal pathway.
“Medicinal Materials-Components-Targets-Pathways” Network Results of Network Topology Parameters.
Preliminary Verification of the Mechanism of Action
The active ingredients (quercetin, luteolin, beta-sitosterol, and kaempferol) were used for molecular docking with core targets (AKT1 and HIF1). When the binding energy is ≤ −4.25 kcal·mol−1, there is a certain binding activity between molecules. In the eight groups of receptor-ligand docking results, there were seven pairs with an affinity ≤ −5 kcal·mol−1, and the scores of the remaining pairs were all less than −4.25 kcal·mol−1, indicating that the active ingredients screened by this particle are similar to core targets. The core targets may have good binding activity, which provides a reference for subsequent experimental verification. The specific molecular docking results are shown in Table 6 and Figure 4.

The result of docking between components and core targets. Red is the component compound, and blue is the protein target. (A) AKT1-Quercetin, (B) HIF1-Quercetin, (C) AKT1-Luteolin, (D) HIF1-Luteolin, (E) AKT1-Kaempferol, (F) HIF1-Kaempferol, (G) AKT1-beta-sitosterol, and (H) HIF1-beta-sitosterol.
Key Component Target Molecular Docking.

In Vivo Validation of the Efficacy of ZMJG
Uterine Anatomy and Morphology Score
Compared with the blank group, no significant difference was observed in the uterine morphology score in the sham surgery group (P > 0.05), but a significantly greater uterine morphology score was observed in the model group (P < 0.01). Compared with the CPID model group, the high-dose ZMJG group (P < 0.01), medium-dose group, Huahong granule group and dexamethasone acetate group (P < 0.05) exhibited significantly higher scores, as shown in Table 7.
Effects of Zhuang Medicine Jin-mu Granules on the Uterine Anatomical Morphology of Rats with Chronic Pelvic Inflammatory Disease (x ± s, n = 10).

Compared with the blank group, + P < 0.05, + +P < 0.01; compared with the model group, *P < 0.05, **P < 0.01.
Blood Routine Test Results
Compared with the blank group, the model group had significantly high leukocyte and neutrophil counts (P < 0.01), indicating that the model was successfully established. However, no significant difference was observed in leukocyte and neutrophil counts between the sham surgery and blank groups (P > 0.05), indicating that abdominal switch did not cause inflammation in rats. Compared with the model group, the high-, medium- and low-dose ZMJG groups showed significantly lower leukocyte counts (P < 0.05), whereas the low- and medium-dose ZMJG groups showed significantly low neutrophils counts (P < 0.05 and P < 0.01, respectively), as shown in Figures 5.

(A) Changes in white blood cell count following routine blood tests. Blank group; B. Sham surgery group; C. Model group; D. Huahong granule group; E. Dexamethasone acetate group; F. High-dose group; G. Medium-dose group; H. Low-dose group; I. Accessories group. Compared with the blank group, + +P < 0.01; compared with the model group, *P < 0.05, **P < 0.01. (B) Changes in the granulocyte count following routine blood tests. A. Blank group; B. Sham surgery group; C. Model group; D. Huahong granule group; E. Dexamethasone acetate group; F. High-dose group; G. Medium-dose group; H. Low-dose group; I. Accessories group. Compared with the blank group, + +P < 0.01; compared with the model group, *P < 0.05, **P < 0.01. (C) Effects of Zhuang Medicine Jin-mu Granules on the thymus and spleen coefficients of rats with chronic pelvic inflammatory disease. Blue: thymus coefficient; Red: spleen coefficient. A. Blank group; B. Sham surgery group; C. Model group; D. Huahong granule group; E. Dexamethasone acetate group; F. High-dose group.; G. Medium-dose group; H. Low-dose group; I. Accessories group. Compared to the blank group, + +P < 0.01; compared to the model group, *P < 0.05, ** P < 0.01. (D) Effects of Zhuang Medicine Jin-mu Granules on the pathological sections of the uterus of rats with chronic pelvic inflammatory disease (HE, × 100). A. Blank group; B. Sham surgery group; C. Model group; D. Dexamethasone acetate group; E. Huahong granule group; F. High-dose group; G. Medium-dose group; H. Low-dose group; I. Accessories group.
Effects on Spleen and thymus Coefficients
Compared with the blank group, the model group had significantly high thymus and spleen coefficients (P < 0.05 and P < 0.01, respectively), but no significant difference was observed between the two groups (P > 0.05). Compared with the model group, the high-dose group had significantly low thymus and spleen coefficients (P < 0.05 or P < 0.01). Compared with the blank and model groups, the dexamethasone acetate group had a significantly low thymus coefficient. Given that dexamethasone acetate is an adrenocortical hormone, it may have more side effects than the Huahong granule (Figure 5).
Uterine Histopathological Score
Pathological Score of Intrauterine Adhesion and Dilatation
Compared with the blank group, the CPID rat model group had a significantly high uterine pathological section score (intrauterine adhesion and dilatation; P < 0.01). No difference in the score was observed between the sham surgery and the blank groups (P > 0.05). Compared with the model group of CPID rats, the high-dose and dexamethasone acetate groups had significantly lower uterine pathological section scores (intrauterine adhesion and dilatation; P < 0.05), as seen in Table 8.
Effect of Zhuang Medicine Jin-mu Granules on Intrauterine Adhesion and Dilatation in Chronic Pelvic Inflammatory Disease Rats.

Compared with the blank group, + P < 0.05, + +P < 0.01; compared with the model group, *P < 0.05, ** P < 0.01.
Effects on a Pathological Section of the Uterus of Rats in Each Group
HE staining and light microscope observation showed that the structure of the uterine cavity and endometrial epithelium of rats in the blank and sham surgery groups remained intact, without congestion and swelling. The main pathological changes in the model group included intrauterine adhesion or dilatation, endometrial congestion, and edema and epithelial cell proliferation with branching papillary, accompanied by a certain degree of inflammatory cell infiltration, which was consistent with the characteristics of clinical pelvic inflammatory disease sequelae. In the high-dose and dexamethasone acetate groups, the structure of the uterine cavity was clear, the endometrial epithelium was intact, intrauterine adhesion or dilatation of less than 1/3 was occasionally present, epithelial cells were highly columnar, and lamina propria was slightly hyperaemic and edematous. In the Huahong granule group, the uterine tissue was slightly dilated without adhesion and endometrium was locally congested and swollen, which could have improved the proliferation of epithelial cells. The intrauterine adhesions or dilations were 1/3 to 2/3 in the middle- and low-dose groups; some of these rats had obvious hyperemia and edema, with highly columnar epithelial cells. In the excipient group, the epithelial cells proliferated in branching papillary shape, uterine cavity adhered or expanded and endometrium was congested and swollen. The location of the lesion is indicated using an arrow, as seen in Figure 5.
Analysis of serum Metabonomics in Rats
LC-MS/MS was used to scan the samples in positive and negative modes to analyze and identify changes in rat blood metabolites. An ion flow diagram is presented in Figure 6. After overlapping the total ion flow chromatograms (TIC) of QC samples, it was found that the retention time reproducibility of the instrument was very good and the instrument was stable, so the reliability of instrument analysis and data results was improved.
Ion flow diagram of serum samples in positive and negative ion modes.
Multivariate statistical analysis was used to analyze the serum samples of each group to determine metabolic differences between the blank and model groups and between the model and administration groups. Figure 7 shows the PCA and OPLS-DA results of the serum samples from each group. Accordingly, the blood metabolism profiles of the model and normal groups differed. Rats treated with Zhuang medicine Jin Mu granule had a serum metabolic profile different from that of the rats in the model group. The serum metabolic profile of rats treated with ZMJG was similar to that of the normal group, whereas the sample points of the medium-dose ZMJG group were more similar to that of the normal group; these results indicated that ZMJG affected the serum metabolic profile of CPID rats such that it showed a tendency to be similar to that of normal rats. OPLS-DA is a supervised prediction model. To verify the reliability of the model and prevent overfitting, permutation verification was performed. The results showed that in the positive mode, one principal component and four orthogonal components were identified (R2X = 0.336, R2Y = 0.975, Q2 = 0.978); however, in the negative mode, one principal component and five orthogonal components were identified (R2X = 0.478, R2Y = 0.96, Q2 = 0.801); these findings indicated that the model had a good prediction ability, as shown in Figure 8.
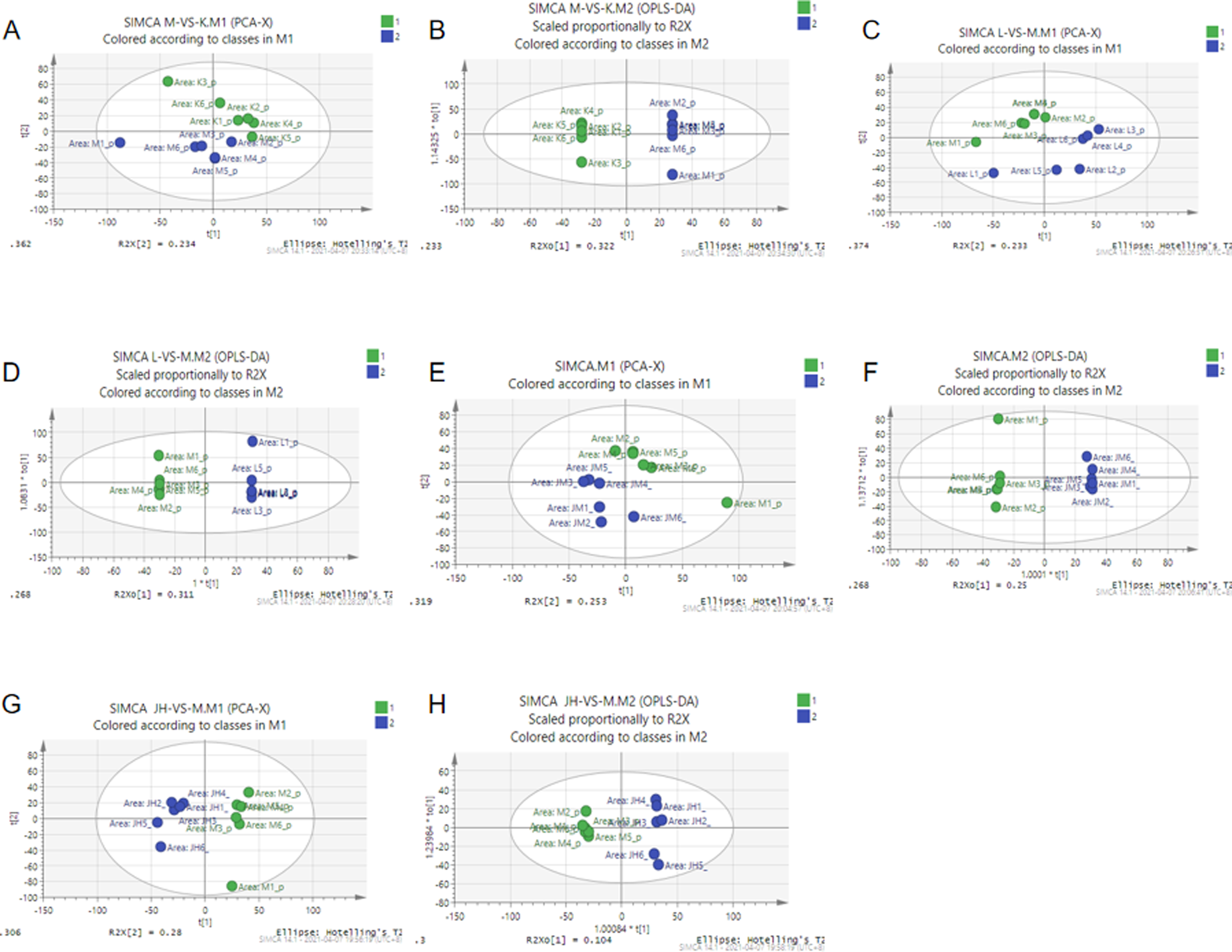
PCA and OPLS-DA scores of serum samples. A. PCA Blank group (green) versus model group (blue). B. OPLS-DA Blank group (green) versus model group (blue). C. PCA Model group (green) versus low dose group (blue). D. OPLS-DA Model group (green) versus low dose group (blue). E. PCA Model group (green) versus medium-dose group (blue). F. OPLS-DA Model group (green) versus medium-dose group (blue). G. PCA Model group (green) versus high-dose group (blue). H. OPLS-DA Model group (green) versus high-dose group (blue).

Verification of permutation. A. Medium-dose group-model group. B. Model group-blank group. C. Low-dose group-model group. D. High-dose group-model group.
Screening and Identification of Different Metabolites
Eight potential metabolites were identified. Compared with the blank group, the model group had significantly high levels of nicotinamide, 1-methyl nicotinamide, L-histidine, alpha-ketoglutaric acid and serotonin, but significantly lower levels of arachidonic acid, L-(-)-methionine and D-(-)-aspartic acid. Compared with the model group, the dexamethasone acetate and Huahong granules (positive control of traditional Chinese and Western medicine) groups showed significant improvement in the eight metabolic markers after ZMJG treatment; this indicates that the correlation between these metabolic markers and pathway changes is the possible mechanism through which ZMJG improves CPID. Table 9 shows the results of serum differential metabolite analysis, and Figure 9 presents the relevant heat map.

Thermograms of the eight potential metabolites. Potential biomarkers are determined by the condition that the fold change is greater than 1.5, P < 0.05 and VIP > 1.0. They are nicotinamide, 1-methylnicotinamide, arachidonic acid, L-histidine, serotonin, L-(-)-methionine, D-(-)-aspartic acid, and alpha-ketoglutaric acid.
Analysis of Serum Differential Metabolites.

Compared to the blank group, + P < 0.05, + +P < 0.01; compared to the model group, *P < 0.05, ** P < 0.01.
Metabolic Pathway Analysis
A total of 13 related metabolic pathways were screened out from the MetaboAnalyst platform, as shown in Figure 10. Taking “Impact > 0.04” as the screening condition, 7 major metabolic pathways—arachidonic acid; nicotinate and nicotinamide; histidine; tryptophan; cysteine and methionine; tricarboxylic acid cycle (TCA cycle); alanine, aspartate, and glutamate—were identified and studied.
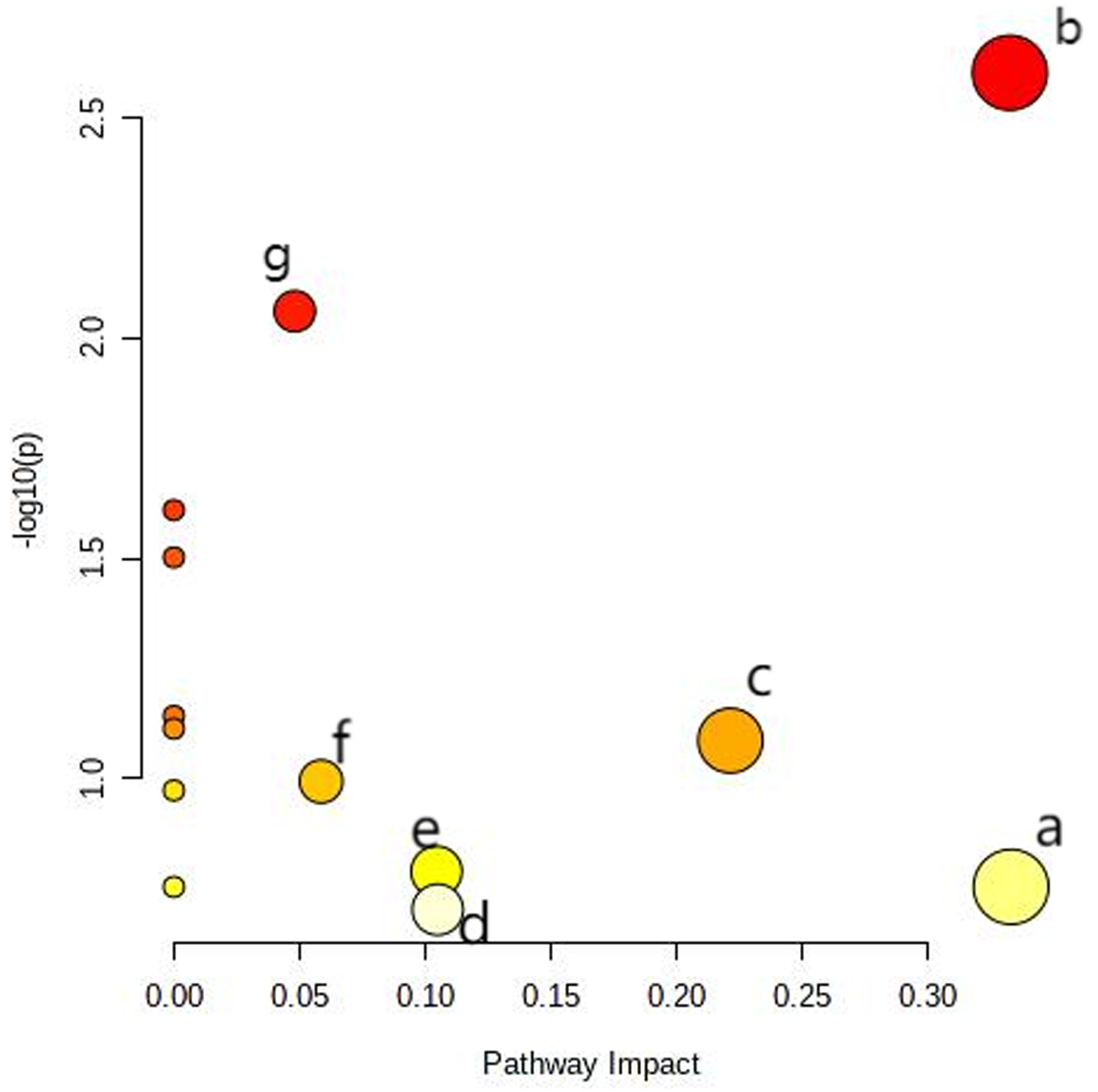
Analysis of the metabolic pathways of different metabolites. A. Arachidonic acid metabolism; B. nicotinate and nicotinamide metabolism; C. histidine metabolism; D. tryptophan metabolism; E. cysteine and methionine metabolism; F. citrate cycle (TCA cycle); G. alanine, aspartate, and glutamate metabolism. In the figure, according to “Impact > 0.04” as the screening condition, 7 main metabolic pathways were selected.
Integration of Network Pharmacology and Metabonomics
We next combined the results of the above-mentioned network pharmacology and metabolomics and conducted a systematic analysis, focusing on the correlation between metabolites and signaling pathways to build an interaction network involved in biological processes (Figure 11). This network indicates that the mechanism of ZMJG in the treatment of CPID is based on the positive regulation of cell proliferation, signal transduction, kinase activity, and hormone responses.

Combination mechanism of network pharmacology and metabolomics analysis. The signal transduction pathway predicted by the target is connected to the differentially regulated metabolic pathway through biological functions. From the signal transduction pathway analysis, the key targets of ZMJG were significantly enriched in HIF-1, TNF, estrogen, PI3k-Akt, VEGF, and Rap1 signaling pathways, which are essential for anti-inflammation, cell proliferation regulation, signal transduction regulation, immune modulation, and other related biological processes. From the perspective of metabolomics, GO and KEGG pathway enrichment analyses for different metabolites showed consistent results. In conclusion, the network pharmacological analysis shows that ZMJG can be used as an immunomodulator, anti-inflammatory agent, cell proliferation mediator, and signal transduction regulator in the treatment of CPID.
Discussion
Previous research has primarily focused on the modernization of traditional Chinese medicine. Indeed, the application of modern science and technology to the field of traditional Chinese medicine remains the only approach for a more scientific approach to traditional Chinese medicine, with research on the mechanisms of traditional Chinese medicinal compounds being the key to realizing this modernization. Traditional Chinese medicine compound preparations are the main form used clinically and are an important carriers for their inheritance and innovation. At present, it is the main direction of the research and development of new Chinese medicines. Network pharmacology reveals the molecular mechanism of drug therapies by analyzing the interaction between chemical components and disease-related macromolecular targets. The active components related to ZMJG are obtained from the TCMSP database, and the disease targets related to CPID are retrieved from the GeneCards database. The GO function enrichment analysis and KEGG pathway analysis found that the main chemical components in ZMJG are quercetin, β2 adrenergic receptors, cyclooxygenase 2, matrix metalloproteinases, isorhamnetin, and berberine. Additionally, these components may act on key proteins, such as AKT1, VEGFA, TP53, EGF, and MAPK1, to mediate a number of biological processes related to anti-inflammation, cell proliferation regulation, signal transduction regulation, and immune modulation, thereby playing a role in the treatment of CPID. Important signaling pathways, including TNF, HIF-1, estrogen, PI3K-Akt, and VEGF, were also revealed. After the preliminary determination of the multi-dimensional regulatory network for the treatment of CPID, the in vivo efficacy of ZMJG was verified in a rat model. By investigating the morphological scoring of the uterus, results of routine blood tests, influence of the spleen and thymus coefficient, and scoring of uterine histopathology, this study demonstrated that ZMJG are indeed effective in the treatment of CPID.
Metabonomics uses the metabolites in the organism as the object and analyzes the changes in metabolites when the organism is stimulated or in different states. Metabonomics can be performed to analyze and detect the level of small molecules in body fluids, determine which compounds have significant abnormalities, and reveal the mechanism underlying disease development. Metabolomics analysis identified 8 different potential metabolites. Notably, a therapeutic effect was achieved by downregulating the levels of nicotinamide, 1-methylnicotinamide, L-histidine, Serotonin, and alpha-ketoglutaric acid and upregulating the levels of arachidonic acid, L-(-)-methionine, and D-(-)-aspartic acid. ZMJG normalized the expression levels of these eight biomarkers, indicating that ZMJG function through a variety of mechanisms and targets in the treatment of CPID.
Our finding suggested that metabolites were involved in seven major metabolic pathways to ameliorate CPID, namely arachidonic acid metabolism, nicotinate and nicotinamide metabolism, histidine metabolism, tryptophan metabolism, cystine and methylene metabolism, citrate cycle (TCA cycle), and alanine, aspartate, and glucose metabolism.
Studies have shown that arachidonic acid is mainly metabolized through the cyclooxygenase (COX) and lipoxygenase (LOX) pathways. COX, also known as prostaglandin G/H synthase, has two types: COX-1 and COX-2. COX-1 is stably expressed and widely present in all body tissues, especially in the gastrointestinal epithelial tissues, platelets, and kidneys. It also participates in maintaining the integrity of the gastrointestinal mucosa and regulating vasomotor function, platelet aggregation, and renal function. 46 COX-2 is an inducible COX that is almost undetectable under normal conditions, but is induced in the presence of various types of damaging stressors. Prostaglandins generated by COX-2 act on arachidonic acid, mainly promoting inflammatory responses and tissue damage. LOX is a heme-free dioxygenase that is mainly expressed in immune, epithelial, and tumor cells. It has a variety of physiological and pathophysiological functions and plays an important role in inflammation, skin diseases, and tumorigenesis.2,47 Histidine is the precursor of carnosine, which plays an important role as a buffer and antioxidant in human muscle and brain. It is produced from histidine and is synthesized in tissues by sarcosine synthase β-alanine, a process that requires ATP hydrolysis and occurs in giant cells of the immune system.48–50 Tryptophan is an essential amino acid for animal growth and development, and its metabolites play an important role in intestinal immunity, intestinal peristalsis, cell secretion, neural response regulation, mood regulation, and anti-oxidation. Evidence has shown that intestinal homeostasis depends on a stable intestinal microenvironment, which is composed of intestinal cells, neurons, and glial, smooth muscle, endocrine, and immune cells. These cells receive and transmit signals from microorganisms to regulate the intestinal microenvironment. Studies have shown that Bifidobacteria and Lactobacilli improve the survival rate of intestinal epithelial cells by inhibiting the apoptosis pathway activated by pathogenic bacteria.51–53 Indole derivatives produced by tryptophan metabolism are important signaling molecules produced by probiotics to maintain the intestinal epithelial barrier and inhibit inflammation.
Conclusion
The present study showed that ZMJG exhibited therapeutic effects against CPID. Network pharmacology and metabolomics were combined to explore the action mechanism of ZMJG in the treatment of CPID. Based on the comprehensive analysis of the network diagram of “medicinal materials-components-targets-pathways” and the enrichment of metabolic pathways, we speculate that the mechanism of ZMJG is attributed to biological processes related to anti-inflammation, cell proliferation regulation, signal transduction regulation, and immune modulation. Moreover, network pharmacology identified 49 active compounds involving 31 common targets. Luteolin and kaempferol in Ku Shen and Huo Tanmu, stigmasterol in Tu Fuling and Huang Bo, and isorhamnetin in Huo Tanmu, were the main components connected to the target sites and involved in the therapeutic effects against CPID. Our results suggest that ZMJG exert their therapeutic effects against CPID through a combination of multiple pathways, metabolic pathways, and multi-component multi-target modes. This research provides a detailed theoretical basis for the development and clinical application of ZMJG, new research ideas for the treatment of CPID using Chinese medicine, and an important reference for the pathogenesis and clinical treatment of CPID.
Footnotes
Acknowledgements
Author note
Zhiping Wang Key Laboratory of Common Technology of Chinese Medicine Preparations, Guangxi University of Chinese Medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major Science and Technology Projects in Guangxi (No. AA18126003) and the Innovation Project of Guangxi Graduate Education (No. YCXJ2021017).
Author Contributions
Zhi-ping Wang and Tan-fang Xie designed the research project; Yu-han Wang, Lin-jie Li, Fang Li, Kai Yu, Xiao Liu, Jun Chen, Xiao-xun Wang, Qiao Feng, and Yan Huang performed the experiments; Yu-han Wang wrote the manuscript; all author contributed to the preparation of the manuscript.
Ethical Approval
All animal experiments were conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee of Guangxi University of Traditional Chinese Medicine. (DW20200415-53).
