Abstract
The novel Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) is causing coronavirus disease 2019 (COVID-19) pandemic. Ancient Chinese herbal formulas are effective for diseases caused by viral infection, and their effects on COVID-19 are currently being examined. To directly evaluate the role of Chinese herbs in inhibiting replication of SARS-CoV-2, we investigated how the phytochemicals from Chinese herbs interact with the viral RNA-dependent RNA polymerase (RdRP). Total 1025 compounds were screened, and then 181compounds were selected for molecular docking analysis. Four phytochemicals licorice glycoside E, diisooctyl phthalate, (-)-medicocarpin, and glycyroside showed good binding affinity with RdRp. The best complex licorice glycoside E/RdRp forms 3 hydrogen bonds, 4 hydrophobic interactions, 1 pair of Pi-cation/stacking, and 4 salt bridges. Furthermore, docking complexes licorice glycoside E/RdRp and diisooctyl phthalate/RdRp were optimized by molecular dynamics simulation to obtain the stable conformation. These studies indicate that they are promising as antivirals against SARS-CoV-2.
Introduction
SARS-CoV-2 mainly infects human respiratory system and has caused the global COVID-19 pandemic. As of January 2022, there have been 3.2 hundred million people infected, including 5.5 million deaths, reported to WHO. Medical scientists did a lot of research in the treatment of COVID-19 in the past 2 years; however, there is still a shortage of effective drugs against the viral infection 1 .
SARS-CoV-2 is genetically related to SARS-CoV; they belong to the genus Betacoronavirus 2 . The viral genome encodes 29 proteins including 4 structural proteins—Membrane, Spike, Envelope, Nucleocapsid, and non-structural proteins (nsps). A polyprotein is cleaved by viral proteases generating 16 small proteins with one called nsp12, or RNA-dependent RNA polymerase (RdRp). A set of nsps forms complex to facilitate viral transcription and replication. RdRp complexed with nsp7/nsp8 catalyzes the viral RNA synthesis, and is a specific RNA polymerase controlling viral replication and transcription. Hence, RdRp is selected as an important target for the novel drug development3,4.
Several drugs, such as remdesivir, nirmatrelvir, and ritonavir, were authorized for emergency use by the US Food and Drug Administration recently. Remdesivir was the first antiviral treatment for COVID 19. It could be covalently bound to the replicating RNA by mimicking an RNA nucleotide, which blocks RNA elongation by RdRp5,6. In another way, medical plants have long been utilized for treating various diseases all over the world. The pharmaceutical active compounds from these herbs could be developed into drugs with minimal or no side-effect. By use of computer-aided drug discovery, medical researchers found some potential inhibitors from African plants for 3CLpro of SARS-CoV-2 7 . Qazt et al reported that marmin, malic acid, benzylamine, and khusinoloxide from Ayurvedic herbs could be potential inhibitors for RdRp and 3CLpro of the virus 8 .
In this study, in silico analysis was applied to explore the natural compounds from Chinese herbs against SARS-CoV-2. With the target protein, thousands of natural compounds could be quickly screened by molecular docking and dynamics simulation9–11. We studied 8 traditional Chinese medicine (TCM) frequently used herbs Allium sativum, Houttuynia cordata, Forsythia suspensa, Scutellaria baicalensis, Lonicera japonica, Polygonum cuspidatum, Glycyrrhiza uralensis, and Gentiana scabra, which have antiviral properties12,13. The selected phytochemicals of the herbs were docked against the viral protein RdRP. RdRP is indispensable for viral transcription and replication, which makes it target for antiviral drugs.
Materials and Methods
Target Protein and Phytochemicals
The target protein RdRp with its PDB ID 6M71 was downloaded from RCSB PDB website 4 .
To select Chinese herbs, TCM references were reviewed carefully to select Chinese herbs that exhibit antiviral activities12–14. Eight TCM frequently used herbs Allium sativum, Houttuynia cordata, Forsythia suspensa, Scutellaria baicalensis, Lonicera japonica, Polygonum cuspidatum, Glycyrrhiza uralensis, and Gentiana scabra were chosen in this study. Subsequently, phytochemicals of these herbs were retrieved from Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP). Compounds were subjected to clusters for absorption, distribution, metabolism and excretion (ADME) analysis. Compounds with oral bioavailability ≥30%, drug-likeness ≥0.18 were subjected to molecular docking.
Molecular Docking and Interaction Analysis
Before starting molecular docking, energy minimization of the phytochemicals was performed using Chem 3D software. Phytochemicals were saved to mol2 format for molecular docking. RdRp was optimized by ligand extraction, water extraction, and hydrogenation; then molecular docking was carried out with the Surflex-Dock module of SYBYL-X 2.0. A protomol was generated for the identification of the active binding pocket. AMBER FF99 force field was used throughout the docking study, and function scoring was applied to evaluate the binding activity of RdRp-ligand complex. Total Score is the total Surflex-Dock score expressed as −log (Kd) 15 .
According to the binding affinities, interaction analysis was applied to the top 4 complexes by protein-ligand profiler 16 .
Molecular Dynamics Simulation
SYBYL-X 2.0 was used for molecular dynamics simulation (MDS) of the 2 best-docked RdRp-ligand complexes. By use of the TRIPOS force field, calculations of molecular mechanics were performed in SYBYL. The energy function of the complex was the sum of parameters (angle bending, bond stretching, electrostatic, planarity, torsion, and van der Waals). Molecular Silverware was used to layer solvent molecule water around the RdRp-ligand complexes. The RdRp-ligand complexes were simulated at constant volume, constant temperature 300 K with 1000 femtoseconds (fs) as coupling constant in water system. With every 5 fs step, molecular simulations were recorded. The MDS was obtained from these snapshots analysis 17 .
The binding energy of a protein-ligand complex was computed by Binding Affinity Prediction of Protein-Ligand (BAPPL) server18,19.
Deformability, B-Factor, and Covariance Computation
The further MDS of 2 complexes was run through iMOD server (iMODS). The iMODS, using normal mode analysis, evaluates complex stability by calculating its internal coordinates. Deformability, B-Factor, and covariance were calculated to elucidate the complex stability using iMODS software 20 .
Results
Phytochemical Screening of Chinese Herbs
The natural products from Chinese herbs with the antiviral activities against SARS-CoV-2 have not been previously examined. Eight TCM frequently used herbs Allium sativum, Houttuynia cordata, Forsythia suspensa, Scutellaria baicalensis, Lonicera japonica, Polygonum cuspidatum, Glycyrrhiza uralensis, and Gentiana scabra were subjected to drug screening in this study. Altogether 1025 phytochemicals of these herbs were retrieved from TCMSP databases. Subsequently, 181 phytochemicals were selected by ADME screening (Supplemental Table 1). Then they were subjected to molecular docking analysis.
Molecular Docking
The aim of this study was to explore phytochemicals that could effectively bind to the viral protein RdRp (Figure 1), and therefore, could potentially be used as drugs inhibiting viral replication. Out of the 1025 ADME screened compounds, 181 of them passed, which were then subjected to molecular docking analysis (Supplemental Table 2). Four out of 181 phytochemicals showed a good binding affinity with the viral protein RdRp. These phytochemicals are licorice glycoside E (Total Score 9.60), diisooctyl phthalate (Total Score 8.49), (-)-medicocarpin (Total Score 8.18) and glycyroside (Total Score 8.17) (Figure 1).

Structures of target protein and 4 phytochemicals. (A) RdRP (PDB ID: 6M71). (B) (a) Licorice glycoside E, (b) Diisooctyl phthalate,(c) (-)-Medicocarpin, and (d) Glycyroside.
Figure 2 presents the docked complexes of phytochemicals/RdRp. Licorice glycoside E, (-)-medicocarpin and glycyroside are phytochemicals from Glycyrrhiza uralensis, diisooctyl phthalate is one of phytochemicals from Scutellaria baicalensis. The top 2 complexes licorice glycoside E/RdRp and diisooctyl phthalate/RdRp were selected for MDS.
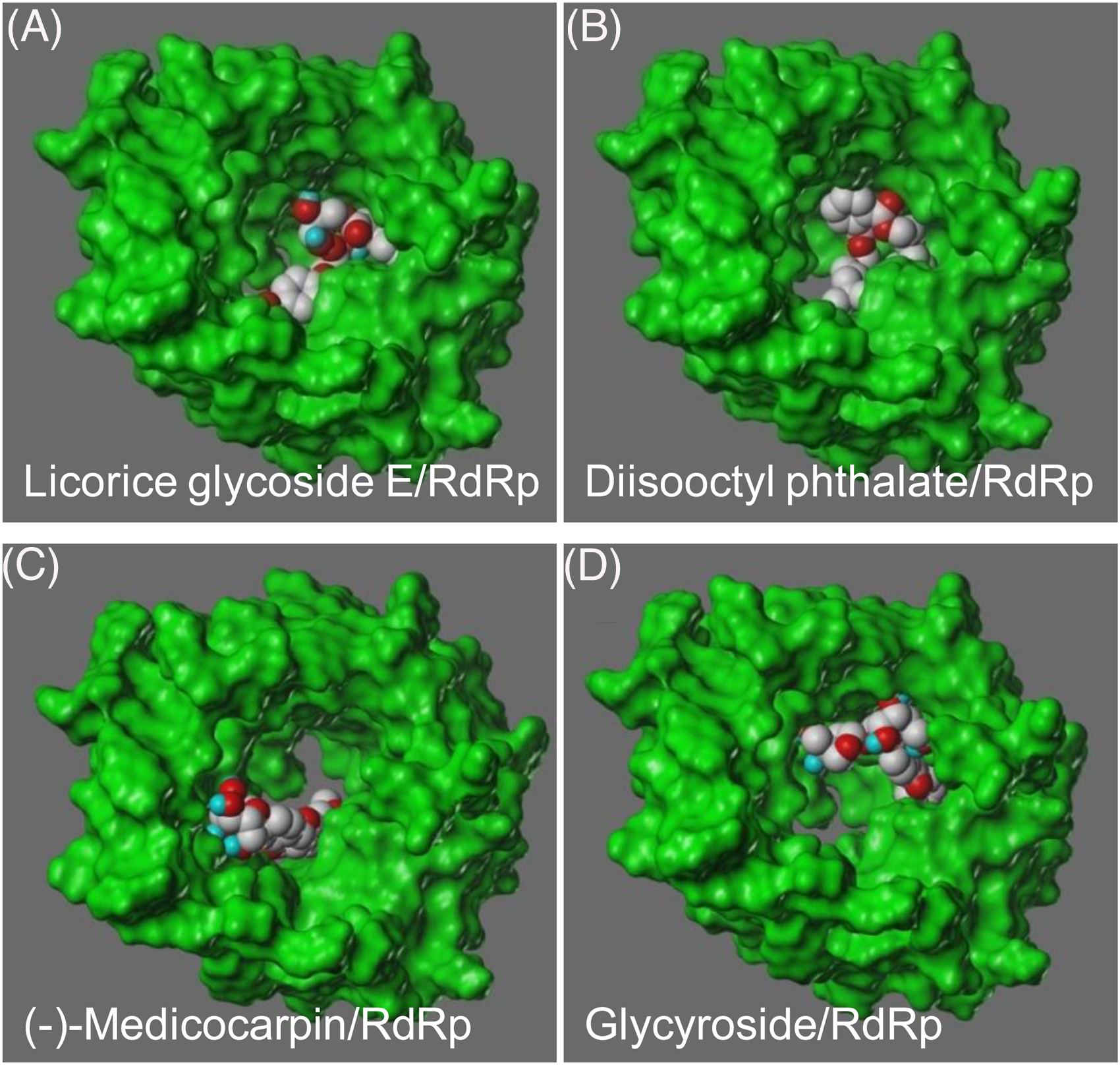
The top 4 docked complexes for the viral RdRp. (A) Licorice glycoside E/RdRp, (B) Diisooctyl phthalate/RdRp, (C) (-)-Medicocarpin/RdRp, and (D) Glycyroside/RdRp.
Interaction Analysis of RdRp/Phytochemicals
To understand the molecular interaction of complexes in detail, protein/ligand interaction analysis was performed to examine the interactions between phytochemicals licorice glycoside E, diisooctyl phthalate, (-)-Medicocarpin, and glycyroside with the target protein RdRp. Table 1 showed the interaction results. Licorice glycoside E formed 3 hydrogen bonds with RdRp residues LYS 621A, ASP 760A, and ARG 836A, 4 hydrophobic bonds with TRP 455A, ALA 550A, ARG 624A, and ASP 833A, a pi–pi stacking bond with HIS 439A, and 4 salt bridges with LYS 551A and ARG 553A. Diisooctyl phthalate formed 3 hydrogen bonds with RdRp residues LYS 621A, LYS 622A, and ARG 623A, 3 hydrophobic interactions with TRP 455A, ARG 553A, and LYS 621A. There was one salt bridge formed between diisooctyl phthalate and RdRp with LYS 621A. (-)-Medicocarpin formed 11 hydrogen bonds with RdRp residues ARG 553A, ARG 553A, ARG 555A, ARG 555A, LYS 813A, SER 814A, ARG 836A, ARG 836A, ASP 865A, ASP 865A, and ASP 865A, a single pi–pi stacking interaction with ARG 555A, and one salt bridges with ARG 555A. Glycyroside formed 11 hydrogen bonds with RdRp residues ARG 553A, ARG 553A, LYS 621A, LYS 622A, LYS 623A, THR 687A, ASN 691A, SER 759A, ASP 760A, 761A, and 761A, and a single hydrophobic interaction with LYS 621A.
Non-Covalent Binding Between the 4 Phytochemicals and the Viral RdRp.

Abbreviations: Dist, distance; ASN, asparagine; TRP, tryptophan; ALA, alanine; LYS, lysine; ARG, arginine; THR, threonine; ASP, aspartic acid; SER, serine.
The interaction analysis indicated that the complexes with a good binding activity were the ones with the higher number of hydrophobic interactions, hydrogen bonds, salt bridges, and pi–pi stacking interactions. Figure 3 presents the non-covalent interactions between RdRp and phytochemicals mentioned above.

Non-covalent binding between the 4 phytochemicals and the viral RdRp. (A) Licorice glycoside E/RdRp, (B) Diisooctyl phthalate/RdRp, (C) (-)-Medicocarpin/RdRp and (D) Glycyroside/RdRp.
Molecular Dynamics Simulation
The MDS studies were performed using licorice glycoside E/RdRp and diisooctyl phthalate/RdRp to obtain the most stable conformation of the complexes. The complexes were simulated to the stable stage, which the structures of complexes reached a good score of root-mean-square deviation (RMSD). The RMSD score of licorice glycoside E/RdRp was 0.0442 Å, and the RMSD score of diisooctyl phthalate/RdRp was 0.0496 Å. It indicated that the 2 complexes changed a little after the refinement (Table 2).
RMSD, ROG, and Binding Energy of the Simulated Complexes.

The radius of gyration or ROG is a useful indicator for complex structure change. The simulation analysis shows that the ROG scores of licorice glycoside E/RdRp (34.74 Å) and diisooctyl phthalate/RdRp(34.55 Å) are satisfactory (Table 2). Based on BAPPL calculation, Table 2 shows that the binding energy of licorice glycoside E/RdRp is the same as that of diisooctyl phthalate/RdRp (−0.21 kCals/mol).
Protein Flexibility Analysis
To evaluate physical motions of the complexes licorice glycoside E/RdRp and diisooctyl phthalate/RdRp, further MDSs were performed by the iMODS. Deformability is a parameter describing the deformation ability of protein residues. The RdRp deformabilities of the complexes were shown in Figure 4A and D. Fluctuations shown in green referred to the location of chain hinges with high deformability, and these were plotted based on each residue of RdRp. The similar deformability was observed in the licorice glycoside E/RdRp complex and the diisooctyl phthalate/RdRp complex. B-factor values quantify the uncertainty of each atom. The B-factor value of licorice glycoside E/RdRp was much the same as that of diisooctyl phthalate/RdRp (Figure 4B and E). The covariance matrix indicates coupling or pairing of the protein residues; the higher the correlation, the better is the complex. The covariance matrix was shown in Figure 4C and F, indicating both licorice glycoside E/RdRp and diisooctyl phthalate/RdRp a good correlation in the majority (red: correlated, blue: anti-correlated, white: uncorrelated). The molecular dynamic results suggest that both complexes are stable.

Protein flexibility analysis. Deformabilities of (A) Licorice glycoside E/RdRp and (D) Diisooctyl phthalate/RdRp, B-factors of (B) Licorice glycoside E/RdRp and (E) Diisooctyl phthalate/RdRp, covariances of (C) Licorice glycoside E/RdRp and (F) Diisooctyl phthalate/RdRp.
Discussion
The goal of these studies was to explore new drugs from TCM frequently used herbs against SARS-CoV-2. To that end, the important Chinese herbs were screened for phytochemicals with antiviral properties though in silico analysis. Previous researches have proved that RdRp was a key target protein for inhibition of the viral replication 21 . In this study, RdRp was served as target to find phytochemicals for the new drug development.
The SARS-CoV-2 RdRp or nsp12 contains a C-terminal polymerase domain (residues SER367 to PHE920), which is important for viral replication and transcription4,22. Residue ASP618 in motif A is responsible for divalent-cation-binding. The catalytic residues in motif C are SER759, ASP760, and 761, which are required for RNA synthesis 4 . In this study, licorice glycoside E, diisooctyl phthalate, (-)-medicocarpin, and glycyroside were found to bind to motif A (LYS621, CYS622, ASP623, and ARG624) and motif C (SER759, ASP760, and 761) through hydrogen bonds, pi–pi interactions, hydrophobic bonds and salt bridges in combination. These phytochemicals may inhibit SARS-CoV-2 genome replication and transcription of genes by blocking the classic divalent-cation-binding or perturbing the catalytic site.
In this study, licorice glycoside E, diisooctyl phthalate, (-)-medicocarpin and glycyroside were also found to bind to the NTP entry channel (ARG553 and 555) through hydrogen bonds, pi–pi interactions, hydrophobic bonds, and salt bridges. These phytochemicals also interact with other residues TRP455, CYS813, SER814, ARG836, and ASP865 in the active site. They may inhibit SARS-CoV-2 replication and transcription by blocking the NTP entry, the template-primer entry, nascent strand exit3,4.
After the MDS, the overall energy of licorice glycoside E/RdRp and diisooctyl phthalate/RdRp complexes was stabilized. The RMSD was used to measure the change in the simulated structure as compared to its initial structure 23 . The similar RMSD scores of licorice glycoside E/RdRp and diisooctyl phthalate/RdRp complexes indicate a little change after the MDS of the complexes. The radius of gyration was calculated to understand the compactness and the whole dimensions of the residues in licorice glycoside E/RdRp and diisooctyl phthalate/RdRp complexes 24 . The analysis shows that both the ROG values of the complexes are satisfactory after the MDS. Next, protein flexibility analysis was carried out for licorice glycoside E/RdRp and diisooctyl phthalate/RdRp complexes25,26. The similar main-chain deformability, pairing of the protein residues was found. Taking together, all these results suggest the both complexes are stabilized structurally after the MDS.
In this molecular docking study, the 4 phytochemicals licorice glycoside E, diisooctyl phthalate, (-)-medicocarpin, and glycyroside show quite good binding affinity with RdRp. Interestingly, 3 of them (licorice glycoside E, (-)-Medicocarpin, and glycyroside) are from Glycyrrhiza uralensis, which is the traditional herbal medicine for treating infections of respiratory tract in China. In addition, they are used as antiviral herbal remedies27,28, against chronic hepatitis B, SARS related coronavirus, herpes simplex as ingredients of preparations in Sini Jia Renshen Decoction 29 , Shenling Baizhu San 30 , Ma Xing Shi Gan Tang 31 , and Feng Re Fan Fei 32 . It is promising that licorice glycoside E, diisooctyl phthalate, (-)-Medicocarpin, and glycyroside have antiviral properties making them ideal drugs for COVID-19 treatment.
Conclusion
In this study, we screened 1025 phytochemicals from 8 Chinese herbs to find natural compounds for inhibiting SARS-CoV-2 replication. A total 181 phytochemicals were qualified from ADME analysis; 4 of them licorice glycoside E, diisooctyl phthalate, (-)-medicocarpin, and glycyroside exhibited good binding with the viral RdRp. They are present in Chinese herbs Glycyrrhiza uralensis and Scutellaria baicalensis. However, the in silico analysis is only the first step for drug discovery. Next, we will perform the viral RdRp activity assay in the cell. Then, the oral activity of the compounds will be analyzed in a mouse adapted SARS-CoV-2 system. These phytochemicals are potential antiviral drugs, which are waiting for experimental and clinical validation.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221105693 - Supplemental material for Molecular Docking and Dynamics of Phytochemicals From Chinese Herbs With SARS-CoV-2 RdRp
Supplemental material, sj-docx-1-npx-10.1177_1934578X221105693 for Molecular Docking and Dynamics of Phytochemicals From Chinese Herbs With SARS-CoV-2 RdRp by Jingyao Lu, Wenpeng Lu, Houli Jiang, Changshui Yang and Xiaoyun Dong in Natural Product Communications
Footnotes
Acknowledgements
We thank Dr Kong Guimei and Dr Wang Chenghai for their academic assistance on this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Jiangsu Key laboratory of integrated traditional Chinese and Western Medicine for prevention and treatment of Senile Diseases (Code: 2015NB005)
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
