Abstract
Ulcerative colitis (UC) is a chronic inflammatory disease of intestinal tract, and Periplaneta americana has been found to be effective in the treatment for UC. The purpose of the study was to investigate the therapeutic effect of Periplaneta americana extract Ento-A on UC in rats induced by 2,4,6-trinitrobenzene sulfonic acid (TNBS) and to explore its mechanism. The Sprague-Dawley (SD) rats were randomly divided into normal control group; TNBS-treated group; sulfasalazine (SASP) treated group; Ento-A low- (50 mg/kg), medium- (100 mg/kg), and high-dose (200 mg/kg) groups, respectively. The UC model of rats was induced via TNBS. Disease activity index (DAI) was used to evaluate the severity of UC in rats. The macroscopic and microscopic damages of colon were accessed by colon mucosa damage index (CMDI) and histopathological score (HS), respectively. The levels of interleukin-4 (IL-4), interleukin-17 (IL-17), tumor necrosis factor-α (TNF-α), and interferon-γ (IFN-γ) in serum and the contents of myeloperoxidase (MPO), transforming growth factor-β1 (TGF-β1), and epidermal growth factor (EGF) in colonic mucosa were measured by enzyme-linked immunosorbent assay (ELISA). Compared with the normal control group, the TNBS-treated group showed increase in DAI, CMDI, HS, IL-17, TNF-α, IFN-γ as well as MPO and decrease in the levels of IL-4, EGF, and TGF-β1. However, Ento-A-administrated groups reversed the changes in the DAI, CMDI, HS, and the cytokines caused by TNBS. The study indicates that Periplaneta americana extract Ento-A can effectively alleviate the inflammation in TNBS-induced UC of rats, and the mechanism of that may be related to restoring the balance of T helper 1 (Th1)/Th2/Th17/T regulatory (Treg) cytokines.
Introduction
Ulcerative colitis (UC) is a chronic non-specific inflammation which begins with inflammation in rectum mucosa and will affect the entire colon. 1 The prevalence of UC has risen all over the world. In China, the morbidity of UC in 2016 was 11.6 per 100,000 people. 2 In addition, it was reported that more than 2 million people had UC in Europe. 3 Up to now, the pathogenesis of UC has not yet been clarified, and it is believed that factors like immunity response, gut microbiota, gene, and environment involve in the occurrence and development of UC. 4 Clinically, patients with UC usually display diarrhea, bloody feces, and abdominal pain. 5 At present, 5-amino salicylic acid, glucocorticoid, and immunosuppressive agents are the main treatments for UC, but they can also lead to some serious toxic side effects such as blood disorders, liver toxicity, and hormone resistance.6,7 Thus, developing novel therapeutic strategies for UC patients is significant.
Periplaneta americana, which belongs to Insecta class, Dictyoptera order, and Blattidae family, is one of the oldest and largest insect groups. 8 In addition, Periplaneta americana is a traditional Chinese medicine that has been used for more than 2000 years. According to the Shen Nong’s Herbal Classic, Periplaneta americana has plenty of biological activities like enhancing blood circulation, dissipating blood stasis, and facilitating digestion. 9 Now, Kangfuxin Liquid that is made from Periplaneta americana has been exploited into clinical drugs. Clinical experiences with more than 20 years have demonstrated that Periplaneta americana can attenuate inflammation, diminish stomach ulcer, eliminate swelling, and accelerate the recovery of wounds. 10 Besides, growing studies have revealed that Periplaneta americana plays an essential role in anti-inflammation,11,12 immune system, 13 and wound healing. 14 Based on the evidences mentioned above, it is indicated that Periplaneta americana may be an underling treatment for UC, but more researches are needed to test the assumption.
Therefore, our present research here was designed to examine the effects of Periplaneta americana extract Ento-A on chronic UC induced by 2,4,6-trinitrobenzene sulfonic acid (TNBS) in rats and explore its mechanism to provide theoretical proof for its clinical application on UC.
Materials and methods
Insect material
The extract Ento-A from Periplaneta americana was prepared according to the method described as follows: First, Periplaneta americana was dried and extracted with water at 95°C for 2 h three times. Then the solvent was filtered and evaporated under reduced pressure. In the end, the extract was precipitated with 95% ethanol for 24 h, filtered and evaporated under reduced pressure to obtain the Periplaneta americana extract Ento-A.
Chemicals and reagents
TNBS was purchased from Sigma-Aldrich (Shanghai, China). Sulfasalazine (SASP) was purchased from Ethypharm Pharmaceutical Co., Ltd. (Shanghai, China). Rat interleukin-4 (IL-4) enzyme-linked immunosorbent assay (ELISA) kit, rat interleukin-17 (IL-17) ELISA kit, rat tumor necrosis factor-α (TNF-α) ELISA kit, and interferon-γ (IFN-γ) ELISA kit were all purchased from Neobioscience Technology Co., Ltd. (Shenzhen, China). Occult blood test kit, myeloperoxidase (MPO) kit, epidermal growth factor (EGF) kit, and transforming growth factor-β1 (TGF-β1) kit were all purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Isoflurane was purchased from Reward Life Technology Co., Ltd. (Shenzhen, China). Chloral hydrate was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). The high-pressure liquid chromatography (HPLC)-grade methanol was purchased from Fisher Scientific (Waltham, MA, USA). The HPLC-grade trifluoroacetic acid (TFA) was obtained from Tedia Company (Fairfield, OH, USA). Pure water (18.2 MW) for the HPLC analysis was obtained from a water purification system purchased from Merck Millipore (Darmstadt, Germany). Xanthine, hypoxanthine, inosine, uracil, and uridine were purchased from National Institutes for Food and Drug Control (Beijing, China).
HPLC analysis of Ento-A
Agilent 1260 (Agilent Technology, Santa Clara, CA, USA) equipped with diode array detector was employed to analyze the chemical composition of Ento-A. A Sepax HP-C18 column (250 mm × 4.6 mm, 5 μm, 120 Å; Agilent Technology, Santa Clara, CA, USA) was used in the analysis under the column temperature at 25°C. The flow rate, the injection volume, and detection wavelength were 1 mL/min, 10 µL, and 254 nm, respectively. Solution A (methanol with 0.1% trifluoroacetic acid (v/v)) and solution B (water with 0.1 % trifluoroacetic acid (v/v)) were utilized as the mobile phase, and the elution gradient was as follow: t = 0 min, eluent A (2%), eluent B (98%); t = 5 min, eluent A (2%), eluent B (98%); t = 20 min, eluent A (20%), eluent B (80%); and t = 25 min, eluent A (25%), eluent B (75%). The samples of Ento-A and standard compounds were dissolved in 50% methanol water solution (w/v) and filtered via 0.22-μm membrane filter.
Animals
Male Sprague-Dawley (SD) rats (n = 70, 200 ± 20 g, Certificate No. SCXK(Xiang)-2011-003) were purchased from Hunan SJA Laboratory Animal Co., Ltd. (Hunan, China). All rats were housed in a room under 25 ± 2°C and 50% humidity. The rats were acclimated to laboratory environment for 1 week. The study was approved by the Animal Ethics Committee of Dali University (ethics permission no. 2018-0627, year: 2018).
Induction of colitis and administration
After 1 week of adaptive feeding, 10 rats were randomly selected as the normal control group based on the weight of rats, and another 60 rats were used to induce the UC model according to the method described by GP Morris et al. 15 Briefly, rats were forced to fast but provided with water for 24 h before they were anesthetized with chloral hydrate (0.3 mL/100g). The rats were placed in prostrate position and 2.0-mm diameter catheter was inserted into the anus at 8 cm. Subsequently, the catheter was connected with 1.0 mL syringe, and 0.6 mL TNBS ethanol solution (15 mg TNBS was dissolved into 30% ethanol) was pushed in the anus through the catheter. Then, the catheter equipped with 1.0 mL syringe was removed and the rats were kept in a vertical position for 10 s to ensure that the solution passed into the anus totally. In addition, the rats in the normal control group received the same equivalent volume of normal saline.
On the first day after modeling, 10 model rats whose disease activity index (DAI) were ⩽6 or ⩾10 were removed in this experiment and the remaining 50 model rats were randomly divided into five groups (10 rats of each group): TNBS-treated group; SASP-treated group; and Ento-A low-, medium-, and high-dose groups. On the eighth day after modeling, the normal group and the TNBS-treated group received normal saline at a dose of 2.5 mL/kg/day; the SASP-treated group received SASP at a dose of 300 mg/kg/day; and the Ento-A low-, medium-, and high-dose groups received 50, 100, and 200 mg/kg/day of the Ento-A, respectively. All agents (normal saline, SASP, and Ento-A) were administered via enema, and the period of administration lasted for 14 days.
Assessment of the DAI
After administration, the body weight, stool consistency, and blood in feces of rats were recorded to evaluate the DAI according to the method described by N Hamamoto et al. 16 in Table 1.
The standard for DAI evaluation.

DAI: disease activity index.
DAI = combined scores of weight loss, stool consistency, and bleeding.
Assessment of the viscera index
After the last administration, the rats were anesthetized with chloral hydrate (0.3 mL/100 g) and dissected. The thymuses, spleens, livers, and colons of rats were cleaned and weighed. Viscera index was calculated with the following equation

Assessment of the CMDI
The colons of rats were cut along the mesentery and cleaned. The length and width of the colons were measured. Then the macroscopic damage of colons was examined and scored by combining the criteria of GM EkstrÖm 17 and HH Luk et al. 18 The criteria were described in Table 2.
The standard for CMDI evaluation.
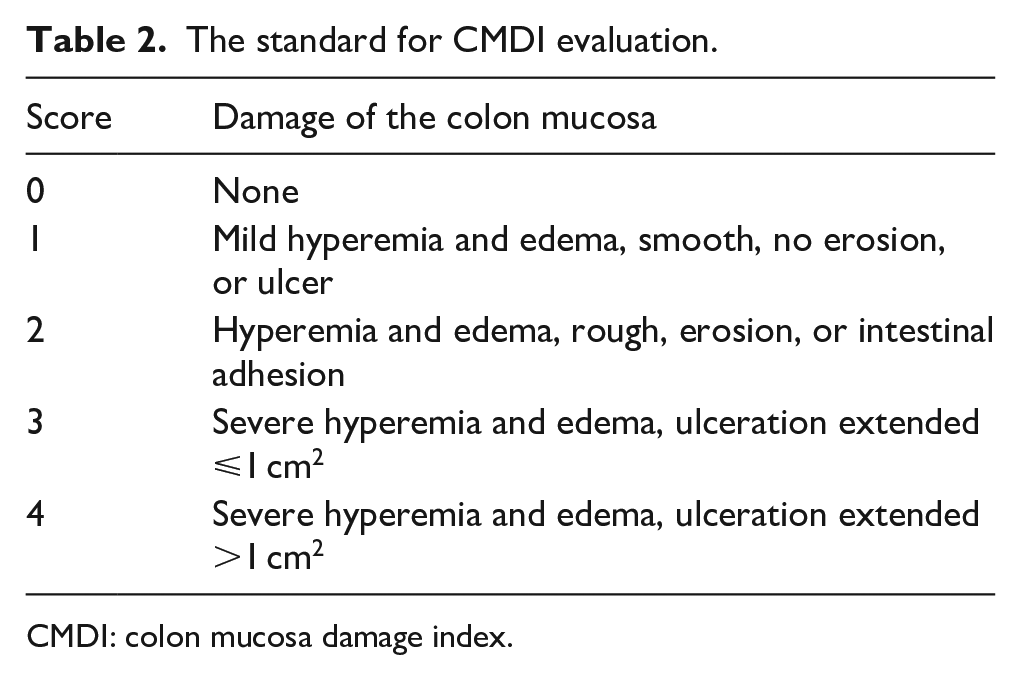
CMDI: colon mucosa damage index.
Assessment of the histological evaluation
The colon tissues of rats were fixed in 10% formalin, dehydrated with ethanol, embedded in paraffin, cut in 3–5 μm, and dyed with hematoxylin and eosin. Then the microscopic changes of colons were observed under microscope and scored by combining the method of N Hamamoto et al. 16 and HH Luk et al. 18 The method was described in Table 3.
The standard for HS evaluation.

HS: histological score.
The HS = combined scores of epithelial morphology and inflammation.
Assessment of the levels of IL-4, IL-17, TNF-α, and IFN-γ in serum
The blood samples of rat were taken from abdominal aorta. Then these blood samples were centrifuged at 4°C and the supernatants were collected to assess the levels of IL-4, IL-17, TNF-α, and IFN-γ via ELISA kits.
Assessment of the contents of MPO, TGF-β1, and EGF in colon mucosa
The colons were homogenized with normal saline and centrifuged at 3500 r/min for 10 min. Then, the supernatants were collected to detect the expressions of MPO, TGF-β1, and EGF in the colon by using ELISA kits.
Statistical analysis
All the data were expressed as mean ± standard deviation and were carried out by SPSS 17.0 (IBM, New York, NY, USA). If the data fit the normal distribution, they were analyzed by one-way analysis of variance (ANOVA) with least significant difference (LSD) t-test as the post hoc test or else by rank test. P value <0.05 was considered statistically significant.
Results
HPLC analysis of Ento-A
After the HPLC analysis of the chemical composition of Ento-A, five compounds were identified in Ento-A. They were uracil, hypoxanthine, uridine, xanthine, and inosine (Figure 1).

HPLC analysis of Ento-A, xanthine, hypoxanthine, inosine, uracil, and uridine. Peaks identified are as follows: uracil (a), hypoxanthine (b), uridine (c), xanthine (d), and inosine (e).
Effects of Ento-A on DAI
The rats in the normal control group exhibited smooth hair, normal food intake, and high spirit without diarrhea and bloody stool. As shown in Figure 2, on the first day after modeling, all the rats in the TNBS-treated group displayed anorexia, low spirit, weight loss, and bloody stool. In addition, there was significant difference in DAI between the TNBS-treated group and the normal control group (P < 0.01), which indicated the success in modeling. On the fourth and seventh day after modeling, the rats in the TNBS-treated group still had diarrhea and weight loss. However, from the 14th day after modeling, the feces of all rats in the TNBS-treated group became harder and the weight of all rats recovered. On the 21th day after modeling, the DAI of the SASP-treated group and all the Ento-A groups were lower than that of the TNBS-treated group (P < 0.01).

Effects of Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on DAI
Effects of Ento-A on viscera index
Thymus, spleen, and liver are the important organs which involve in the immune and drug metabolism. Therefore, their weight and general appearance, to some degree, indicate the severity of inflammation, immune responses, and drugs effects. As shown in Figure 3, no significant difference was found in thymus, spleen, and liver indices between any two groups. However, due to the local ulceration and edema, the colon index of the TNBS-treated group was higher than that of the normal control group (P < 0.01). Compared with the TNBS-treated group, there was a decreasing tendency in the colon index of the drug-administered groups though no significant difference was observed.

Effects of Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on viscera indices
Effects of Ento-A on the length, the ratio of the length to width of colon and CMDI
As shown in Figure 4, in the TNBS-treated group, both the colon length and the ratio of the length to width of the colon significantly decreased (P < 0.01 vs normal control group) while colon mucosa damage index (CMDI) increased obviously (P < 0.01 vs normal control group). As shown in Figure 5, with naked eyes, the colon of the normal control group showed distinct plica along the lumen surface without visible swelling and ulceration whereas obvious ulceration and swelling were found in the distant colon of the TNBS-treated group. In addition, compared with the TNBS-treated group, the SASP-treated group and Ento-A-administrated groups decreased the CMDI and displayed slight swelling.

Effects of Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on rats colon length, ratio of colon length to width and CMDI

Macroscopic appearance of colon in the normal control group, TNBS-treated group, SASP-treated group, Ento-A low-dose group (50 mg/kg), Ento-A middle-dose group (100 mg/kg), and Ento-A high-dose group (200 mg/kg).
Effects of Ento-A on serum levels of IL-4, IL-17, TNF-α, and IFN-γ
As shown in Figure 6, the TNBS-treated group showed decrease in the level of IL-4 (P < 0.01 vs normal control group) and increase in the levels of IL-17, TNF-α, and IFN-γ (P < 0.05 or P < 0.01 vs normal control group). However, the SASP-treated group and all the Ento-A groups showed obvious increase in the level of IL-4 (P < 0.05 or P < 0.01 vs TNBS-treated group) and decrease in the levels of TNF-α and IFN-γ (both P < 0.01 vs TNBS-treated group). Besides, down-regulation of IL-17 was found in all the drug-given groups although no obvious difference was found when compared with the TNBS-administrated group.

Effects of Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on levels of IL-4, IL-17, TNF-α, and IFN-γ in serum
Effects of Ento-A on the levels of MPO, TGF-β1, and EGF
As shown in Figure 7, compared with the normal control group, significant increase of MPO (P < 0.01) and obvious decrease of the levels of TGF-β1 and EGF (both P < 0.01) were found in the TNBS-treated group. However, the SASP-treated group, the Ento-A medium- and high-dose groups showed decrease in the level of MPO when compared with the TNBS-treated group. In addition, the SASP-treated group and all the Ento-A groups significantly increased the expression of TGF-β1 (both P < 0.01 vs TNBS-treated group). Besides, only the SASP-treated group and the Ento-A low-dose group obviously increased the content of EGF (P < 0.01 or P < 0.05 vs TNBS-treated group).

Effects of Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on levels of MPO, TGF-β1, and EGF in colon mucosa
Effects of Ento-A on colon HS
As shown in Figure 8, under microscope, the colon in the normal control group displayed intact mucosal epithelium, no inflammatory infiltration, and no loss of goblet cells and crypts. However, the colon in the TNBS-treated group showed severe changes such as great loss of goblet cells, disappearance of crypts, and a large amount of inflammatory cells infiltration in mucosa layer. In addition, as shown in Figure 9, HS of the TNBS-treated group was higher than that of the normal control group (P < 0.01). Interestingly, significant decrease of HS was found in the SASP-treated group and all the Ento-A-given groups when compared with the TNBS-treated group (all P < 0.01).
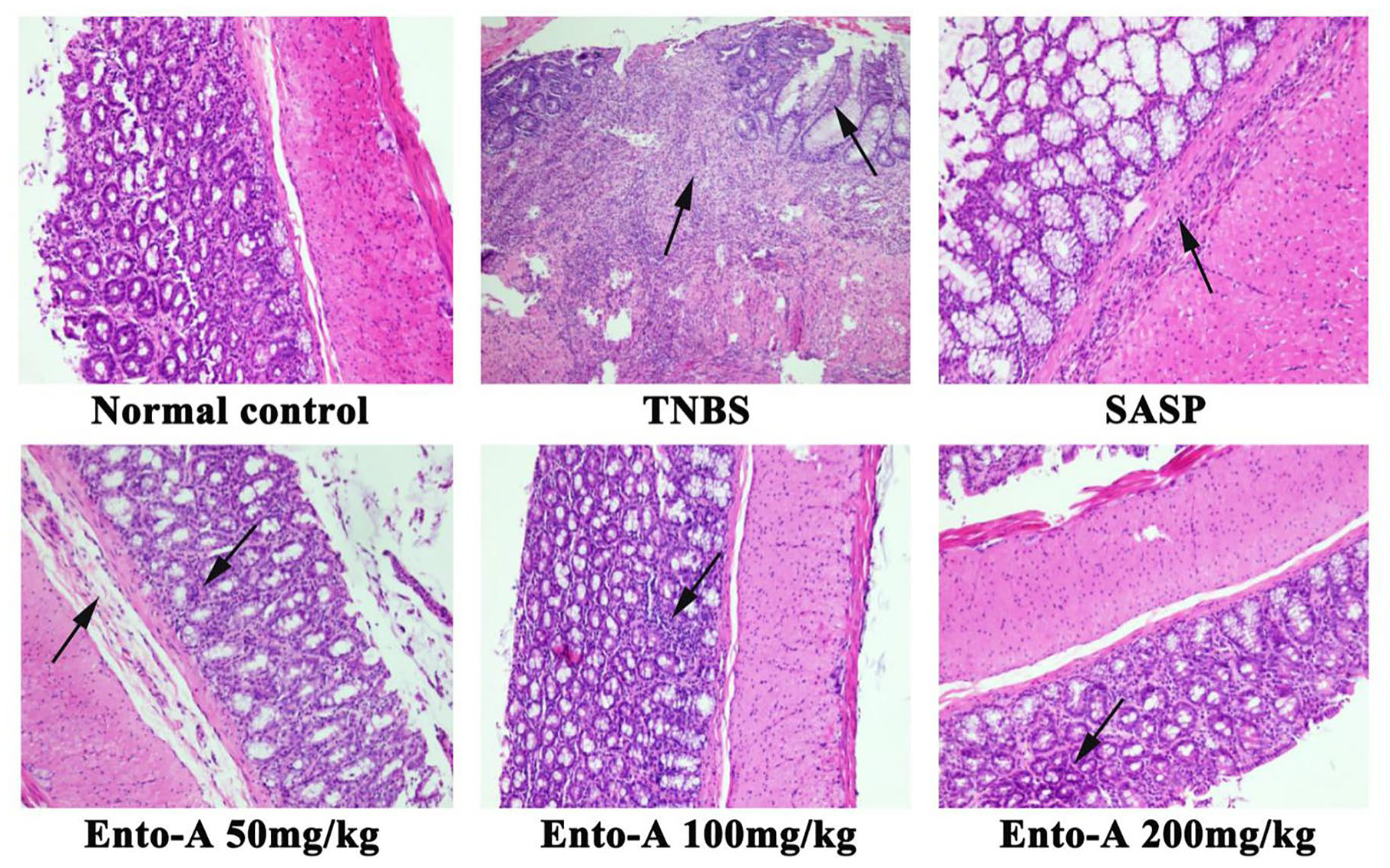
Results from the H&E staining (100×) in the normal control group, TNBS-treated group, SASP-treated group, Ento-A low-dose group (50 mg/kg), Ento-A middle-dose group (100 mg/kg), and Ento-A high-dose group (200 mg/kg).
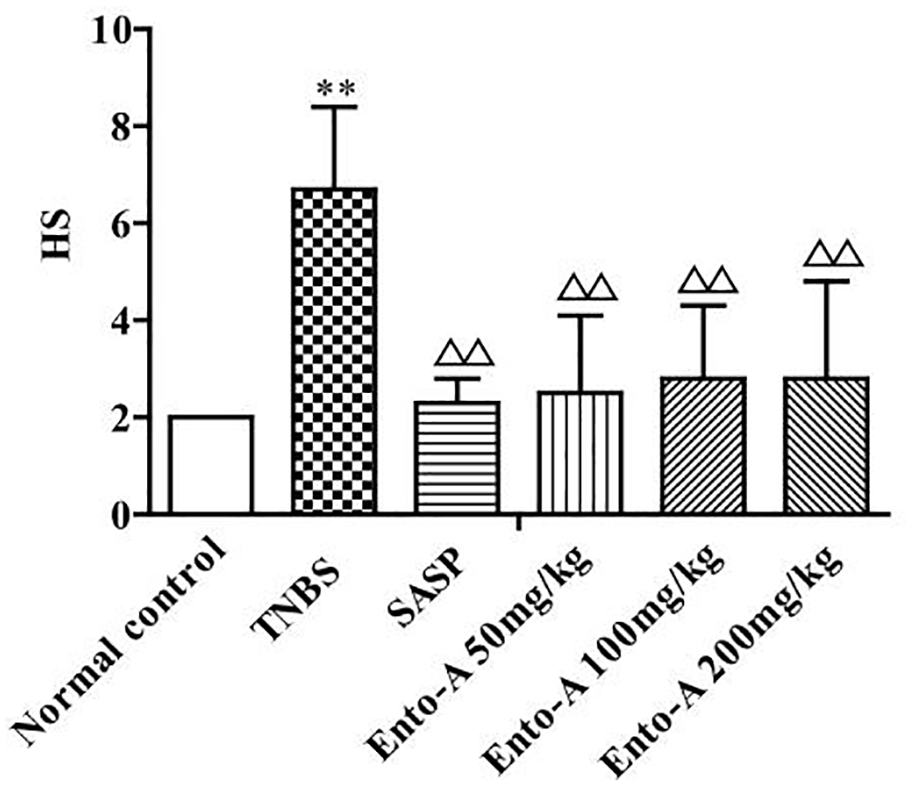
Effects on Ento-A (low dose: 50 mg/kg, middle dose: 100 mg/kg, high dose: 200 mg/kg) on HS of colon
Discussion
UC now becomes a global problem because of the increasing morbidity. 19 One of the important characters of UC is the imbalance of inflammatory cytokines, which will cause serious inflammation. 20 In this study, the Periplaneta americana extract Ento-A exhibited a beneficial effect in attenuating the inflammation of UC rat induced via TNBS.
TNBS was used to establish the UC rat model in the study. TNBS is a hapten which can cause delayed hypersensitivity reaction when it binds to animal colon autologous cells. The effect of TNBS contributes to long-lasting inflammation and ulceration in rat colon, which will led to neutrophil infiltration and damage to the colon epithelial cells. Subsequently, it will elicit the elevated expression of MPO, the decrease of EGF, and the loss of goblet cells as well as crypts. Importantly, the histological characteristics and distribution of the inflammation and ulceration are similar to the clinical manifestations of UC.15,21,22 In this study, serious ulceration and edema on the colon were found in the TNBS-administrated group while the situation was improved significantly by the treatment of Ento-A. Histologically, the TNBS-given group caused a large loss of goblet cells and crypts together with the severe inflammatory cell infiltration, whereas only mild inflammatory cell infiltration and swell were observed in the Ento-A-treated groups. The evidences suggested that Ento-A had an certain effect in preventing inflammation in TNBS-induced colitis.
To ascertain the role of Ento-A on the inflammation in TNBS-induced colitis, this study next explored the effect of Ento-A on the balance of Th1/Th2/Th17/Treg cytokines. It was reported that the balance of Th1/Th2/Th17/Treg cells involved in the etiology of ulcerative colon. 23 The immune system of host plays a vital role in protecting against pathogenic microorganisms. 24 As one of the main subsets of T cells, CD4+ T cells exert an essential function in regulating the immune responses to pathogens. 25 Activated through T-cell receptor and cytokine-mediated signaling, naive CD4+ T cells can differentiate into Th1, Th2, Th17, and Treg cells. 26 Characterized by unique cytokines, the CD4+ T cells subtypes have specific function.27–29 Th1 cells activate classical macrophages and regulate the immune responses to intracellular pathogenic microorganisms by expressing IL-2, IL-12, TNF-α, and IFN-γ. Besides, Th2 cells are responsible for mediating the immune responses against extracellular parasites and allergic inflammation via the secretions of IL-4, IL-5, IL-10, IL-13, and IL-25. In addition, Th17 cells produce IL-17 and IL-22, which led to the recruitment of neutrophils and control the immune responses to extracellular pathogenic microorganisms. However, Treg cells have an essential role in suppressing immune responses from effector T cells and maintaining immune homeostasis by expressing the anti-inflammatory cytokines like IL-10 and TGF-β. Thus, the balance of these CD4+ T cells subtypes is significant for the UC. In this present research, compared with the normal control group, the TNBS-treated group not only markedly increased the levels of IL-17, TNF-α, and IFN-γ but also remarkably decreased the expressions of IL-4 and TGF-β, which suggested that the balance of Th1/Th2/Th17/Treg cytokines was broken. Interestingly, the Ento-A-administrated groups showed a total opposite trend in the contents of the IL-17, TNF-α, IFN-γ, IL-4, and TGF-β when compared with the TNBS-given group. The results demonstrated that the mechanism of Ento-A in protecting against the inflammation of TNBS-induced colitis may be related to maintaining the balance of Th1/Th2/Th17/Treg cytokines.
Periplaneta americana inhabits in the warm and humid tropical and subtropical regions and is the important source of indoor allergen.30,31 It was reported that the allergic proteins named Per a 1-12 from Periplaneta americana contributed to the allergic diseases like asthma. 32 In this study, we prepared the Ento-A by water extraction and ethanol precipitation, which removed the macromolecular substances. Besides, we also observed the effect of Ento-A on liver, thymus, and spleen. Compared with the normal control group, the Ento-A treated group had no significant changes in the viscera indexes of liver, thymus, and spleen. The evidences indicated that Ento-A had lower toxicity on other organs.
In this research, the sample size was determined by collecting literatures from other researchers who conducted similar experiments. 33 Although the sample size of the method was enough to detect the effects of the drugs, it took more time, resources, and money. Thus, it is better estimate the sample size of a study by combining different methods to minimize the sample size and ensure the effects of drugs. In addition, although Ento-A showed beneficial effects on TNBS-induced colitis in rats, no dose dependence was found in the results of Ento-A with different doses. This may be caused by the complex chemical composition of traditional Chinese medicine acting on a variety of targets. Characterized with multiple compounds and targets, traditional Chinese medicines treat plenty of diseases in a holistic way, making it difficult to elucidate the effects of traditional Chinese medicines on various diseases. 34 Therefore, more researches are needed to further investigate the active compounds of Ento-A and their specific molecular mechanisms on UC.
Conclusion
In summary, it is demonstrated that Ento-A can antagonize the inflammation on UC rat caused by TNBS through balancing the Th1/Th2/Th17/Treg cytokines, which suggests that Ento-A can become a possible medicine for UC induced by TNBS in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Animal Ethics Committee of Dali University (ethics permission number: 2018-0627, year: 2018). The present study followed national and institutional guidelines for humane animal treatment and complied with relevant legislation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by several funds as the following: The National Natural Science Fundation of China (No. 81660605, 81860742, 81860765, 81660186); Yunnan Fundamental Research Projects (No. 2017FH001-108, 2018FB143, 2019FH001-003 and 2020FB022); Innovation Team of Dali University for Advanced Pharmaceutics of Entomological Bio-pharmacy R&D (No. ZKLX2019101); Innovative Team of Dali University for Digital Development of Medicinal Insects and Arachnids Resources (No. ZKLX2020303); and Special Fund for Traditional Chinese Medicine Pieces of Yunnan Industry and Information Commission (No. 2019-YG-067); Yunnan Provincial Department of Science and Technology - Kunming Medical University Joint Special Project on Applied Basic Research (No. 2017FE468 (-009)).
