Abstract
Gastric cancer (GC) is one of the most common and deadly malignant tumors worldwide. While the application of herbal drugs for GC treatment is increasing, the multicompound–multitarget pharmacological mechanisms involved are yet to be elucidated. By adopting a network pharmacology strategy, we investigated the properties of the anticancer herbal drug FDY003 against GC. We found that FDY003 reduced the viability of human GC cells and enhanced their chemosensitivity. We also identified 8 active phytochemical compounds in FDY003 that target 70 GC-associated genes and proteins. Gene ontology (GO) enrichment analysis suggested that the targets of FDY003 are involved in various cellular processes, such as cellular proliferation, survival, and death. We further identified various major FDY003 target GC-associated pathways, including PIK3-Akt, MAPK, Ras, HIF-1, ErbB, and p53 pathways. Taken together, the overall analysis presents insight at the systems level into the pharmacological activity of FDY003 against GC.
Introduction
Gastric cancer (GC) is the fourth most frequent cause of cancer mortality, at a global rate of 0.77 million deaths per year 1 . The current standard treatment options for GC are chemotherapy, targeted therapy, and immunotherapy 2 ; however, the development of resistance and occurrence of side effects limit their therapeutic success3,4. Herbal drugs are also being widely applied as GC treatment as they exert potent anticancer activity, improve the therapeutic efficacy of standard cancer therapeutics, and ameliorate adverse events5-8. Additionally, they can prolong survival, increase cure rate, improve prognosis, alleviate symptoms, and enhance recovery in patients with GC5-8.
FDY003 is an anticancer herbal drug composed of
Network pharmacology has become one of the most efficient methodologies used in herbal drug-associated studies owing to its ability to effectively elucidate the complex multicompound–multitarget mechanisms of herbal drugs using related comprehensive pharmacological data12-14. The polypharmacological effects of herbal drugs are coordinated through complex interactions between the chemical compounds and their diverse therapeutic targets 12-14. Thus, network pharmacology investigates the active pharmacological compounds in herbal drugs and their disease-related therapeutic targets12-14. This is done by analyzing the topological and functional features of diverse types of herbal drug-associated networks that consist of the interactions between their associated pharmacological components and targets12-14. By adopting a network pharmacology strategy, we investigated the properties of FDY003 as a treatment for GC.
Materials and Methods
Cell culture
AGS human GC cells were purchased from the Korean Cell Line Bank (Seoul, Korea) and cultured in Dulbecco's modified Eagle's medium (WELGENE Inc.) supplemented with 10% fetal bovine serum (WELGENE Inc.), 100 μg/mL streptomycin, and 100 U/mL penicillin (Life Technologies Corp.) at 37°C in a humidified atmosphere containing 5% CO2.
Preparation of FDY003
Dried Cm (6.25 g), AcT (6.25 g), and LjT (4.16 g), which were purchased from Hanpure Pharmaceuticals (Pocheon, Korea), were ground, mixed, and placed in 70% ethanol (500 mL). The herbal extract was obtained by refluxing the mixture at 80°C for 3 h. Next, the extract was filtered, purified with 80% and 90% ethanol consecutively, lyophilized at 80°C, and stored at − 20 °C. The lyophilized samples were dissolved in distilled water prior to performing the experiments.
Cell viability analysis
Cell viability was measured using a water-soluble tetrazolium salt (WST-1) assay. In total, 1.0 × 104 cells were seeded in a 96-well plate and incubated at 37°C for 72 h with FDY003 and/or 5-fluorouracil (5-FU; Sigma-Aldrich). Afterward, the cells were incubated with WST-1 solution (Daeil Lab Service Co., Ltd,) for 2 h at 37°C and 5% CO2. Absorbances at 450 nm were measured using an xMark microplate absorbance spectrophotometer (Bio-Rad).
Bioactive compounds screening
Detailed data on the chemical compounds present in FDY003 were retrieved from the Traditional Chinese Medicine Systems Pharmacology
15
, Anticancer Herbs Database of Systems Pharmacology
16
, and Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine
17
. Then, the pharmacologically bioactive compounds were identified based on their Caco-2 permeabilities, druglikenesses, and oral bioavailability parameters, which are widely used in network pharmacological analysis for compound screening14,15,18. Caco-2 permeability determines whether a compound has pharmacologically suitable intestinal permeability15,19-21, in which a value ≥ −0.4 is generally used as an indicator for strong
Target investigation
The simplified molecular-input line-entry system (SMILES) notations of the active compounds of FDY003 were collected from the PubChem database 26 . Then, they were imported to SwissTargetPrediction 27 , Search Tool for Interactions of Chemicals 5 28 , Similarity Ensemble Approach 29 , and PharmMapper 30 to identify the human targets of each compound. Information on GC-associated genes and proteins was investigated using the search keyword “gastric cancer” on the following databases: DrugBank 22 , Therapeutic Target Database 23 , DisGeNET 31 , Comparative Toxicogenomics Database 32 , Online Mendelian Inheritance in Man 33 , Human Genome Epidemiology Navigator 34 , GeneCards 35 , and Pharmacogenomics Knowledgebase 36 .
Herbal medicine-related networks
A network is composed of nodes (eg, herbal medicines, compounds, targets, and pathways) and edges (or links) describing their pharmacological interactions and associations 37 , in which the No. of edges of a given node is called the degree 37 . The herbal medicine-compound-target (H-C-T) network depicts the association between the FDY003-constituting herbal medicines, their active phytochemical compounds, and the GC-associated genes and proteins targeted by the compounds. The H-C-T-pathway (H-C-T-P) network depicts the association between the targets of the H-C-T network and their related GC-associated pathways. The protein–protein interaction (PPI) network depicts the interactions between GC-associated targets using the STRING database 38 . The networks were built, drawn, and analyzed using Cytoscape 39 .
Survival analysis
The prognostic associations between the expression of FDY003 targets and the survival of patients with GC were computed using a Kaplan–Meier Plotter 40 .
Investigation of functional enrichment for the FDY003 targets
Gene ontology (GO) and pathway enrichment for the FDY003 targets were investigated using g:Profiler 41 .
Analysis of molecular docking interaction
Information on the molecular structures of the active compounds of FDY003 and their targets was obtained from RCSB Protein Data Bank 42 and PubChem 26 . Then, the binding affinities between the compounds and their targets were evaluated based on the molecular docking scores obtained by importing their structural information into Autodock Vina 43 . Docking scores of ≤ −5.0 indicated strong binding affinities between compound–target pairs44,45.
Results
Pharmacological effects of FDY003 for gastric cancer
To examine the pharmacological activity of FDY003 in GS, the viabilities of AGS cells were measured after treatment with FDY003 and/or 5-FU, a chemotherapeutic agent used in clinics for GC treatment 46 . FDY003 reduced the viability and increased the 5-FU sensitivity of AGS cells (Supplementary Figure S1A and B), suggesting that FDY003 exhibits anti-GC effects.
The active phytochemical compounds and targets of FDY003
The active phytochemical compounds of FDY003 were determined based on their pharmacokinetic criteria (Supplementary Table S1)11,14,15,18. Moreover, some of them were determined to be active based on their reported potent pharmacological effects, although they did not meet the aforementioned criteria. In total, 18 FDY003 compounds were considered active (Supplementary Table S2). The targets of the active compounds were investigated using their structural information and, in total, 270 targets for FDY003 were identified, of which 70 were GC-associated targets (Supplementary Table S3).
Network-perspective pharmacological properties of FDY003 against gastric cancer
To investigate the pharmacological properties of FDY003 at the network level, we generated an H-C-T network using comprehensive FDY003-related data (Figure 1). The generated network was composed of 81 nodes (3 herbal medicines, 8 active phytochemical compounds, and 70 GC-associated targets) and 121 links (Figure 1 and Supplementary Table S3). The compounds quercetin, luteolin, and kaempferol had relatively many targets compared to other compounds (Figure 2 and Supplementary Table S3), demonstrating their potential importance in the anti-GC activities of FDY003. In addition, 78.6% of the FDY003 targets (55 of 70 targets) interacted with two or more compounds (Figure 1), demonstrating the multicompound–multitarget characteristics of the herbal drug.
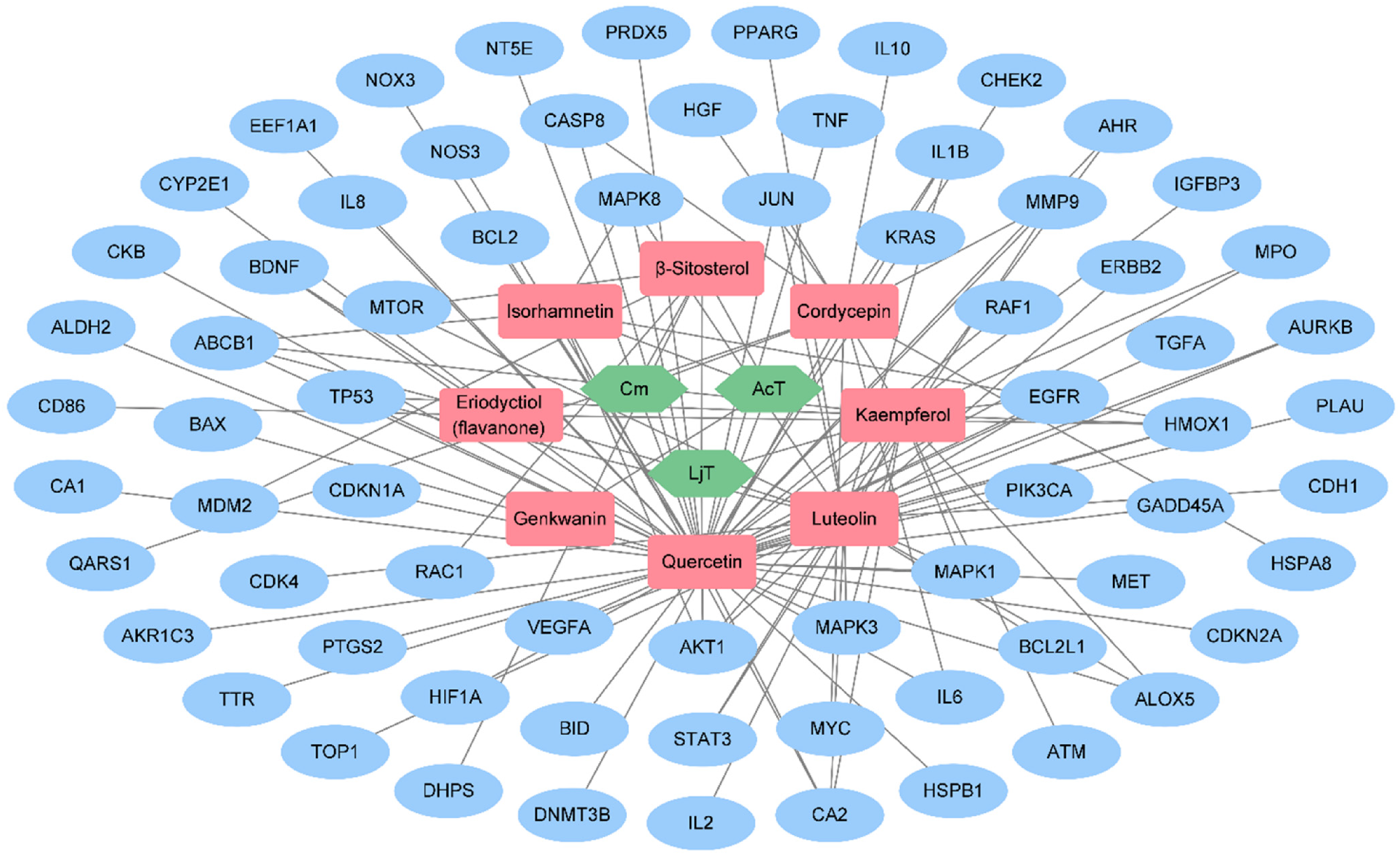
The herbal medicine-active phytochemical compound-target (H-C-T) network of FDY003. Green hexagons, herbal medicines; red round rectangles, active phytochemical compound; blue ellipses, gastric cancer (GC)-associated targets.

The protein–protein interaction (PPI) network for gastric cancer (GC)-associated targets of FDY003. Pink ellipses, hub GC-associated targets.
As the pharmacological effects of drugs are conferred through interactions with disease-related target genes and proteins47-49, a PPI network (60 nodes and 272 links) consisting of FDY003 targets was built, in which the large-degree hub nodes with topological significance and potential as effective therapeutic targets were investigated50,51. Following previous studies, the hubs were considered as nodes with degrees equal to or greater than twice the average degrees of all the nodes in a network52,53. AKT1, MAPK1, MAPK3, MAPK9, PIK3CA, STAT3, and TP53 were identified as hubs in the PPI network (Figure 2), indicating that they are key targets in the anti-GC mechanism of FDY003. The survival analysis further suggested that the targets function as determinants for survival outcome in patients with GC (Figure 3), demonstrating their clinical significance.

Survival analysis of gastric cancer (GC)-associated targets of FDY003. Kaplan–Meier curves for the overall survival of patients with GC with respect to the expression status of the indicated FDY003 targets.
Functional analysis of the FDY003 mechanisms
To gain insight into the pharmacological mechanisms involved in the anti-GC activities of FDY003, GO and pathway enrichment analyses were performed on the FDY003 targets. The targets were found to potentially be involved in regulating various cellular processes, such as cellular proliferation, survival, and death (Supplementary Figure S2). Furthermore, the FDY003 targets were found to be important constituents of the following diverse GC-associated signaling pathways: “Chemokine signaling pathway”, “Epithelial cell signaling in Helicobacter pylori infection”, “Epstein-Barr virus infection”, “Erythroblastic leukemia viral oncogene homolog (ErbB) signaling pathway”, “Estrogen signaling pathway”, “Gastric cancer”, “Hypoxia inducible factor-1 (HIF-1) signaling pathway”, “Janus kinase (JAK)-signal transducer and activator of transcription (STAT) signaling pathway”, “Mitogen-activated protein kinase (MAPK) signaling pathway”, “Mammalian target of rapamycin (mTOR) signaling pathway”, “Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kappa B) signaling pathway”, “p53 signaling pathway”, “Pathways in cancer”, “Programmed death ligand 1 (PD-L1) expression and programmed death-ligand 1 (PD-1) checkpoint pathway in cancer”, “Phosphoinositide 3-kinase (PI3K)-Akt signaling pathway”, “Ras signaling pathway”, “Tumor necrosis factor (TNF) signaling pathway”, and “Vascular endothelial growth factor (VEGF) signaling pathway” (Figure 4 and Supplementary Figure S2). These findings demonstrated the molecular- and signaling-perspective mechanistic properties of FDY003 in GC treatment.

The herbal medicine-active phytochemical compound-target-pathway (H-C-T-P) network of FDY003. Green hexagons, herbal medicines; red round rectangles, active phytochemical compound; blue ellipses, gastric cancer (GC)-associated targets; orange nodes, GC-associated signaling pathways.
Molecular docking analysis for the FDY003 components
To verify the binding potential of the FDY003 components, the binding affinities between the active compounds of FDY003 and their targets were measured based on molecular docking analysis. The active compounds and their direct hub targets of the herbal drug [ie, AKT1-isorhamnetin (score = −6.4), AKT1-kaempferol (score = −6.6), AKT1-luteolin (score = −7.1), AKT1-quercetin (score = −6.9), MAPK1-kaempferol (score = −7.4), MAPK1-luteolin (score = −8.1), MAPK1-quercetin (score = −7.8), MAPK3-kaempferol (score = −8.4), MAPK3-luteolin (score = −8.8), MAPK3-quercetin (score = −9.1), MAPK8-isorhamnetin (score = −8.6), MAPK8-luteolin (score = −8.2), MAPK8-quercetin (score = −8.0), PIK3CA-quercetin (score = −8.5), STAT3-kaempferol (score = −7.7), STAT3-luteolin (score = −7.5), TP53-kaempferol (score = −8.7), TP53-luteolin (score = −9.0), and TP53-quercetin (score = −9.2)] had docking scores of ≤ −5.0 (Figure 5A-S), suggesting their binding activities.

Molecular docking analysis for the active phytochemical compounds of FDY003 and the gastric cancer (GC)-associated targets. (A) AKT1-isorhamnetin (score = −6.4). (B) AKT1-kaempferol (score = −6.6). (C) AKT1-luteolin (score = −7.1). (D) AKT1-quercetin (score = −6.9). (E) MAPK1-kaempferol (score = −7.4). (F) MAPK1-luteolin (score = −8.1). (G) MAPK1-quercetin (score = −7.8). (H) MAPK3-kaempferol (score = −8.4). (I) MAPK3-luteolin (score = −8.8). (J) MAPK3-quercetin (score = −9.1). (K) MAPK8-isorhamnetin (score = −8.6). (L) MAPK8-luteolin (score = −8.2). (M) MAPK8-quercetin (score = −8.0). (N) PIK3CA-quercetin (score = −8.5). (O) STAT3-kaempferol (score = −7.7). (P) STAT3-luteolin (score = −7.5). (Q) TP53-kaempferol (score = −8.7). (R) TP53-luteolin (score = −9.0). (S) TP53-quercetin (score = −9.2).
Discussion
Although the application of herbal drugs for GC treatment has gained increasing attention5-8, their complex multicomponent–multitarget mechanistic properties have yet to be investigated. In this study, we dissected the anti-GC properties of the herbal drug FDY003 through a systems perspective9-11. We found that FDY003 suppressed the viability and enhanced the chemosensitivity of human GC cells. Based on the network pharmacological investigation, we identified 8 active phytochemical compounds in FDY003 and 70 GC-associated therapeutic targets that may confer the pharmacological effects of the herbal drug. The GO enrichment analysis suggested that the FDY003 targets are involved in various cellular processes, such as cellular proliferation, survival, and death. We further identified various GC-associated pathways that are major pharmacological targets of FDY003, such as the PIK3-Akt, MAPK, Ras, HIF-1, ErbB, and p53 pathways. These results demonstrate the comprehensive polypharmacological anti-GC mechanisms of the herbal drugs.
The molecular docking analysis revealed that the active compounds of FDY003 and their direct hub targets show molecular docking scores of ≤ −5.0 (Figure 5), suggesting that the targets have strong binding affinities with their interacting active compounds and that these interactions confer the treatment effects of FDY003 against LC. The major targets of FDY003 were found to be genes and proteins associated with GC mechanisms and relevant therapeutic targets for GC treatment. The protein kinases AKT1 (encoded by
The target pathways of FDY003 are also involved in the key signaling mechanisms of GC pathology. The chemokine signaling pathway plays a role in the regulation of the immune system and tumor microenvironment, and it is further involved in GC tumorigenesis by modulating tumor transformation, growth, survival, metastasis, invasion, and angiogenesis
82
. The MAPK, HIF-1, ErbB, PI3K-Akt, mTOR, NF-kappa B, and Ras pathways are key modulators of GC carcinogenesis and progression through the regulation of malignant cellular behaviors such as proliferation, stem-like characteristics, migration, invasion, survival, angiogenesis, metastasis, apoptosis, chemoresistance, and EMT of GC cells46,83-87. These oncogenic pathways have gained much attention as potential drug targets for targeted GC therapies46,83-87. The estrogen signaling pathway controls proliferation, apoptosis, and invasion of GC cells, and it may act as a prognostic factor for the progression and prognosis of GC88-91. The dysregulation of JAK-STAT, TNF, and NF-kappa B signaling also leads to uncontrolled proliferation, survival, EMT, and protumorigenic inflammation in GC cells92,93. Moreover, functional deficiency of the p53 pathway may induce stemness and EMT in gastric epithelial cells, which ultimately leads to the formation, development, and metastasis of malignant GC tumors, and the genetic alterations in the components of this pathway are further associated with increased risk of GC78-81,94. Additionally, the PD-1/PD-L1 pathway regulates the promotion and persistence of immune responses in the tumor microenvironment, making it a potential target for immune checkpoint blockade therapy in treating GC95-97. The expression levels and activity of the components of this pathway may also serve as prognostic indicators for GC95-97. Furthermore, the VEGF pathway is important for tumor metastasis and growth by promoting angiogenesis, with its activity being associated with decreased survival in patients with GC98,99. Lastly, infection with
The phytochemicals of FDY003 have also been reported to function as anti-GC compounds. AcT and Cm exhibit anti-proliferative and pro-apoptotic effects on GC cells102,103. Cordycepin induces apoptosis and cell cycle arrest as well as inhibits proliferation, survival, and migration in human GC cells by targeting PI3K/Akt, caspase, and CLEC2 signaling104-106. Isorhamnetin exhibits anti-migratory, anti-invasive, anti-proliferative, pro-apoptotic, and chemosensitizing effects by regulating peroxisome proliferator-activated receptor γ (PPAR-γ) and NF-kappa B cascades107,108. Kaempferol regulates the activities of Akt, cyclooxygenase-2 (COX-2), ERK, caspase, and endoplasmic reticulum (ER) stress signaling, leading to anti-proliferation and autophagic cell death in GC cells109,110. Luteolin inhibits proliferation, cell cycle progression, survival, migration, invasion, EMT, and angiogenesis as well as enhances the efficacy of chemotherapy and radiotherapy on GC cells by altering PI3K-Akt-mTOR, MAPK, STAT3, HIF-1, NF-kappa B, p53, and NOTCH1 signaling111-117. Quercetin reduces the survival, proliferation, stemness, metastasis, migration, invasion, and chemoresistance activities of GC cells by modulating PI3K-Akt-mTOR, HIF-1, Wnt, VEGF receptor (VEGFR), MAPK, caspase, protein kinase C (PKC), NF-kappa B, adenosine monophosphate (AMP)-activated protein kinase (AMPK), matrix metalloproteinases (MMP), uPAR, p53, and Hippo pathways118-125. It has also been reported that a higher intake of quercetin is associated with a decreased risk of developing GC 118 . β-Sitosterol suppresses the proliferation and survival capacity of GC cells through the modulation of AMPK, PTEN, heat shock protein 90 (HSP90), and caspase signaling 126 .
In summary, the network pharmacology analysis presented system-level insights into the pharmacological mechanisms underlying the anti-GC activity of FDY003. We identified the active phytochemical compounds and therapeutic targets responsible for the multicompound–multitarget pharmacological effects of FDY003. Through the functional analysis of FDY003 components, we identified the molecular- and signaling-level mechanisms of the herbal drug from a comprehensive systems perspective. Future studies are needed to elucidate the anticancer effects of FDY003 on various cancerous cellular behaviors such as migration, invasion, stemness, and angiogenesis, which may broaden the therapeutic usage of herbal drugs for cancer treatment.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211073030 - Supplemental material for A Network Pharmacology Perspective Investigation of the Pharmacological Mechanisms of the Herbal Drug FDY003 in Gastric Cancer
Supplemental material, sj-docx-1-npx-10.1177_1934578X211073030 for A Network Pharmacology Perspective Investigation of the Pharmacological Mechanisms of the Herbal Drug FDY003 in Gastric Cancer by Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon and Dae-Yeon Lee in Natural Product Communications
Footnotes
Acknowledgments
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (grant No. 2021R1F1A1049472).
Data Availability
All data generated or analyzed during this study are included in this published article and its Supplemental material file.
Author Contributions
Conceptualization: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee.
Methodology: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee.
Data collection: Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, and Tae-Wook Kwon.
Data analysis and investigation: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee.
Writing: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee.
All authors read and approved the final manuscript.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
