Abstract
Herbal drugs are continuously being developed and used as effective therapeutics for various cancers, such as cervical cancer (CC); however, their mechanisms of action at a systemic level have not been explored fully. To study such mechanisms, we conducted a network pharmacological investigation of the anti-CC mechanisms of FDY2004, an herbal drug consisting of Moutan Radicis Cortex, Persicae Semen
Cervical cancer (CC), a malignant tumor that occurs owing to the abnormal control of the cellular behavior of cervical cells, is a highly incident gynecologic cancer possessing high mortality with more than 0.5 million cases and 0.3 million deaths per year worldwide. 1,2 Key molecular and pathway mechanisms for CC pathology include the dysregulation of multiple oncogenes and tumor suppressors, and their associated signaling and gene regulatory cascades. 3,4 Growing knowledge regarding the pathomechanisms of CC has advanced the design and development of chemotherapy, targeted therapy, and cancer immunotherapy, which are the most widely used CC therapies at present 5,6 ; however, they frequently cause resistance and side effects. 7 Along with these concerns, substantial attention has been given to the development and usage of herbal drugs, the multiple component-multiple target polypharmacological agents with potent efficacy and safety, for cancer treatment. 8 -10 Herbal drugs elevate the clinical outcome and lower the toxicity of anticancer treatments, thereby improving the cancer-related symptoms and health status of cancer patients. 11
FDY2004 is an herbal drug consisting of Moutan Radicis Cortex (MRC), Persicae Semen (PS)
Because herbal drugs show their therapeutic activities in a complex multiple phytochemical component-multiple targeted-manner, network pharmacology, a research concept that combines pharmacology, medicine, and network science and effectively facilitates the mechanistic investigation of polypharmacological drugs, is widely used to uncover the comprehensive mechanisms of herbal drugs. 9,13 -16 Network pharmacology analysis identifies the major bioactive phytochemical components and therapeutic targets that are important for the pharmacological action of herbal drugs, integrates herbal drug-associated comprehensive data into diverse types of networks, and investigates pharmacological mechanisms of herbal drugs by analyzing their network properties. 9,13 -16 Using this network pharmacology methodology, we studied the therapeutic activity of FDY2004 12 in CC treatment.
Materials and Methods
Preparation of FDY2004
Dried raw plant materials of MRC (6.67 g), PS (6.67 g), and RRR (6.67 g) were purchased from Green Myeong-poom Pharm. (Namyangju, South Korea). Then, they were ground and mixed, and further added to 200 ml of distilled water and boiled for 120 minutes at 100 °C. After filtration with a 1–μm-pore filter (Hyundai Micro, Seoul, South Korea), the herbal extract was lyophilized at −80 °C. The lyophilized samples were kept at −20 °C and dissolved in distilled water for experimental use.
Cell Culture
HeLa human CC cells were purchased from the Korean Cell Line Bank (Seoul, South Korea). The cells were cultured in Dulbecco’s modified Eagle’s medium (WELGENE, Daegu, South Korea) supplemented with 100 µg/mL streptomycin, 100 U/mL penicillin, and 10% fetal bovine serum (Thermo Fisher Scientific, Waltham, MA, USA) in a humidified atmosphere in 5% CO2 at 37 °C.
Cell Viability Assay
The 3-(4,5-dimethyl-2-thiazolyl)−2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich, St. Louis, MO, USA) assay was conducted to measure cell viability. 17 Cells were seeded at 5.0 × 104 cells per well in 48-well plates and treated with FDY2004 for 48 hours in 5% CO2 at 37 °C. Cells were then incubated for 2 hours after adding 200 µL of MTT to the contents of each well. Afterwards, the MTT solution was removed and dimethyl sulfoxide was added to dissolve the formed formazan crystals. The absorbance was assessed using an Epoch 2 microplate reader (BioTek, Winooski, VT, USA) at 550 nm.
Determination of the Bioactive Phytochemical Components of FDY2004
The pharmacokinetic information (eg, oral bioavailability, drug-likeness, and Caco-2 permeability) of the phytochemical components of FDY2004 was investigated using the Traditional Chinese Medicine Systems Pharmacology 18, Traditional Chinese Medicine Integrated Database, 18 Anticancer Herbs Database of Systems Pharmacology, 19 and Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine 20 databases. Then, bioactive phytochemical components were determined according to the following criteria, as previously described: oral bioavailability ≥30%, Caco-2 permeability ≥−0.4, and drug-likeness ≥0.18. 14 Oral bioavailability refers to the quantitative proportion of the chemical components reaching the target sites (eg, tissues and organs) after oral administration; those with oral bioavailability ≥30% are expected to possess sufficient pharmacological absorption capability in the body. 21,22 Drug-likeness is a parameter indicating the qualitative assessment of chemical components to determine their suitability for drug usage based on their molecular information and Tanimoto coefficients 21,23 ; chemical components having drug-likeness ≥0.18 are considered to retain drug-like properties because the value of 0.18 is the average drug-likeness of available drugs. 21,23 Caco-2 permeability is a coefficient for evaluating the intestinal permeability, flux rate, and ability to transport chemical components using human Caco-2 intestinal cells; generally, those with Caco-2 permeability ≥−0.4 are considered to exhibit suitable permeability in the intestinal epithelium. 24,25
Targets of the Bioactive Phytochemical Components of FDY2004
The simplified molecular-input line-entry system (SMILES) notation of the phytochemical components of FDY2004 were extracted from the PubChem database, 26 and imported into the following databases to obtain the human molecular targets: Similarity Ensemble Approach, 27 SwissTargetPrediction, 28 Search Tool for Interactions of Chemicals 5, 29 and PharmMapper. 30 The targets associated with initiation, development, and progression of CC were collected from the following databases using “Uterine Cervical Neoplasms” (Medical Subject Headings Unique ID: D002583) as the search keyword: Therapeutic Target Database, 31 Comparative Toxicogenomics Database, 32 DrugBank, 33 Human Genome Epidemiology Navigator, 34 DisGeNET, 35 Online Mendelian Inheritance in Man, 36 GeneCards, 37 and Pharmacogenomics Knowledgebase. 38
Construction of the Herbal Drug-Related Networks
In network pharmacology, a network consists of nodes (eg, herbal medicines, bioactive phytochemical components, targets, or pathways), and edges (or links) representing interactions between them. 39 The number of edges connected to a node is called the degree. 39 Comprehensive data regarding the relationship between the herbal medicines and their bioactive phytochemical components and their components and therapeutic targets were merged into an herbal medicine-bioactive phytochemical component-target (H-C-T) network. The herbal medicine-bioactive phytochemical component-target-pathway (H-C-T-P) network was generated by linking the targets in the H-C-T network with their enriched pathways. A protein-protein interaction (PPI) network was created using the paired interaction between the targets with a confidence score ≥0.9 from the STRING database. 40 Nodes were considered as hubs if their degrees met the following criteria, ≥2 × average degree of all nodes in the PPI network, as previously described. 41,42 The constructed H-C-T, H-C-T-P, and PPI networks were visualized using Cytoscape software. 43
Survival Analysis
We used Kaplan-Meier Plotter 44 to perform the survival analysis of the FDY2004 targets.
Functional Analysis of the FDY2004 Targets
We used g:Profiler 45 and Kyoto Encyclopedia of Genes and Genomes 46 for the gene ontology (GO) and pathway enrichment analysis of the FDY2004 targets, respectively. Functional interaction analysis was conducted using GeneMANIA. 47
Molecular Docking Analysis
Information on the 3-dimensional structures of the bioactive phytochemical components and targets of FDY2004 was surveyed from the PubChem database 26 and the RCSB Protein Data Bank database, 48 respectively. Next, the molecular docking scores between them were evaluated by importing their structural information into Autodock Vina. 49 We considered the phytochemical component-target pairs with docking scores ≤ −5 to exhibit stable physicochemical binding affinities, as previously described. 50,51
Results
Effects of FDY2004 Treatment on Cervical Cancer Cells
To investigate the inhibitory effects of FDY2004 against CC, we treated HeLa cells with the herbal drug and observed their cell viability. The HeLa cells showed a significant reduction in their viability after FDY2004 treatment (Supplemental Figure S1), indicating the anticancer property of the herbal medicine against CC.
Bioactive Phytochemical Components of FDY2004 and Their Targets
Among the phytochemical components of FDY2004 investigated from herbal medicine-associated databases (Supplemental Table S1), we screened the bioactive components among those using the criteria mentioned earlier (see Materials and Methods). 52,53 In addition, some phytochemical components that unsatisfied the criteria but are present in large quantities in the herbal medicines and possess effective therapeutic activity were also regarded as bioactive components. Consequently, we identified 35 bioactive components for FDY2004 (Supplemental Table S2). Thereafter, using various databases for the investigation of protein-chemical interactions and disease-associated genes and proteins, we obtained 212 targets (116 CC-associated and 96 non-CC-associated) for the 29 bioactive phytochemical components (Supplemental Table S3).
Network-Perspective Analysis of the Mechanism of FDY2004
The FDY2004-related detailed information was integrated into the H-C-T network with 148 nodes (3 herbal medicines, 29 bioactive phytochemical components, and 116 CC-related targets) and 271 links between them (Figure 1 and Supplemental Table S3). Quercetin and kaempferol were the bioactive components with the largest number of targets (Figure 1, Supplemental Table S3), suggesting their crucial pharmacological roles. Furthermore, 93.10% (27 out of 29) of the phytochemical components shared one or more target, and 77.59% (90 out of 116) of the CC-associated targets interacted with2 or more herbal medicines (Figure 1), indicating the multiple component-multiple target effects of FDY2004.
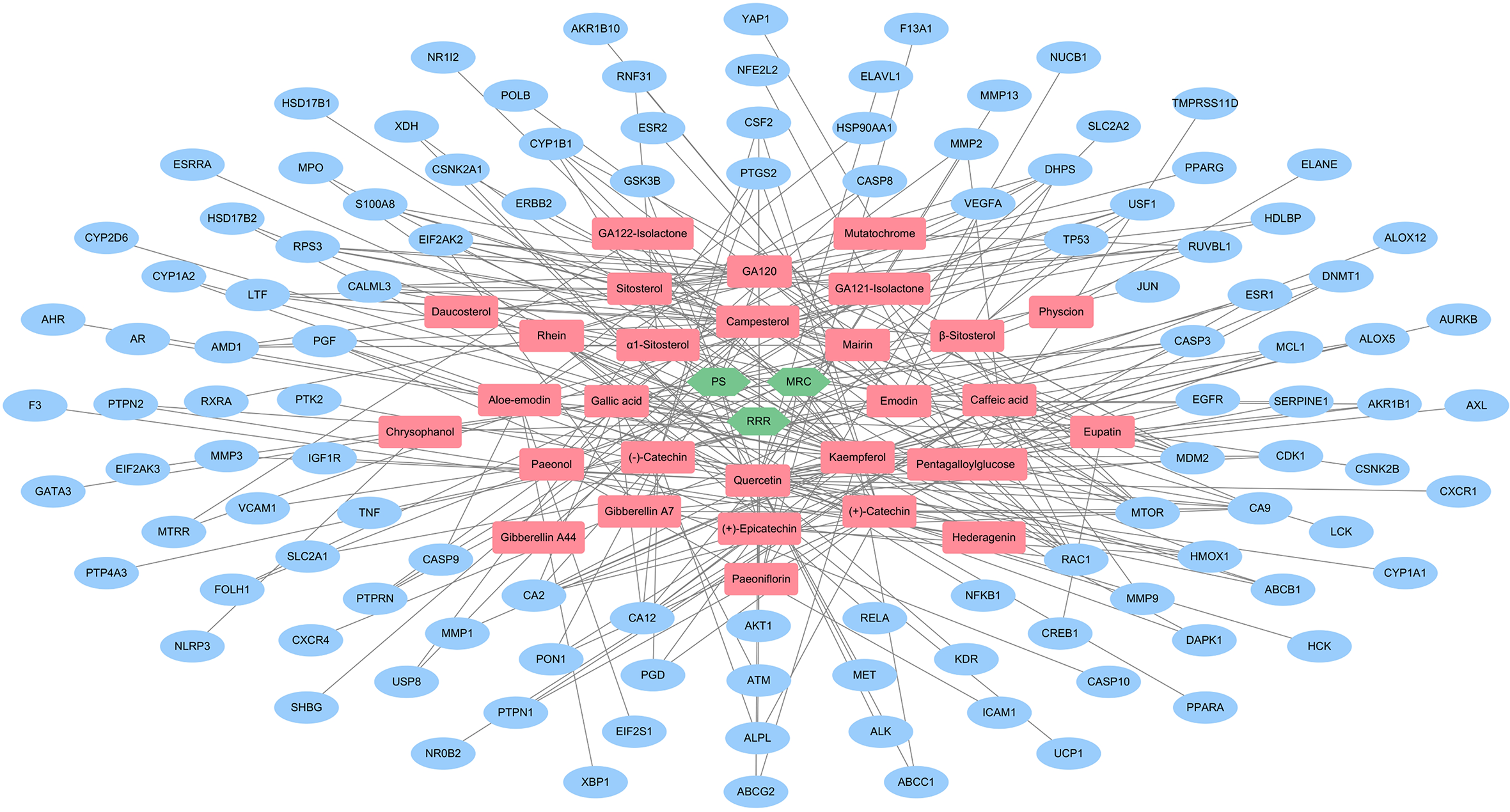
Herbal medicine-bioactive phytochemical component-target network of FDY2004. Green nodes, herbal medicines; red nodes, bioactive phytochemical components, blue nodes, targets.
To gain insight into the interactions between the CC-associated FDY2004 targets, we investigated the topological features of the PPI network (92 nodes and 231 links) in which the targets serve as nodes and their interactions as links (Figure 2). In the network analysis, we identified the high-degree hub nodes that have important biological functions and potential as key therapeutic targets (see Materials and Methods). 54,55 As a result, the nodes AKT1, EGFR, ESR1, HSP90AA1, JUN, PTK2, TNF, TP53, and VEGFA were determined as hubs, indicating that they are the major targets for the therapeutic activities of FDY2004 against CC (Figure 2). Furthermore, the expression levels of these nodes correlated with the survival outcome of patients with CC (Figure 3), which indicates their clinical significance. In addition, GeneMANIA analysis indicated the biological mechanisms underlying the interaction between the hubs, including the physical interaction, co-expression, and genetic interaction (Supplemental Figure S2). Altogether, the results demonstrate the network-perspective characteristics underlying the pharmacological activity of FDY2004 for CC treatment.

Protein-protein interaction network for cervical cancer-associated targets of FDY2004. Pink nodes, hub cervical cancer-associated targets; blue nodes, non-hub cervical cancer-associated targets that interact with a single hub node; purple nodes, non-hub cervical cancer-associated targets that interact with 2 or more hub nodes; orange nodes, non-hub cervical cancer-associated targets that do not interact with hub nodes.

Survival analysis of the cervical cancer-related targets of FDY2004. Kaplan-Meier curves for the survival of patients with cervical cancer having high or low expression levels of the indicated targets.
Investigation of Functional Enrichment of FDY2004 Networks
To dissect the biological mechanisms of the anti-CC activity of FDY2004, we analyzed the GO terms enriched for its targets. We found that the modulation of cellular behaviors, such as apoptosis, growth, proliferation, and survival, may be mediated by the FDY2004 targets (Supplemental Figure S3), thus demonstrating the molecular mechanisms of FDY2004.
Aberrant activation and/or inactivation of various signaling pathways is the key mechanism of cancer pathology. 56 By analyzing the pathway enrichment for the FDY2004 targets, we found that the targets were major components of the following CC-associated pathways: “Apoptosis,” “Cellular senescence,” “ErbB signaling pathway,” “Estrogen signaling pathway,” “Focal adhesion,” “HIF-1 signaling pathway,” “Human papillomavirus infection,” “IL-17 signaling pathway,” “MAPK signaling pathway,” “NF-kappa B signaling pathway,” “p53 signaling pathway,” “Pathways in cancer,” “PD-L1 expression and PD-1 checkpoint pathway in cancer,” “PI3K-Akt signaling pathway,” “Platinum drug resistance,” “Prolactin signaling pathway,” “Ras signaling pathway,” “TNF signaling pathway,” “VEGF signaling pathway,” and “Viral carcinogenesis” (Figure 4 and Supplemental Figure S3).

Herbal medicine-bioactive phytochemical component-target-pathway network of FDY2004. Green nodes, herbal medicines; red nodes, bioactive phytochemical components; blue nodes, targets; orange nodes, pathways.
The overall functional enrichment analysis suggests the pharmacological regulatory properties of FDY2004 for CC treatment at the molecular and pathway levels.
Molecular Docking Analysis
Using in silico molecular docking techniques, we examined the binding affinities of the chemical constituents and their CC-associated targets. The interactions between the bioactive phytochemical components and their hub targets showed molecular docking scores ≤ −5.0 (Figure 5), which suggests the potential binding capacities between them and further confirms the results of the network pharmacological analysis.
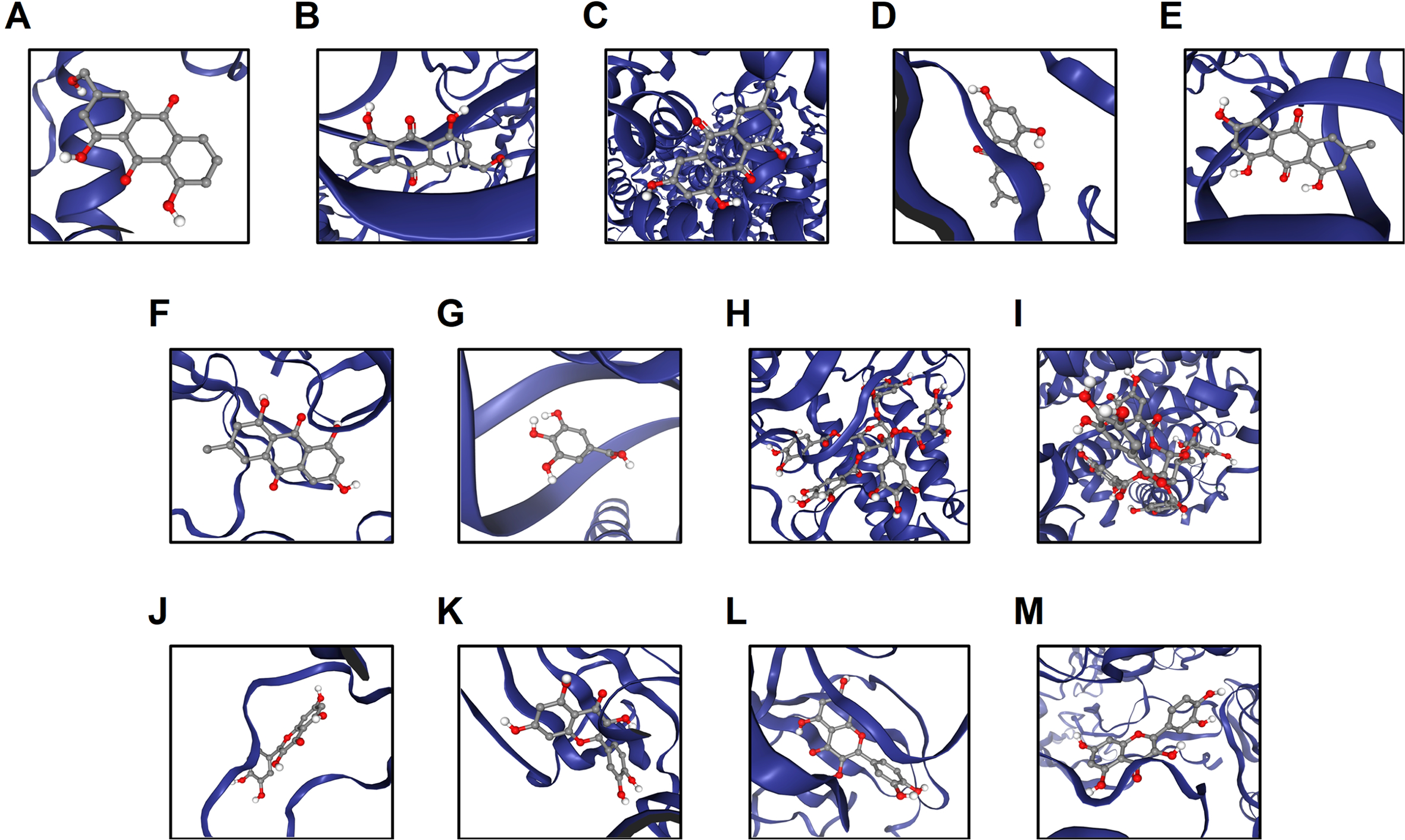
Molecular docking analysis for the bioactive phytochemical components of FDY2004 and their hub targets. (A) Aloe-emodin-HSP90AA1 (score = −7.2). (B) Aloe-emodin-TP53 (score = −7.1). (C) Emodin-ESR1 (score = −7.8). (D) Emodin-TNF (score = −6.8). (E) Emodin-TP53 (score = −7.3). (F) Emodin-VEGFA (score = −7.0). (G) Gallic acid-JUN (score = −5.1). (H) Pentagalloylglucose-EGFR (score = −8.9). (I) Pentagalloylglucose-ESR1 (score = −7.0). (J) Quercetin-AKT1 (score = −6.4). (K) Quercetin-EGFR (score = −7.9). (L) Quercetin-PTK2 (score = −6.1). (M) Rhein-VEGFA (score = −7.1).
Discussion
Globally, CC is among the most incident, prevalent, and fatal malignant cancer affecting women. 1 Herbal drugs are increasingly being considered as effective cancer therapeutics for their pharmacological efficacy and reduced toxicity. 8,9 Using a network pharmacology-based methodology, we investigated the therapeutic activity of FDY2004 against CC. We found that FDY2004 exerted anticancer effects on CC cells. The overall analysis results identified 29 bioactive phytochemical components of FDY2004 that interact with 116 CC-related targets. GO enrichment investigation suggested that the targets may participate in the modulation of apoptosis, growth, proliferation, and survival. Diverse signaling associated with CC initiation and progression, including phosphoinositide 3-kinase (PI3K)-Akt, human papillomavirus (HPV) infection, interleukin (IL)−17, mitogen-activated protein kinase (MAPK), tumor necrosis factor (TNF), focal adhesion, and viral carcinogenesis pathways, may confer the polypharmacological anti-CC effects of FDY2004.
The hub targets of FDY2004 may serve as key regulators underlying the pathological processes of CC and have druggable potential. AKT (encoded by
The FDY2004-targeted pathways are the key signaling mechanisms mediating CC pathology and its therapeutic strategies. Dysregulation of erythroblastic leukemia viral oncogene homologue (ErbB), focal adhesion, hypoxia-inducible factor 1 (HIF-1), MAPK, PI3K-Akt, and Ras pathways are important for CC initiation, development, and progression. 4,122 -127 Abnormal activity of the estrogen signaling pathway is one of the major drivers of CC, and high expression levels of its pathway components are related to the risk of CC. 73 -75,128 Viral infection with HPV is one of the most crucial contributors to CC carcinogenesis. 129 The key components of the IL-17 pathway play a role in the carcinogenesis of CC with viral infection, and their activity functions as a prognostic indicator for patients with CC. 130 -134 Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling modulates the growth, cell cycle, migration, invasion, survival, apoptosis, immunomicroenvironment, autophagy, and the pathomechanisms associated with the viral infection of CC cells. 135 -141 The p53 pathway is a pharmacological mediator of anticancer therapy-induced intervention of the survival, migration, invasion, and proliferation of CC cells. 142 -144 The upregulated activity of programmed death-ligand 1 (PD-L1)/programmed cell death protein 1 (PD-1) is implicated in the decreased survival of patients with CC and targeting this pathway can elevate anti-tumor immunity. 145,146 The hyperactivation of the anti-apoptotic prolactin pathway may enhance the survival and proliferative capacity of CC cells. 147,148 The TNF pathway controls the pro-tumorigenic inflammatory processes of CC cells and is linked with HPV-associated CC progression. 91,149 -151 Dysregulated activation of the VEGF pathway promotes the metastasis and angiogenesis of tumors, which in turn promotes CC progression and advancement. 152,153
The bioactive phytochemical components of the herbal medicines comprising FDY2004 have been reported to exhibit pharmacological effects against CC, which supports the anti-CC activity of this herbal drug. Aloe-emodin inhibits cell cycle progression, proliferation, growth, and survival, but enhances the apoptosis and radiosensitivity of CC cells. 154 -156 The anticancer mechanisms of caffeic acid include the enhancement of apoptotic cell death, chemosensitivity, and cell cycle arrest and the blockage of the proliferation and EMT of CC cells; these pharmacological effects are exhibited via the modulation of transforming growth factor (TGF)-β, HIF-1, p53, mitochondrial, and adenosine monophosphate-activated protein kinase (AMPK) pathways. 157 -161 Catechins possess anticancer potential that may cause growth-suppression and apoptosis of CC cells mediated by caspase activation and induction of oxidative stress. 162,163 Chrysophanol, paeoniflorin, paeonol, and pentagalloylglucose can repress the survival and proliferation of CC cells while promoting cell cycle arrest and apoptosis. 164 -167 Emodin suppresses the migration, invasion, and proliferation, and causes the oxidative stress, DNA damage, and apoptosis of CC cells by modulating the PI3K-Akt, HIF-1, VEGF, TGF-β, mitochondrial, and death receptor signaling pathways. 168 -171 Gallic acid is an anticancer compound that modulates the activities of oncogenic ErbB, PI3K-Akt, MAPK, p53, caspase, and HPV signaling, which leads to the augmentation of apoptosis and necrosis and suppression of growth, proliferation, angiogenesis, and chemoresistance. 172 -174 The pro-apoptotic and anti-proliferative role of hederagenin is mediated by the modulation of signal transducer and activator of transcription (STAT)−3 pathway. 175 Kaempferol regulates the activities of PI3K-Akt and telomerase reverse transcriptase (hTERT) pathways to promote apoptotic cell death and suppress the growth and survival of CC cells. 176 Mairin (betulinic acid) stimulates anti-tumor processes such as apoptosis, oxidative stress, and antiproliferation of CC cells that are conferred through the HIF, VEGF, TNF, PI3K-Akt, caspase, reactive oxygen species (ROS)-mediated mitochondrial, and endoplasmic reticulum (ER) pathways. 177 -179 Rhein-induced growth repression and apoptosis of CC cells may occur through the caspase and calcium pathways. 180 The anticancer component quercetin inhibits cell cycle progression, proliferation, survival, viability, metastasis, migration, invasion, and EMT of CC cells by modulating diverse signaling mechanisms, including HPV, p53, ER stress, ROS, PI3K-Akt, mitochondrial, DNA damage response, epigenetic, p53, caspase, MAPK, HIF-1, Wnt, and NF-κB pathways. 144,181 -186 β-Sitosterol controls the expression and activity of HPV E6 and p53, which induces the apoptosis and arrests the growth of CC cells. 187,188
Limitations of the current study include the lack of experiments that evaluate the anti-CC effects and toxicity of combined treatment of FDY2004 with other anticancer agents involved in chemotherapy, targeted therapy, and cancer immunotherapy. Further investigations are needed to expand the therapeutic application of herbal drugs as anticancer therapies.
In summary, the overall network pharmacology-based analysis demonstrated the anti-CC properties of FDY2004. Our study offers an in-depth and systematic understanding of the polypharmacological mechanisms of herbal drugs, which will contribute to the design and development of improved anticancer herbal agents.
Supplemental Material
Online supplementary file 1 - Supplemental material for A Comprehensive Understanding of the Anticancer Mechanisms of FDY2004 Against Cervical Cancer Based on Network Pharmacology
Supplemental material, Online supplementary file 1, for A Comprehensive Understanding of the Anticancer Mechanisms of FDY2004 Against Cervical Cancer Based on Network Pharmacology by Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon and Dae-Yeon Lee in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
