Abstract
Ovarian cancer (OC) is one of the deadliest gynecological tumors responsible for 0.21 million deaths per year worldwide. Despite the increasing interest in the use of herbal drugs for cancer treatment, their pharmacological effects in OC treatment are not understood from a systems perspective. Using network pharmacology, we determined the anti-OC potential of FDY003 from a comprehensive systems view. We observed that FDY003 suppressed the viability of human OC cells and further chemosensitized them to cytotoxic chemotherapy. Through network pharmacological and pharmacokinetic approaches, we identified 16 active ingredients in FDY003 and their 108 targets associated with OC mechanisms. Functional enrichment investigation revealed that the targets may coordinate diverse cellular behaviors of OC cells, including their growth, proliferation, survival, death, and cell cycle regulation. Furthermore, the FDY003 targets are important constituents of diverse signaling pathways implicated in OC mechanisms (eg, phosphoinositide 3-kinase [PI3K]-Akt, mitogen-activated protein kinase [MAPK], focal adhesion, hypoxia-inducible factor [HIF]-1, estrogen, tumor necrosis factor [TNF], erythroblastic leukemia viral oncogene homolog [ErbB], Janus kinase [JAK]-signal transducer and activator of transcription [STAT], and p53 signaling). In summary, our data present a comprehensive understanding of the anti-OC effects and mechanisms of action of FDY003.
Introduction
Ovarian cancer (OC) is one of the deadliest gynecological tumors responsible for 0.21 million deaths per year worldwide 1 . Several cytotoxic and molecular-targeted drugs have been developed and used for OC treatment in clinics2-4; however, their treatment rates and efficacies remain limited, and they may further generate harmful side effects2-4. To overcome these issues, herbal drugs are gaining attention as effective and complementary anticancer therapeutics that can improve the efficacy of anticancer agents and alleviate their toxicities5,6. Herbal drugs may decrease mortality, cancer symptoms, and therapy-related adverse events, and improve the prognosis and survival of patients with OC5,6.
FDY003, a combination of Artemisia capillaris Thunberg (AcT), Cordyceps militaris (Cm), and Lonicera japonica Thunberg (LjT), is a prescription that displays growth-suppressive and cell-death-inducing effects against diverse types of cancer7-9. These pharmacological outcomes result from the targeting of various molecules and pathways related to important cancerous and tumorigenic mechanisms7-9; however, the pharmacological properties of FDY003 for OC treatment have not been elucidated from a systems perspective.
Network pharmacology is a research paradigm that seeks to elucidate the polypharmacological regulatory mechanisms of herbal drugs by analyzing and studying the integrated large-scale pharmacological data relevant to them; it is proven to be the most efficient methodology for herbal-drug-associated studies10-17. This research approach produces distinct types of herbal-drug-associated networks by merging relevant comprehensive pharmacological, medicinal, and biomolecular data10-17. This is followed by an investigation of the topological and functional characteristics of the networks, which contributes to the systematic understanding of the treatment mechanisms of herbal drugs10-17. Using network pharmacology, we determined the anti-OC potential of FDY003 from a comprehensive systems view.
Results and Discussion
Anti-Ovarian Cancer Activities of FDY003
To confirm the pharmacological activities of FDY003 against OC, SK-OV-3 human OC cells were treated with FDY003 and/or paclitaxel, a frontline drug for OC chemotherapy 18 , and their viability was assessed. The viability of SK-OV-3 cells was significantly (P < .05) reduced after FDY003 treatment compared to the untreated cells, and the reduction in viability was significantly (P < .05) greater after co-treatment with paclitaxel and FDY003 (Supplementary Figure S1A and B). These results suggest that FDY003 exhibits pharmacological activity against OC.
Active Ingredients of FDY003 and Their Targets
Among the various ingredients of FDY003, those with Caco-2 permeability ≥ −0.4, druglikeness ≥ 0.18, and oral bioavailability ≥ 30%, were defined as active (Supplementary Table S1)7,12,19,20. Notably, some were further regarded as active ingredients, despite failing to meet the aforementioned requirements on the basis of their previously reported anti-OC effects. From the pharmacokinetic analysis, we obtained 20 active ingredients for FDY003 (Supplementary Table S2). Subsequently, by analyzing the protein–chemical interactions, we obtained 270 targets for 18 of the FDY003 active ingredients (none were identified for loniceracetalides B_qt and demethoxycapillarisin) (Supplementary Table S3). Among the 18 active ingredients having interacting targets, 16 (excluding 4'-methylcapillarisin and isoarcapillin) interacted with 108 targets related to the pathophysiological mechanisms of OC (Supplementary Table S3).
Network-Level Effect of FDY003 Against Ovarian Cancer
We initiated the network pharmacology study for FDY003 by integrating comprehensive FDY003-related information into an herb–active chemical ingredient–target (H-I-T) network (Figure 1 and Supplementary Table S3). In the H-I-T network of FDY003, kaempferol, luteolin, and quercetin displayed the largest number of targets (Figure 2 and Supplementary Table S3), which suggests that these may be the major phytochemicals responsible for the anti-OC activity of FDY003. Among the FDY003 targets, 79.6% (86 of 108 targets) had 2 or more interacting ingredients (Figure 1), implying the multiple ingredient–multiple target pharmacological nature of FDY003.
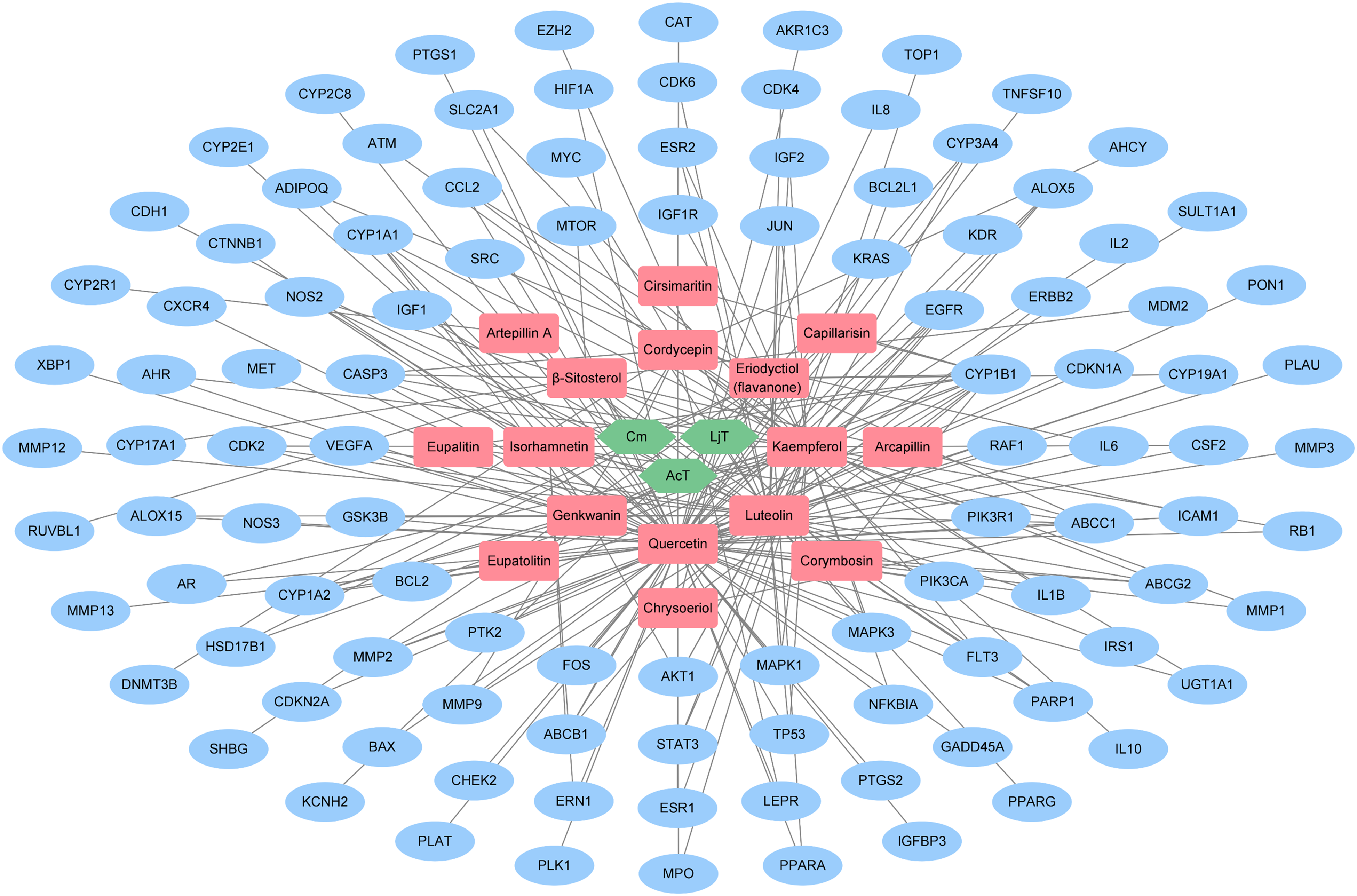
Herb–active chemical ingredient–target (H-I-T) network for FDY003. Green nodes, drug constituents; red nodes, active constituents; blue nodes, ovarian cancer (OC)-associated targets.

Protein–protein interaction (PPI) network for ovarian cancer (OC)-associated targets of FDY003. Purple nodes, hub targets; blue nodes, non-hub targets.
Since the pharmacological effects of a given drug are exerted and conferred through the molecular, genetic, and functional interactions among the therapeutic targets21-25, we merged and integrated the interaction information between the OC-related targets of FDY003 into a protein–protein interaction (PPI) network (Figure 2). Subsequently, we analyzed the topological characteristics of the FDY003-associated PPI network and explored the hub nodes in the network; notably, hub nodes possessing the largest number of degrees and serving crucial roles as effective therapeutic targets26,27. On the basis of previous analyses, hubs were determined to have a degree at least twice the average degree of all nodes in the network28,29. The hub nodes of the PPI network were AKT1, CASP3, CTNNB1, EGFR, ESR1, IL6, JUN, MAPK3, MYC, SRC, STAT3, TP53, and VEGFA (Figure 2), which may be the major targets for the pharmacological activities of FDY003 against OC. Furthermore, the expression status of these targets was shown to serve as a significant (P < .05) predictor of survival and mortality of patients with OC (Figure 3), suggesting the clinical and therapeutic importance of the hub targets.

Survival analysis associated with the ovarian cancer (OC)-related targets of FDY003. Kaplan–Meier curves depicting the survival of patients with OC in relation to the expression status of indicated targets.
To gain insights into the regulatory effects of FDY003 at the molecular and signaling levels in OC treatment, we explored the functional enrichment of FDY003 targets. Functional enrichment investigation revealed that the targets may coordinate diverse cellular behaviors of OC cells, including their growth, proliferation, survival, death, and cell cycle regulation (Supplementary Figure S2). Furthermore, the FDY003 targets were important constituents of diverse signaling pathways implicated in the OC mechanisms (Figure 4 and Supplementary Figure S2), which suggests that FDY003 may target these key pathways, exhibiting its anti-OC activities.

Herb–active chemical–ingredient–target–pathway (H-I-T-P) network for FDY003. Green nodes, drug constituents; red nodes, active constituents; blue nodes, ovarian-cancer-associated targets; orange nodes, ovarian-cancer-related pathways.
The overall analysis suggests that FDY003 exhibits multiple-ingredient, multiple-target, and multiple-pathway polypharmacological features, with multiple modes of action, against OC.
Molecular Docking Analysis of FDY003 Ingredients and Their Targets
To verify the interactions between the FDY003 ingredients and their targets, we performed molecular docking analysis and assessed their binding affinities. The active ingredients of FDY003 and their hub targets exhibited chemical–protein docking scores < − 5.0 (Figure 5 (A–L) and Supplementary Figure S3), which suggests their potent binding affinities.
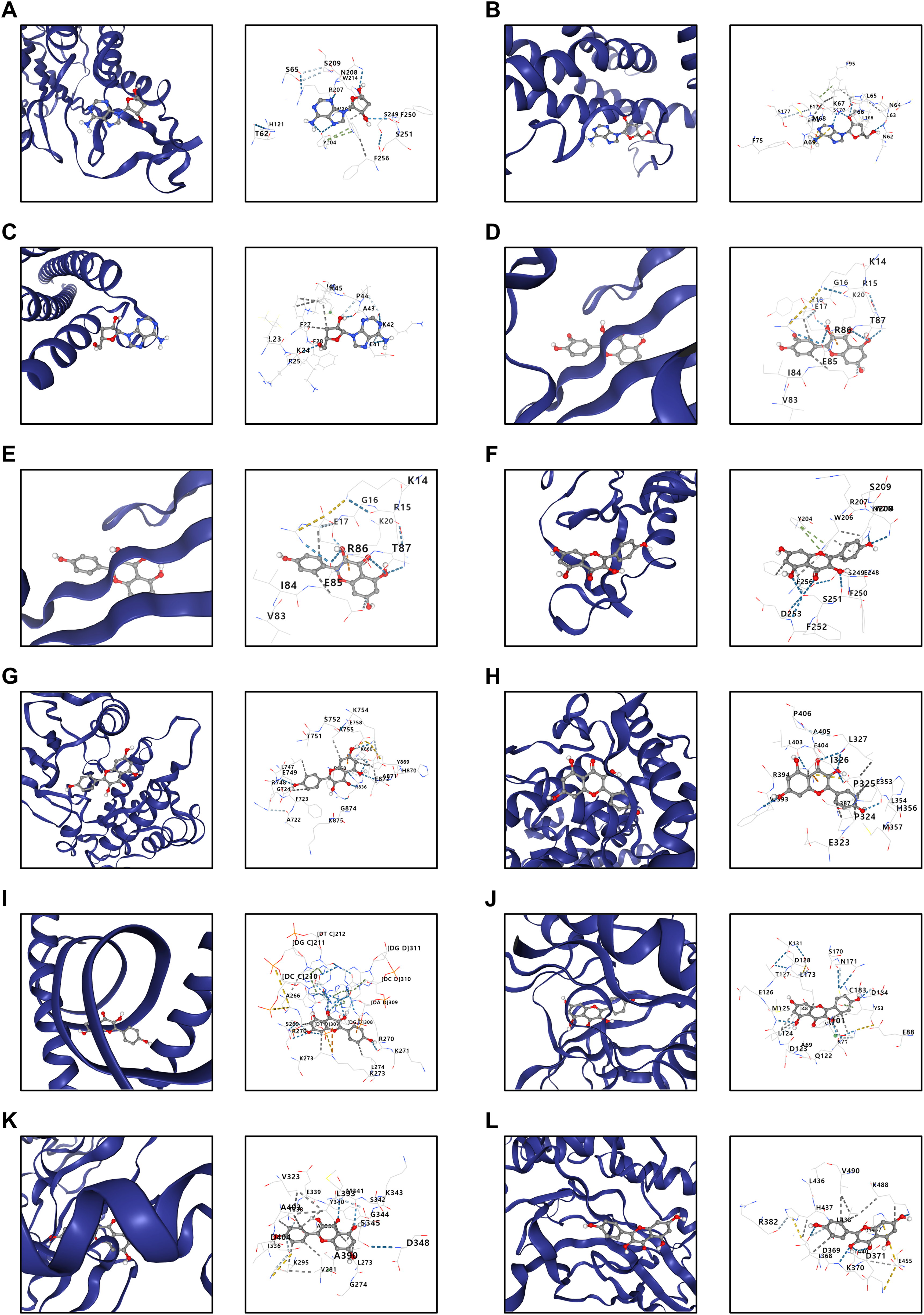
Molecular docking study for the active ingredients of FDY003 and their targets. (A) Cordycepin–CASP3 (score = − 5.6). (B) Cordycepin–IL6 (score = − 6.1). (C) Cordycepin–MYC (score = − 5.1). (D) Isorhamnetin–AKT1 (score = − 6.4). (E) Kaempferol–AKT1 (score = − 6.5). (F) Kaempferol–CASP3 (score = − 6.8). (G) Kaempferol–EGFR (score = − 8.1). (H) Kaempferol–ESR1 (score = − 8.6). (I) Kaempferol–JUN (score = − 9.0). (J) Kaempferol–MAPK3 (score = − 8.4). (K) Kaempferol–SRC (score = − 8.6). (L) Kaempferol–STAT3 (score = − 7.4).
In the present study, we found that FDY003 suppressed the viability of human OC cells and further chemosensitized them to cytotoxic chemotherapy. Functional enrichment investigation revealed that the targets may coordinate diverse cellular behaviors of OC cells, including their growth, proliferation, survival, death, and cell cycle regulation. Furthermore, the FDY003 targets were found to be important constituents of diverse signaling pathways implicated in OC mechanisms, that is, phosphoinositide 3-kinase (PI3K)-Akt, mitogen-activated protein kinase (MAPK), focal adhesion, hypoxia-inducible factor (HIF)-1, estrogen, mitogen-activated protein kinase (TNF), erythroblastic leukemia viral oncogene homolog (ErbB), Janus kinase-signal transducer and activator of transcription (JAK-STAT), and p53 signaling. These analyses present the anti-OC effects of FDY003 and its regulatory mechanisms.
The important FDY003 hub targets have been reported to coordinate diverse cancerous and pro-tumorigenic events of OC and serve as potent targets for pharmacological interventions against OC. More specifically, the oncogene AKT1 enhances growth, migration, epithelial-to-mesenchymal transition (EMT), glycolysis, and chemoresistance of OC cells; it functions as a biomarker for therapeutic response to OC treatment30-33. CASP3 expression and activity are correlated with apoptosis, migration, proliferation, growth, viability, and chemosensitivity of OC cells; it is further associated with metastasis, survival, and clinical and prognostic outcomes of patients with OC34-39. The dysregulated expression and activity, and the genetic mutation of CTNNB1 are implicated in the development, progression, metastasis, recurrence, and prognosis of OC40-42. Overexpression, amplification, and mutations of EGFR are associated with OC pathomechanisms; targeting EGFR may lead to antitumor effects 43 . ESR1 is involved in the proliferation and invasion of OC cells and its genetic features are correlated with the risk, progression, and overall survival of patients with OC44-46. IL6 induces angiogenesis, pro-tumorigenic inflammation, infiltration of tumor-promoting macrophages, chemoresistance, survival, proliferation, and immune surveillance of OC cells and tumors; its increased expression is related to a worse prognosis for patients with OC47-49. The proto-oncogene JUN stimulates chemoresistance, survival, migration, proliferation, angiogenesis, growth, EMT, invasion, recurrence, and metastasis of OC cells50-55. Expression of JUN is further elevated in OC cells and tumors, which correlates with carcinogenesis, progression, and prognosis of OC50-55. Extracellular signal-regulated kinase (ERK)-2 (encoded by MAPK3) regulates survival, growth, proliferation, cell cycle process, migration, invasion, anoikis resistance, and stemness of OC cells56-59. Targeting MYC can suppress cell cycle progression, growth, proliferation, viability, survival, and chemoresistance of OC cells; MYC is a predictor of prognosis and therapy response in patients with OC60-63. Genetic mutation and aberrant expression and activity of TP53 are important events that contribute to OC initiation and progression64,65. Src (encoded by SRC) is a key oncogenic kinase that is hyperactivated in OC cells and tumors and is involved in various pro-tumorigenic mechanisms; targeting the kinase can enhance therapy responsiveness66-69. STAT3 is a promising drug target for OC treatment because it is frequently overexpressed in OC cells and promotes angiogenesis, cancer stemness, proliferation, metastasis, survival, and drug resistance 70 . VEGFA is overexpressed in OC cells and tumors and it modulates angiogenesis, lymphangiogenesis, tumor vascularization, carcinogenesis, migration, invasion, progression, proliferation, tumorigenicity, and therapeutic sensitivity71-77.
The targeted OC-related pathways of FDY003 represent important signaling mechanisms of OC, and they may serve as targets for pharmacological modulation in OC treatment. The 5' adenosine monophosphate-activated protein kinase (AMPK), estrogen, gonadotropin-releasing hormone (GnRH), and mammalian target of rapamycin (mTOR) pathways are involved in the regulation of proliferation, survival, invasion, migration, and metastasis of OC cells78-82. Imbalanced activity of the chemokine pathway contributes to metastasis, enhanced proliferation and survival, angiogenesis, vascularization, and pro-tumorigenic inflammation of OC cells and tumors, which makes the pathway a potential therapeutic target 83 . Aberrations in the functional regulation of ErbB, MAPK, PI3K-Akt, and Ras pathways may promote uncontrolled cell growth and proliferation, survival, cell cycle progression, metastasis, angiogenesis, and malignant progression of OC cells, which can be inhibited by their targeting84-86. Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and TNF pathways stimulate pro-tumorigenic inflammation that is suppressed by pharmacological intervention, and their expression levels are relevant to OC carcinogenesis87,88. The focal adhesion pathway modulates migration, angiogenesis, proliferation, metastasis, survival, invasion, and anoikis resistance; inhibition of this pathway can augment therapeutic sensitivity89-92. The HIF-1 and vascular endothelial growth factor (VEGF) pathways confer angiogenic and metastatic properties to OC cells and tumors, and their expression levels are correlated with malignancy, prognosis, and metastasis of OC93-95. Activation of the JAK-STAT pathway results in EMT, chemoresistance, stemness, migration, metastasis, invasion, proliferation, and survival of OC cells; it is related to the poor survival outcome of patients with OC96-99. The prolactin pathway drives hyperproliferation, pro-survival, and malignant progression of OC cells and tumors; its activity is associated with the risk of OC100-102. The programmed death-ligand 1 (PD-1)/programmed cell death-ligand 1 (PD-L1) pathway, which regulates antitumor immune response, is a prognostic indicator for the survival of patients with OC 103 . Genetic mutations and improper functional modulation of the p53 pathway may result in metastasis, EMT, proliferation, and increased survival and motility of OC cells and tumors; these are key mechanisms in the incidence, prognosis, and disease progression104-110.
FDY003 and its active ingredients possess anti-OC properties. The growth-arrest and pro-apoptotic effects of Cm in OC cells are mediated by the pharmacological regulation of TNF-α, TNF receptor 1, NF-κB, activating transcription factor 3 (ATF3), caspase, poly ADP-ribose polymerase (PARP)-1, B-cell lymphoma (Bcl)-2, and Bcl-2-associated X protein (Bax) pathways111,112. Cordycepin targets Dickkopf-related protein 1 (Dkk1), autophagy-related protein 8 (ATG8), beclin, microtubule-associated protein 1A/1B-light chain 3 (LC3), β-catenin, matrix metalloproteinase (MMP), transforming growth factor (TGF)-β, vimentin, peroxisome proliferator-activated receptor-gamma coactivator (PGC)-1α, E-cadherin, estrogen receptor (ER)-α, mitochondrial, CC chemokine ligand 5 (CCL5), Akt, NF-κB, the long form of cellular FLICE-inhibitory protein (c-FLIPL), and caspase signaling of OC cells; these pharmacological effects inhibit the survival, self-renewal potential, viability, EMT, metastasis, therapeutic resistance, migration, and invasion of OC cells113-116. Kaempferol modulates the activities of p53, p21, Bad, Bax, caspase, Bcl-2, B-cell lymphoma-extra large (Bcl-xL), c-Myc, HIF, VEGF, Akt, estrogen-related receptor alpha (ESRRA), ERK, STAT, p38, c-Jun N-terminal kinases (JNK), checkpoint kinase 2 (Chk2), cell division cycle 25C (CDC25c), cyclin-dependent kinase 1 (Cdc2), Fas-associated protein with death domain (FADD), death receptor, TNF-related apoptosis-inducing ligand (TRAIL), CCAAT-enhancer-binding protein homologous protein (CHOP), and endoplasmic reticulum (ER) stress pathways, thereby suppressing viability, growth, cell cycle progression, chemoresistance, angiogenesis, and proliferation of OC cells108,117-122. Luteolin inhibits invasion, migration, survival, drug resistance, autophagy, metastasis, EMT, and proliferation of OC cells by targeting Bcl-2, MMP, FAK, ERK, Snail, Slug, and Twist1 pathways123-126. Quercetin pharmacologically modulates autophagy, survival and death, viability, invasion, migration, proliferation, radio- and chemoresistance, cell cycle processes, and stem-like features of OC cells127-129. These processes are mediated by ER stress, STAT, Bcl-2, Bcl-xL, urokinase-type plasminogen activator (uPA), MMP, caspase, mitochondrial, BH3-interacting domain death agonist (Bid), Bad, Bax, survivin, induced myeloid leukemia cell differentiation protein (Mcl-1), p53, p21, cyclins, and TGF-β pathways127-129.
Conclusions
In summary, the network pharmacological analysis enabled us to dissect the regulatory mechanisms of FDY003 against OC. FDY003 decreased the viability of human OC cells and increased their chemosensitivity. The drug contains 16 potentially active ingredients that may target various key OC-associated genes, proteins, and pathways. Follow-up studies need to assess the pharmacological action of FDY003 on important cancerous behaviors of cancer cells and tumors (eg, angiogenesis, anoikis resistance, cancer stem-like features, and metastasis) and chemo- and radiosensitivity. Such studies will advance the role of natural medicines as effective anticancer therapies.
Materials and Methods
Cell Culture
The SK-OV-3 human OC cell line was purchased from the Korean Cell Line Bank (Seoul, Korea) and incubated in Dulbecco’s modified Eagle’s medium (WELGENE Inc.) supplemented with antibiotics (penicillin-streptomycin) (Thermo Fisher Scientific, Inc.) and 10% fetal bovine serum (WELGENE Inc.) at 37°C under 5% CO2.
Preparation of FDY003
The dried raw herbal constituents of FDY003 (eg, Cm [100 g], LjT [150 g], and AcT [150 g]) were acquired from Hanpure Pharmaceuticals (Pocheon, Korea). After grinding and mixing, the herbal samples were placed in 70% ethanol (500 mL), and subjected to a 3 h reflux extraction at 80°C. The herbal extracts were successively purified with 80% and 90% ethanol and lyophilized at − 80°C. The final 50.4 g of dried extract was stored at − 20°C and dissolved in distilled water prior to use.
Evaluation of Cell Viability in Response to Drug Treatment
Cell viability in response to drug treatment was determined using water-soluble tetrazolium salt (WST-1) assays. Cells (1.0 × 10 4 ) were seeded in a 96-well plate and cultured with the indicated drug combinations consisting of FDY003 and paclitaxel (Sigma-Aldrich) for 72 h, and subsequently with WST-1 solution (Daeil Lab Service Co. Ltd) for an additional 2 h, at 37°C under 5% CO2. The viability of the drug-treated cells was determined by measuring the absorbance at 450 nm using an xMark microplate absorbance spectrophotometer (Bio-Rad).
Active Ingredient Screening
Detailed information on the ingredients of FDY003 was obtained from the Anticancer Herbs Database of Systems Pharmacology 130 , Traditional Chinese Medicine Systems Pharmacology 20 , and Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine 131 . We identified bioactive ingredients by evaluating their oral bioavailability, druglikeness, and Caco-2 permeability, which are pharmacokinetic parameters widely considered for active ingredient screening12,19,20. Oral bioavailability indicates the efficiency of delivery of orally administered compounds to their target organs and tissues20,132; oral bioavailability ≥ 30% is an indicator of a suitable absorption rate in the human body20,132. Druglikeness indicates the potential of compounds to act as drugs with respect to their structural, functional, physicochemical, and molecular characteristics20,133; a druglikeness ≥ 0.18 (the average druglikeness value of whole available pharmacological agents) is an indicator of reasonable druggability20,133. Caco-2 permeability indicates the intestinal permeability and absorptivity of compounds20,134-136; Caco-2 permeability ≥ − 0.4 is an indicator of potent intestinal permeability required by a druggable compound29,30. In summary, the active ingredients selected were those with oral bioavailability ≥ 30%, druglikeness ≥ 0.18, and Caco-2 permeability ≥ − 0.412,19,20.
Exploration of FDY003 Targets
The simplified molecular-input-line entry system notation of the active ingredients of FDY003 searched from PubChem 137 was entered into the following databases: SwissTargetPrediction 138 , PharmMapper 139 , Search Tool for Interactions of Chemicals 5 140 , and Similarity Ensemble Approach 141 , and the targets of the active ingredients were obtained. In addition, we investigated the targets associated with OC pathophysiology by entering the search term “ovarian cancer” into the following databases: Human Genome Epidemiology Navigator 142 , DrugBank 143 , Therapeutic Target Database 144 , Pharmacogenomics Knowledgebase 145 , DisGeNET 146 , Comparative Toxicogenomics Database 147 , Online Mendelian Inheritance in Man 148 , and GeneCards 149 .
Network Generation
A network contains nodes (which indicate herbal medicines, ingredients, genes/proteins, and pathways) and edges (which indicate node-to-node interactions) 150 . The degree of a node is the number of its edges 150 . H-I-T network contains edges connecting the medicines of FDY003 to their active constituents, and edges connecting the active ingredients to the OC-associated target interacting with them. The H-I-T-pathway (H-I-T-P) network additionally contains edges connecting the FDY003 targets to the OC-associated pathways in which they are involved. The PPI network contains edges connecting the individual FDY003 targets to their interacting partners, and information on the target-to-target interactions was investigated using STRING 151 . Network generation was conducted using Cytoscape 152 .
Survival Assessment of Patients with Ovarian Cancer According to the Expression of FDY003 Targets
The survival rates and survival periods of patients with OC according to the expression status of OC-related targets of FDY003 were investigated using the Kaplan–Meier Plotter 153 .
Assessment of Functional Enrichment of the Ovarian Cancer-Related Targets of FDY003
The OC-related targets of FDY003 were imported into g:Profiler 154 , a widely used web-based database for the investigation of functional enrichment, and gene ontology (GO) and pathway enrichment analysis results were obtained. Differences at a level of P < .05 were considered significant.
Molecular Binding Evaluation
We explored the molecular structures of the chemical ingredients and targets of FDY003 from the PubChem 137 and RCSB Protein Data Bank 155 , respectively. The binding affinities of the targets with their interacting ingredients were evaluated on the basis of molecular docking scores of individual chemical ingredient–target pairs using Autodock Vina 156 . Those with molecular docking scores ≤ -5.0 were regarded to exhibit potent binding affinities, as previously described 9 .
Statistical Analysis
Statistical analysis was conducted using GraphPad Prism software (GraphPad Software Inc.). The test methods and number of replicates are provided in the figure legends. P-values < .05 were considered significant.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221075432 - Supplemental material for A Network Pharmacology Study to Uncover the Mechanism of FDY003 for Ovarian Cancer Treatment
Supplemental material, sj-docx-1-npx-10.1177_1934578X221075432 for A Network Pharmacology Study to Uncover the Mechanism of FDY003 for Ovarian Cancer Treatment by Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon and Dae-Yeon Lee in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (grant number 2021R1F1A1049472).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Author Contributions
Conceptualization: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee
Methodology: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee
Data collection: Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, and Tae-Wook Kwon
Data analysis and investigation: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee
Writing: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee
All authors read and approved the final manuscript.
Data Availability Statement
All data either generated or analyzed during this study are included in this published article and its Supplemental materials file.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
