Abstract
Tyrosinase catalyzes hydroxylation of L-tyrosine and dehydrogenation of L-DOPA in the melanin biosynthesis pathway. Tyrosinase inhibitors have potential use as cosmetic whitening agents and for preventing seafood deterioration. In this report, tyrosinase inhibitors extracted from brown alga Colpomenia bullosa (Scytosiphonaceae, Scytosiphonales) were investigated. Inhibitory principles were isolated from the extract and identified as phlorotannins, phloroglucinol (
Tyrosinase (EC 1.14.18.1), a copper-containing oxidase, catalyzes hydroxylation of L-tyrosine to L-3,4-dihydroxyphenylalanine (L-DOPA) and dehydrogenation of L-DOPA to L-dopaquinone, which is further converted to melanin. 1 Tyrosinase is responsible for the first step in melanin production by organisms. This pigment is distributed in human skin, eyes and hair and protects the body from ultraviolet radiation and free radicals. 1 However, dysfunctional or uncontrolled tyrosinase activity is associated with risks such as hyperpigmentation, melanoma, Parkinson’s disease, and alkaptonuria. 2 Melanin production is also associated with the deterioration of crustaceans such as lobster, 3 with the resulting black-colored crustaceans having reduced market value. Thus, inhibiting tyrosinase activity can play an important role in protecting the deterioration of seafood.
Several natural and synthetic tyrosinase inhibitors are known. 4 Various inhibitors such as kojic acid, arbutin and ellagic acid are used as whitening agents in cosmetics. 4 There are numerous studies that have characterized tyrosinase inhibitors from marine organisms. Moreover, various phlorotannins derived from brown algae have been identified as tyrosinase inhibitors. 5 -11 Most of these phlorotannins have been obtained from Ecklonia and Agarum sp. belonging to Laminariales, Ishige sp. belonging to Ishigeales and Fucus sp. belonging to Fucales. During our investigation of tyrosinase inhibition, we have obtained tyrosinase inhibitors from algal extracts. 12 -14 In the present report, an extract of Colpomenia bullosa (C. bullosa) (Scytosiphonaceae, Scytosiphonales) showed relatively high inhibitory activity against tyrosinase when compared with that of other algal extracts. The inhibitory principles were isolated from the extract and identified as phlorotannins. The inhibitory activity of these previously unknown tyrosinase inhibitors was compared.
Results and Discussion
Dried methanol (MeOH) extracts from C. bullosa, which possesses tyrosinase inhibitory activity, was separated into ethyl acetate (EtOAc)- and n-butanol (n-BuOH)-soluble fractions. Analytical thin layer chromatography (TLC) analysis with detection using the 2,4-dimethoxybenzaldehyde (DMBA) test revealed the presence of several low molecular weight phlorotannins in both fractions.
15
Compounds
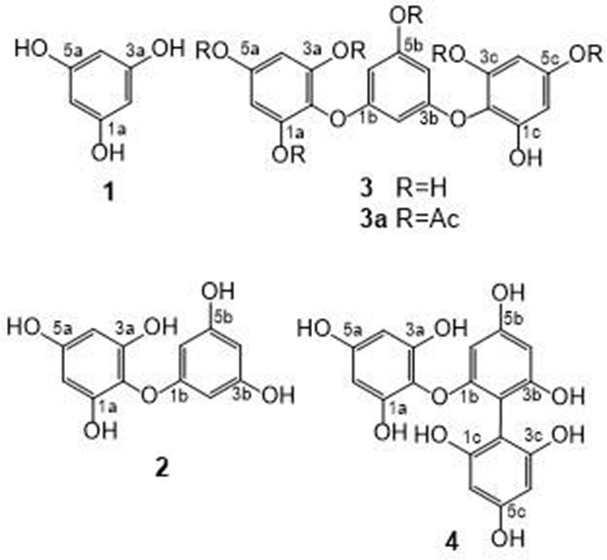
Phlorotannins isolated from C. bullosa.
The determined molecular weight (126) of compound
The determined molecular weight (250) and formula (C12H10O6) of compound

Possible isomers of the phloroglucinol dimer.

Inhibitory activity of phlorotannins 1
The determined molecular weight (374) and formula (C18H14O9) of compound
Compound
Compounds
The tyrosinase inhibitory activity of compounds
Inhibition Parameters of Phlorotannin 1

aL-Tyrosine concentration was 184 µM.
bNot tested.
cLiterature data. 21
Conclusions
Phlorotannins, phloroglucinol (
Experimental
Algal Material
Whole body of C. bullosa was collected at the coast of Hakodate, Hokkaido, Japan in April 2019. A voucher specimen (19US1B01) was deposited at Faculty of Fisheries Sciences, Hokkaido University.
General Procedures
NMR spectra were recorded on a JOEL JNM-ECZ400S NMR spectrometer (Tokyo, Japan) operating at 400 MHz (1H) and 100 MHz (13C). (HR-)FD-MS was measured on a JEOL JMS-T100GCV mass spectrometer (Tokyo, Japan). Column chromatography was performed on silica gel (Wakogel C-100, Kyoto, Japan) or microcrystalline cellulose (Avicel, Merck KGaA, Darmstadt, Germany). TLC was performed on pre-coated silica gel 60 F254 plates (Merck KGaA, Darmstadt, Germany). HPLC was carried out on a SHIMADZU LC-10ATVP apparatus (Kyoto, Japan) equipped with a SHIMADZU SPD-M10AVP photodiode array detector A Mightysil RP-18 HPLC column (5 µm, 250 × 10 mm, Kanto Chemical, Tokyo, Japan) was connected to the HPLC system. Mushroom tyrosinase (T3824) was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Extraction, Isolation and Identification of Phlorotannins
The whole body (402 g semi-air-dried weight) C. Bullosa was cut and extracted twice with MeOH (5 L) at room temperature for 3 days. The extracting solution was filtered through filter paper. The solvent was removed under reduced pressure to yield dark extracts. Combined MeOH extracts (9.60 g) were separated into an EtOAc-soluble fraction (1.10 g) and a n-BuOH-soluble fraction (1.02 g). Both fractions containing low-molecular phlorotannins were evaluated by analytical TLC with DMBA reagent. Both fractions were then combined, and silica gel column chromatography was conducted with a combination of n-hexane and acetone to elute several active fractions. The low-polar fraction was separated by preparative TLC developed with toluene/EtOAc/acetic acid (AcOH) = 2/3/1 (v/v/v) to obtain compound
Phloroglucinol (
Diphlorethol (
Triphlorethol C (
Hepta-O-acetyl triphlorethol C (
Fucophlorethol C
4
(
Tyrosinase Activity Assay
The tyrosinase assay was performed by the method of Islam et al. 13 with slight modifications. In brief, test compounds were dissolved in MeOH as the test solution. L-tyrosine was dissolved in 50 mM phosphate buffer (pH 6.8) to a concentration of 0.100 mg/mL (184 µM for inhibitor screening or measurement of IC50 values) or 0.100, 0.150 and 0.200 mg/mL (184, 276 and 368 µM for Lineweaver-Burk and Dixon plot analyses), as the substrate solution. Tyrosinase was dissolved in the phosphate buffer to a concentration of 200 units/mL as the enzyme solution. A mixture of test solution or MeOH as a control (15 µL), substrate solution (500 µL) and the buffer (780 µL) was preincubated at 25 °C for 5 minutes. The reaction was initiated by adding enzyme solution (205 µL). As soon as possible, the reaction mixture was transferred to a cuvette and the absorbance at 490 nm was measured as the blank. The remaining reaction mixture was kept at 25 °C for 30 minutes to allow the tyrosinase reaction to progress. The absorbance of the reaction mixture was then measured at 490 nm immediately. Values are shown as the means ± standard errors of means (SEMs) from three independent experiments.
Supplemental Material
Supplementary Material 1 - Supplemental material for Phlorotannins Derived From the Brown Alga Colpomenia bullosa as Tyrosinase Inhibitors
Supplemental material, Supplementary Material 1, for Phlorotannins Derived From the Brown Alga Colpomenia bullosa as Tyrosinase Inhibitors by Hideyuki Kurihara and Kazuki Kujira in Natural Product Communications
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
