Abstract
N ε-(Carboxymethyl)lysine (CML), which is formed by the glycation of collagen, is a skin-accumulating advanced glycation end product and has been shown to be deeply involved in diabetic osteopenia and skin aging. In this study, we prepared the phlorotannins of marine algal polyphenols from Japanese Lessoniaceae (Ecklonia cava, Ecklonia kurome, cultured E. kurome, Ecklonia stolonifera, Eisenia nipponica, and Eisenia bicyclis) and evaluated their inhibitory activities against CML formation in a collagen-glyoxal environment. The level of CML formed from the glycation of collagen by glyoxal was detected using an enzyme-linked immunosorbent assay. Except for E. stolonifera, the level of CML formation in the treatment with crude phlorotannins at 0.16 µg/mL was found to be comparable to that in the treatment with 0.40 mM aminoguanidine hydrochloride (AG) which is a typical antiglycation agent. In the test using phloroglucinol and isolated eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8’-bieckol) at a concentration of 0.80 µg/mL, the level of CML formed was lower for each compound, except for phlorofucofuroeckol A, than the data obtained with the addition of 2.0 mM AG. The mass concentration of 0.80 µg/mL was converted to 6.3 µM for phloroglucinol, 2.2 µM for eckol, 1.7 µM for fucofuroeckol A, 1.3 µM for phlorofucofuroeckol A, and 1.1 µM for dieckol and 8,8’-bieckol. From a comparison of the molar concentrations, it was found that phloroglucinol and the eckols inhibited the formation of CML resulting from glycation of collagen by glyoxal at concentrations of approximately 317 to 1818 times lower than AG.
Reducing sugars (eg, glucose and fructose) in the blood react nonenzymatically with lysine and arginine residues of proteins in the body, causing irreversible denaturation of the proteins. This reaction is called glycation of proteins, which leads to the production of advanced glycation end products (AGEs). 1 In previous studies, nonfluorescent compounds such as N ɛ-(carboxymethyl) lysine (CML) and N ω-(carboxymethyl) arginine, and fluorescent compounds such as pentosidine and pyropyridine have been identified as major structures of AGEs. CML, one of the most well-known AGEs, is formed by the direct reaction of glyoxal, which is formed by the oxidation of glucose, with the ε-amino group of lysine. 2 In addition, it has been found that CML was also formed from the oxidation of N ɛ-fructoselysine by hydroxyl radicals, 3 peroxynitrite derived from nitric oxide, 4 and hypochlorous acid 5 generated by inflammatory reactions. Collagen, a structural protein, is one of the proteins that partially constitute the dermis, tendons, bones, and organs in vertebrates. In the human body, collagen makes up about 30% of the total proteins. It has been shown that CML-collagen reduces the ability of keratinocytes in the epidermis to adhere to collagen 6 and to induce apoptosis in human dermal fibroblasts. 7 In addition, it has been pointed out that CML-collagen inhibits collagen cross-link formation via Discoylidine Domain Receptor 2 of lysyl oxidase in osteoblasts, resulting in diabetic osteopenia. 8 Thus, collagen-derived CML is deeply involved in osteoporosis in diabetic patients in addition to skin aging, so it is important to suppress its formation.
In the past, some antiglycation agents such as aminoguanidine hydrochloride (AG) 9 and thiazolidine derivative OPB-9195 (2-isopropylidenehydrazono-4-oxo-thiazolidin-5-ylacetanilide) 10 have been developed, but none of them have clinical applications because of their side effects (eg, anemia and hepatic injury). In previous studies, it has been shown that some extracts containing phenolic compounds from terrestrial plants demonstrate inhibitory activity against CML formation. Shin et al 11 have conducted experiments on the inhibition of CML formation using Silybum marianum flower extracts and isolated silibinin as an active substance. In addition, although the inhibitory activity has been reported on extracts of wild berries 12 and grape skins, 13 there are few findings on natural products that have an inhibiting effect on CML formation. Phlorotannins, polyphenols specific to brown algae, have a structure in which phloroglucinol (1,3,5-trihydroxybenzene) is polymerized. 14 Recently, phlorotannins have attracted attention as a new functional polyphenol because of their various physiological functions 15 including antioxidant properties. 16 -18 In preceding studies, we reported 2 findings regarding the physiological function of phlorotannins as antiglycation substances: (1) eckol and its oligomers had superior inhibition of fluorescent AGEs formation compared with AG in albumin-methylglyoxal 19 and albumin-glyceraldehyde environments 20 and (2) eckol has the activity of scavenging methylglyoxal, an α-dicarbonyl compound that is highly reactive with proteins, and its activity is superior to AG. 19 In this study, the inhibitory activities of phlorotannins on CML formation were evaluated by measuring the amount of CML produced from the glycation reaction of collagen and glyoxal using enzyme-linked immunosorbent assay (ELISA).
Results and Discussion
The usefulness of phlorotannins as inhibitors of CML formation was determined by comparison with the data of AG used as a positive control. Figure 1 shows the effects of crude phlorotannins on CML formation in a collagen-glyoxal environment. At a concentration of 0.16 µg/mL, the level of CML formed by glycation of collagen was significantly lower than control in all samples except for Ecklonia stolonifera (Figure 1). Furthermore, the level of CML formation in the treatment with crude phlorotannins was found to be comparable to that in the treatment with 0.40 mM AG. (Figure 1). This molar concentration of AG corresponds to a mass concentration of 44.0 µg/mL. Liu et al 21 reported on the antiglycation activities of mogroside extract prepared from Siraitia grosvenorii, a medicinal and edible plant. The report showed that mogroside extract at 500 µg/mL had the same degree of inhibition on CML formation as AG at 500 µg/mL. In the test using a bovine serum albumin-glucose environment, silibinin at 100 µg/mL has been shown to have a slightly better inhibition on CML formation than AG at 500 µg/mL. 11 The obtained data strongly suggest that phlorotannins may have an inhibitory activity on CML formation at extremely low concentrations compared with AG.

The effect of crude phlorotannins prepared from Lessoniaceae on N ɛ-(carboxymethyl)lysine formation in a collagen-glyoxal environment. Each crude phlorotannin sample was prepared at a concentration of 0.16 µg/mL and used for the enzyme-linked immunosorbent assay. The data are expressed as mean ± SE (n = 3). The mass concentration of 0.40 mM aminoguanidine hydrochloride (AG) is 44.0 µg/mL, that of 2.0 mM AG is 220 µg/mL, and that of 10 mM AG is 1.10 mg/mL. * P < .05, ** P < .005, *** P < .001, compared with control. a: Eisenia bicyclis (from Fukuoka), b: Eisenia nipponica (from Mie), c: Ecklonia cava (from Mie), d: Ecklonia kurome (from Fukuoka), e: E. kurome (from Kumamoto) f: cultured E. kurome (from Kumamoto), g: Ecklonia stolonifera (from Yamaguchi).
To further analyze the inhibitory activity of phlorotannins against CML formation in a collagen-glyoxal environment, a test was carried out using phloroglucinol and 5 kinds of isolated eckols (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8′-bieckol). Figure 2 shows the chemical structures of phloroglucinol and eckols used in the test. Except for fucofuroeckol A, these compounds are commonly found in Japanese Lessoniaceae. Figure 3 shows the effects of phloroglucinol and isolated eckols on CML formation in a collagen-glyoxal environment. Each compound was prepared at concentrations of 0.16 µg/mL or 0.80 µg/mL. All compounds at the concentrations tested significantly reduced the level of CML formed by glycation of collagen compared with control (Figure 3). In the test at a concentration of 0.80 µg/mL, the level of CML formed was lower for all compounds, except for phlorofucofuroeckol A, than the data obtained with the addition of 2.0 mM AG (Figure 3). The mass concentration of 0.80 µg/mL was converted to 6.3 µM for phloroglucinol, 2.2 µM for eckol, 1.7 µM for fucofuroeckol A, 1.3 µM for phlorofucofuroeckol A, and 1.1 µM for dieckol and 8,8′-bieckol. From a comparison of the molar concentrations, it was found that phloroglucinol and the eckols inhibited the formation of CML resulting from glycation of collagen by glyoxal at concentrations of approximately 317 to 1818 times lower than AG.
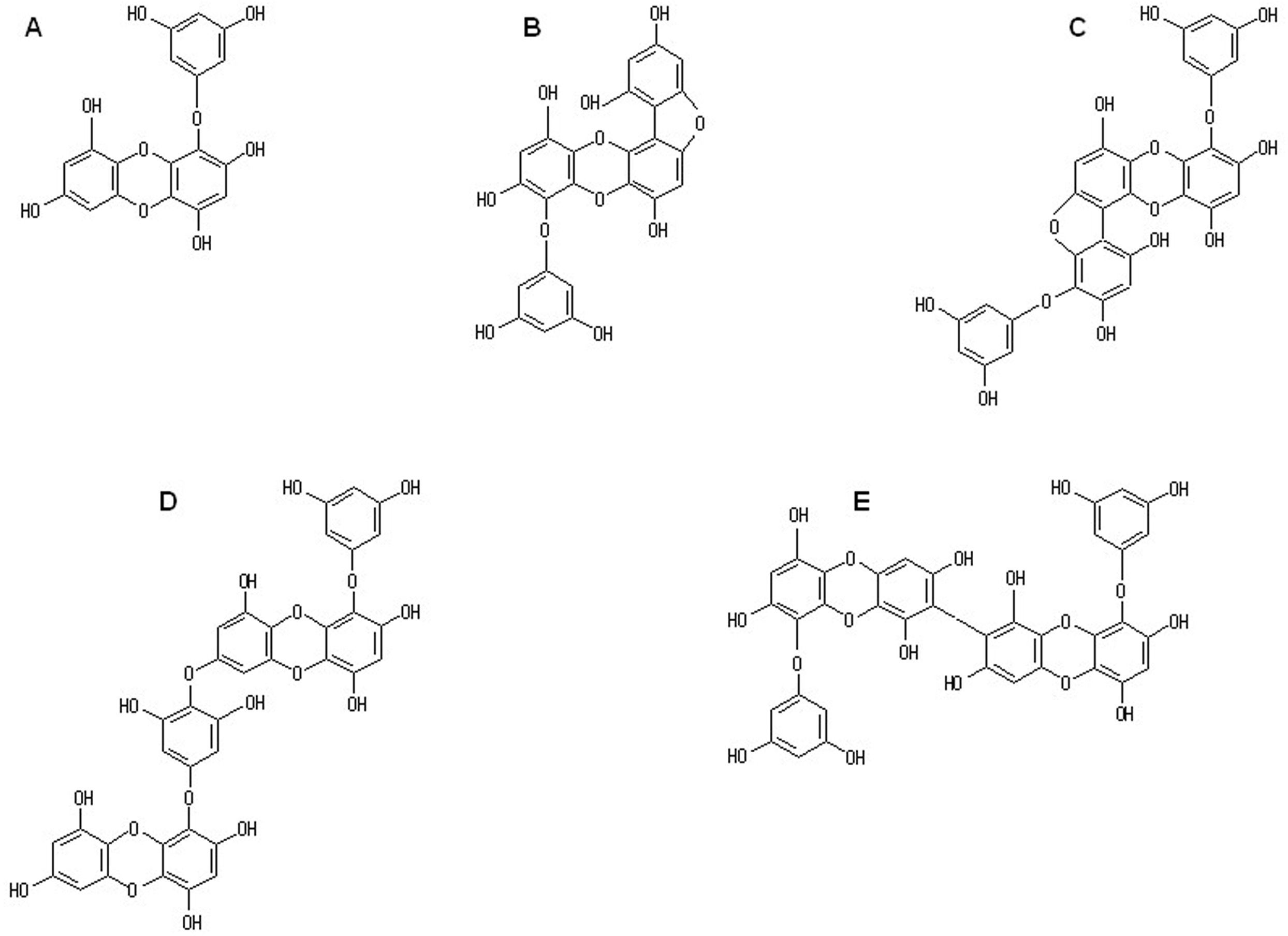
Chemical structure of eckols isolated from Eisenia bicyclis. (A) eckol, (B) fucofuroeckol A, (C) phlorofucofuroeckol A, (D) dieckol, and (E) 8,8′-bieckol.

The effect of phloroglucinol and isolated phlorotannins on N ɛ-(carboxymethyl)lysine formation in a collagen-glyoxal environment. Each compound was prepared at concentrations of 0.16 µg/mL and 0.80 µg/mL and used for the enzyme-linked immunosorbent assay. The molar concentrations converted for each compound are shown in the figure. The data are expressed as mean ± SE (n = 3). * P < .05, ** P < .01, *** P < .005, **** P < .001, compared with control.
To our knowledge, there is no report on a natural product having an inhibitory activity of CML formation exceeding that of AG, a typical antiglycation agent. In this study, it was found that phlorotannins had an excellent inhibitory effect on glyoxal-induced CML formation from collagen. Thomas et al 22 reported 2 things about the accumulation of CML in human cortical bone: (1) CML was present in bone in quantities 40-100 times greater than pentosidine, the fluorescent AGEs and (2) the amount of CML in bone increased with age and correlated to a decrease in crack propagation toughness. The Lessoniaceae used for the extraction of phlorotannins in this study has long been used as a food ingredient, mainly in Japan and Korea. In our previous study, it was confirmed that phlorotannins prepared from E. kurome do not exhibit biotoxicity on a single dose oral toxicity test in mice. 23 The beneficial effect of phlorotannins, in particular eckols, may be applicable to the prevention or management of diabetic osteopenia mediated by CML-collagen, in addition to skin aging.
Experimental
Algal Samples
The Lessoniaceae (Ecklonia cava Kjellman, Ecklonia kurome Okamura, cultured Ecklonia kurome, Ecklonia stolonifera Okamura, Eisenia nipponica, and Eisenia bicyclis Kjellman), used for the preparation of phlorotannins, was identical to the samples used in the previous reports. 19,20 Each of the air-dried algae were pulverized via a pulverizing mill (ABS-W, Osaka Chemical) and stored at −30 °C until use.
Preparation of Phlorotannins
Extraction of crude phlorotannins from the algal powder was performed according to the method described in the previous report. 18 Phlorotannins were isolated using silica gel column chromatography (1.5 cm i.d. × 150 cm) on a Wakogel C-300HG (Fujifilm Wako) and a preparative high-performance liquid chromatography (HPLC) system (Prominence, Shimadzu) with an Inertsil ODS-3 column (10 mm i.d. × 250 mm, GL Sciences) under the same conditions as previously reported. 18 Crude phlorotannins from E. bicyclis was used for the isolation of phlorotannins. Each of the isolated phlorotannins (eckol, fucofuroeckol A, phlorofucofuroeckol A, dieckol, and 8,8′-bieckol) was confirmed to have a purity of 98% or more by a 3-dimensional HPLC (SPD-M10AV, Shimadzu) with an Inertsil ODS-3 column (4.6 mm i.d. × 250 mm, GL Sciences). 18
Each amount of phlorotannin was completely dissolved in phosphate-buffered saline (PBS) containing dimethylsulfoxide at a final concentration of 0.001% and sterilized using a filter (polar diameter: 0.2 µm, Minisart, Sartorius) for sterilization. The sterilized solutions were then used as samples for the ELISA.
Measurement of CML Using ELISA Method
CML formed by the glycation reaction between collagen and glyoxal was detected using an ELISA kit (Cosmo Bio). To each well of a collagen-coated 96-well microplate, 50 µL of sample solution (0.16 µg/mL or 0.80 µg/mL) or AG (Fujifilm Wako) solution as a positive control was added. Then, fifty microliters of 20 mM glyoxal (product maker) was added to the plate, sealed with a microplate seal, and incubated for 24 hours at 37 °C. After incubation, the wells were washed 3 times using PBS containing 0.05% Tween 20 (washing buffer), and 200 µL of 0.5% gelatin in 50 mM sodium carbonate buffer (pH 9.6) (blocking buffer) was added to each well and left to stand for 1 hour at room temperature. After standing, the wells were washed 3 times with the washing buffer, and 100 µL of primary antibody (anti-CML antibody) in the blocking buffer was added to each well, and the incubation was carried out for 1 hour at room temperature. The solution of the primary antibody was removed, and each well was washed 3 times with the washing buffer. One hundred microliters of secondary antibody (horseradish peroxidase-conjugated antimouse immunoglobulin G antibody) in the blocking buffer was added to each well and further incubated for 1 hour at room temperature. After removing the solution of the secondary antibody, the wells were washed 3 times using the washing buffer, and 100 µL of 1,2-phenylenediamine dihydrochloride was added to each well and allowed to react at room temperature for 20 minutes. Finally, the enzyme reaction was stopped by adding 100 µL of 1 M sulfuric acid to each well. The absorbance of each well was measured at a measurement wavelength of 450 nm using a microplate reader (Infinite F50, Tecan). For the control wells, PBS was used instead of sample solutions. All data are expressed as mean ± SE (n = 3). The differences between the control and samples were analyzed by t-test.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by an Adaptable and Seamless Technology transfer Program (A-STEP) through Target-driven R&D from the Japan Science and Technology Agency (JST).
