Abstract
A new β-carboline alkaloid, 10-hydroxy-capitelline (
Introduction
Hedyotis is a genus of the flowering plants belonging to the family Rubiaceae.
1
The Hedyotis plants are widely distributed in tropical and subtropical regions with about 500 different species.
1
The previous studies showed that they have diverse chemical constituents with mainly secondary metabolite classes of alkaloids,2-5 anthraquinones,5-8 flavonoids,5,6 lignans,
9
iridoids, and triterpenoids.5,6,10 The diversity in chemical composition has brought various important pharmacological activities such as antibacterial, antidiabetic, antioxidant, anti-inflammatory, and anticancer.6,11,12 In Vietnam, Hedyotis capitellata is a typical species of the genus Hedyotis and is a traditional medicine as diuretic, anti-toxicity, and anti-inflammatory for the stomach, tongue, and throat.
10
In this study, we reported the isolation and structure elucidation of a new β-carboline alkaloid and three known anthraquinones from the methanol extract of the aerial part of H capitellata. The inhibition of nitric oxide (NO) production in lipopolysaccharide-stimulated RAW 264.7 cells by compounds
Results and Discussion
Compound
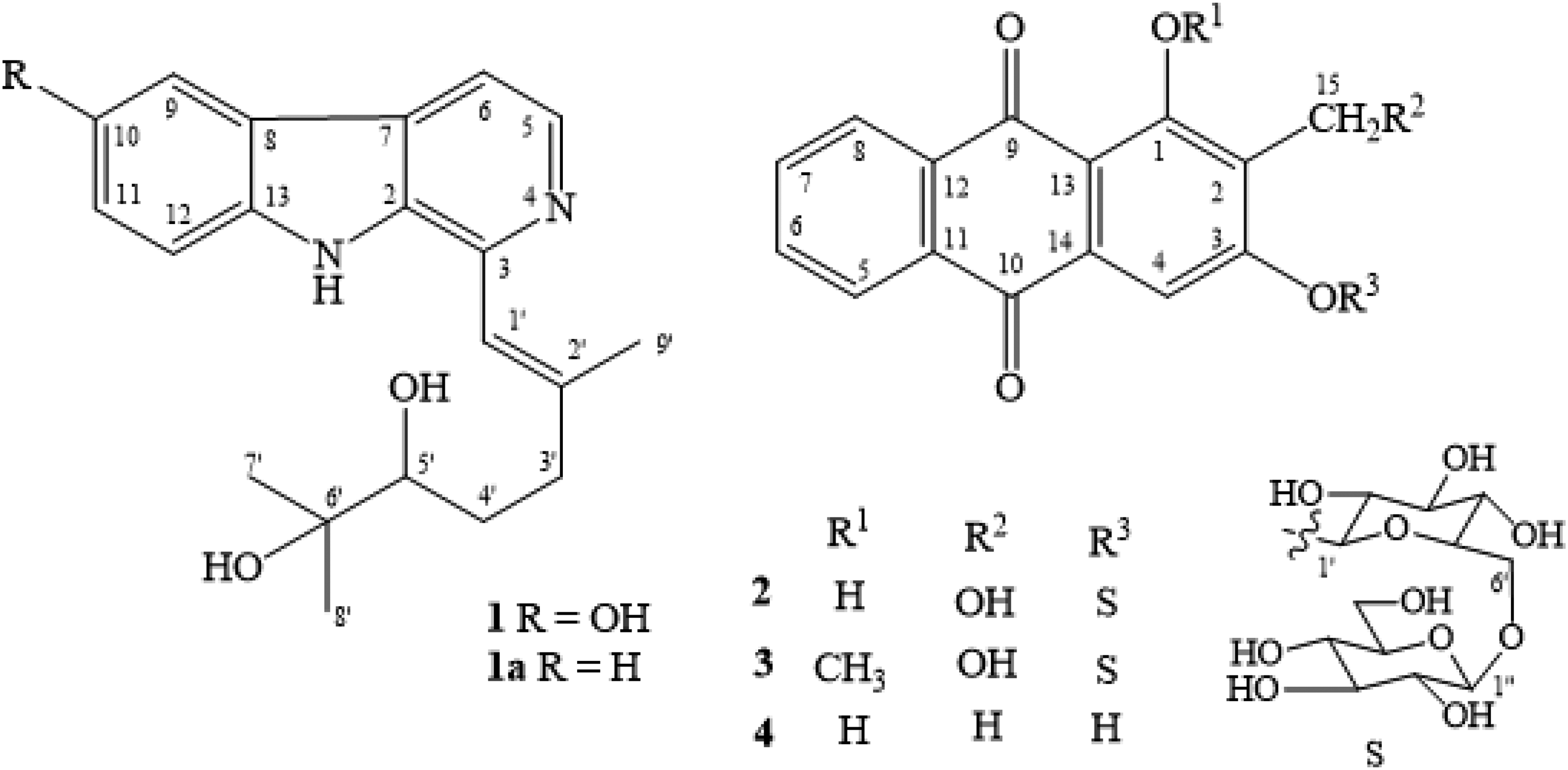
Chemical structures of compounds

Key HMBC and H-H COSY correlations of compound
1H NMR and
13
C NMR Data for Compounds

Abbreviations: 1H NMR, proton nuclear magnetic resonance; 13C NMR, carbon nuclear magnetic resonance.
Measured at 125 MHz.
Measured at 500 MHz.
Measured in deuterated methanol.
Measured in deuterated dimethyl sulfoxide. 4
Compounds
β-Carboline alkaloid and anthraquinone compounds have been known for many interesting biological activities including anti-inflammatory activity.6,11,12 So that, compounds
Material and Methods
General Experimental Procedures
Optical rotation was measured on a Jasco P-2000 polarimeter (589 nm). Circular dichroism (CD) spectra were recorded on a Chirascan CD spectrometer (Applied Photophysics). NMR spectra were studied on a Bruker 500 MHz spectrometer, and HR-ESI-MS on an Agilent 6530 Accurate Mass quadrupole time-of-flight liquid chromatography/mass spectrometry (Q-TOF LC/MS). Flash column chromatography was performed using either silica gel or reversed phase (RP-18, mesh) resins as adsorbent. Thin layer chromatography (TLC) was carried out on pre-coated silica gel 60 F254 and/or RP-18 F254S plates. TLC plates were visualized under ultraviolet irradiation (254 and 365 nm) or by spraying with H2SO4 solution (5%) followed by heating with a heat gun. High-performance liquid chromatography (HPLC) was carried out using an AGILENT 1100 HPLC system.
Plant Material
The plant H capitellata Wall. ex G. Don was collected in Nam Dinh in August 2018 and identified by Dr Nguyen The Cuong, Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology (VAST). A voucher specimen (HC08.2018-1) was deposited at the Lab of Pharmaceutical Chemistry, VNU University of Science, Hanoi.
Extraction and Isolation
The dried and powdered aerial parts of H capitellata (3.6 kg) were ultrasonically extracted with hot methanol at 50oC, (each batch: 0.6 kg, 2L MeOH, for 30 min, three times). After filtering, the filtrate was evaporated at 60oC to give the methanol extract (HC, 170 g). This extract was suspended in water (2 L) and successively partitioned with hexane, CH2Cl2, and EtOAc to obtain corresponding extracts HCH (26.8 g), HCD (1.9 g), HCE (19.9 g), and the water layer. The HCE fraction (19.5 g) was chromatographed on a silica gel column (500 g, mesh) eluting with CH2Cl2-EtOAc (1:1.5) to obtain five subfractions (HCE1-HCE5). The HCE4 (2.1 g) was chromatographed on a silica gel column (70 g, mesh) eluting with CH2Cl2–MeOH (7:1) to yield compound
10-Hydroxy-Capitelline (1 )
A yellowish-brown colloidal.
Nitric Oxide Assay
The detailed description of nitric oxide assay test is presented in Supplemental Material. 15
Conclusions
In summary, this report describes the isolation and structural elucidation of a new β-carboline alkaloid named 10-hydroxy-capitelline (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211047705 - Supplemental material for A New β-Carboline Alkaloid From the Aerial Part of Hedyotis capitellata
Supplemental material, sj-docx-1-npx-10.1177_1934578X211047705 for A New β-Carboline Alkaloid From the Aerial Part of Hedyotis capitellata by Le Thi Huyen, Nguyen Thi Thu Hau, Hoang Son Vu, Nguyen Thi Lan Huyen, Bui Huu Tai, Nguyen Xuan Nhiem and Phan Van Kiem in Natural Product Communications
Footnotes
Author Contributions
Isolation and research idea, LT Huyen, NTT Hau, VH Son, NTL Huyen; Structure elucidation and writing, BHTai, NX Nhiem, PV Kiem.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
