Abstract
Trifuhalol A, a phlorotannin, was extracted from Agarum cribrosum with ethyl acetate and fractionated using Sephadex LH-20 column chromatography (SF1-SF6). The ethyl acetate fraction (EAF) and SF5-containing trifuhalol A exhibited strong inhibitory activity against hyaluronidase. The anti-inflammatory activity of the phlorotannin, EAF, and SF5 was determined through the inhibition of nitric oxide (NO) production in lipopolysaccharide-stimulated RAW264.7 cells. Furthermore, the inhibition of NO production was validated by confirming the appreciable downregulation of inducible nitric oxide synthase expression. Agarum cribrosum phlorotannin also markedly suppressed the expression of cyclooxygenase-2, interleukin (IL)-1β, IL-6, and tumor necrosis factor-α. In addition, the anti-inflammatory action was verified by examining its effects on proinflammatory signaling pathways. The activation of nuclear factor (NF)-κB and mitogen-activated protein kinases (MAPKs) was attenuated via the inhibition of NF-κB p-65, c-Jun N-terminal kinase, extracellular signal-regulated kinase 1/2, and p38 MAPK phosphorylation. Therefore, trifuhalol A is a potential source for either the prevention or the treatment of inflammation.
Inflammation is normally a physiological response to harmful stimuli. Although the inflammatory response is protective, the excessive or prolonged production of inflammatory mediators can lead to human inflammatory pathologies, including arthritis, asthma, cancers, and cardiovascular diseases. 1 Lipopolysaccharide (LPS) is one of the most important bacterial components contributing to the induction of proinflammatory factors, as well as bacterial infections. 2 In the inflammatory process, LPS triggers macrophages to produce several inflammatory mediators, such as nitric oxide (NO), prostaglandin E2 (PGE2), proinflammatory cytokines, tumor necrosis factor-α (TNF-α), interleukin 1β (IL-1β), and IL-6. 3 However, these inflammatory mediators need to be tightly controlled to avoid excessive inflammation, tissue damage, and toxicity. The production of these mediators is primarily controlled at transcriptional levels. 3 At the cellular level, signal transduction leads to the activation of both mitogen-activated protein kinases (MAPKs) and the transcription factor nuclear factor (NF)-κB, thereby resulting in pro-inflammatory cytokine expression. 4 Therefore, substances to regulate the production of these inflammatory mediators are considered as potential anti-inflammatory agents.
Phlorotannins, major polyphenols of brown algae, are formed via polymerization of phloroglucinol (1,3,5-trihydroxybenzene) monomer units. 5 Among the brown algal phlorotannins, phlorofucofuroeckol A (PFF-A), PFF-B, dieckol, and 6,6′-bieckol are considered to be major anti-inflammatory components. 3 PFF-A inhibits LPS-stimulated inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) expressions in macrophages via the inhibition of NF-κB, Akt, and p38 MAPK. 6 Besides, 6,6′-bieckol exerts anti-inflammatory activity by downregulating iNOS, COX-2, and pro-inflammatory cytokines in LPS-stimulated macrophages through the NF-κB pathway. 7 Thus, phlorotannins have attracted much attention as anti-inflammatory agents. Phlorotannins are structurally diverse and more than 150 are known. 8 Agarum cribrosum Bory (Costariaceae) has an amphi-Pacific distribution in Korea, Japan, Russia, Alaska, British Columbia, and northern Washington. 9 Although, A. cribrosum is commonly found along the eastern coast of Korea, it has been poorly utilized for human consumption and for bio-industrial use. Moreover, little information exists about the extraction, purification, and biological activity of phenolic compounds of A. cribrosum. In a previous study, a phlorotannin extracted was identified as trifuhalol A. 10 In the present study, the hyaluronidase inhibitory and anti-inflammatory activities of trifuhalol A were determined using reverse transcription-polymerase chain reaction (RT-PCR) and western blot analyses. In addition, the specific signal pathways responsible for the inflammatory activity in LPS-stimulated RAW264.7 macrophage cells were investigated.
In the previous study, 10 A. cribrosum phlorotannin was efficiently extracted through ultrasound and purified using organic solvents and Sephadex LH-20 column chromatography, resulting in 6 subfractions (SF1-SF6), of which SF5, showing strong antioxidant and cosmeceutical activities, contained trifuhalol A (Figure 1). Therefore, SF5 was further studied for hyaluronidase inhibitory and anti-inflammatory activities using RAW264.7 cells through NF-kB and MAPK signaling pathways.

Structure of trifuhalol A.
Hyaluronidase depolymerizes hyaluronic acid, which induces pro-inflammatory immune responses. Thus, the inhibition of hyaluronidase activity may represent a useful method for protecting against allergy and inflammation. 11 The hyaluronidase inhibitory activities of A. cribrosum extract, solvent-partitioned fractions, and purified fraction (SF5) at different concentrations are shown in Figure 2. The A. cribrosum extract, ethyl acetate (EAF), and SF5 inhibited the hydrolysis of hyaluronic acid dose-dependently, in which the EAF and SF5 were the most effective. The IC50 values of the EAF, SF5, and disodium cromoglycate (DSCG) against hyaluronidase were 349.6 ± 7.8, 353.2 ± 9.2, and 535.2 ± 6.9 µg/mL, respectively. The inhibitory effect of EAF and SF5 was about 2 times stronger than that of DSCG, a positive control. However, SF5 with trifuhalol A (390 Da) exhibited lower hyaluronidase inhibitory activity than did PFF-A (602 Da) and 8,8'-bieckol (742 Da), 12 which was in agreement with the results of Shibata et al, 12 in which phlorotannins with higher molecular weight were more active than those with lower molecular weight.
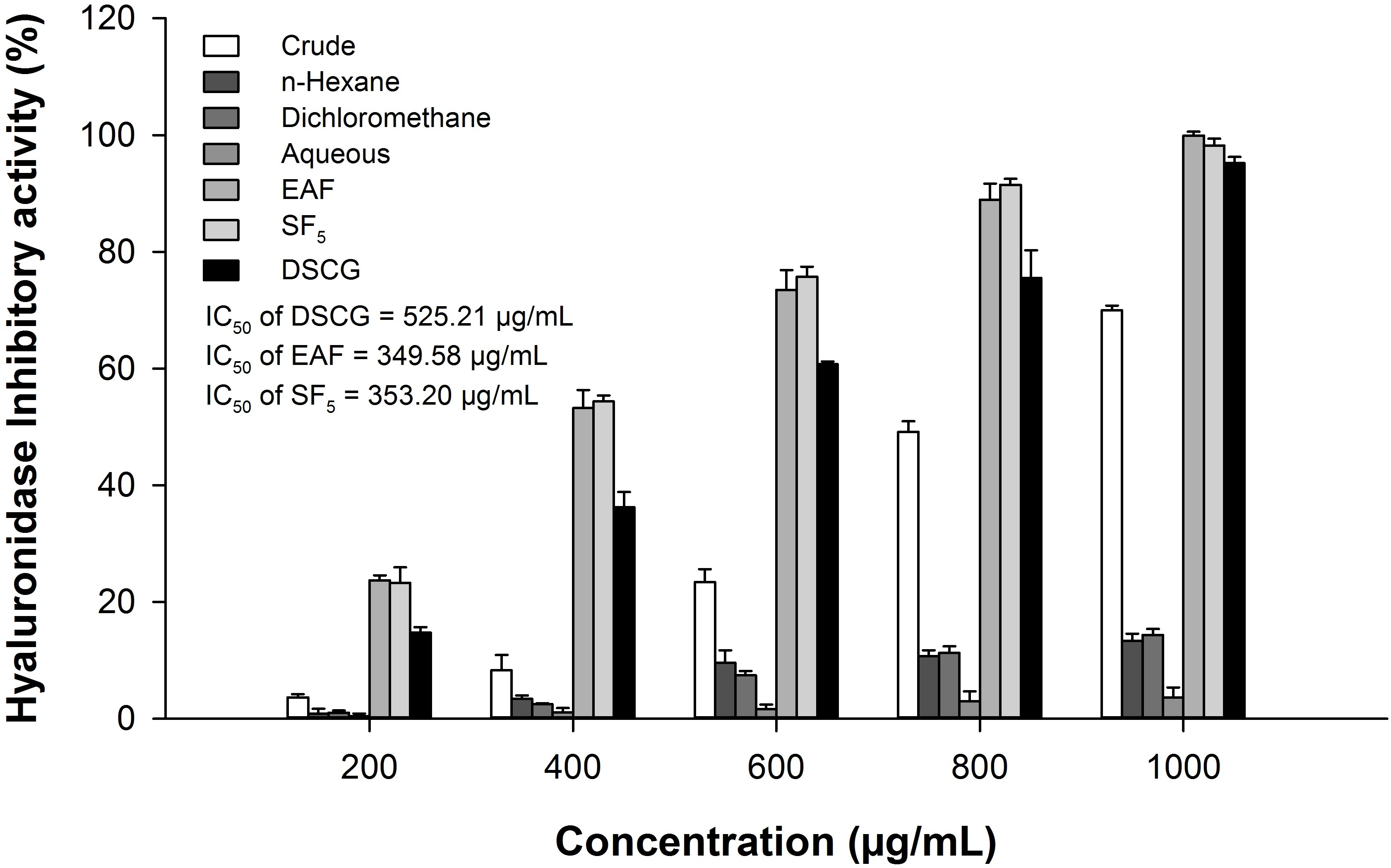
Hyaluronidase inhibitory activity of Agarum cribrosum extract, solvent-partitioned fractions, and SF5 at different concentrations. SF, subfraction.
Macrophages function as scavenger cells, immune mediator cells, as well as a source of chemotactic molecules and cytokines. 13 RAW264.7, a murine macrophage cell line, has been frequently used for screening anti-inflammatory compounds. Likewise, LPS has been frequently used for activating macrophages, which leads to the production of many inflammatory mediators. 3,14,15 In this study, the anti-inflammatory effect of A. cribrosum phlorotannin, EAF, and SF5 was analyzed using LPS-stimulated RAW264.7 cells because they were the most effective hyaluronidase inhibitors. The effects of EAF and SF5 on cell viability were evaluated at concentrations of 5 to 20 μg/mL (Figure 3a). EAF exhibited very low cytotoxicity (90% cell viability) at a concentration of 20 µg/mL, whereas SF5 did not affect the proliferation of RAW264.7 cells at concentrations up to 20 µg/mL. NO is a signaling molecule that plays a crucial role as an inflammatory mediator. Upon phagocytosis, macrophages produce NO, which is directly toxic to bacteria. 16 Furthermore, the overproduction of NO is involved in the pathogenesis of septic shock and several inflammatory disorders of the joint, gut, and lungs. 17 An increase in NO production is a typical phenomenon that occurs in LPS-stimulated macrophages and is used as an indicator of an inflammatory response. As shown in Figure 3(b), LPS treatment exhibited a significant increase in the amount of NO compared with the control treatments in RAW264.7 cells. Both EAF and SF5 treatment significantly reduced the amount of NO in a dose-dependent manner. Although the more prominent was shown by EAF when compare to that of SF5, EAF was more cytotoxic than SF5. EAF and SF5 decreased the NO production in LPS-stimulated RAW264.7 cells by 56.8% and 36.6% at 20 μg/mL, respectively. The inhibitory activity of EAF on NO production is higher compared with that of Eisenia bicyclis MeOH extract, in which NO production was inhibited by 45.3% at 400 µg/mL in LPS-stimulated RAW264.7 cells. 15 However, SF5 with trifuhalol A exhibited lower NO production inhibitory activity than did PFF-A and dieckol in LPS-stimulated RAW264.7 cells. 6

Effects of EAF and SF5 of Agarum cribrosum on (a) proliferation and (b) NO production in LPS-stimulated RAW264.7 cells. EAF, ethyl acetate; SF, subfraction; NO, nitric oxide; LPS, lipopolysaccharide.
The effects of EAF and SF5 on LPS-stimulated expression of proinflammatory mediators were further investigated in RAW264.7 cells. During the progression of an inflammatory process, LPS-activated macrophages express the inducible isoform of COX-2 and iNOS and produce excessive amounts of PGE2 and NO, which play key roles in the processes of inflammation. 18 As shown in Figure 4(a) and (b), LPS stimulation significantly increased iNOS and COX-2 expressions, whereas the EAF and SF5 treatments significantly decreased the inducible levels of iNOS and COX-2, which was in agreement with the results of Kim et al, 6 who found that PFF-A exerted anti-inflammatory activity by inhibiting iNOS and COX-2 expressions.

Effects of EAF and SF5 of Agarum cribrosum on the cytokines mRNA expression in LPS-stimulated RAW264.7 cells. (a-e) Graphic representation of the PCR product for RNA levels of different cytokines. (f) Expression of iNOS, COX-2, IL-6, IL-1β, and TNF-α assessed by RT-PCR. x,y,zSignificantly different (P < 0.05) compared with the EAF and SF5 at the same concentration. a,b,cSignificantly different (P < 0.05) compared with each concentration in the same sample. EAF, ethyl acetate; SF, subfraction; LPS, lipopolysaccharide; RT-PCR, reverse transcription-polymerase chain reaction; mRNA, messenger ribonucleic acid; iNOS, inducible nitric oxide synthase; COX, cyclooxygenase; IL, interleukin; TNF, tumor necrosis factor.
However, although EAF exhibited stronger inhibition of iNOS and COX-2 expressions, it also had a more cytotoxic effect than SF5. In addition, LPS also stimulates the release of proinflammatory cytokines such as TNF-α, which induces a number of physiological effects including septic shock, inflammation, and cytotoxicity; IL-1β, which is an important component in the initiation and enhancement of inflammatory response to microbial infection; and IL-6, which is a pivotal proinflammatory cytokine mainly synthesized by macrophages and which plays an important role in the acute phase immune response. 6 As shown in Figure 4(c-e), LPS stimulation significantly increased the expression levels of IL-6, IL-1β, and TNF-α, whereas the EAF and SF5 treatments significantly decreased the expression levels of these proinflammatory cytokines. Therefore, EAF and SF5 significantly inhibited IL-6, IL-1β, and TNF-α secretions in LPS stimulation, suggesting that they were potential inhibitors for the initial phase of inflammatory cascades under LPS stimulations.
NF-κB and MAPK play an important role in relaying external inflammatory signals to the nucleus and promoting the expression of proinflammatory genes. 19 LPS stimulation triggers signaling pathways leading to the phosphorylation and activation of three members of the MAPK family: c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase 1/2 (ERK1/2), and p38. 20 These signaling pathways in turn mediate multiple downstream events leading to the activation and translocation of NF-κB into the nucleus for transcriptional initiations. 6 The activated NF-κB acts as a transcription factor, leading to an increase in the expression of several inflammatory genes. 21 Therefore, the NF-κB and MAPK activations were determined in order to investigate the effect of EAF and SF5 on the activation of intracellular signaling protein in LPS-stimulated RAW264.7 cells. As shown in Figure 5, LPS treatment induced a significant increase in the expression of phosphorylated ERK1/2, JNK, p38 MAPK, and NF-κB p-65. In contrast, EAF and SF5 inhibited the phosphorylation of NF-κB p-65 in a dose-dependent manner. As in the MAPK pathway, EAF and SF5 downregulated the phosphorylation of JNK, ERK1/2, and p38 MAPK. Therefore, EAF and SF5 inhibited the inflammation through NF-κB and MAPK pathways in LPS-stimulated RAW267.4 cells.

Effects of EAF and SF5 of Agarum cribrosum on the activation of NF-κB and MAPK phosphorylation in LPS-stimulated RAW264.7 cells. The expression of NF-κB and phosphorylation of ERK, JNK, and p-38 detected by western blot of (a) EAF and (c) SF5. Graphic representation of the expression for the phosphorylation of NF-κB, ERK, JNK, and p-38 of (b) EAF and (d) SF5. The letters a, b, c, and d indicate a significant difference (P < 0.05) between the treatments. EAF, ethyl acetate; SF, subfraction; LPS, lipopolysaccharide; NF-κB, nuclear factor-κB; MAPK , mitogen-activated protein kinase; ERK, extracellular signal-regulated kinase; JNK, c-Jun N-terminal kinase.
In summary, trifuhalol A possesses anti-inflammatory activity sufficient to inhibit hyaluronidase activity and produces nitric oxide (NO) and inflammatory mediators such as iNOS, IL-1β, IL-6, TNF-α, as well as COX-2 by downregulating their messenger ribonucleic acid (mRNA) expression through NF-κB and MAPK pathways, resulting in alleviation of the immune response. These results might be helpful to understand the anti-inflammatory mechanism of EAF and SF5 with trifuhalol A on immune cells. Therefore, A. cribrosum phlorotannin is a potential source for the treatment of inflammation.
Experimental
Chemicals and Reagents
Hyaluronic acid, hyaluronidase, DSCG, Griess reagent (modified), LPS, and Tween® 20 were obtained from Sigma-Aldrich (St Louis, MO, USA). The murine macrophage cell line, RAW264.7, was obtained from American Type Culture Collection (ATCC, Rockville, MD, USA). Roswell Park Memorial Institute (RPMI)−1640 medium was obtained from Capricorn Scientific GmbH (Ebsdorfergrund, Germany), fetal bovine serum (FBS) from Welgene Inc. (Daegu, Korea), trypsin and penicillin/streptomycin from Lonza (Walkersville, MD, USA), and the EZ-Cytox cell viability assay kit (high-sensitive water-soluble tetrazolium salt, WST-1) from Dogen (Seoul, Korea). The following antibodies were used in this study: phospho-p38 MAPK (Thr180/Tyr182) rabbit mAb, phospho-p44/42 MAPK (Erk1/2) (Thr202/Tyr204) rabbit mAb, phospho-SAPK/JNK (Thr183/Tyr185) (81E11) rabbit mAb, phospho-NF-κB p65 (Ser536) rabbit Ab (Cell Signaling Technology, Beverly, MA, USA), goat anti-rabbit IgG H&L (horseradish peroxidase), and anti-alpha tubulin (Abcam, Dawinbio Inc., Seoul, Korea). All other chemicals and reagents used were of analytical grade.
Sample Preparation
Agarum cribrosum was collected from rocky subtidal sites along the coast of Gangneung, Korea, in December 2015. Salt, sand, and epiphytes were removed with tap water. The samples were rinsed with distilled water and dried at room temperature in the dark for 5 days. The dried samples were ground and passed through a 300 µm sieve. The ground powder was stored at −20°C in airtight bags until further use.
Extraction and Fractionation of A. c ribrosum Phlorotannin
The phlorotannin was prepared according to the method of Phasanasophon and Kim. 10 Briefly, A. cribrosum powder was extracted with 70% acetone using ultrasound at a frequency of 35 kHz and an intensity of 56.58 W/cm2 for 5.75 hours at 30°C. Afterwards, the extract was fractioned with EAF and purified by Sephadex LH-20 column chromatography using stepwise gradient water/methanol (1/1 to 1/3 to 0/1) and methanol/acetone (5/1 to 3/1 to 1/1) solvent systems, yielding 6 subfractions (SF1-SF6). Only trifuhalol A was identified from SF5 using 1H NMR, 13C NMR, and liquid chromatography-mass spectroscopy analyses. 10 Therefore, SF5 with trifuhalol A (purity, 83%) was used as the purified phlorotannin in this study.
Hyaluronidase Inhibitory Activity
Hyaluronidase activity was determined according to the slightly modified method of Ferreres et al. 22 Briefly, 50 µL hyaluronic acid (5 mg/mL), 100 µL buffer (0.2 M sodium formate, 0.1 M NaCl, and 0.2 mg/mL bovine serum albumin, pH 3.68), and 200 µL distilled water were mixed and incubated at 37°C for 10 minutes, following which 50 µL sample and 50 µL hyaluronidase (600 U/mL prepared in 0.9% NaCl) were added. The solution was then incubated at 37°C for 40 minutes. The reaction was stopped by adding 25 µL 0.8 M disodium tetraborate and subsequent heating in a boiling water bath for 3 minutes. The reaction mixtures were cooled to room temperature and 750 µL p-dimethylaminobenzaldehyde (DMAB) solution was added (2 g of DMAB dissolved in a mixture of 2.5 mL of 10 N HCl and 17.5 mL of glacial acetic acid and further diluted 1:2 with glacial acetic acid immediately before use), and then incubated at 37°C for 20 minutes. The absorbance of the reaction mixture at 585 nm was measured using a UV-VIS spectrophotometer (V-730; Jasco, Tokyo, Japan), with DSCG acting as a positive control. The percentage inhibition of hyaluronidase activity was calculated using the equation: % Inhibition = [[(A–B)–(C –D)]/(A–B)] ×100, where A, B, C, and D represent absorbance of the control, control blank, sample, and sample blank, respectively.
Cell Culture
RAW267.4 cells were cultured in RPMI medium supplemented with 10% FBS and 1% penicillin/streptomycin (100 U/mL) at 37°C in a humidified atmosphere containing 5% CO2.
Inhibition of Cellular NO Production and Cell Proliferation
RAW264.7 cells (100 µL, 1.0 × 106 cells/well) were seeded into a 96-well plate, pretreated with the sample (100 µL) for 1 hour, stimulated with LPS (100 µL, 1 µg/mL), and then incubated at 37°C in the presence of 5% CO2 for 24 hours. The anti-inflammatory activity of the sample was determined on the basis of NO production in the macrophage culture supernatant, in which the nitrite concentration was measured using the Griess reaction assay. 23 After incubation, each 100 µL of the supernatants and Griess reagent were mixed and then incubated at room temperature for 10 minutes. The absorbance of the mixture at 540 nm was measured using a microplate reader (EL-800; BioTek Instruments, Winooski, VT, USA). The proliferation of cells was determined using the WST-1 colorimetric assay as described by Qi and Kim. 24 The optical density at 450 nm was measured using a microplate reader (EL-800; BioTek Instruments). The absorbance was translated into proliferation (%) = A t/A c × 100, where A t and A c represent absorbance of the test and control groups, respectively.
Reverse Transcription-Polymerase Chain Reaction
RAW264.7 cells (500 µL, 1.0 × 106 cells/well) were seeded into a 24-well plate, pretreated with the sample (500 µL) for 1 hour, stimulated with LPS (500 µL, 1 µg/mL), and then incubated at 37°C in the presence of 5% CO2. Total RNA was isolated using the TRIzol reagent (Invitrogen Co., Carlsbad, CA, USA). The concentration of total RNA was quantified using a nanophotometer (P330; Implen GmbH, Munich, Germany). The first-strand complementary deoxyribonucleic acid (cDNA) was converted using an oligo-(dT) 20 primer and Superscript III reverse transcriptase (Invitrogen). The resulting cDNA was amplified by PCR using GoTaq Flexi DNA polymerase (Promega, Madison, WI, USA) with an initial denaturation at 95°C for 5 minutes, followed by 35 cycles of denaturation (95°C for 30 seconds), annealing (60°C for 20 seconds), extension (72°C for 30 seconds), and a final extension step at 72°C for 5 minutes. PCR amplification of cDNA was carried out with specific primers for iNOS, COX-2, IL-1β, IL-6, TNF-α, and β-actin (Table 1). After amplification, the RT-PCR products were electrophoresed on 1% agarose gels at 100 V for 25 minutes, visualized using GelRed staining, imaged and analyzed using a Gel Doc XR + Imaging System (BIO RAD, Jupiter, FL, USA).
The Sequences of Oligonucleotide Primers for RT-PCR.

RT-PCR, reverse transcription-polymerase chain reaction; IL, interleukin; iNOS, inducible nitric oxide synthase; COX, cyclooxygenase; TNF, tumor necrosis factor.
Western Blot Analysis
RAW264.7 cells (2 mL, 2.0 × 106 cells/well) were seeded into a 6-well plate, pretreated with the sample (2 mL) for 1 hour, stimulated with LPS (2 mL, 1 µg/mL), and then incubated at 37°C in the presence of 5% CO2. The cells were harvested, washed twice with ice-cold phosphate-buffered saline, and lysed in lysis buffer (1X RIPA, T&I, Seoul, Korea) containing protease and phosphatase inhibitor for 30 minutes with agitation. The lysates were then centrifuged at 15, 928 × g at 4°C for 15 minutes. Protein concentrations were determined using a Pierce BCA Protein Assay Kit (Thermo Scientific, Rockford, IL, USA). The protein equivalents of samples (30 µg protein/lane) were separated using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes. The membranes were blocked with 5% skim milk in TBST (Tris-buffered saline, 0.1% Tween 20) for 1 hour, incubated with specific primary antibody (anti-phopspho-p38, -ERK1/2, -JNK, -p65, or anti-α-tubulin) at 4°C overnight, and then incubated with goat anti-rabbit IgG secondary antibody conjugated to horseradish peroxidase at room temperature for 1 hour. Antibody-specific proteins were visualized using ECL advance Western blotting detection reagents (Thermo Scientific). The expression levels of each protein were imaged and analyzed using a ChemiDoc XRS + with Image Lab Software (BIO RAD).
Statistical Analysis
All experiments were performed in triplicates. Statistical comparisons between the different treatments were performed with one-way analysis of variance and Duncan’s multiple range test using SPSS software (v 17.0, SPSS Inc., Chicago, IL, USA), in which a probability value of P < 0.05 was considered to be statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was part of the project titled ‘Future Marine Technology Development’ funded by the Ministry of Oceans and Fisheries, Republic of Korea.
