Abstract
Chemical analysis of the aerial parts of Artemisia argyi H. Lév. & Vaniot led to the isolation of 6 lignans, including a new lignan glycoside, artemisiaside A, using various chromatographic techniques. Detailed spectroscopic (including 1D, 2D- nuclear magnetic resonance) and high resolution mass spectroscopy procedures, and electronic circular dichroism were used to ascertain the structural orientations of these compounds. The anti-inflammatory activities of compounds
Introduction
Compositae, the largest family of flowering plants, encompasses more than 1600 genera and 22,000 species, with a worldwide distribution in all continents except Antarctica.
1
This family has enormous economic benefits by providing food, cosmetics, and pharmaceuticals. Artemisia is a large genus in the Compositae family, with more than 350 species distributed across parts of the northern hemisphere temperate regions.
2
Artemisia species have been used to treat a variety of diseases, from simple fevers to malaria.3,4 Secondary metabolites of Artemisia species reported in the literature include phenolic compounds, alkaloids, terpenoids, sterols, and coumarins.
5
Especially, the discovery of artemisinin from Artemisia annua has brought more attention to this genus. Artemisia argyi, a perennial plant, known as wormwood in traditional Chinese medicines, is mainly found across parts of eastern Asia and Europe.
6
The stems and leaves of A argyi have a particularly strong fragrance. The whole plant was generally used as a Chinese traditional herb to treat warming meridians to dissipate cold, to stop bleeding, as an anti-inflammatory, to relieve cough-melt sputum, and so on. In addition, it can also be used as a dietary source for treating gastric mucosal injury or unhealthy eating habits.
7
Previous study has shown that the leaves and stems of A argyi mainly contain volatile oil, sesquiterpenoids, flavonoids, phenols, alkaloids, and sugars.
8
In our phytochemical investigation of the methanol extract of the aerial parts of A argyi, we report the isolation and structure determination of 3 lignans and 3 lignan glycosides, including 1 previously undescribed compound (
Result and Discussion
Compound
1H and 13C NMR Data of Compounds

The 1H and 13C NMR data of compound 1 were recorded at 600 MHz and 150 MHz, with CD3OD as solvent, respectively.
The 1H and 13C NMR data of the compound reported in the literature were recorded with a Bruker DRX-360, with (CD3)2CO as solvent. 9 .
data (Table 1) were reminiscent of 2-acetoxymethyl-1,2,3,4-tetrahydro-7-hydroxy-1-(4-hydroxy-3,5 -dimethoxyphenyl)-3-hydroxymethyl-6,8-dimethoxynaphthalene (compound

Chemical structures of compounds
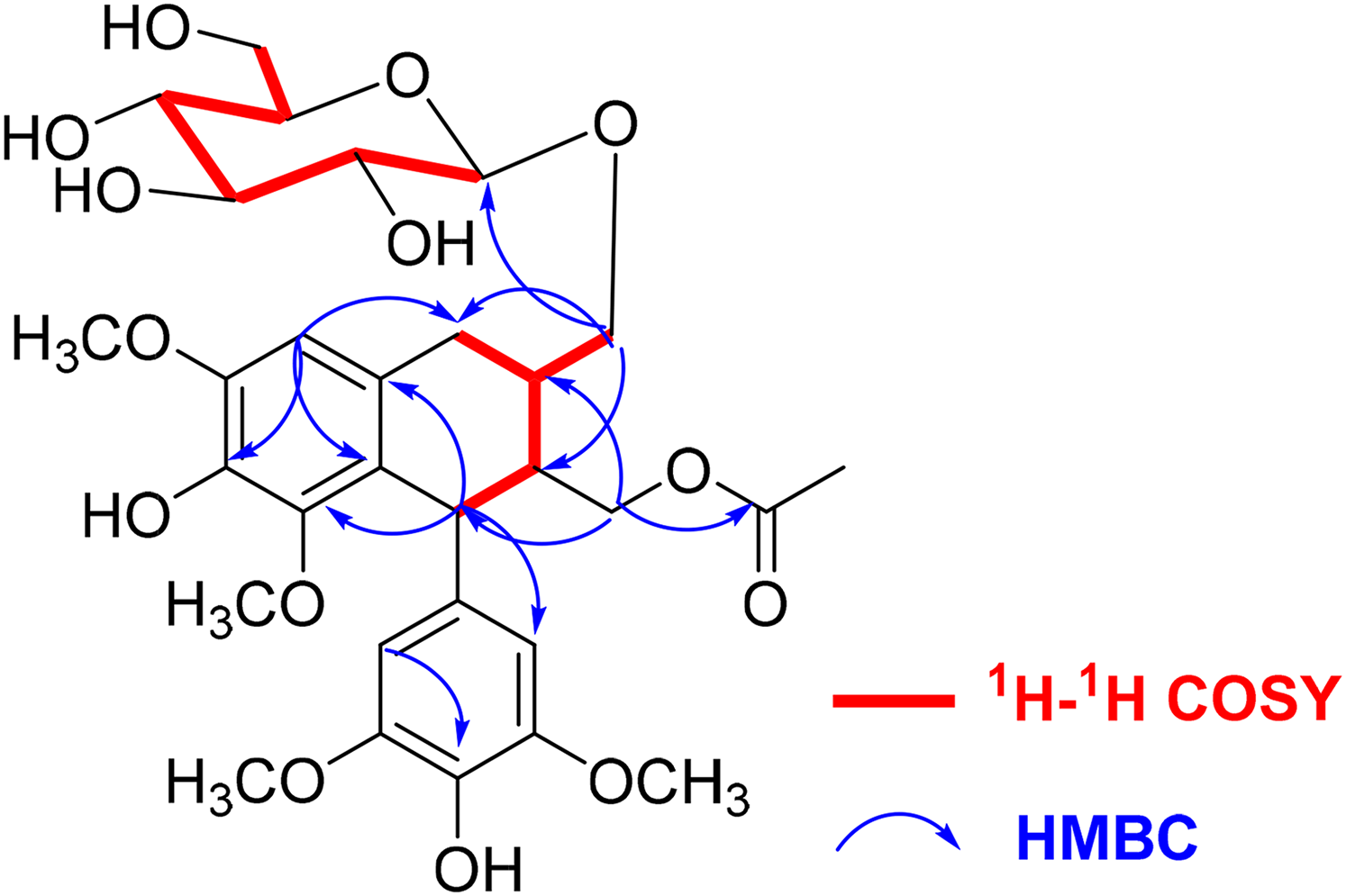
1H-1H COSY and key HMBC correlations of compound

Experimental ECD spectra of compound
By comparing their spectroscopic data with those reported in the literature, the other 5 known compounds
As a traditional (folk) medicine, A argyi exhibits multifarious bioactivities and is used to control dysmenorrhea, abdominal pain, and inflammation.
15
For instance, the essential oil from A argyi (AAEO) dose-dependently suppressed the release of pro-inflammatory mediators (nitric oxide [NO], PGE2, and ROS) and cytokines (TNF-α, IL-6, IFN-β, and MCP-1) in LPS-induced RAW264.7 macrophages.
16
As well as the crude extract, dehydromatricarin A was also identified as an active component of this plant against inflammation of the lung.
17
Moreover, several sesquiterpenoids obtained from A argyi have shown an enormous potential for inhibitory activity of NO production in vitro.18–20 Therefore, in order to explore and enrich the variety of compounds with anti-inflammatory activity from A argyi, the anti-inflammatory activities of compounds
Inhibitory Rate of Lipopolysaccharide (LPS)-Induced Nitric Oxide (NO) Production in RAW246.7 Cells Treated With Compounds

Conclusion
Six lignan analogues, including 1 new lignan glycoside, were obtained from the aerial parts of A argyi. Their structures were determined from extensive spectroscopic and mass spectrometric data and ECD. Compounds
Experimental
General Experimental Procedures
Column chromatography (CC) was performed on silica gel (200-300 mesh, Qingdao Marine Chemical Factory), Sephadex LH-20 (20-100 μm, Amersham Pharmacia Biotech), and MCI gel CHP-20P (70-150 μm, Mitsubishi Chemical Corp.). Standard thin-layer chromatography (TLC) was carried out on silica gel (GF254, 10-40 μm, Qingdao Marine Chemical Factory). Compounds were visualized on the TLC plates under UV light and by spraying with 5% H2SO4 in EtOH (v/v) followed by heating. HPLC analyses were performed on an Agilent 1260 HPLC system using an ODS column (C18, 250 × 4.6 mm, YMC Pak, 5 μm; detector: UV) and DAICEL chiral column (AGP 100.4 100 × 4.0) with a flow rate of 1.0 mL/min. Preparative HPLC was performed on an Agilent 1100 series with a Zorbax SB-C18 column (5 μm, 9.4 × 150 mm, Agilent) with a flow rate of 8.0 mL/min. NMR experiments were carried out using a cryogenic Bruker AV-600 spectrometer with TMS as internal standard. Mass spectra were measured on an Agilent G6230 spectrometer. UV spectra were obtained on either a Shimadzu 2700 or 2401 PC double-beam spectrophotometer. Optical rotations were measured with a Horiba SEPA-300 polarimeter. ECD experiment was conducted with an Agilent Applied Photophysics circular dichroism spectrometer.
Plant Material
The aerial parts of A argyi (Compositae) were collected from Xichang (GPS, 30o52′N/104o44′E) of Sichuan province, China, in July 2021 (wet season). The plant was identified by Prof. Qing-Shan Yang (Anhui University of Chinese Medicine), and an authentic sample (A20210708) is kept in the Laboratory of Research and Development of Medicinal Plants in the Panxi Region.
Extraction and Isolation
Air-dried and powdered aerial parts of A argyi (2 kg) were milled and soaked in methanol (3L) at room temperature for 48 h, and the extraction was conducted 3 times under the same conditions. The combined extract was evaporated to dryness under reduced pressure to yield an oily residue (106 g), which was further subjected to silica gel CC eluting with CH2Cl2 and then with CH2Cl2/MeOH stepwise-gradient (50:0, 25:1, 20:1, 15:1, 10:1, 5:1, 2:1, 0:1, v/v) to give 3 fractions (Frs. I-III).
Fr. I (15 g) was fractionated by silica gel CC with CH2Cl2/MeOH (from 50:1 to 20:1, v/v) as the eluent to give 3 subfractions (Frs. IA-IC). Fr. IC (500 mg) was applied to a silica gel column and eluted with CH2Cl2/MeOH (30:1, v/v) to afford 4 additional subfractions (Fr. IC-1-Fr. IC-4). Fr. IC-2 (108 mg) was passed through a Sephadex LH-20 column eluting with CH2Cl2/MeOH (1:1, v/v) to obtain 3 additional subfractions (Fr. IC-2a-Fr. IC-2C). Fr. IC-2b (30 mg) was separated on a Sephadex LH-20 column eluting with CH2Cl2/MeOH (1:1, v/v) and then subjected to semi-preparative RP-HPLC using MeCN-H2O (15:85-25:75, v/v) as the mobile phase to yield
Fr. III (12 g) was separated on a silica gel column eluting with CH2Cl2/MeOH (from 15:1 to 1:1, v/v) to afford 3 subfractions (Frs. IIIA-IIIC). Fr. IIIA (3 g) was subjected to silica gel CC with CH2Cl2/MeOH (5:1, v/v) as the eluent and then was chromatographed over a Sephadex LH-20 column eluting with CH2Cl2/MeOH (1:1, v/v) to obtain 3 additional subfractions (Fr. IIIA-1-Fr. IIIA-3). Fr. IIIA-2 (76 mg) was separated on a Sephadex LH-20 column eluting with CH2Cl2/MeOH (1:1, v/v) and then was applied to semi-preparative RP-HPLC using MeCN-H2O (5:95-20:80, v/v) as the mobile phase to yield
Artemisiaside A (1 )
Colorless amorphous powder, [α] D25 −54 (c 0.1, MeOH); UV (MeOH) λmax (log ε) 275 (3.54) nm; ECD (MeOH) λmax (Δε) 216 ( + 8.31), 244 (−3.68), 273 (−0.92), 287 ( + 0.86) nm; HRESIMS m/z 623.2341[M-H]− (calc. for C30H39O14−, m/z 623.2340); 1H and 13C NMR data; see
Aglycone (1c ) of Compound 1
Colorless amorphous powder, [α] D25 −4 (c 0.02, MeOH); ESIMS m/z 463 [M + H]+ (calc. for C24H31O9+, m/z 463), CD (c = 0.0667mg/mL, CH3OH) (Δε) 312 ( + 0.33), 299 ( + 0.33), 274 (−0.91), 265 (−0.36), 244 (−3.68), 225 ( + 2.92), 213 ( + 4.96).
Determination of Sugar Components
Compound
Measurement of Nitric Oxide Production
NO production was assayed in supernatants of cultured RAW264.7 cells using an NO assay kit (Beyotime Institute of Biotechnology). Cells were seeded in 96 well culture plates, and pretreated with the tested compounds (50 µM) and stimulated with LPS (1 μg/mL) for 24 h. The supernatant was mixed with an equal volume of Griess reagent (1% sulfanilamide, 0.1% naphthylethylenediamine dihydrochloride, and 2.5% phosphoric acid) and incubated at room temperature for 24 h. The concentration of nitrite was measured by reading the absorbance at 570 nm.21,22 L-NMMA was used as a positive control.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221118552 - Supplemental material for Artemisiaside A: A new Lignan Glycoside and its Analogues From the Aerial Parts of Artemisia argyi
Supplemental material, sj-docx-1-npx-10.1177_1934578X221118552 for Artemisiaside A: A new Lignan Glycoside and its Analogues From the Aerial Parts of Artemisia argyi by Chuan-Wang Tong, Ming Tao, En-Bo Zhang, Yi Huang, Hao Geng and Yang Yu in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank the members of the analytical center of Kunming Institute of Botany, Chinese Academy of Science, for the measurement of all spectra. This study was financially supported by the Natural Science Key Research Program of Anhui Province University (KJ2020A0913, KJ2020A0915), Foundation of Anhui Province Key Laboratory of Research & Development of Chinese Medicine (AKLPDCM202004), the Doctoral Scientific Research and Double High Talent Project of Xichang University (YBZ202114, LGLZ201811), and the Key Research and Development Program of Science and Technology Bureau of Liangshan Yi Autonomous Prefecture (21ZDYF0019). Computational resources used in this work were supported in part by SciGrid, Chinese Academy of Sciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Doctoral Scientific Research and Double High Talent Project of Xichang University, Key Research and Development Program of Science and Technology Bureau of Liangshan Yi Autonomous Prefecture, Natural Science Key Research Program of Anhui Province University, Foundation of Anhui Province Key Laboratory of Research & Development of Chinese Medicine (grant numbers YBZ202114, LGLZ201811, 21ZDYF0019, KJ2020A0913, KJ2020A0915, gxgnfx2022133 and AKLPDCM202004).
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
