Abstract
The volatile constituents, toxicity, antinociception, and anti-inflammatory activities of the essential oil obtained from the leaf of
Introduction
Traditional use of the plant as antivenom is prevalent among the Northern tribes of Nigeria. The methanolic extract of the seed successfully neutralizes the effect of
Phytochemical screening of
Materials and Methods
Collection of Leaf Sample
The leaves of
Preparation of Plant Sample
Prior to the hydrodistillation process, the plant samples were air-dried under laboratory shade for 2 weeks (at 25°C) to reduce the moisture contents. In addition, sediments and other unwanted materials were separated from the samples. Afterward, samples were pulverized to a coarse powder.
Hydrodistillation of the Essential Oil
In this process, 345.0 g of air-dried and pulverized leaves of
Chemical Analysis of the Essential Oil
GC analysis of the oils
GC analysis was accomplished with an HP-5890 Series II instrument equipped with an HP-Wax and HP-5 capillary columns (both 30 m × 0.25 mm, 0.25 µm film thickness), working with the following temperature program: 60°C for 10 minutes, rising at 5°C/minute to 220°C. The injector and detector temperatures were maintained at 250°C; carrier gas nitrogen (2 mL/minute); detector dual, flame ionization detector (FID); split ratio 1:30. The volume injected was 0.5 µL. The relative proportions of the oil constituents were percentages obtained by FID peak area normalization without the use of a response factor.
GC–MS analysis of the oils
GC-electrospray ionization-MS analysis was performed with a Varian CP-3800 gas-chromatograph equipped with an HP-5 capillary column (30 m × 0.25 mm; film thickness 0.25 µm) and a Varian Saturn 2000 ion trap mass detector. Analytical conditions: injector and transfer line temperature 220°C and 240°C, respectively; oven temperature programmed from 60 to 240°C at 3°C/minute; carrier gas helium at a flow rate of 1 mL/minute; injection volume 0.2 µL (10%
Identification of the constituents was based on a comparison of the retention times with those of authentic samples, comparing their linear indices relative to a series of
Drug and Chemicals
Carrageenan of analytical grade was obtained from Sigma-Aldrich Chemical Co. (St Louis, MO, USA; Batch Number: SLBR0530V). Piroxicam (May and Baker; Batch Number: MT2056) and ibuprofen injection (Dizpharm, Nigeria Ltd., Batch Number: 180606) were purchased from Juta Pharmacy, Lagos, Nigeria.
Animal Study
Wistar rats of about 150-200 g of Wistar rats of both sexes were provided by the Biochemistry Department animal unit and accommodated within the animal facility of Lagos state University, Ojo-Lagos. Animals were assigned at random to a group of 5 consisting of 6 animals per group and kept in a metal steel cage, with unlimited supply to water and standard pellet food. They were acclimatized for 2 weeks before the commencement of the experiment.
Group 1—ibuprofen/piroxicam-treated group 100 mg/kg (standard group); group 2—control group (saline solution, 0.9%); group 3—100 mg/kg of
An ethical clearance certificate was obtained from the Research Ethical Clearance Committee (RECC) of the University (approval no: 012/2019/LASU/BCH).
Carrageenan-Induced Paw Edema in Rats (Anti-Inflammatory Analysis)
Animals in each aforementioned groups were induced subcutaneously with 0.1 mL of 1% freshly prepared carrageenan in the right hind paw of rats treated by oral administration of vehicle normal saline (10 mL/kg; control), sodium diclofenac (10 mg/kg; positive control), and MPEO doses (100, 200 and 400 mg/kg) with normal saline as vehicle following Avoseh et al’s method with slight modification. 25,26 Paw volume of the injected rats was measured hourly on a plethysmometer (Ugo Basile, Italy mod. 7140) commencing 1 hour before (basal values) and up to 4 hours following carrageenan injection. Edema was calculated as the difference (L) between injected and control paw. The area under the curve (AUC) time versus change (Δ) in paw volume was calculated for each animal, and the edema was expressed as the mean ± standard error of the mean (SEM) of AUC. 27
Toxicity Assay
The essential oil was tested for acute toxicity study. Twenty-five Wistar rats (both sexes, 150-200 g each) divided into 5 animals into each group were used for the toxicity study. Wistar rats were administered 500, 1000, 1500, and 2000 mg/kg of the MPEO peroral route. One group received normal saline that served as a negative control. 23
Hot-Plate Test for Antinociceptive Study
The experiment was carried out according to the modified method.
25
Thirty mature Wistar rats (both sexes) were randomly divided into 5 groups of 6 rats per group. The animals were fasted for 12 hours with the provision of clean water ad libitum. Each rat was placed upon the heated metal plate (hot plate) maintained at the temperature of about 50-55°C
15
within the restraining glass cylinder. Group 1 rats received 10 mL/kg of saline solution and served as control. Group 2 rats received 10 mg/kg of piroxicam (standard control), and groups 3, 4, and 5 received 100, 200, and 400 mg/kg of
Formalin Test
This test was based on the method of Avoseh et al’s with slight modification. 28 Briefly, formalin solution (10 mg/L in normal saline solution 0.5 mL/paw) was injected into the hind paw plantar surface (intraplantar injection) of the grouped animals. The time (seconds) spent in intense licking or biting the affected paw was rated during 2 time intervals: 0-5 minutes (first phase or neurogenic pain) and 15-30 minutes (second phase or inflammatory pain). Oral treatment (p.o.), with piroxicam (10 mg/kg; positive control), vehicle normal saline (10 mL/kg), and MPEO (100, 200, 400 mg/kg), was given 60 minutes prior to formalin injection. Percentage inhibitions are evaluated using:

where
Statistical Analysis
Unless otherwise specified, data represented the mean ± EM of the evaluated parameter and were analyzed for statistical significance by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple for the formalin-induced assay. At the same time, 2-way ANOVA and post hoc Bonferroni tests were used for the carrageenan and the hot-plate test using GraphPad Prism (version 7.02; San Diego CA, USA, www.graphPad.com). The minimum level of significance considered was
Results and Discussion
The average yield of the light-yellowish essential oil was 0.2 (v/w), calculated on a dry weight basis. The chemical constituents present in oils were indicated by their percentages as well as linear retention indices on the HP-5 column. The GC/GC–MS of essential oils of
The essential oils of
Toxicity Effect
The acute toxicity of the essential oils evaluated at 500, 1000, 1500, and 2000 mg/kg body weight showed no contrary effects on the behavioral responses in the tested rats following 14 days of observation. No mortality, size, or weight change observed. Therefore, the highest dose of 400 mg/kg was adopted to be administered to rats in this study were considered safe. This result is consistent with the earlier report by Nweze et al
41
who reported no mortality or toxicity when 10, 100, 1000, 1600, 2900, and 5000 mg/kg body weight of
Antinociceptive Assay
This assay is to evaluate the analgesic ability of plant extracts. It also depicts the strength of the plant extract to act as an opioid antagonist. Opioid receptor antagonists block one or more of the opioid receptors in the central or peripheral nervous system. The three most clinically relevant opioid receptors are the µ, κ, and δ receptors. 42 In the hot-plate test, pain induced by the thermal stimulus is to evaluate the centrally mediated analgesia characteristics of opioid. The tests are objective, quantifiable, and can be administered repeatedly without causing inflammation, and assesses supraspinally organized responses to a noxious stimulus. 43 Figure 1 shows the effect of MPEO on the thermal stimuli of Wistar rats at different exposure times.
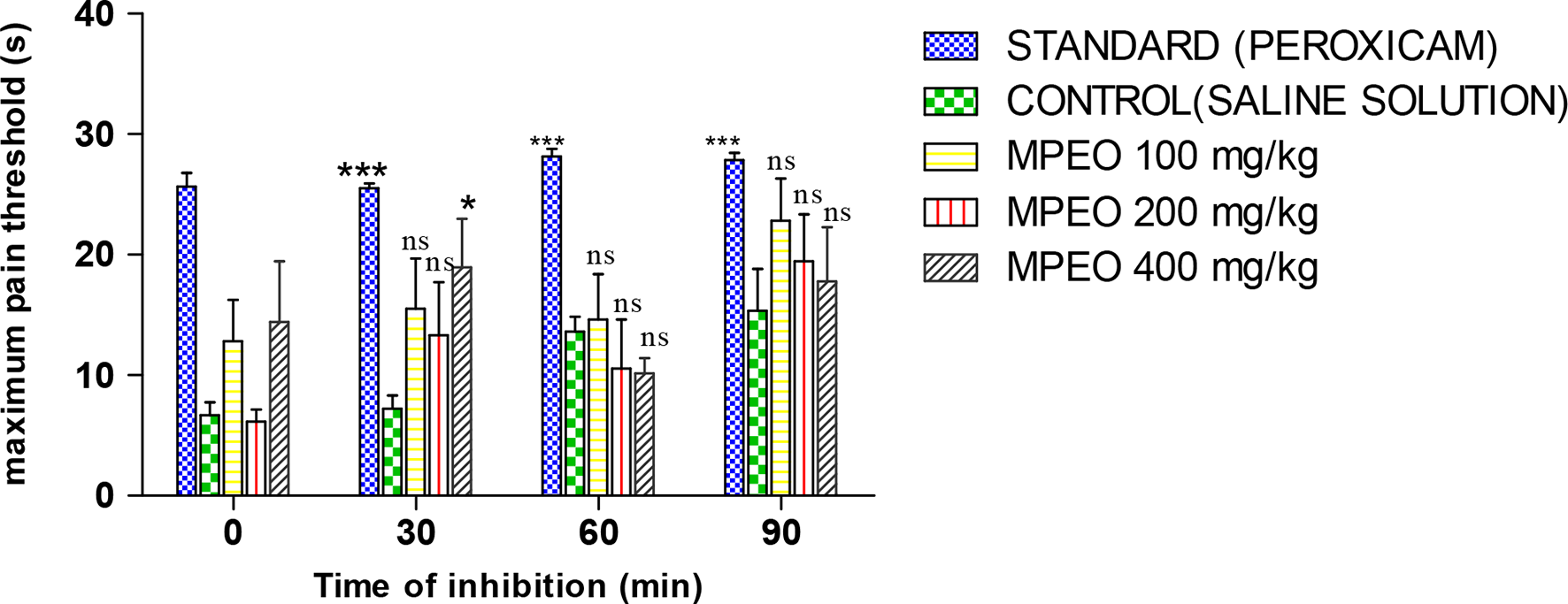
Effect of
The result shows that the time taken by the saline-treated Wistar rats for which it remained on the hot plate was significantly low compared with the standard (piroxicam). Whereas, when the rats were treated with MPEO, mice stayed on the hot plate significantly longer than the control rats (
Formalin-Induced Assay
The formalin test for nociception, usually used with rats and mice, involves moderate, continuous pain generated by injured tissue. This test is a handy tool for assessing the antinociceptive properties of drugs and for elucidating the action mechanism. The responses to formalin-induced pain, such as licking and biting of the injected paw, are biphasic. The first (early) phase is caused predominantly by C-fiber activation reflecting centrally mediated pain with the release of substance P. On the other hand, the second (late) phase depend on a combination of ongoing inputs from nociceptive afferents, due to the release of excitatory amino acids, prostaglandin E2, nitric oxide (NO), and other inflammation mediators. 48 Therefore, it allows analysis of drug actions relevant to acute and toxic pain during one test. Reports have shown that drugs that act mainly centrally, such as opioids and narcotics, inhibit both phases of formalin-induced pain while peripherally acting drugs, only restrain the late phase.
In the present study, the reactions of MPEO induced Wistar rats to formalin inhibition are shown in Table 2. At the early phase (neurogenic), the essential oils show a low to moderate inhibition averaged between 31.9% and 54.6%, which is relatively small. The highest peak is by the 400 mg/kg essential oils dose with an inhibition rate of 54.6%. The reduced activities recorded are in agreement with the hot-plate model earlier reported in this study. At the latter phase (anti-inflammatory), the essential oils significantly inhibited the release of inflammation mediators at a maximal value for all the doses >95.6%. Since essential oil of
The Statistical Result of the Formalin-Induced Assay of MPEO on Wistar Rats.
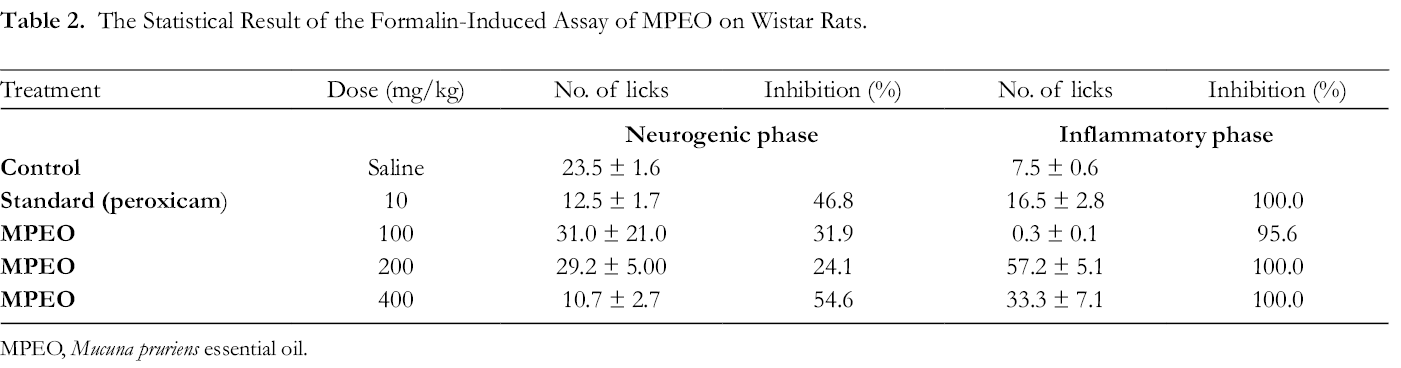
MPEO,
Anti-Inflammatory Assay
To evaluate the anti-inflammatory activity of MPEO, the carrageenan-induced model was adopted. Carrageenan is large, highly flexible molecules that form curling helical structures that are used as phlogistic agents for the release of inflammation and proinflammatory mediators such as prostaglandins, leukotrienes, histamine, cytokines, etc. It is a well-defined model for acute inflammation, and it is used in the study of the antiedematous effect of extracts due to the production of different inflammatory mediators in the Wistar rat. The model is time-dependent, characterized by the biphasic release of mediators. The initial phase involves the release of mediators such as histamine, serotonin, and bradykinin; it lasts within the first 1 hour, while 2-4 (latter period) is characterized by infiltration of leukocytes and prostaglandins biosynthesis. 51
In this study, MPEO significantly inhibits the development of edema at all stages of exposure except for the 400 mg/kg extract, as shown in Figure 2. The results show that MPEO exhibit maximum activity within the 1-3 hours of administration, indicating its ability to suppress the expression of histamine, serotonin, and bradykinin (0-1 hour post-treatment), while the latter phase is suspected to be inhibition of leukocytes and elevated prostaglandins biosynthesis(second to fourth hour post-treatment). The reduction in activity at longer hours can be a result of the high absorption capacity of the oil by the macrophages because of its high volatility and its inhibitory capabilities.

Effect of
Ethanolic extract of
The activities of essential oils against inflammatory conditions is majorly attributed to the plant’s major constituents or in synergy with other constituents. The volatile oils of
Conclusion
Essential oils were successfully isolated from the leaves of
Footnotes
Acknowledgements
The authors would like to express their appreciation to the Department of Biochemistry, Lagos State University, for their technical assistance during the animal study and Prof. Guido for technical assistance in GC/MS analysis. The authors gratefully acknowledge the financial support received from the Office of Research Directorate, Vaal University of Technology, Vanderbijlpark, South Africa.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

