Abstract
An amino acid-directed strategy has been developed to explore the potential of marine fungi to produce alkaloids. The marine fungus Lecanicillium fusisporum was cultured in glucose-peptone-yeast (GPY) medium to which were added L-tryptophan, L-phenylalanine, L-threonine, D, L-methionine, L-serine, L-lysine and L-valine. A new indole alkaloid, lecasporinoid (
Introduction
Marine-derived fungi have become an important source of structurally novel and pharmacologically active metabolites. Among them, alkaloids are a class of marine fungal natural products that show unique promise in the development of new drug leads. Biogenetically, alkaloids are derived from an amino acid pathway. 1 Thus, an amino acid-directed strategy, that is adding various natural amino acids, such as L-phenylalanine, L-tryptophan, and L-valine into the regular culture medium, may induce the marine-derived fungi to produce alkaloids. For example, 23 indole alkaloids were obtained from the culture of the marine-derived fungus Fusarium sp. in glucose-peptone-yeast (GPY) medium supplied with L-tryptophan. 2 Among them, fusaindoterpene B, JBIR-03 and 1,2-bis(1H-indol-3-yl)ethane-1,2-dione showed inhibitory activity against the Zika virus (ZIKV). 2 Thirteen indole alkaloids, including two new compounds, were discovered from the marine-derived fungus Pseudallescheria boydii F44-1 in GPY medium with L-tryptophan, L-phenylalanine, L-methionine, and L-threonine. 3 3,3’-Cyclohexylidenebis(1H-indole) showed cytotoxic activity against various cancer cell lines. 3 Seventeen new fumiquinazoline alkaloids were isolated from the marine fungus Scedosporium apiospermum F41-1 by feeding various amino acids.4,5 Scedapin C and scequinadoline D displayed significant antiviral activity against hepatitis C. 4 Additionally, scequinadolines D, E, J and quinadoline A were found to strongly promote triglyceride accumulation in 3T3-L1 cells. Furthermore, qRT-PCR analysis suggested that scequinadoline D might be a potent antidiabetic agent by activating the PPARγ pathway. 5 These results demonstrate the great potential of alkaloid production in marine fungi.
Recently, a marine fungus Lecanicillium fusisporum was isolated from the inner tissue of a sea cucumber collected from Xisha Islands in China. To induce this fungal strain to produce alkaloids, L. fusisporum was fermented in GPY medium supplemented with L-tryptophan, L-phenylalanine, L-threonine, D, L-methionine, L-serine, L-lysine and L-valine. By tracking the aromatic 1H NMR signals at δH 6.50 − 8.50 in the EtOAc extract of the culture broth, one new indole alkaloid, lecasporinoid (
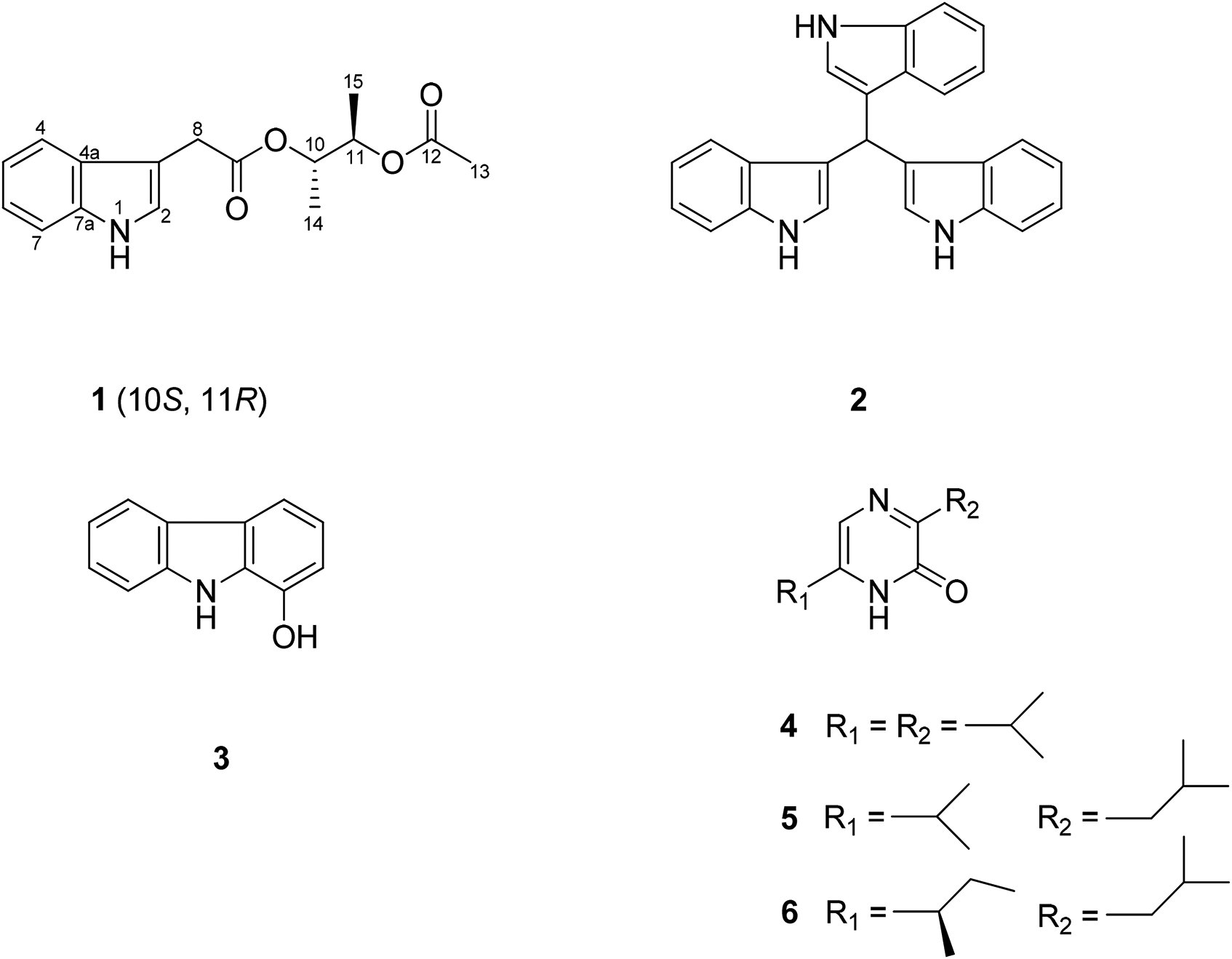
Chemical formulas of compounds
Results and Discussion
Structural Elucidation
Lecasporinoid (

Key 1H–1H COSY and HMBC correlations of compound
1H NMR (500 MHz) and 13C NMR (125 MHz) Data for

To determine the absolute configuration of

13C NMR calculation results for two possible isomers of
Finally, in order to validate further the calculation, we turned our attention to synthesize (10R,11R)-lecasporinoid (

The synthetic route of
1,1,1-Tris(3-indolyl)methane (
Anti-DENV Activity
To investigate the antidengue virus 2 activity of compounds
Experimental
General Experimental Procedures
1D and 2D NMR spectra were measured in CDCl3 using Bruker Avance II 400 and 500 spectrometers (Bruker Bio Spin AG, Industriestrasse 26, Fällanden, Switzerland), and the chemical shifts are given relative to the residual solvent signals (CDCl3: δH 7.26, δC 77.2). UV spectra were recorded using a Shimadzu UV-Vis-NIR spectrophotometer (Shimadzu Corporation, Nakagyo-ku, Kyoto, Japan), optical rotations (OR) were measured with an Anton Paar MCP500 polarimeter at 25 °C, and IR spectra were recorded on a Bruker tensor-27 spectrophotometer with KBr discs. Mass spectra were acquired on Thermo DSQ EI low-resolution and the Thermo MAT95XP EI high-resolution mass spectrometers (Thermo Fisher Scientific Inc., Waltham, MA, USA). Preparative HPLC was performed using a Shimadzu LC-20AT HPLC pump (Shimadzu Corporation, Nakagyo-ku, Kyoto, Japan) installed with an SPD-20A dual λ absorbance detector (Shimadzu Corporation, Nakagyo-ku, Kyoto, Japan) and a Shim-pack PRC-ODS HPLC column (250 × 20 mm, Shimadzu Corporation, Nakagyo-ku, Kyoto, Japan). Column chromatography was performed over silica gel (200 − 300 mesh, Qingdao Marine Chemical Inc., Qingdao, China) and Sephadex LH-20 (Green Herbs, Beijing, China).
Fungal Strain and Culture Method
The marine fungus Lecanicillium fusisporum was isolated from the inner tissue of a sea cucumber collected from Xisha Islands in China. The fungal strain was maintained on potato dextrose agar (PDA) slants. Analysis of the ITS rDNA by BLAST database screening provided a 99.9% match with Lecanicillium fusisporum. The fermentation medium contained glucose 10 g/L, peptone 5 g/L, yeast extract 2 g/L, sea salt 20 g/L, L-tryptophan 2 g/L, L-phenylalanine 2 g/L, L-threonine 2 g/L, D, L-methionine 2 g/L, L-serine 2 g/L, L-lysine 2 g/L, L-valine 2 g/L, and H2O 1 L (pH 7.5). The mycelia were aseptically transferred to 500 mL Erlenmeyer flasks containing 200 mL of the liquid medium, then sterilized at 120 °C for 30 min. The flasks were statically incubated at 28 °C for 40 days.
Extraction and Isolation
Fifty liters of culture broth were filtered through cheesecloth. The culture broth was extracted three times with 60 L EtOAc. The combined EtOAc extract was concentrated under reduced pressure to afford 17 g crude extract. This was chromatographed on a silica gel column (diameter: 8 cm, length: 80 cm, silica gel: 170 g) with a stepwise gradient of light petroleum–EtOAc (100:0–0:100), followed by EtOAc–MeOH (100:0–0:100). The eluents were collected every 400 mL, and similar fractions were pooled to afford six fractions (code Fr.1–Fr.6) by TLC monitoring. Fr.3 was further chromatographed using Sephadex LH-20 (MeOH) to give three subfractions (Fr.3-1–Fr.3-3). Fr.3-2 was further purified by preparative HPLC (73% MeOH–H2O) to yield compounds
Procedure for the Preparation of Compounds 7 and 1a
(2R,3R)-3-Hydroxybutan-2-yl-2-(1H-indol-3-yl)acetate (
(2R,3R)-Lecasporinoid (
Spectroscopic Data
Lecasporinoid (
Antiviral Activity Against DENV2
A549 cells were pretreated with compounds
Computational Section
The absolute configuration of compound
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211046072 - Supplemental material for Alkaloids From the Marine Fungus Lecanicillium fusisporum Using an Amino Acid-Directed Strategy
Supplemental material, sj-docx-1-npx-10.1177_1934578X211046072 for Alkaloids From the Marine Fungus Lecanicillium fusisporum Using an Amino Acid-Directed Strategy by Qin Huang, Hou-Jin Li, Chang-Bai Huang, Zi-Han Wang, Wen-Jian Lan and Lai-You Wang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Provincial Science and Technology Research Program (grant number Nos. 2015A020216007, Nos. 2016A020222004), and the National Science Foundation of China (grant number No. 81673689, No. 81872795).
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
