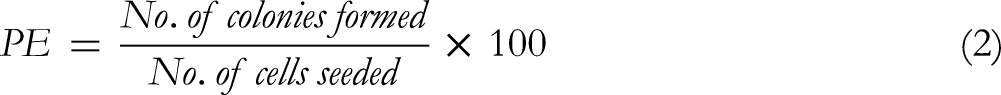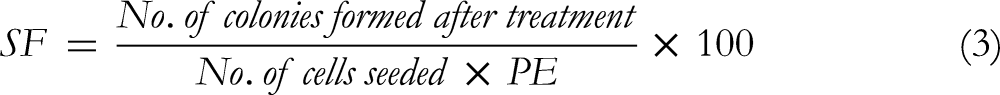Abstract
Introduction
Ovarian cancer, caused by malignant transformation of ovarian epithelial cells, ranks fifth among cancers commonly observed in females. Since this malignancy has nonspecific symptoms and no suitable screening method for timely detection, most cases are diagnosed in advanced stages with metastasis.1,2 Common therapeutic options are surgery, along with chemotherapy and radiotherapy. However, their clinical outcome is low and only 30% of all patients experience a full recovery.3,4 As a result, the search for effective treatment methods is still ongoing. Radiosensitizers are either natural or synthetic agents that enhance the effects of ionizing radiation and consequently, improve the life expectancy of cancer patients. Due to their high efficiency and low toxicity, the focus of numerous studies has shifted towards the use of natural products as radiosensitizers. 5 Galbanic acid (GBA) is a sesquiterpene coumarin with myriad therapeutical applications including antimicrobial, anticoagulant, antiviral, antileishmania, and anti-inflammatory properties. 6 In addition, anticancer effects of GBA have been reported in numerous studies. For instance, it has been shown that GBA suppressed tumor angiogenesis, exerted inhibitory effects on tumor cell proliferation, and induced apoptosis.7–9 So far, no study has been conducted to assess the possible synergy between GBA and irradiation, therefore, the present study aimed to examine whether GBA could induce radiosensitizing effects on an ovarian cancer cell line in vitro.
Materials and Methods
Cell Culture
The human ovarian carcinoma cell line, OVCAR-3, was obtained from the National Cell Bank of Iran (Pasture Institute). The cells, cultured in RPMI 1640 culture medium (Invitrogen) containing 10% fetal bovine serum (Gibco), 100 μg/mL streptomycin (Gibco), and 100 units/mL penicillin (Gibco), were incubated at 37°C in a humidified atmosphere of 95% air and 5% CO2.
Determination of GBA Cytotoxicity
GBA (Figure 1, Golexir pars, CAS number: 3566-55-0) was dissolved in dimethyl sulfoxide (DMSO) (0.4%) and final concentrations (5, 10, 20, and 40 μg/mL) were prepared using a culture medium. Half-maximal inhibitory concentration (IC50) of GBA on OVCAR-3 cells was determined after 24, 48, and 72 h by alamar Blue assay.

Chemical structure of galbanic acid (GBA).
For alamar Blue assay, a total of 6 × 103 cells/well were cultured in 96-well plates (SPL Lifescience). Then, the cells were treated with 0, 5, 10, 20, and 40 μg/mL GBA while the wells containing 0.4% DMSO were used as the solvent control group. Wells containing solely culture medium were utilized as blanks. Afterward, the cells were incubated at 37°C for 24, 48, and 72 h. At the end of each incubation time, 20 µL alamar Blue (Invitrogen) was added and the cells were incubated in the dark for 2 h. Then, optical density (OD) was recorded by a spectrophotometer (BioTek) at 600 nm, and cell viability was calculated using the following equation:
Treatment With GBA and Irradiation
OVCAR-3 cells were cultured overnight in T25 flasks and incubated in 5% CO2 at 37°C. Then, the cells were treated with 10 μg/mL GBA for 24 h. Flasks without GBA were also incubated for the same duration. Afterward, the medium of all flasks was removed and replaced with a fresh medium, and the cells were irradiated with 0, 1, 2, and 4 Gy 100 kV X-rays (Philips, serial number: 2/625). For irradiation, flasks were placed on a coach and surrounded with a 20 × 20 cm2 applicator attached to the X-ray tube. The dose rate was 1.307 Gy/min and the distance between the source and surface of media in the flasks was 30 cm. Flasks were divided into 4 groups based on their treatments: without any treatment (control group), treated with GBA (GBA), treated with irradiation (R), and treated with GBA and irradiation (GBA + R).
Clonogenic Assay
Twenty-four hours after irradiation, the cells were trypsinized and cultured in 6-well plates at different densities. Numbers varied according to different doses (700 for the control group, 700 for the cells treated with GBA, 1000 for 1 Gy, 1200 for 2 Gy, and 1400 for 4 Gy). Then, the cells were incubated at 37°C for 8 days in a 5% CO2 incubator. Lastly, colonies were fixed with 1 mL methanol (Merck), stained with 10% Giemsa (Merck), and then counted. The number of colonies in the control group was used to calculate the plating efficiency (PE) (equation (2)). Survival fraction (SF), as the rate of colonies formed after irradiation, was determined using the following equation
10
:
Determination of Synergistic Effects of GBA
Synergism occurs when the combined effect of 2 treatments exceeds the sum of their individual effects. To determine the synergy between GBA and irradiation, synergistic effect (SE) was calculated by equation (4), in which an SE higher than 1 indicates synergism.
Statistical Analysis
The IC50 value of GBA at different time points was calculated by Origin Pro2019b (9.65) software. All data were analyzed by SPSS, software version 16.0, using one-way ANOVA and Tukey tests to determine the significant difference between the groups. Values were expressed as mean ± SD, and a P value less than .05 was considered to be statistically significant.
Results
Toxicity of GBA
As illustrated in Figure 2, anti-proliferative effects and cytotoxicity of GBA on OVCAR-3 cells were assessed by alamar Blue assay. At all incubation periods (24, 48, and 72 h), the viability of the cells treated with 20 and 40 μg/mL GBA was significantly lower than that of the solvent and main control (without any treatments) groups (P < .001). However, no significant difference was observed between the cells receiving 5 or 10 μg/mL GBA and the solvent control group (P > .05), except for the group receiving 10 μg/mL GBA for 72 h (P < .05). Moreover, IC50 values of GBA after 24, 48, and 72 h were determined to be 15.4, 23, and 21 μg/mL, respectively.

Viability percentage of the cells treated with different concentrations of GBA for 24, 48, and 72 h (mean ± SD), * P < .05, ** P < .01, *** P < .001, **** P < .0001 indicate significant difference compared to DMSO group. Viability percentage of the groups is normalized to the viability of the main control group.
Treatment duration with GBA caused no impact on cell viability, as there was no significant difference between the groups receiving a particular concentration of GBA for 24 h or longer (P > .05).
Toxicity of Irradiation With and Without GBA
Considering the mentioned results above, GBA, at a concentration of 10 μg/mL, did not significantly decrease the survival rate of the cells. Besides, the duration of treatment with GBA had no impact on the survival rate of the cells. Therefore, 10 μg/mL GBA and a 24 h treatment time were chosen as the optimal concentration and period for the combinatorial treatment with GBA and irradiation. To evaluate their combined efficacy and compare it to irradiation alone, clonogenic assay was performed (Figure 3); Figure 4 highlights the morphology of the colonies generated in different groups; and Table 1 demonstrates the SF values after different treatments. As illustrated, survival of the cells co-treated with irradiation and GBA (R + GBA) was significantly lower than that of the cells treated with radiation alone (R). The calculated PE values (%) of the control groups for GBA + 1 Gy, +2 Gy, and +4 Gy were 41.1 ± 1.9, 42.6 ± 1.5, and 43.9 ± 6.2, respectively. SF values also confirm morphological observations. Of note, the effect of our combinatorial approach was most prominent at 1 Gy.
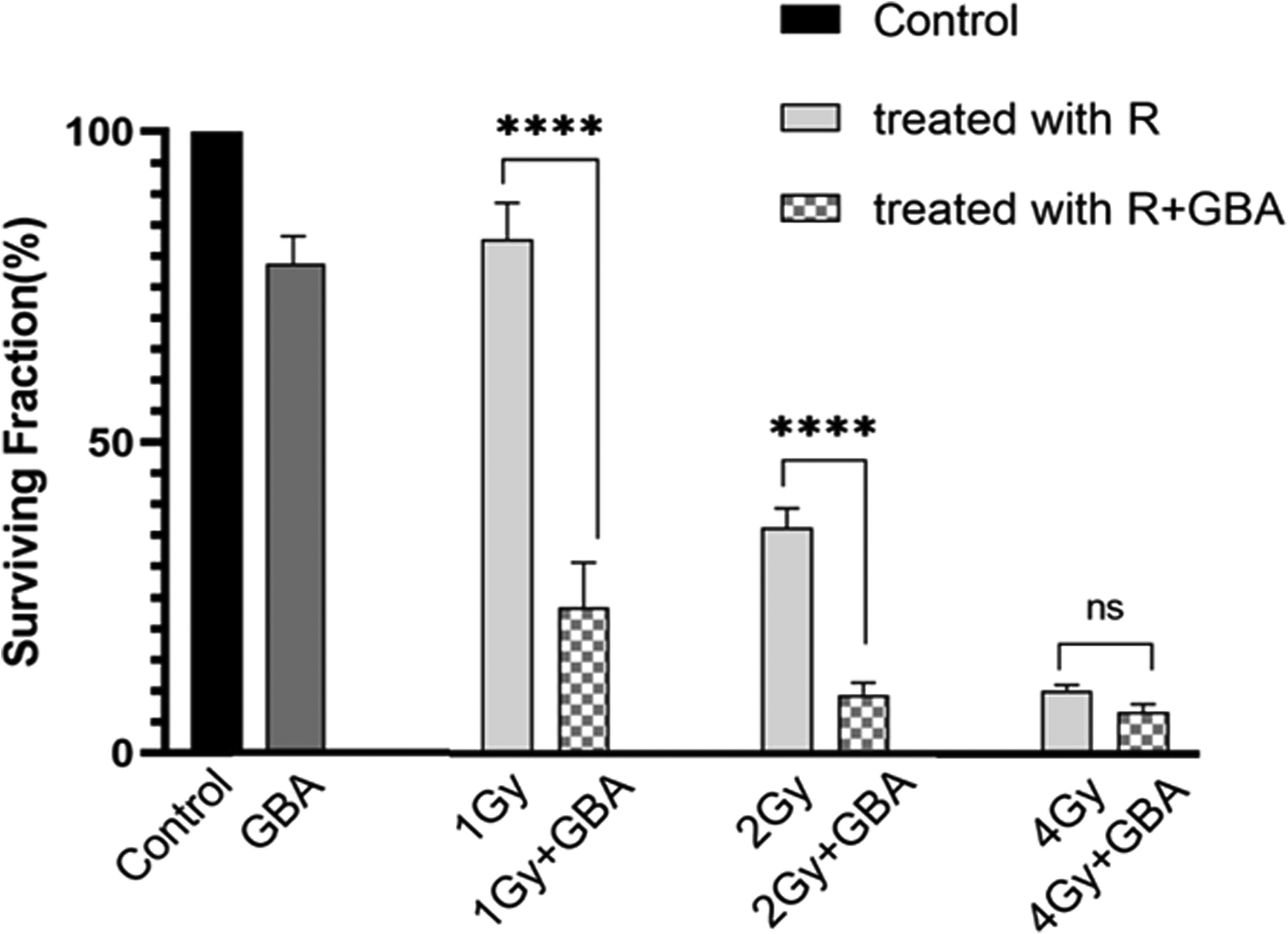
Survival fraction (mean ± SD) of different groups. **** P < .0001 indicates significant difference with control group. Survival fraction of the groups is normalized to the viability of the control group.

Photomicrographs of colonies after 8 days. (A) control group, (B) GBA at a concentration of 10 µg/mL, (C) OVCAR-3 cells treated with irradiation (1 Gy), (D) OVCAR-3 cells treated with a combination of GBA and irradiation (1 Gy), (E) OVCAR-3 cells treated with irradiation (2 Gy), (F) OVCAR-3 cells treated with a combination of GBA and irradiation (2 Gy), (G) OVCAR-3 cells treated with irradiation (4 Gy), (H) OVCAR-3 cells treated with a combination of GBA and irradiation (4 Gy).
SF Values (%) for Cells Treated With GBA and Irradiation, Alone and in Combination, at Different Doses (1, 2, and 4 Gy).
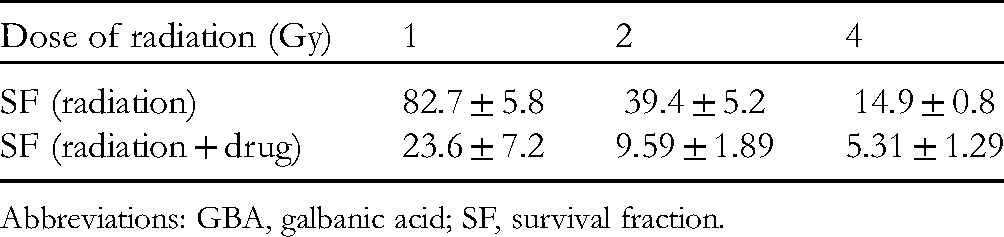
Abbreviations: GBA, galbanic acid; SF, survival fraction.
To determine whether GBA sensitizes the cells to irradiation, SE values were determined. As shown in Table 2, SE values demonstrated that GBA, at a concentration of 10 µg/mL, had synergistic effects with irradiation, most noticeable with 1 Gy irradiation.
Values Calculated Based on Equation (4) to Evaluate SEs of GBA.

Abbreviations: GBA, galbanic acid; SE, synergistic effect.
Discussion
Radiotherapy has been used for decades as a treatment modality for patients with ovarian cancer. Nonetheless, radioresistant tumor cells can cause a relapse. Since the use of compounds with radiosensitizing and anti-cancer properties is crucial, recent findings indicate that natural compounds may fill that niche 11,12 and the present study investigated whether GBA, a natural compound with anti-proliferative properties derived from Ferula asafoetida and Ferula szowitsiana could act as a radiosensitizer in vitro.
Alamar Blue assay was performed to evaluate the toxic effects of GBA on OVCAR-3 cells. Viability assessment of the cells indicated that GBA caused toxicity in a dose-dependent manner. The calculated IC50 value for GBA after 24 h in our study was 15.45 μg/mL (38.82 µM), which was consistent with the results reported by Eskandani et al 13 . GBA was dissolved in DMSO, but final concentrations were prepared with complete medium just before each experiment. Therefore, similar cytotoxicity of GBA after 24 h treatment, in comparison with 48 and 72 h treatment periods, might be due to reduced stability of this agent in an aqueous cell culture medium.
Accordingly, 10 μg/mL GBA, a dose lower than the calculated IC50, was selected as the optimal concentration for our combinatorial approach. Furthermore, the cells were only pretreated with GBA for 24 h. As mentioned before, at the specified GBA concentration, prolongation of treatment time had no impact on cell viability. The radiosensitizing potential of GBA was then evaluated by clonogenic assay and the results revealed that treatment with GBA prior to irradiation reduced cell growth and increased radiosensitivity. SE values obtained in the present study were higher than 1, indicating that GBA had synergistic effects with irradiation, most considerable with 1 Gy irradiation.
Irradiation may activate multiple signaling pathways relevant to proliferation or apoptosis. An increase in cell proliferation is dependent on the activation of various mitogenic signaling pathways, such as the EFGR/Ras/Raf/MAPK signaling pathway, through EGFR kinase expression. This is one of the clinically relevant mechanisms implemented by tumor cells to escape from irradiation-induced apoptosis.14–16 Since induced expression of EGFR by irradiation results in poor clinical outcomes, inhibition of EGFR may improve the efficacy of treatment. It has been previously reported that GBA reduced EGFR expression 13 ; therefore, downregulation of EGFR by GBA might, to some extent, be responsible for the observed radiosensitizing effects.
The PI3K/AKT/mTOR signaling pathway plays an important role in cell growth and survival, as well as resistance to ionizing irradiation. Since it has been shown that GBA reduced the phosphorylation of AKT protein, 8 the synergy observed in our combinatorial approach might also be due to GBA effects on the PI3K/AKT signaling pathway.
Conclusion
In the present study, we reported the radiosensitizing effects of GBA on human ovarian cancer cells for the first time, although understanding the mechanism of such action needs to be explained in future research. Our findings also suggest that the radiosensitizing effect of GBA is more prominent when the radiation dose is 1 Gy. However, these results remain to be confirmed in in vivo experiments, and also the investigation of the radiosensitizing effects of GBA on other cell types is suggested for future studies.
Footnotes
Acknowledgments
The authors thank the Office of the Vice-President for Research Affairs of Mashhad University of Medical Sciences for funding this work. The authors are also grateful to Mrs Malihe Rostamyari and Mrs Atefeh Vejdani who helped us in the absence of some of the researchers. This article is based on the results extracted from an MSc thesis (code no: 971840) presented to the Medical Physics Department of Mashhad University of Medical Sciences.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mashhad University of Medical Sciences
Supplemental Material
Supplemental material for this article is available online.