Abstract
Docetaxel is the first-line chemotherapeutic drug for ovarian cancer. However, its clinical use is limited owing to its serious side effects. Therefore, it is of great clinical significance to enhance the efficacy of docetaxel at lower doses in a less-toxic manner. In this study, we investigated whether δ-tocopherol could enhance the anti-tumor effects of docetaxel on ovarian cancer SKOV3 cells in vitro. For docetaxel and δ-tocopherol, IC50 values of 1.89 nM and 11.41 µM, respectively, were obtained, in SKOV3 cells. The combination of δ-tocopherol and docetaxel had a synergistic cell growth inhibition effect, with lower cell viability and more cell arrest at the S phase compared to either δ-tocopherol or docetaxel alone. In addition, the combination of δ-tocopherol and docetaxel had a synergistic cell apoptosis induction effect, with more apoptotic cells and reduced anti-apoptotic protein expression compared to either δ-tocopherol or docetaxel alone. Furthermore, we identified 3 hoursub genes (CAT, EP300, CREBBP), which predicted the prognosis of ovarian cancer, which correlated with δ-tocopherol and docetaxel. In conclusion, the combination of δ-tocopherol and docetaxel presented synergistic cell growth inhibition and cell apoptosis induction effects in SKOV3 cells at a low dose, which suggesting that δ-tocopherol could improve the serious side effects of docetaxel.
Ovarian cancer is a very common malignant tumor in females and has the highest mortality rate of all gynecological cancers. 1 The Global Cancer Statistics report that approximately 295,414 new cases were diagnosed and 184,799 women died of this disease, accounting for 1.6% and 1.9% of all tumors in 2018. 2 Chemotherapy remains the effective treatment for ovarian cancer. Docetaxel is the “gold standard” first-line chemotherapy agent and remains the backbone for current development of treatment regimens.
Docetaxel is a cell-cycle-specific drug that binds to tubulin, inhibits spindle formation during mitosis, induces cell cycle arrest, and activates the apoptosis-related pathway. 3 It is widely used for the treatment of ovarian, breast and non-small cell lung cancers. Unfortunately, almost all patients treated with docetaxel either develop resistance to the drug or have serious side effects. 4,5 The side effects include fluid retention, skin toxicity and bone marrow suppression. 6 Studies have shown that the first-line chemotherapy using docetaxel combined with carboplatin in ovarian cancer patients had a total effective rate of 73%, the median survival time without disease progression was 18 months, and the total survival time was more than 24.4 months. 7 The combination of docetaxel with other drugs exhibited synergistic effects. Therefore, if we identified some candidates which could enhance the efficacy of docetaxel at lower doses in a less-toxic manner and reduce the resistance, it would be of great clinical significance.
Tocopherols, the major forms of vitamin E, are a group of fat-soluble phenolic compounds. Tocopherols, which comprise alpha, beta, gamma, and delta forms, determined by the number and position of methyl groups on the chromanol ring, show strong antioxidative activities. 8 α-Tocopherol is methylated at the 5-, 7-, and 8-positions of the chromanol ring; β-tocopherol is methylated at the 5- and 8-positions; γ-tocopherol is methylated at the 7- and 8-positions; and δ-tocopherol is methylated at the 8-position. 9
Recent reports have shown that δ-tocopherol can prevent hormone-dependent breast cancer, 10 colon carcinogenesis, 11 lung tumorigenesis 12 and prostate cancer progression, 13 with rarely adverse reactions. The aim of this study was to determine whether δ-tocopherol could enhance the anti-tumor effects of docetaxel at low dose.
In this study, we compared the anti-tumor effects on ovarian cancer SKOV3 cells of a combination of δ-tocopherol and docetaxel to docetaxel alone. The results showed that the combination enhanced cell cycle arrest and cell apoptosis induction induced by docetaxel. In addition, 3 hoursub genes were identified, which correlated with the pathology and prognosis of ovarian cancer and interacted with δ-tocopherol.
Results and Discussion
Effects of δ-Tocopherol and Docetaxel Either Alone or in Combination on the Cell Growth of SKOV3 Cells
Docetaxel is widely used for the first-line treatment of ovarian cancer. However, the high dose required eventually leads to toxicity and drug resistance, which limits the use of this drug. 6 To overcome this, we have applied a combination strategy by using a low or non-cytotoxic drug in combination with docetaxel to improve its toxicity and resistance. δ-Tocopherol showed broad-spectrum anti-tumor activity. 14 -18 In the current study, we determined whether a combination of δ-tocopherol with docetaxel could enhance the anti-tumor activities of docetaxel. We used different concentration of docetaxel and δ-tocopherol to treat SKOV3 cells for 72 hours, and cell viabilities were measured by MTT assay. The results showed that docetaxel (Figure 1(A)) and δ-tocopherol (Figure 1(B)) significantly inhibited SKOV3 cell growth in a concentration-dependent manner. Treatment with docetaxel from 0.5 to 8 nM for 72 hours resulted in 4.7% to 81.5% decrease in the number of viable cells compared to control cells, which were treated with DMSO only (Figure 1(A)). Similarly, treatment with δ-tocopherol from 2.5 to 40 µM caused a 12% to 93.9% decrease in the number of viable cells compared to control cells (Figure 1(B)). The IC50 was 1.89 nM for docetaxel and 11.41 µM for δ-tocopherol. We defined 5 µM of δ-tocopherol and 1 nM of docetaxel as low dose, following the drug combination theory. 19 Furthermore, a combination of δ-tocopherol (5 µM) with docetaxel (1 nM) was used to treat SKOV3 cells. As shown in Figure 1(C), the combination treatment caused a 47.2% decrease in the number of viable cells, which was significantly higher than treatment with either docetaxel or δ-tocopherol alone. In addition, the CI, which was calculated by CompuSyn software, was 0.58 in the combination treatment (Figure 2). All these results indicated that the combination of δ-tocopherol with docetaxel showed a synergistic inhibition of the growth of SKOV3 cells.

Effects of δ-tocopherol and docetaxel either alone or in combination on the cell growth of SKOV3 cells SKOV3 human ovarian cancer cells were treated with docetaxel (A) and δ-tocopherol (B) at various concentrations, and docetaxel (1 nM) and δ-tocopherol (5 µM) either alone or in combination of docetaxel (1 nM) + δ-tocopherol (5 µM) (C) for 72 hours. Data shown are mean ± SD of triplicate experiments. *P < 0.05, **P < 0.01, ***P < 0.001.

Isobologram curves and combination index curves for docetaxel and δ-tocopherol either alone or in combination Isobologram curves for docetaxel, δ-tocopherol, and the 2 combined (A). Combination index (CI) for docetaxel and/or δ-tocopherol on inhibition of SKOV3 cells (B).
Effects of δ-Tocopherol and Docetaxel Either Alone or in Combination on the Cell Cycle of SKOV3 Cells
Docetaxel caused cell growth inhibition by inducing cell cycle arrest. 3 Our results showed that δ-tocopherol enhanced the cell growth inhibition induced by docetaxel. We hypothesized that δ-tocopherol may enhance docetaxel to induce cell cycle arrest at the S phase. To test this hypothesis, we used flow cytometry to determine the cell cycle of SKOV3 cells which had been treated with δ-tocopherol and docetaxel either alone or in combination. Docetaxel induced cell cycle arrest of SKOV3 cells at the S phase, and the δ-tocopherol and docetaxel combination treatment increased this effect (Figure 3).

Effects of δ-tocopherol and docetaxel either alone or in combination on the cell cycle of SKOV3 cells by FACS analysis. SKOV3 human ovarian cancer cells were treated with δ-tocopherol (5 µM) and docetaxel (1 nM) either alone or in combination for 72 hours. The cell cycle was analyzed using FACS. Untreated SKOV3 cells were used as the negative control. ***P < 0.001.
Effects of δ-Tocopherol and Docetaxel Either Alone or in Combination on Apoptosis of SKOV3 Cells
As well as suppressing cell growth, docetaxel also initiates apoptosis of many types of cancer cells. 20 -22 Furthermore, δ-tocopherol also exhibits cell apoptosis induction activity . 20 So, we investigated the apoptosis of SKOV3 cells treated with a δ-tocopherol and docetaxel combination. The cell apoptosis was measured by FACS. Treatment with docetaxel (1 nM) and δ-tocopherol (5 µM) separately resulted in late apoptotic cells of 12.55 ± 1.01% and 12.01 ± 0.74%, respectively, whereas the value was 30.58 ± 0.84% for the combination of docetaxel (1 nM) with δ-tocopherol (5 µM) (Figure 4(A–B)). The cell morphology was also studied by microscopy; representative micrographs of apoptotic cells are shown in Figure 5(A). The cell apoptotic rates, treated with either docetaxel (1 nM) or δ-tocopherol (5 µM) alone, were 10.43 ± 1.10% and 6.67 ± 1.33%, respectively, lower than the 30.57 ± 1.92% caused by combination treatment of δ-tocopherol (5 µM) and docetaxel (1 nM) (Figure 5(B)). These results suggest that the combination of δ-tocopherol and docetaxel increase apoptosis of SKOV3 cells.

Effects of δ-tocopherol and docetaxel either alone or in combination on the cell apoptosis of SKOV3 cells by FACS analysis. SKOV3 human ovarian cancer cells were treated with δ-tocopherol (5 µM) and docetaxel (1 nM) either alone or in combination for 72 hours. The apoptosis of cells was analyzed using FACS. Untreated SKOV3 cells were used as the negative control. Representative images are shown (A); Data shown are mean ± SD of triplicate experiments (B). ***P < 0.001.
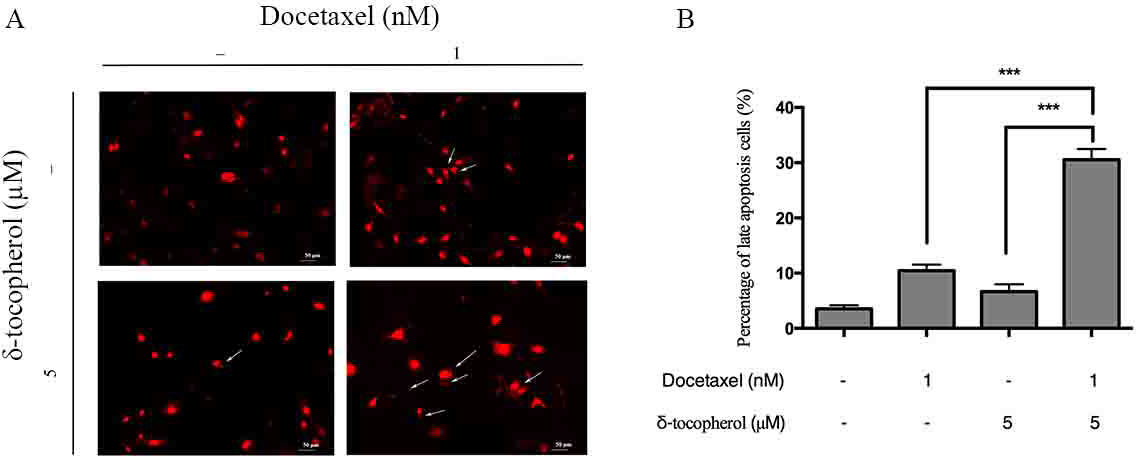
Effects of δ-tocopherol and docetaxel either alone or in combination on cell apoptosis of SKOV3 cells by morphological assessment. SKOV3 human ovarian cancer cells were treated with δ-tocopherol (5 µM) and docetaxel (1 nM) either alone or in combination for 72 hours. The apoptosis of cells was measured by PI staining and morphological assessment. Untreated SKOV3 cells were used as the negative control. Representative micrographs of apoptotic cells are marked by white arrows (A); Data shown are mean ± SD of triplicate experiments (B). *P < 0.0001.
Effects of δ-Tocopherol and Docetaxel Either Alone or in Combination on the Levels of Survivin and Bcl-2
δ-Tocopherol induces cell apoptosis in different cancer cell lines 23 through a variety of molecular mechanisms. It inhibits COX2 activity, 24 modulates nuclear receptors, upregulates PPARγ, 10 downregulates ER α-dependent estrogen signaling and downregulates the cell survival marker Akt. 25
We investigated the relative mechanisms of δ-tocopherol combined with docetaxel treatment in ovarian cancer cells. The anti-apoptotic protein Survivin and Bcl-2 in SKOV3 cells were measured by Western blot. As shown in Figure 6, the expressions of Survivin and Bcl-2 were significantly reduced after treatment with a combination of δ-tocopherol (5 µM) and docetaxel (1 nM) compared with either δ-tocopherol (5 µM) or docetaxel (1 nM) alone.

Effects of δ-tocopherol and docetaxel either alone or in combination on the expression of Survivin and Bcl-2 SKOV3 human ovarian cancer cells were treated with δ-tocopherol (5 µM) and docetaxel (1 nM) either alone or in combination for 72 hours. Survivin and Bcl-2 expression were determined by Western blot and analyzed by optical density measurement and normalized for actin.
PPI Network and Hub Gene Analysis
A string protein interaction database was used to analyze the interaction relationship of overlapped DEGs and to construct a protein interaction network. We screened combined scores higher than 0.4 in PPI (Figure 7). The results of string protein interaction database analysis were imported into Cytoscape software (Figure 8). As shown in Figures 8 and 9 hub genes were identified by the CytoHubba plugin.

PPI network of overlapping genes. The lines represent the regulation of the relationship between 2 genes.

Hub gene screening by CytoHubba plug in.

Effects of δ-tocopherol and docetaxel alone or in combination on the expression of EP300, CAT and CREBBP. SKOV3 human ovarian cancer cells were treated with docetaxel (1 nM) and δ-tocopherol (5 µM) either alone or in combination for 72 hours. Protein expression was determined by Western blot and analyzed by optical density measurement and normalized for actin.
Survival and Clinicopathological Analysis
To verify the results from bioinformatics analysis, the TCGA database was used to predict the prognostic of these hub genes. As shown in Figure 8, we identified 10 hub genes, and the role of 3 hub genes (CAT, EP300 and CREBBP) in ovarian cancer have not yet been reported. The Kaplan-Meier plotter was used to predict the overall survival (OS) and disease-free survival (DFS) of CAT, EP300, CREBBP genes in ovarian cancer. We found that ovarian cancer patients with CAT, EP300, CREBBP and mRNA high expression had lower OS and DFS than those with low expression (Figure 10).

Kaplan-Meier analysis for the OS and RFS of EP300 (A), CAT (B), and CREBBP (C) in ovarian serous cystadenocarcinoma (TCGA, PanCancer Atlas).
Effects of δ-Tocopherol and Docetaxel Either Alone or in Combination on the Levels of CAT, EP300 and CREBBP
To determine the relationship between the 3 hub genes and the actions of δ-tocopherol and docetaxel, we determined the effects of these 2 drugs alone and in combination on the expression of CAT, EP300, and CREBBP. As shown in Figure 9, although treatment with either δ-tocopherol or docetaxel alone had little or no effect on the expression of CAT and EP300,the combination treatment showed a significant reduction of the expression of CAT and EP300. In addition, treatment with either δ-tocopherol or decetaxel alone or in combination did not change the expression of CREBBP (Figure 9).
Conclusions
In this study, the combination of δ-tocopherol and docetaxel presented strong effects on cell growth inhibition and cell apoptosis induction in ovarian cancer SKOV3 cells in vitro. Furthermore, we also found that the combination of docetaxel and δ-tocopherol significantly inhibited the expression of the apoptosis-related protein Survivin, Bcl-2, CAT and EP300. These results provide a powerful basis for the clinical evaluation of the efficacy of docetaxel combined with δ-tocopherol in the treatment of ovarian cancer.
Materials and Methods
Cells and Reagents
The SKOV3 human ovarian cancer cell line was purchased from American Type Culture Collection (ATCC, MD, USA). According to the manufacturer’s instruction, the cells were cultured in 90% RPMI-1640 (HyClone, USA) supplemented with 10% fetal bovine serum (Gibco, USA) and were maintained in a humidified atmosphere of 5% CO2 at 37 °C. Docetaxel and δ-tocopherol were purchased from Sigma-Aldrich (St. Louis, MO, USA), and dissolved in dimethyl sulfoxide (DMSO).
Cell Viability Assay
To detect cell viability after treatment with the drugs, cells were seeded at a density 5 × 104 cells per well. After recovery for 24 hours, the cells were treated with either docetaxel at various concentrations (8 nM, 4 nM, 2 nM, 1 nM, 0.5 nM), or δ-tocopherol (40 µM, 20 µM, 10 µM, 5 µM, 2.5 µM) or docetaxel + docetaxel (8 nM +40 µM, 4 nM +20 µM, 2 nM +10 µM, 1 nM +5 µM, 0.5 nM +2.5 µM) for 72 hours. At the indicated times, 100 µl of MTT (Sigma-Aldrich, MO, USA) solution was added to each well and incubated for another 4 hours at 37 °C. Absorbance readings were measured with a microplate reader (Bio-Rad Laboratories Inc.) at 490 nm. The sensitivity of tumor cells to the drugs and cell viability were determined by estimating the IC50 values and cell growth inhibition, respectively. The impact of background absorbance was eliminated by using the absorbance percentage of drug-treated cells and untreated cells to obtain cell viability.
Cell Cycle Analysis
SKOV3 cells were seeded in 6-well plates at a density 1 × 105 cells per well and incubated for 24 hours. The cells were then treated with either docetaxel (1 nM) or δ-tocopherol (5 µM) alone or in combination with 1% FBS culture medium for 72 hours. After addition of 10 µL of PI, the samples were incubated for 15 minutes at room temperature and analyzed by FACS (BD Bioscience Mountain View, CA, USA).
Cell Apoptosis Analysis
SKOV3 cells were seeded in 6-well plates at a density 1 × 105 cells per well and incubated for 24 hours. The cells were then treated with either docetaxel (1 nM) or δ-tocopherol (5 µM) alone or in combination with 1% FBS culture medium for 72 hours. After addition of 5 µL FITC Annexin V and 10 µL of PI, the samples were incubated for 15 minutes at room temperature and analyzed with FACS (BD Bioscience Mountain View, CA, USA).
Morphological Assessment of Apoptosis
SKOV3 cells were seeded in 6-well plates at a density 2.5 × 104 cells per well and incubated for 24 hours. The cells were then treated with either docetaxel (1 nM) or δ-tocopherol (5 µM) alone or in combination in 1% FBS culture medium for 72 hours. After treatment, cytospin slides were fixed with a mixture of acetone-methanol (1:1) for 10 minutes and stained with propidium iodide (PI) (1 µg/mL) for 10 minutes. The percentages of apoptotic cells were measured by morphological assessment using a fluorescence microscope (TS100/100 F, Nikon, Japan). These experiments were performed in triplicate.
Western Blotting Assay
All the samples were lysed in radioimmunoprecipitation assay (RIPA) (Beyotime Institute of Biotechnology, China) medium containing the protease inhibitor phenylmethanesulfonyl fluoride (PMSF) (Beyotime Institute of Biotechnology, China). The protein concentration was determined using a bicinchoninic acid (BCA) protein assay kit (Beyotime Institute of Biotechnology, China). Equal amounts of protein (20 µg) in 10 µL volume were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, USA). The membranes were blocked with 5% non-fat milk and then incubated with primary antibodies at 4 °C overnight. Secondary antibodies were incubated for 1 hours at room temperature. Protein bands were visualized using an enhanced chemiluminescence detection kit (Thermo scientific, USA).
PharmMapper
This is an updated integrated pharmacophore matching platform with statistical methods for potential target identification.
STRING Database and Cytoscape Software Analysis
STRING (https://string-db.org/cgi/input.pl) is a database of known and predicted protein-protein interactions. The thresholds were set as follow: minimum required interaction score: medium confidence (0.4); Hide disconnected nodes in the network.
Cytoscape (https://cytoscape.org/) is an open source software platform for visualizing molecular interaction networks and biological pathways and these networks are integrated with annotations, gene expression profiles, and other stated data.
Statistical Analyses
All data are presented as the mean ± SD. The analyses were performed using SPSS 17.0 software (SPSS Inc, USA) and P values of <0.05 were considered statistically significant. For the comparison of growth inhibition and cell apoptosis, data were analyzed by ANOVA. The combination index (CI) was used to analyze the potential synergism by CompuSyn software. The combined effects of the drugs are considered to be synergistic if CI is less than 1. Otherwise, they are considered to be either antagonistic or additive if CI is >1 or =1, respectively. The analysis was performed at least in triplicate.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was supported by grants from the Provincial Health and Family Planning Commission of Hainan (18A200155).
