Abstract
Estrogen receptor-positive breast cancer (ERPBC) is the commonest subtype of breast cancer, with a high prevalence, incidence, and mortality. Herbal drugs are increasingly being used to treat ERPBC, although their mechanisms of action are not fully understood. Therefore, in this study, we aimed to analyze the therapeutic properties of FDY003, a herbal anti-ERPBC prescription, using a network pharmacology approach. FDY003 decreased the viability of human ERPBC cells and sensitized them to tamoxifen, an endocrine drug that is widely used in the treatment of ERPBC. The network pharmacology analysis revealed 18 pharmacologically active components in FDY003 that may interact with and regulate 66 therapeutic targets. The enriched gene ontology terms for the FDY003 targets were associated with the modulation of cell survival and death, cell proliferation and growth arrest, and estrogen-associated cellular processes. Analysis of the pathway enrichment of the targets showed that FDY003 may target a variety of ERPBC-associated pathways, including the PIK3-Akt, focal adhesion, MAPK, and estrogen pathways. Overall, these data provide a comprehensive mechanistic insight into the anti-ERPBC activity of FDY003.
Keywords
Introduction
Breast cancer (BC) is a potentially fatal malignancy, with 2.3 million new cases and 0.7 million deaths reported globally every year. 1 BCs can be divided into three types: estrogen receptor-positive BC (ERPBC), human epidermal growth factor receptor 2 (HER2)-amplified BC, and triple-negative BC; ERPBC is the most commonly occurring type. 2 Selective estrogen receptor modulators, aromatase inhibitors, and selective estrogen receptor down-regulators are commonly used for the treatment of ERPBC. 3 These endocrine therapies exhibit low clinical efficacy due to the development of drug resistance, and cause adverse events, including hot flashes, osteoporosis, arthralgia, and cardiovascular symptoms. 4 To overcome these challenges, herbal drugs that have substantial efficacy and fewer side effects are now being prescribed for the treatment of cancer.5,6 Studies have demonstrated the effects of herbal drugs for improving survival and clinical outcomes, enhancing the treatment rate, reducing adverse effects, and augmenting the health status and quality of life of cancer patients.7,8
FDY003 is a herbal anticancer prescription containing Lonicera japonica Thunberg (LjT), Artemisia capillaris Thunberg (AcT), and Cordyceps militaris (Cm).9-11 FDY003 can suppress cell proliferation and induce apoptosis in various cancers by modulating important signaling pathways and regulators that are involved in the coordination of cellular proliferation, growth, survival, and apoptosis.9-11 However, the systemic role of FDY003 in the treatment of ERPBC has not been investigated.
The complex systematic multicompound-multitarget pharmacological characteristics of herbal drugs present a major challenge in the investigation of their therapeutic mechanisms and effects.6,12-18 To overcome this hurdle, the network pharmacology approach has been applied to facilitate the study of the comprehensive systemic mechanisms of herbal drugs.6,12-18 The therapeutic activity of polypharmacological herbal drugs is exerted through the interactions of their constituent phytochemicals with multiple biological components.6,12-18 Therefore, the goal of network pharmacology is to identify the bioactive components of herbal drugs and the disease-associated genes and proteins that they target.6,12-18 Thereafter, the multicompound-multitarget features of the herbal drugs are analyzed by exploring the mechanisms underlying the interactions between the compounds and their biological targets, as well as the biomedical functions and characteristics of the targets.6,12-18 In this study, we assessed the therapeutic effects of FDY003 against ERPBC and employed network pharmacology to explore its comprehensive anticancer mechanisms.
Materials and Methods
Cell Culture
MCF-7 human ERPBC cells were obtained from the Korean Cell Line Bank (Seoul, Korea). The cells were cultured in Dulbecco's modified Eagle's medium (WELGENE Inc., Daegu, Korea) with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin (Thermo Fisher Scientific Inc., Waltham, MA, USA) and incubated at 37 °C in a humidified atmosphere containing 5% CO2.
Preparation of FDY003
The herbal medicines LjT, Act, and Cm were purchased from Hanpure Pharmaceuticals (Pocheon, Korea). Dried LjT (4.16 g), AcT (6.25 g), and Cm (6.25 g) were ground and immersed in 70% ethanol (500 mL). An extract was prepared by refluxing the mixture at 80 °C for 3 h. The extract was then filtered, purified with 80% and 90% ethanol successively, and lyophilized at −80 °C. The samples were stored at −20 °C and dissolved in distilled water for use in further experiments.
Cell Viability Assay
Cell viability was assessed using the WST-1 assay. Briefly, 1.0 × 104 cells were seeded in a 96-well plate and treated with the indicated doses of FDY003 and tamoxifen (Sigma-Aldrich, St. Louis, MO, USA) for 72 h. WST-1 solution (Daeil Lab Service Co., Ltd, Seoul, Korea) was added to each well, and the cells were incubated for an additional 2 h. The absorbance was measured at 450 nm using an xMark microplate absorbance spectrophotometer (Bio-Rad, Hercules, CA, USA).
Bioactive Phytochemical Compounds of FDY003
A list of the chemical components of FDY003, and their absorption, distribution, metabolism, and excretion profiles were obtained from the Traditional Chinese Medicine Systems Pharmacology, Anticancer Herbs Database of Systems Pharmacology, 19 Anticancer Herbs Database of Systems Pharmacology, 20 Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine, 21 and Traditional Chinese Medicine Integrated Database, 22 Next, we assessed the parameters that are widely used in network pharmacology studies (Caco-2 permeability, drug-likeness, and oral bioavailability)13,19,23 to determine which components were most likely to be pharmacologically active. Caco-2 permeability is an index for assessing the intestinal permeability of a compound by using a Caco-2 human intestinal cell model.19,24-26 A Caco-2 permeability ≥0.4 is indicative of good in vivo intestinal permeability.27,28 Drug-likeness is an index of the potential of a compound to be used as a drug, based on its structural and physicochemical features, molecular descriptors, and Tanimoto coefficient.19,29 The mean drug-likeness score of approved drugs is reported to be 0.18; therefore, compounds with a score ≥0.18 were considered potentially druggable.19,29 Oral bioavailability describes a compound's ability to reach the target site (tissues and organs) after oral administration.19,30 A compound with oral bioavailability ≥30% is considered to be effectively absorbed and distributed in the body.19,30 Therefore, phytochemical compounds with Caco-2 permeability of −0.4 or higher, drug-likeness ≥0.18, and oral bioavailability ≥30% were considered bioactive.13,19,23
Targets of Bioactive Phytochemical Compounds of FDY003
The canonical simplified molecular input line entry specification (SMILES) notations of the bioactive phytochemical components of FDY003 were obtained from the PubChem database. 31 The SMILES notations were imported into SwissTargetPrediction, 32 PharmMapper, 33 Search Tool for Interactions of Chemicals 5, 34 and Similarity Ensemble Approach 35 to determine the biological target of each compound in Homo sapiens by investigating the chemical-protein interactions. The genes and proteins associated with ERPBC in humans were retrieved from a previous network pharmacology study 36 and from other databases (DisGeNET, 37 Therapeutic Target Database, 38 Online Mendelian Inheritance in Man, 39 GeneCards, 40 and DrugBank 41 ) using the search term “Estrogen receptor-positive breast cancer”.
Herbal Prescription-Associated Networks
Pharmacological networks were constructed to facilitate further data analysis. The herbal medicine-bioactive compound-target (H-C-T) network mapped the links among herbal medicines, their bioactive phytochemical constituents, and their ERPBC-related therapeutic targets. The H-C-T-P network outlined the connections between the targets in the H-C-T network, and the pathways in which they are involved. The protein–protein interaction (PPI) network summarized paired interactions between ERPBC-associated targets retrieved from the STRING database. 42 All networks were constructed, visualized, and analyzed using the Cytoscape software. 43 The herbal prescription-associated network is composed of nodes (herbal and bioactive phytochemical constituents, targets, and pathways) and links (or edges) that represent the interactions between them. 44 The degree indicates the number of links that the nodes contain. 44
Survival Analysis
To assess the clinical significance and therapeutic importance of the FDY003 targets, we conducted a survival analysis. The correlation between the survival rates of patients with ERPBC and the FDY003 targets was analyzed with the Kaplan–Meier Plotter using auto-selected best cutoffs and the log-rank test. 45 Differences with P < 0.05 were considered statistically significant.
Functional Enrichment Analysis
To explore the key molecular and pathway mechanisms of action of FDY003 against ERPBC, we conducted a functional enrichment analysis for the FDY003 targets. Therefore, we imported the targets into g:Profiler, a useful tool for the functional investigation of genes of interest, 46 and obtained the gene ontology terms and pathways wherein they are significantly enriched (P < 0.05).
Molecular Docking Assessment
The structural information of phytochemical compounds and targets was retrieved from PubChem 31 and RCSB Protein Data Bank, 47 respectively. These data were imported into Autodock Vina 48 and the molecular docking scores for interactions between the phytochemical compounds and targets were calculated. A docking score of − 5.0 or less was considered indicative of strong binding.49,50
Results
Anticancer Properties of FDY003 Against BC
To assess the anticancer effects of FDY003 on ERPBC, MCF-7 cells were treated with FDY003 in the presence or absence of tamoxifen, an endocrine drug that is clinically used for the treatment of ERPBC, 51 and the cellular responses were evaluated. FDY003 was found to decrease the viability of MCF-7 cells and enhance the anti-proliferative effect of tamoxifen (Supplementary Figure S1), indicating that FDY003 may possess anti-ERPBC activity.
Identification of Bioactive Phytochemical Compounds of FDY003 and Their Targets
We assessed the pharmacokinetic properties of the phytochemical components of FDY003 (Supplementary Table S1), and shortlisted potentially bioactive compounds that satisfied the set criteria (Caco-2 permeability of −0.4 or higher, and drug-likeness ≥0.18, oral bioavailability ≥30%), as previously described.11,13,19,23 Some compounds were additionally considered bioactive because of their promising anti-ERPBC properties. 11 Therefore, 18 bioactive compounds were determined for FDY003 (Supplementary Table S2). Then, the human targets of FDY003 were screened based on the molecular structures of the bioactive compounds (see Materials and Methods). In total, 66 ERPBC-associated and 130 non-ERPBC-related targets were determined for FDY003 (Supplementary Table S3).
Network Pharmacological Mechanisms of FDY003 Against ERPBC
The comprehensive data associated with the bioactive phytochemical components and targets of FDY003 were integrated into an H-C-T network (Figure 1). The network was composed of 87 nodes (3 herbal medicines, 18 bioactive phytochemical compounds, and 66 ERPBC-associated targets) and 134 edges (Figure 1 and Supplementary Table S3). The bioactive compounds with the highest number of targets were kaempferol, luteolin, and quercetin (Figure 2 and Supplementary Table S3), suggesting that these agents may be the major pharmacological components. Moreover, 83% of the targets (55 out of 66 nodes) interacted with two or more compounds (Figure 2), implying the polypharmacological features of FDY003.

The herbal medicine-bioactive phytochemical compound-target network of FDY003. Green nodes, herbal medicines; red nodes, bioactive phytochemical compounds; blue nodes, estrogen receptor-positive breast cancer-related targets.
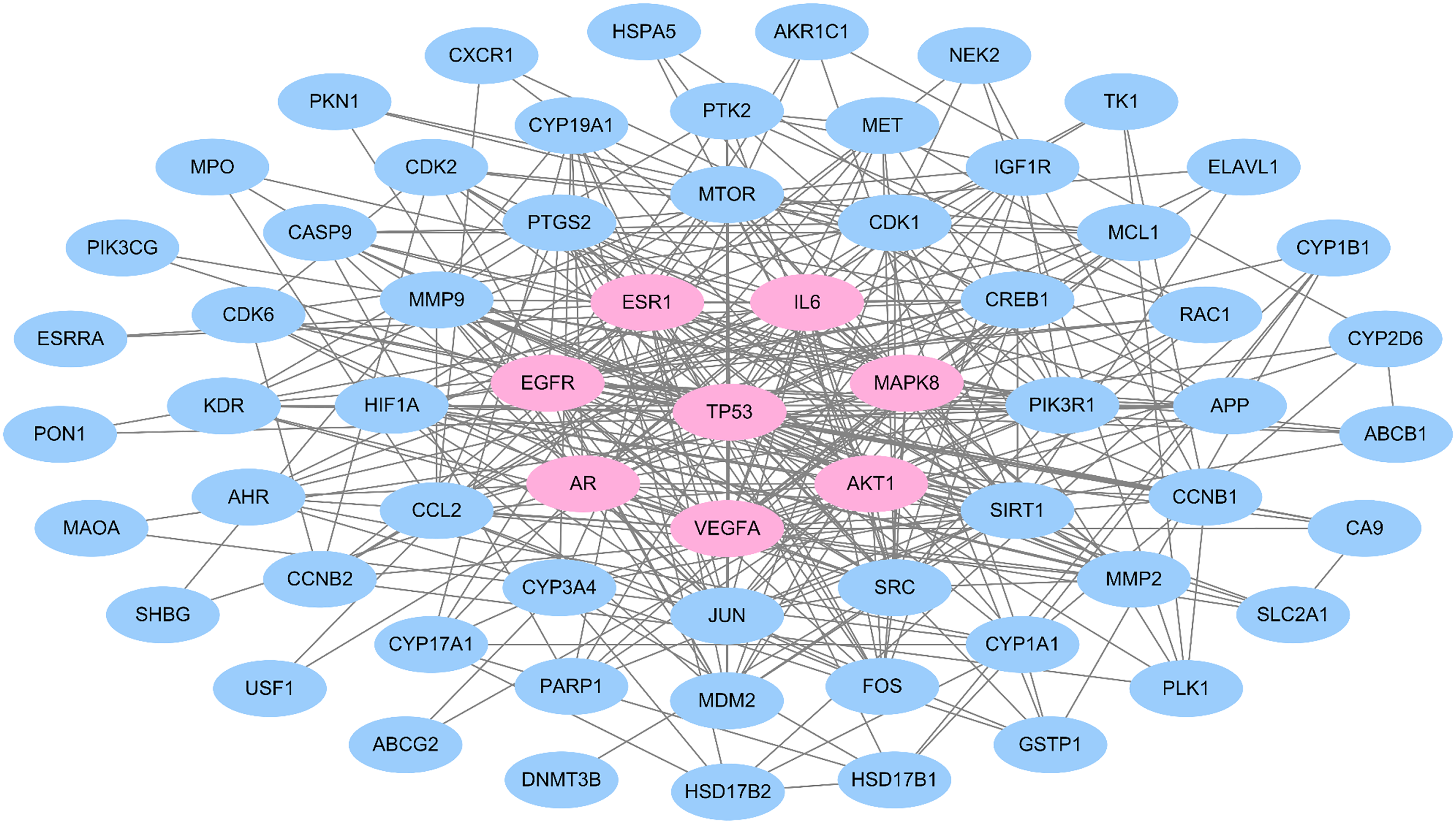
The protein-protein interaction network for estrogen receptor-positive breast cancer-related targets of FDY003. Pink nodes, hub estrogen receptor-positive breast cancer-related targets.
Mechanistic investigation of the complex interactions between target genes and proteins is essential for unraveling the therapeutic properties of a drug.52-56 Therefore, we constructed a PPI network (65 nodes and 332 links) using the ERPBC-associated targets as nodes (Figure 2). Then, we explored the hubs within the PPI network, nodes that possess relatively many links and function as crucial biological regulators and also as promising drug targets.57,58 As previously described, hubs were defined as nodes where the number of links was greater than or equal to twice the mean node degree of the network.59,60 AKT1, AR, EGFR, ESR1, IL6, MAPK8, TP53, and VEGFA were identified as high-degree hub nodes (Figure 2), suggesting their importance in mediating the anti-ERPBC activity of FDY003. The level of expression of these targets was found to correlate with the overall, recurrence-free, and distant metastasis-free survival of patients with ERPBC (Figure 3), suggesting that these targets may function as potential prognostic indicators.

Survival analysis of estrogen receptor-positive breast cancer-associated targets of FDY003. Kaplan-Meier curves for the survival of patients with estrogen receptor-positive breast cancer according to the expression levels of the indicated estrogen receptor-positive breast cancer-associated targets of FDY003.
The results indicate the network-perspective properties of the anti-ERPBC mechanisms of FDY003.
Functional Analysis of the Pharmacological Properties of FDY003
To dissect the key mechanisms of action of FDY003 against ERPBC, we conducted a functional enrichment analysis. Gene ontology enrichment analysis demonstrated that the targets of FDY003 participate in the regulation of cell survival and death, cell proliferation and growth arrest, and estrogen-associated cellular processes (Supplementary Figure S1). Furthermore, pathway enrichment analysis revealed that the targets are major constituents of the pathways associated with ERPBC pathology (Figure 4 and Supplementary Figure S2), suggesting the key signaling mechanisms of FDY003. The adenosine monophosphate (AMP)-activated protein kinase (AMPK) pathway exerts tumor-suppressing effects by inducing cell cycle arrest and apoptosis, and inhibits the growth and survival of ERPBC cells. 61 The chemokine, erythroblastic leukemia viral oncogene homolog (ErbB), focal adhesion, mitogen-activated protein kinase (MAPK), PI3K-Akt, and Ras pathways play important roles in the initiation and progression of ERPBC by controlling diverse tumorigenic behaviors in cells (eg, angiogenesis, cancer stemness, metastasis, migration, invasion, proliferation, survival, and tumor-initiating capacity).62-67 The dysregulated synthesis and secretion of estrogen and its downstream activity are the primary drivers of tumorigenesis and progression of ERPBC; pharmacological modulation of estrogen and the pathways in which it is involved is a key strategy in anti-ERPBC therapeutics.68-70 The hypoxia-inducible factor (HIF)-1 pathway coordinates the self-renewal potential, cancer stemness, proliferation, and metastasis of ERPBC cells.71,72 Furthermore, insulin resistance may be associated with the incidence, recurrence, and survival rates of BC patients.73-76 The loss of p53 pathway function remains one of the most important events in the pathophysiology of ERPBC and is associated with disease aggressiveness and survival outcomes in patients with BC.77,78 The activity of the PD-1/PD-L1 pathway is an indicator of survival in BC patients, and blocking this pathway may strengthen anti-tumor immune function.79-81 The prolactin pathway functions as a survival and migration factor for ERPBC cells and is a risk factor for BC; therefore, inhibition of this pathway can reduce the cancerous ability of ERPBC cells and make them more susceptible to anticancer drugs.82-85 The tumor necrosis factor (TNF) pathway modulates pro-tumorigenic inflammation, and its activity is associated with carcinogenesis, malignant tumor development, metastasis, induction of therapeutic resistance, and prognosis of ERPBC.86,87 The VEGF pathway contributes to ERPBC progression by stimulating metastasis, cell survival, and angiogenesis in ERPBC tumors.88,89 The misregulation of important cellular processes, including apoptosis, cell cycle processes, and senescence is associated with uncontrolled growth, proliferation, and survival of ERPBC cells.90-93 The development of resistance against anticancer agents, such as endocrine therapeutics, platinum-based cytotoxic drugs, and EGFR inhibitors, is largely responsible for the failure of ERPBC treatment.94-100

The herbal medicine-bioactive phytochemical compound-target-pathway network of FDY003. Green nodes, herbal medicines; red nodes, bioactive phytochemical compounds; blue nodes, estrogen receptor-positive breast cancer-related targets; orange nodes, estrogen receptor-positive breast cancer-associated signaling pathways.
Overall, these results reveal the key molecular mechanisms and pathways underlying the anti-ERPBC activity of FDY003.
Molecular Docking Assessment of Bioactive Compounds and the Targets of FDY003
Molecular docking analysis is a reliable and convenient method to investigate and predict the intermolecular interactions between drug molecules and targets by evaluating their potential binding affinities.101-104 To assess the binding capacity of the bioactive components of FDY003 with their biological targets, we conducted molecular docking studies (see Materials and Methods). The docking scores of the bioactive compounds with their hub targets were found to be −5.0 or less (Figure 5), indicating the binding potential of the compound-target pairs. With regard to the binding interaction sites of the compound-target pairs, kaempferol mainly interacted with the amino acid residues Ala748, Met745, and Trp751 in androgen receptor (AR); amino acid residues Ala722, Arg836, Arg858, Arg860, Glu758, Gly724, Leu747, and Phe723 in EGFR; and amino acid residues Arg394, Leu387, and Phe404 in ESR1 (Figure 5). Luteolin may bind to MAPK8 by interacting with its amino acid residues such as Ala113, Asp112, Asp169, Leu178, and Val158 (Figure 5). Quercetin primarily formed interactions with amino acid residues Glu85, Ile84, and Val83 in Akt1; amino acid residue Met745 in AR; amino acid residues Glu872 and Lys875 in EGFR; amino acid residues Ala69, Asn62, Asn64, Leu65, Lys67, Met68, and Pro66 in IL6; amino acid residues Ala53, Gly33, Ile32, Ile39, Lys55, Ser34, and Val40 in MAPK8; and amino acid residues Gln165 and Gln167 in TP53 (Figure 5).

Molecular docking analysis for the bioactive phytochemical compounds of FDY003 and their estrogen receptor-positive breast cancer-associated targets. (A) Kaempferol-AR (score = −8.3); (B) Kaempferol-EGFR (score = −7.9); (C) Kaempferol-ESR1 (score = −6.6); (D) Luteolin-MAPK8 (score = −8.0); (E) Quercetin-AKT1 (score = −6.2); (F) Quercetin-AR (score = −8.5); (G) Quercetin-EGFR (score = −8.6); (H) Quercetin-IL6 (score = −7.2); (I) Quercetin-MAPK8 (score = −8.1); (J) Quercetin-TP53 (score = −6.9); (K) Quercetin-VEGFA (score = −7.4).
Discussion
There has been a growing interest in the use of medicinal herbs for the treatment of ERPBC;105,106 however, their poly-pharmacological mechanisms have not yet been explored from a network perspective. In this study, we dissected the multicompound-multitarget anti-ERPBC mechanism of the herbal prescription, FDY003.9-11 FDY003 decreased the viability of human ERPBC cells and sensitized them to endocrine therapy. Evaluation of the network pharmacology revealed that FDY003 contains 18 pharmacologically active compounds that may interact with and regulate 66 potential therapeutic targets. The enriched gene ontology terms of the FDY003 targets were found to be associated with the modulation of cell survival and death, cell proliferation and growth arrest, and estrogen-associated cellular processes. Pathway enrichment investigation revealed the crucial ERPBC-associated signaling pathways that may be targeted by FDY003: the PI3K-Akt, focal adhesion, MAPK, and estrogen pathways. Overall, the network pharmacological analysis revealed the molecular and signaling mechanisms underlying the anti-ERPBC activity of the herbal prescription.
The crucial ERPBC-associated hub targets of FDY003 are the key mediators that coordinate the pathological mechanisms involved in ERPBC and serve as potential therapeutic targets. The abnormal activation and deactivation of AKT1 (encoded by AKT1) and epidermal growth factor receptor (EGFR; encoded by EGFR) induce cancerous cellular behavior in ERPBC cells (eg, survival, metastasis, proliferation, cancer stemness, invasion, and migration); their expression, activity, and mutation status are prognostic predictors of therapeutic sensitivity and survival in patients with BC.65,107-120 Furthermore, these kinases are implicated in the occurrence of therapeutic resistance to anti-ERPBC treatment, and thus can be targeted to overcome this resistance.65,109-111,117,121 The activation of AR (encoded by AR) weakens the invasion, proliferation, migration, and tumor-initiating properties of ERPBC cells, 122 and the expression of AR correlated with reduced mortality and improved prognostic outcome of ERPBC patients,123,124 Genetic alterations in and over-activation of estrogen receptor α (ERα; encoded by ESR1) are the key driving forces for the tumorigenesis and progression of ERPBC, and ERα is the main therapeutic target for endocrine therapies. 125 In addition, ESR1 expression and mutation status are the determinants of endocrine resistance and clinical prognosis of ERPBC patients.126-130 Interleukin-6 (IL-6; encoded by IL6) is a cytokine that is involved in the growth, malignancy, invasiveness, and metastatic capability of ERPBC tumors, and its expression is highly upregulated in cancer patients.131-135 The expression, activity, and genetic variants of IL-6 are further associated with the risk of cancer incidence, survival, recurrence, and the development of therapy-induced toxicity in patients with BC.136-139 c-Jun N-Terminal Protein Kinase 1 (JNK1; encoded by MAPK8) regulates the survival, apoptosis, metastasis, and inflammation of ERPBC cells, and it also mediates the pharmacological effects of anticancer drugs.140-145 Malfunction and loss-of-function polymorphisms of TP53 induce the development and progression of ERPBC, and the expression and mutation status of this gene are correlated with the survival and prognostic outcomes of BC patients.77,146-154 Vascular endothelial growth factor (VEGF)-A (encoded by VEGFA) may contribute to the metastasis, angiogenesis, growth, and drug resistance of ERPBC cells,89,155-157 and its expression level and polymorphisms may act as genetic markers for the risk, progression and recurrence, clinical outcomes for endocrine and antiangiogenic therapies, and survival of patients.158-162 These results provide a pharmacological basis for the anti-ERPBC mechanisms of and further suggest the need for an in-depth investigation of FDY003's therapeutic effects on multiple malignant cellular behaviors, including invasion, migration, metastasis, anoikis resistance, cancer stemness, and angiogenesis in ERPBC cells as investigated from the functional enrichment analysis (Supplementary Figure 2).
The components of FDY003 have previously been shown to exhibit anti-ERPBC activity. AcT has been found to reduce the proliferation and survival capacity of ERPBC cells. 163 Cm represses the migration, survival, and growth of ERPBC cells by activating caspase-dependent mitochondrial signaling.164,165 Capillarisin exhibits anticancer effects by blocking the invasion and proliferation of ERPBC cells. 166 The inhibitory effects of cordycepin on the invasion, survival, and proliferation of ERPBC cells are mediated by the caspase and MAPK pathways.167-169 Cordycepin can enhance the anticancer effects of radiotherapy on ERPBC cells. 170 Genkwanin targets PI3K-Akt signaling to exhibit its anti-growth and pro-apoptotic activities in ERPBC cells.171,172 Isorhamnetin inhibits cell cycle progression and proliferation of ERPBC cells. 173 β-Sitosterol promotes the death of ERPBC cells 174 and improves the efficacy of endocrine therapy in the treatment of ERPBC. 175 Quercetin, luteolin, and kaempferol induce cell-cycle arrest, growth arrest, and cell death, while weakening proliferation, angiogenesis, metastasis, survival, tumor-initiating capacity, invasion, and migration by targeting various signaling pathways associated with key ERPBC pathomechanisms.176-190 In addition, these compounds function as chemosensitizers that make ERPBC cells more susceptible to other anticancer drugs.191-193 Furthermore, the compound-target interaction relationship that was identified from the network pharmacological analysis (Supplementary Table S3) was previously reported. Cordycepin and quercetin increased the activity of caspase-9 (encoded by CASP9) to induce apoptosis of ERPBC cells.168,177 Kaempferol inactivates glucose transporter 1 (GLUT1; encoded by SLC2A1) and ERα, which suppresses the growth and survival capacity of ERPBC cells.176,180 Luteolin and quercetin target matrix metalloproteinase 2 (MMP2; encoded by MMP2) and MMP9 (encoded by MMP9) and exhibit anti-invasive effects on ERPBC cells.181,194 Quercetin, a pharmacological flavonoid, 195 possesses anti-angiogenic ability against ERPBC cells by targeting c-JUN (encoded by JUN), VEGF-A, and VEGF receptor 2 (VEGFR-2; encoded by KDR).190,196,197 The suppression of cyclooxygenase-2 (COX-2; encoded by PTGS2) by quercetin contributes to the reduction of the invasiveness of ERPBC cells. 198 Moreover, this active compound can downregulate AKT1, EGFR, and HIF-1α (encoded by HIF1A), while upregulating p53 (encoded by TP53) to inhibit the proliferation, chemoresistance, survival, cancer stemness, and invasion of ERPBC cells.177,181,183,188,199 Further experimental studies are warranted to verify the physicochemical interactions and biological regulations between the active compounds and their targets for the investigation of the precise mechanisms of the herbal prescription.
In conclusion, this network pharmacological study gave us a comprehensive view of the molecular and signaling mechanisms involved in the anti-ERPBC activity of the herbal prescription, FDY003. The overall analysis results provide a systematic insight that can advance our understanding of the multicompound-multitarget properties of herbal drugs as anticancer agents. To broaden the potential pharmacological applications of FDY003, the effects of FDY003 must be studied further, and the agent's activity must be evaluated in combination with other anticancer agents (eg, chemotherapeutics, targeted therapeutics, and cancer immunotherapeutics) in other BC subtypes, including HER2-positive BC and triple-negative BC.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211044377 - Supplemental material for Network Pharmacology-Based Dissection of the Comprehensive Molecular Mechanisms of the Herbal Prescription FDY003 Against Estrogen Receptor-Positive Breast Cancer
Supplemental material, sj-docx-1-npx-10.1177_1934578X211044377 for Network Pharmacology-Based Dissection of the Comprehensive Molecular Mechanisms of the Herbal Prescription FDY003 Against Estrogen Receptor-Positive Breast Cancer by Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon and Dae-Yeon Lee in Natural Product Communications
Footnotes
Acknowledgments
Not applicable.
Author Contributions
Conceptualization: Ho-Sung Lee, In-Hee Lee, Dae-Yeon Lee; Methodology: Ho-Sung Lee, In-Hee Lee, Dae-Yeon Lee; Data collection: Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon; Data analysis and investigation: Ho-Sung Lee, In-Hee Lee, Dae-Yeon Lee; Writing: Ho-Sung Lee, In-Hee Lee, Dae-Yeon Lee; All authors read and approved the final manuscript.
Data Statement
All data either generated or analyzed during this study are included in this published article and its supplementary materials file.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2021R1F1A1049472).
Statement of Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
