Abstract
Despite accumulating evidence for the value of herbal drugs for cancer treatment, the mechanisms underlying their effects have not been fully elucidated in a systematic manner. In this study, we performed a network pharmacological analysis to elucidate the anti-esophageal cancer (EC) properties of the herbal drug FDY003, a mixture of
Introduction
Esophageal cancer (EC), an aggressive gastrointestinal cancer, is the 10th most frequently diagnosed malignant tumor worldwide, accounting for 0.60 million new cases and 0.54 million deaths per year. 1 The major treatment strategies for EC include immunotherapy, targeted therapy, and chemotherapy. 2 However, treatment options are limited and their efficacies are generally insufficient. 2 Herbal drugs are increasingly recognized as potent agents for cancer treatment based on evidence that they can increase the therapeutic success rate, promote recovery after anticancer therapies, suppress recurrence and adverse events, and alleviate cancer-associated symptoms in patients with EC.3,4
FDY003—an herbal mixture made up of
Network pharmacology is a powerful approach to dissect the complex and synergistic therapeutic effects of herbal drugs, as demonstrated by extensive studies.9,10 Based on the “drug-targeted gene/protein-disease” perspective, network pharmacology attempts to explore systematic relationships among pharmacological targets, genes, and proteins involved in the pathological mechanisms of a given disease.9,10 Using network pharmacology, the active pharmacological chemicals with major roles in the pharmacological action of herbal drugs are screened, targets of active chemicals are identified, and the molecular regulatory mechanisms underlying the effects of herbal drugs are determined via the analysis of networks formed by the intricate interactions among drug targets and disease-associated genes and proteins.9,10 We conducted a network pharmacology-driven study to evaluate the anti-EC activity of FDY003.
Materials and Methods
Cell Culture
The EC9706 human EC cell line was obtained from the American Type Culture Collection (Rockville, MD, USA). Cells were cultured in Dulbecco's modified Eagle's medium (WELGENE Inc.) that contained 10% fetal bovine serum (WELGENE Inc.) and 1% streptomycin-penicillin (Thermo Fisher Scientific, Inc.). The cells were then incubated at 5% CO2 and 37 °C.
Preparation of FDY003
LjT (150.0 g), Cm (100.0 g), and AcT (150.0 g), components of FDY003, were obtained from Hanpure Pharmaceuticals (Pocheon, Korea). They were mixed and ground, and the resulting mixture was submerged in 70% ethanol (500 mL). After reflux extraction at −80 °C for 3 h, the extracts were dissolved in 80% and 90% ethanol consecutively. Next, the extracts were cryodesiccated at −80 °C to obtain 50.4 g of a freeze-dried form, stored at −20 °C, and dissolved in purified water before experiments.
Determination of EC Cell Viability After Drug Treatment
The effects of drug treatment on the viability of EC cells were determined using water-soluble tetrazolium salt (WST-1) assays. In brief, 1.0 × 104 cells were plated onto 96 well-plates and treated for 72 h with FDY003 and/or 5-FU (Sigma-Aldrich) as a single drug treatment and also in a combination. Then, WST-1 solution (Daeil Lab Service Co. Ltd, Seoul, Korea) was added to the cells before incubation for 2 h at 37 °C with 5% CO2. The absorbance at 450 nm was measured using an xMark microplate absorbance spectrophotometer (Bio-Rad) to assess viability.
Uncovering Active Pharmacological Chemicals and Targets of FDY003
Detailed information on the chemicals contained in FDY003 and their pharmacological properties was retrieved from large-scale herbal medicine-relevant databases (eg, Traditional Chinese Medicine Systems Pharmacology, 11 Anticancer Herbs Database of Systems Pharmacology, 12 and Bioinformatics Analysis Tool for Molecular Mechanism of Traditional Chinese Medicine 13 ). Their oral bioavailability, druglikeness, and Caco-2 permeability—widely used parameters for network pharmacology studies—were analyzed, and the chemicals were classified as active pharmacological chemicals if they were able to satisfy the following conditions as reported previously 11 : oral bioavailability ≥30%, druglikeness ≥0.18, and Caco-2 permeability ≥−0.4. Oral bioavailability determines whether a chemical taken orally has the potential to effectively reach relevant organs and tissues it aims to.11,14 Oral bioavailability ≥30% is a general criterion for screening chemicals with effective delivery in the human body.5,11,15 Druglikeness determines whether a chemical possesses pharmacologically suitable properties in terms of structures and physicochemical and molecular activities.5,11,15 Druglikeness ≥0.18 is a general criterion for screening chemicals with strong potential as drug candidates.5,11,15 Caco-2 permeability determines whether a chemical shows reasonable permeability in the human intestinal systems. 11 Caco-2 permeability ≥−0.4 is a typical criterion for screening chemicals that may permeate the intestines to exert pharmacological action.16,17
Next, the simplified molecular-input line-entry system (SMILES) notations for the individual active pharmacological chemicals were acquired from PubChem 18 and entered into databases of protein–chemical interactome data and algorithms (eg, SwissTargetPrediction, 19 PharmMapper, 20 Search Tool for Interactions of Chemicals 5, 21 and Similarity Ensemble Approach 22 ) to identify the targets of the active chemicals of FDY003. Targets that have been shown to contribute to the carcinogenesis and malignant development and progression of EC were screened using comprehensive data obtained from various relevant databases (eg, DisGeNET, 23 Human Genome Epidemiology Navigator, 24 Comparative Toxicogenomics Database, 25 Pharmacogenomics Knowledgebase, 26 GeneCards, 27 Therapeutic Target Database, 28 DrugBank, 29 and Online Mendelian Inheritance in Man 30 ).
Generation of Networks and Analysis of their Properties
Networks are generally composed of nodes that represent individual components and edges (or links) that represent interactions, relationships, and associations between nodes. 16 The term “degree” describes the No. of edges a given node has. 16 The herbal constituents, active pharmacological chemicals, and targets of FDY003 serve as nodes in the herbal medicine-active pharmacological chemical-target (H-A-T) network; various forms (ie, molecular, functional, and physicochemical mechanisms) of interactions and associations between the nodes serve as edges. In the case of the H-A-T-Pathway (H-B-T-P) network, the herbal constituents, active pharmacological chemicals, targets, and pathways enriched by the targets of FDY003 serve as nodes, and various forms (ie, molecular, functional, and physicochemical mechanisms) of interactions and associations between the nodes serve as edges. The protein–protein interaction (PPI) network uses FDY003 targets as nodes and diverse mechanistic interactions and relationships between the nodes as edges based on PPI interaction data with confidence scores ≥0.7 from the STRING database. 17 Network generation and all related analyses were performed using Cytoscape. 31
Associations Between Expression Levels of FDY003 Targets and EC Survival
Associations between the expression levels of FDY003 targets and EC survival were determined by importing the targets to the Kaplan–Meier Plotter,
32
a effective tool for the survival analysis across various cancer types. The results were obtained with an auto-selected best cut-off and those with
Functional and Pathway Enrichment Analyses of FDY003 Targets
Enriched Gene Ontology terms and pathways for the EC-related FDY003 targets were identified by importing them to the g:Profiler,
33
and results with
Molecular Binding and Docking Between Active Pharmacological Chemicals and Targets of FDY003
Results
Pharmacological Action of FDY003 in EC
For the evaluation of the pharmacological action of FDY003 in EC, either FDY003, 5-FU (a cytotoxic anti-EC drug 37 ), or their combination was administered to EC9706 human EC cells. FDY003 was able to inhibit the viability of EC9706 cells and further raised the inhibitory effect of 5-FU on EC cells (Figure 1A and B). These data may indicate that FDY003 acts as an anticancer inhibitor and chemosensitizer for EC treatment.

Inhibition of viability of human esophageal cancer (EC) cells by the FDY003 treatment. (A) EC9706 human EC cells were treated with varying doses of FDY003, and their viability was monitored. (B) EC9706 human EC cells were treated with either FDY003, 5-FU (a cytotoxic agent against EC), or both FDY003 and 5-FU, for 72 h, and their viability was monitored. Data represent the mean + SD of 5 replicates.
Active Pharmacological Chemicals and Targets of FDY003
Following previous studies,5,11 the following threshold values of pharmacochemical parameters were applied for the determination of active pharmacological chemicals of FDY003: druglikeness ≥0.18, Caco-2 permeability ≥−0.4, and oral bioavailability ≥30% (Supplementary Tables S1 and S2). Some chemicals with previously established potent antitumor efficacy were added to the list of active pharmacological chemicals (Supplementary Tables S1 and S2). The targets of these active chemicals were then identified using various tools and algorithms to search human gene and protein targets of certain chemicals (Supplementary Table S3). The targets that were closely related to the pathological processes and mechanisms in EC were screened using databases related to cancer biology and medical oncology (Supplementary Table S3).
Network-Perspective Analysis of Mechanisms Underlying FDY003 Effects Against Esophageal Cancer
To analyze the anticancer mechanisms of FDY003 against EC from a network perspective, its H-A-T network was created by integrating the comprehensive data of the herbal drug (Figure 2 and Supplementary Table S3). The H-A-T network indicated that quercetin interacted with the largest number (51) of the 61 targets (Figure 2 and Supplementary Table S3), supporting its key role in the anti-EC activity of FDY003. Additionally, 80.32% (49 of 61) of targets interacted with 2 or more active pharmacological chemicals (Figure 2).
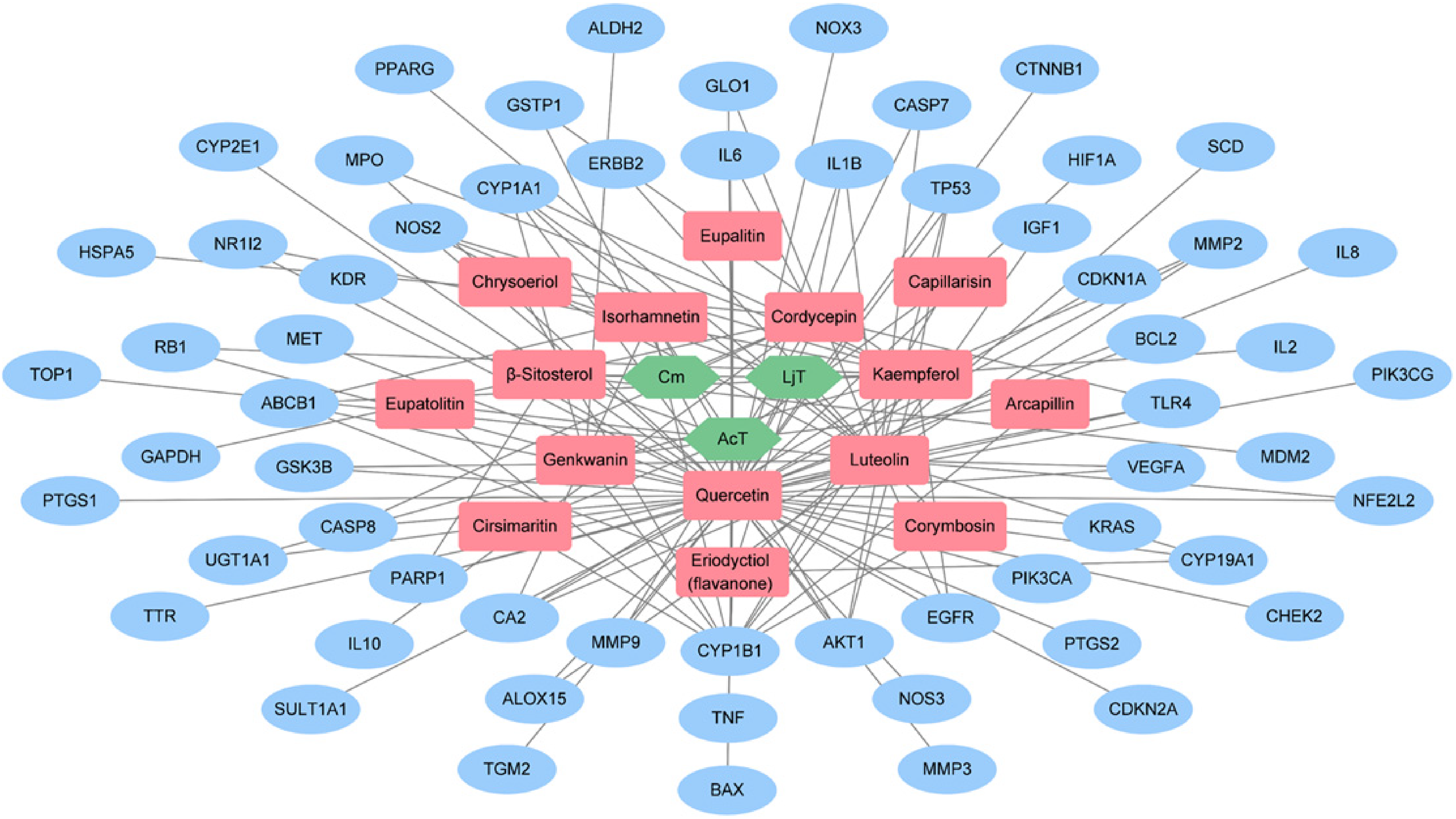
Herbal medicine-active pharmacological chemical target network for FDY003. Green node, herbal medicine; red node, active pharmacological chemical; blue node, esophageal cancer (EC)-related target.
It is essential to investigate the network-level features of the molecular targets to elucidate the regulatory mechanisms of a given drug.
38
Thus, a PPI network of the FDY003 targets was created based on molecular and functional interactions, and hub nodes with a relatively large number of links and pharmacological importance39,40 were searched in the network (Figure 3). Hubs were defined using the following criterion: nodes whose No. of links is equal to, or greater than, 2 times the average No. of links across all nodes in the network.5–8 The nodes AKT1, epidermal growth factor receptor (EGFR), glyceraldehyde 3-phosphate dehydrogenas (GAPDH), IL6, tumor necrosis factor (TNF), TP53, and VEGFA were identified as hubs (Figure 3), demonstrating that these loci may be key determinants of the anti-EC functions of FDY003. Additionally, a survival analysis indicated a significant (

Protein–protein interaction network for esophageal cancer (EC)-related targets of FDY003. Claret node, hub; blue node, non-hub.

Associations of the expression levels of the FDY003 targets with esophageal cancer (EC) survival. Kaplan–Meier curves for the association of the expression levels of the indicated FDY003 targets with the EC survival.
The analysis of functional enrichment of the FDY003 targets further revealed the mechanism underlying its effects in EC. The targets were potent coordinators of the malignant behaviors of EC cells, having roles in cell death, survival, division, proliferation, and growth (Supplementary Figure S1). In addition, the FDY003 targets were key regulators of the activities of major oncogenic signaling pathways associated with EC pathomechanisms (Figure 5 and Supplementary Figure S1), such as phosphoinositide 3-kinase (PI3K)-Akt, hypoxia-inducible factor (HIF)-1, mitogen-activated protein kinase (MAPK), TNF, p53, Janus kinase (JAK)-signal transducer and activator of transcription (STAT), erythroblastic leukemia viral oncogene homolog (ErbB), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kappa B), and vascular endothelial growth factor (VEGF) cascades. These results provide insight into the polypharmacological activity of FDY003 against EC.

Herbal medicine-active pharmacological chemical target pathway network for FDY003. Green node, herbal medicine; red node, active pharmacological chemical; blue node, esophageal cancer (EC)-related target; orange node, EC-related pathway.
Verification of the Molecular Binding Between Targets and Active Pharmacological Chemicals of FDY003
To confirm the molecular binding between the targets and active pharmacological chemicals of FDY003 and the investigation of their active binding sites, we evaluated the molecular docking scores for target-active chemical pairs. The scores for target–chemical binding interactions were less than or equal to −5.0 (Figure 6A-L and Supplementary Figure S2A-D), suggesting strong pharmacological binding.

Binding affinities of the active pharmacological chemicals of FDY003 and esophageal cancer (EC)-associated targets by molecular docking analysis. (A) Cordycepin-IL6 (score = −6.1). (B) Cordycepin-GAPDH (score = −8.0). (C) Isorhamnetin-AKT1 (score = −6.4). (D) Kaempferol-AKT1 (score = −6.4). (E) Kaempferol-TP53 (score = −8.8). (F) Kaempferol-EGFR (score = −8.2). (G) Luteolin-AKT1 (score = −6.6). (H) Luteolin-TP53 (score = −9.0). (I) Luteolin-EGFR (score = −8.4). (J) Luteolin-VEGFA (score = −7.9). (K) Quercetin-AKT1 (score = −6.6). (L) Quercetin-TP53 (score = −9.1).
Discussion
We elucidated the anti-EC properties of the herbal drug FDY003 by a comprehensive network pharmacological approach. FDY003 reduced the viability of human EC cells and improved the efficacies of anticancer chemotherapeutic drugs. Fifteen active pharmacological chemicals in FDY003 targeted 61 EC-associated genes and proteins involved in the regulation of major oncogenic signaling pathways (eg, the PI3K-Akt, HIF-1, MAPK, TNF, p53, JAK-STAT, ErbB, NF-kappa B, and VEGF pathways). These EC-associated genes, proteins, and pathways targeted by FDY003 contribute to various processes in EC cells, including cell death, survival, division, proliferation, and growth. Overall, the network pharmacological investigation provided integrative and detailed insight into the mechanisms underlying the therapeutic effects of FDY003 in EC.
Key targets of FDY003 that possessed hub-like properties are involved in the initiation and progression of malignant pathological processes in EC and are candidate drug targets with high efficacies for EC treatment.
The pathways by which FDY003 exerts anticancer effects are crucial mediators of EC carcinogenesis and progression. The ErbB, MAPK, p53, and PI3 K-Akt pathways are involved in the risk, tumor-node-metastasis (TNM) staging, lymph node and distant metastasis, tumor progression and development, tumor depth and invasion, tumor stage, and overall and disease-free survival of patients with EC.41–51,64–68 Targeting these pathways may lead to the inhibition of therapeutic resistance, metastasis, migration, survival, invasion, and proliferation of EC cells and tumors.41–51,64–68 The HIF-1 pathway is associated with the modulation of proliferation, treatment sensitivity, death, and survival of EC cells and tumors.69,70 The JAK-STAT pathway participates in the regulation of angiogenesis, inflammation, colony formation, stemness, invasion, motility, metastasis, EMT, cell cycle progression, growth, anoikis resistance, proliferation, migration, malignant formation, and sensitivity to chemotherapy and radiotherapy in EC cells and tumors.71–78 It has also been reported that patient survival, TNM stage, lymphatic metastasis, and degree of infiltration in patients with EC are associated with the expression, genetic, and activation statuses of this pathway.71–78 The NF-kappa B pathway has prognostic value in EC; it represses angiogenesis, metastasis, growth, inflammation, treatment resistance, carcinogenesis, tumorigenesis, and survival of EC cells and tumors. 79 The programmed death-ligand 1 (PD-L1)/programmed death-1 (PD-1) checkpoint pathway may be important for the prediction of disease-free survival, overall survival, cancer relapse, treatment response rate and susceptibility, metastasis, lymph node invasion, tumor stage, and incidence of post-therapeutic adverse effects in patients with EC.80–82 The modulation of this pathway can inhibit immunosuppression, tumorigenicity, self-renewal, immune escape, proliferation, metastasis, EMT, and treatment resistance of EC cells and tumors, and can enhance the antitumor immune response and survival in EC.80–82 The TNF pathway determines the treatment sensitivity, viability, and growth of EC cells and tumors.58–61 It also serves as an indicator of tumor aggressiveness, survival durations, immune status, and complications and symptoms in patients with EC.58–61 The VEGF pathway stimulates various malignant behaviors of EC cells and tumors, such as growth, metastasis, and angiogenesis.62,63
The chemical and herbal components of FDY003, uncovered from the network pharmacological elucidation, have been reported to exhibit anticancer activities against EC. Cm, genkwanin, and isorhamnetin repress the proliferation of EC cells.83–85 Cordycepin intervenes in the activities of the cyclin, B-cell lymphoma 2 (Bcl-2), and extracellular signal-regulated kinase (ERK) pathways of EC cells to decrease their proliferative and survival abilities. 86 Kaempferol promotes EC cell proliferation arrest and apoptosis by regulating Bcl-2-associated X protein (Bax), Bcl-2, caspase, EGFR, and hexokinase-2 cascades. 87 Luteolin suppresses cell cycle progression, growth, survival, EMT, migration, and chemoresistance by the modulation of cyclin, survivin, Myc, p21, p53, poly (adenosine diphosphate [ADP]-ribose) polymerase (PARP), Bim, caspase, SIRT1, c-Jun N-terminal kinase (JNK), and ROS signaling. 88 Quercetin targets cyclin, DNA methyltransferase 1, histone deacetylase 1, caspase, p16, NF-kappa B, growth arrest and DNA damage-inducible protein (GADD)-β, p53-inducible gene 3, p18, E-cadherin, VEGF-A, matrix metalloproteinase (MMP), miR-1-3p/ transgelin-2, and phosphatase and tensin homolog (PTEN) pathways 89 ; these effects result in the suppression of drug resistance, growth, proliferation, invasion, angiogenesis, and cancer stemness of EC cells. 89
The anticancer properties of FDY003 against diverse kinds of cancer cells have been previously investigated.5–8 FDY003 may intervene in the expression and activity of important regulators for determining proliferation, cell cycle progression, cell division, survival and death of cancer cells, such as Bax, caspases, p21, and p53.5–8 This herbal prescription was able to repress the growth rate of human tumors transplanted into xenograft mice,
8
which suggests its
There are several limitations in the present study. First, the pharmacologically active chemicals and targets of FDY003 identified by the network pharmacology methodology were explored based on data analysis and modeling, and which may require experimental validation to enhance the reliability of the network pharmacological findings. Second, this study lacks animal experiments and clinical trials to validate the network pharmacology analysis results. These issues may emphasize the necessity of further experimental researches to (i) validate the presence of active chemicals in FDY003, (ii) evaluate the contribution of individual active chemicals to the pharmacological effects of the herbal drug, (iii) explore the anticancer efficacy of FDY003 in terms of cancer mechanisms other than proliferation and survival/apoptosis, including migration, angiogenesis, invasion, cancer stemness, and immunomodulation of cancer cells, and (iv) assess the safety and effectiveness of FDY003 and its use in combination with current standard anticancer strategies (eg, chemotherapy, cancer immunotherapy, radiotherapy, and targeted therapy) through animal experiments and clinical trials. These efforts would improve the rationality of the present network pharmacological findings.
In conclusion, we conducted a network pharmacological analysis to systematically elucidate the anti-EC properties of the herbal drug FDY003. We found that FDY003 reduces the viability of human EC cells and increases the pharmacological effects of anticancer chemotherapeutic drugs. Furthermore, 15 active pharmacological chemicals targeting 61 EC-associated genes and proteins were identified. These targets of FDY003 were key regulators of the activities of such major oncogenic EC-associated signaling pathways as PI3K-Akt, HIF-1, MAPK, TNF, p53, JAK-STAT, ErbB, NF-kappa B, and VEGF cascades. These FDY003 targets are potent determinants of the malignant behaviors of EC cells, having regulatory roles in the death, survival, division, proliferation, and growth of cells. Further studies are needed to determine the therapeutic effects of herbal drugs and the mechanisms by which they increase the efficacy of anticancer strategies, including surgery, chemotherapy, and radiotherapy.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221105362 - Supplemental material for A Network Pharmacological Elucidation of the Systematic Treatment Activities and Mechanisms of the Herbal Drug FDY003 Against Esophageal Cancer
Supplemental material, sj-docx-1-npx-10.1177_1934578X221105362 for A Network Pharmacological Elucidation of the Systematic Treatment Activities and Mechanisms of the Herbal Drug FDY003 Against Esophageal Cancer by Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, Tae-Wook Kwon and Dae-Yeon Lee in Natural Product Communications
Footnotes
Acknowledgments
Not applicable.
Author’s Contribution
Conceptualization: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee. Methodology: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee. Data collection: Ho-Sung Lee, In-Hee Lee, Kyungrae Kang, Sang-In Park, Minho Jung, Seung Gu Yang, and Tae-Wook Kwon. Data analysis and investigation: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee. Writing: Ho-Sung Lee, In-Hee Lee, and Dae-Yeon Lee. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (grant number 2021R1F1A1049472).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Data Statement
All data generated or analyzed during this study are included in this published article and its Supplemental materials file.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
