Abstract
Bletilla striata is a dried tuber of B striata (Thunb.) Reichb.f. of Orchidaceae plant, which is mainly used for hemoptysis, vomiting blood, trauma bleeding, sore swollen poison, and cracked skin. There have been few research reports on the effect of this herb on cytochrome P450 (CYP), therefore, the study was aimed to investigate the effects of the B striata extracts on the activity of 6 subtypes (CYP2D6, 1A2, 2C19, 2E1, 3A4, and 2C9) using a cocktail method. The B striata extracts were administrated to rats in 0.21 or 0.63 g/kg once a day for 7 or 14 days. The 3 control groups were used to ensure the accuracy of the results. Subsequently, a cocktail of tolbutamide, chlorzoxazone, midazolam, metoprolol, omeprazole, and caffeine was injected. A ultra performance liquid chromatography–tandem mass spectrometer was developed and validated to investigate the concentration of the probes and the pharmacokinetic parameters were calculated to investigate the effects of the extracts on the activity of 6 enzymes under different doses and different dosing periods. The results suggested that the B striata extracts could induce the activities of CYP2D6, 1A2, and 2C19 and could inhibit the activities of CYP2E1, 3A4, and 2C9. When used in combination with drugs that are metabolized by CYP2D6, 1A2, 2C19, 2E1, 3A4, and 2C9, appropriate dose adjustments were needed to avoid toxic side effects caused by drug interactions.
Introduction
Bletilla striata (Thunb.) Reichb.F is a commonly used medicinal plant contained within the Chinese Pharmacopoeia. Its effect is to converge and stop bleeding, reduce swelling, and grow muscles, and is mainly used for hemoptysis, vomiting, traumatic bleeding, sores, swelling, and skin cracks. It belongs to medicine, not to the category of dietary supplements.1,2 At present, there are over 20 products which take B striata as the main medicine, such as Baibai anti-tuberculosis® granules, Baibeiyifei® capsules, and others. As one of the key traditional Chinese medicinal plants being cultivated and developed in Guizhou industry, it has strong regional characteristics, being an advantageous resource, and having the potential for further development. Presently, there have been many studies analyzing its chemical composition and compounds such as bibenzyl, bibenzyl glucoside, biphenanthrene, etc, have been isolated from different parts of the plant. 3 It has pharmacological effects such as hemostasis, antibacterial, antitumor, etc. 4 However, the published literature mainly focuses on the separation and identification of its chemical components as well as the preliminary exploration of various crude extracts of therapeutic substances, there have been few reports on the interaction between B striata and other drugs. Therefore, this study aims to evaluate its effect on cytochrome P450 (CYP) activity in rat by using a Cocktail approach, as well as evaluate its safety, and predict its interaction with other drugs.5,6
CYP is the most important family of liver microsomal mixed-function oxidases and plays an extremely important role in the metabolism of exogenous and endogenous substances.7,8 The activity of the CYPs can be induced or inhibited by many compounds thereby causing drug–drug interactions. 9 Among them, CYP1A2, 2C9, 2C19, 2E1, 2D6, and 3A4 are the 6 most important CYP enzymes, accounting for ∼80% of the total amount of CYP enzymes in the liver and being responsible for the metabolization of >90% of clinical drugs.10,11 Worldwide, the effect of drugs on CYP enzyme activity has been used as an important basis for evaluating the safety of drug combination, combination of food and medicine, and combination of herbs. 12
The cocktail approach for studying drug effects indirectly reflects the activity of various CYP enzymes by determining the blood drug concentration of specific probes in vivo. 13 Due to rapidity, accuracy, and high sensitivity, it has become an effective method to study the effect of drugs on CYP activity as well as drug interactions.13,14 A probe is a substance that is catalyzed by either only 1 specific enzyme or by several enzymes, but whose metabolic pathways and their products are well known. As such, the activity of the enzyme is evaluated by measuring its metabolic rate. According to previously published literature, this study has selected caffeine, tolbutamide, omeprazole, metoprolol, chlorzoxazone, and midazolam as specific probes of enzymes of CYP1A2, 2C9, 2C19, 2D6, 2E1, and 3A4, respectively.15-23
Results and Discussion
Determination of Main Components in B striata
The main indicator components in the effective fraction of B striata are α-isobutylmalic acid (B6), blestroside (B12), militarine (B14), dihydrophenanthrenes 1 (B19), and gymnosides Ⅸ (B23). The content of each component in the extracts was B6 (2.36%), B12 (2.64%), B14 (26.37%), B19 (0.86%), and B23 (2.09%).24,25
Method Validation
According to the methodological evaluation guidelines of Food and Drug Administration, specificity, linearity, low limit of detection, precision, stability, recovery, and matrix effect were investigated, and the pharmacokinetics parameters of 6 probe drugs were determined using this method. Specific results were shown in Figure 1 and Tables 1 to 4.

Typical chromatograms. (A) Blank plasma. (B) Blank plasma spiked with 6 probes and IS. (C) Rat plasma sample collected at 5 min after intravenous administration of 6 probes: (1) puerarin, (2) metoprolol, (3) omeprazole, (4) caffeine, (5) midazolam, (6) chlorzoxazone, and (7) tolbutamide.
Standard Curve and LLOD of 6 Probe Drugs in Rat Plasma.

Abbreviations: SD, standard deviation; n, number of replicates.
Stability of the 6 Probes in Rat Plasma Under Various Storage Conditions (mean ± SD, n = 5).

Abbreviations: SD, standard deviation; n, number of replicates.
As shown in Figure 1, the endogenous substances in the plasma did not interfere with the determination of the analyzed components from the retention time, indicating that the experimental conditions had good specificity.
The linearity was evaluated by analyzing standard curves by weighted least squares linear regression at each concentration on 3 days. The accuracy and precision were assessed by analyzing quality control (QC) samples in 6 replicates at 3 concentrations on 3 days. Standard curves indicated good linearity within the concentration ranges. As shown in Tables 1 and 2, all parameters met the requirements, suggesting that the method used was accurate, reliable, and reproducible.
Precision and Accuracy of UPLC-MS Method for the Assay of 6 Probes in Rat Plasma (mean ± SD, n = 6).

Abbreviations: SD, standard deviation; n, number of replicates.
The extraction recoveries and matrix effect were determined at 3 QC levels. The average extraction recovery rates of 3 different concentrations, as shown in Table 3, were between 84.3% and 105.3%, and the matrix effect of the 5 probe drugs was between 84.7% and 106.0%, indicating that the extraction yield of each indicator component was good and there was no obvious matrix effect.
Extraction Recoveries and Matrix Effect of the 6 Probes in Rat Plasma (Mean ± SD, n = 5).
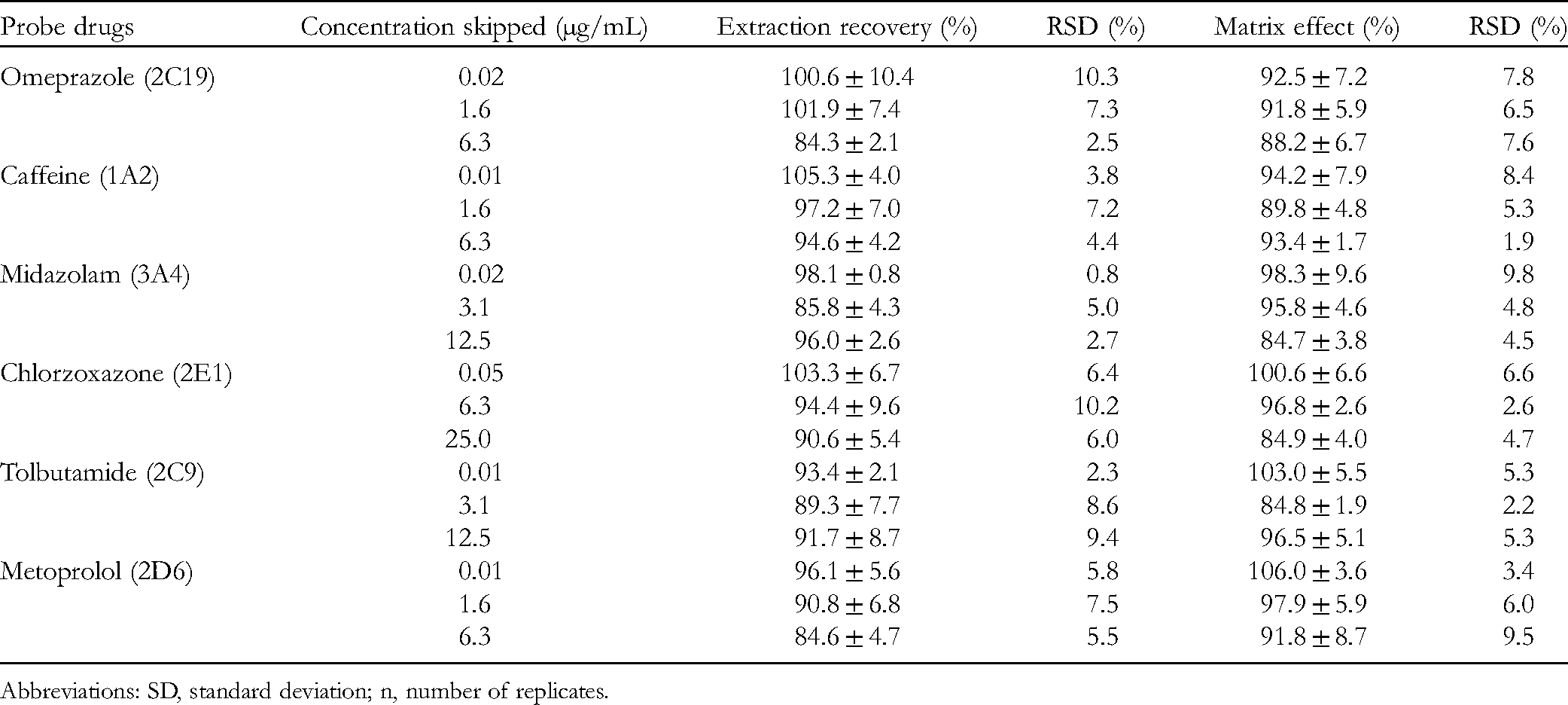
Abbreviations: SD, standard deviation; n, number of replicates.
Stability was analyzed by placing the QC samples of 3 concentrations under different conditions. The stability rate, as shown in Table 4, was ≤ 10.61%, indicating that rat plasma samples containing probes were stable under the different conditions.
Effects of the B striata Extracts on the 6 cytochrome P450
The pharmacokinetic parameters of caffeine, metoprolol, omeprazole, midazolam, chlorzoxazone, and tolbutamide in the 4 treatment groups were used to describe the effect of the B striata extracts on CYP1A2, 2D6, 2C19, 3A4, 2E1, and 2C9, respectively. The results were shown in Table 5 and Figure 2.

Mean-time curve of blood concentration in the BCG and the experimental group after intravenous administration of cocktail probe drugs (n = 6).
Pharmacokinetic Parameters Describing the Disposition of 6 Probes After Intravenous Administration (n = 6).

Note: *P <0.05, **P <0.01 compared with the BCG.
Abbreviations: SD, standard deviation; n, number of replicates; BCG, blank control group; DCG, inducer control group; HCG, inhibitor control group.
When compared with blank control group (BCG): Under the index of caffeine, AUCs decreased significantly in 7d-H and 14d-H, while mean residence times (MRTs) increased distinctly in 14d-L and 14d-H, and CL z significantly increased in 7d-H and 14d-H. Under the index of metoprolol, areas under the curve (AUCs) were significantly reduced in some treatment groups, while CL z increased distinctly in 7d-L and 7d-H. Under the index of omeprazole, AUCs were found to be significantly decreased in 7d-H and 14d-H, while t1/2z was significantly decreased in all 4 treatment groups. Moreover, CL z increased significantly in 7d-H and 14d-H. Under the index of midazolam, the AUCs of each experimental group increased significantly, while the CL z was significantly decreased in all 4 treatment groups. Under the index of chlorzoxazone, AUCs were significantly increased in some groups. Moreover, CL z s in 14d-L and 14d-H were found to be significantly decreased. Under the index of tolbutamide, AUCs, t1/2zs were significantly increased in some groups. In addition, CL z was found to be significantly decreased in 3 groups. These results were statistically significant, indicating that the B striata extracts had an induction effect on CYP1A2, 2D6, and 2C19 and had the potential to inhibit 3A4, 2E1, and 2C9.
Effects of Different Dosing Periods and Dosage of the B striata Extracts on the Pharmacokinetic Parameters of Probes
The pharmacokinetics parameters of 6 probes in the 4 experimental groups were analyzed by 2 × 2 factorial analysis of variance in order to determine the effects of the dosing period and administered dosage of the B striata extracts. Results were shown in Table 6. Under the index of dosing period, P values of some parameters in 6 probes were significantly different. When the dose administered was different, P values of some parameters in 6 probes were significantly different. And all probes except metoprolol were subject to period and dosage interactions.
Factorial Analysis of Variance (2 × 2) for the Effects of Dosing Period and Administered Dosage.

Since human beings and rats possess orthologous CYP enzymes, CYP1A2, 2C9, 2C19, 2D6, and 2E1 are expressed in both species, while rat CYP3A1/2 is equivalent to human CYP3A4. Therefore, in this article, CYP3A4 is used to represent rat CYP3A1/2. CYP1A2 is specifically expressed in liver tissue and is one of the important liver P450 oxidases, accounting for about 13% of the total liver P450 oxidase content, and its content is ranked third after CYP3A and CYP2C subfamily. CYP2D6 is an important member of the CYP450 family, which is mainly distributed in liver, small intestine, and brain tissue. Although it only accounts for 4% of the total liver CYP enzyme protein, it can metabolize about 30% of the commonly used clinical drugs.26-28
The Cocktail method was chosen in this study as multiple data could be obtained for the same animal, thereby reducing experimental variations due to differences between individuals. 29 Based on the previous results obtained in vitro, we simultaneously set up inducer control group (DCG) and inhibitor control group (HCG) together with treatment groups and BCG. In order to improve the accuracy and reliability of the results, the pharmacokinetic parameters of BCG were compared with DCG and HCG. Our results (Table 5) showed that phenobarbital could induce CYP1A2, 3A4, 2E1, and 2C9 activity, while ketoconazole could inhibit CYP3A4. However, CYP2C19 and CYP2D6 were not affected by either compound. These results are consistent with previously published literature, 30 indicating that the Cocktail method was successful and could be used to study the effect of the B striata extracts on CYPs activity.
The results suggested that the B striata extracts might induce CYP1A2, 2D6, and 2C19 activity indicating that B striata combined with the drugs metabolized by these 3 enzymes might accelerate the metabolism of the combined drug and decrease the blood drug concentration, thereby reducing their therapeutic effect. While the extracts could inhibit CYP3A4, 2E1, and 2C9 activity indicating that when combined with the drugs metabolized by these 3 enzymes, the metabolism of the combined drugs might be slowed down, and the blood concentration might be increased, thereby increasing the therapeutic effect or possibly causing side effects. The parameters of each treatment group of each index were analyzed by factorial analysis of variance, which revealed that the “dosing period” and “administration dosage” had an effect on the induction or inhibition of each of the above enzyme. Moreover, it was shown that they had a specific interaction effect on 5 enzymes, other than CYP2D6. It is well known that many of the drugs commonly used in clinical practice are metabolized by these 6 enzymes, including antipyrine, ibuprofen, clomipramine, amitriptyline, paracetamol, carbamazepine, and others.18,31,32 Therefore, in order to ensure the efficacy and safety of these drugs when used in combination with the B striata extracts, the dosage and administration period of the drug should be appropriately adjusted.
Experimental
Plants and Chemicals
The tubers of B striata were collected from Zheng’an of Guizhou Province and identified by Professor Qinde Long from the Department of Pharmacognosy, Guizhou Medical University, Guiyang, China. A voucher specimen (BJ20160718) was deposited at the herbarium of Guizhou Medical University.
Puerarin reference substance (0752-9605) was purchased from the China National Institute for the Control of Pharmaceutical and Biological Products. Omeprazole, tolbutamide, chlorzoxazone, caffeine, ketoconazole, and metoprolol (purity > 98%) were purchased from Dalian Meilun Biotechnology Co., Ltd. Midazolam and phenobarbital (purity > 98%) were obtained from Fujian Mindong Rejuvenation Pharmaceutical Co., Ltd. The chromatography methanol was purchased from Tianjin Kemiou Chemical Reagent Co., Ltd and the chromatography acetonitrile was purchased from Merck. All other reagents were of analytical grade and were obtained from Tianjin Chemical Reagent No.1 Factory.
Animals and Treatment
Healthy male and female Sprague-Dawley (SD) rats, of SPF grade, and with a weight of 250 ± 20 g were provided by Changsha Tianqin Biotechnology Co., Ltd. The rats were randomly divided into 7 groups (n = 6): BCG, DCG, HCG, and 4 treatment groups. BCG received an intragastric administration of 1% sodium carboxymethylcellulose solution, which was the same volume as the extracts, once per day for 7 days. DCG was intraperitoneally injected with a phenobarbital injection of 50 mg/kg per day for 7 days. HCG received a ketoconazole solution of 100 mg/kg once a day for 7 consecutive days. The 4 treatment groups were 7d-L, 7d-H, 14d-L, and 14d-H, with each group receiving either a low dose of 0.21 or a high dose of 0.63 mg/kg of the B striata extracts for 7 or 14 consecutive days, respectively.2,24,33
The rats in each group fasted for 12 h following the first administration, and received water ad libitum. On the second day, the rats were injected with the Cocktail in the tail vein. At 0.083, 0.167, 0.333, 0.667, 1, 1.5, 2, 3, 5, 8, 12, and 24 h after administration, ∼0.3 mL of blood was drawn from the tail vein, and then placed in a heparin-coated centrifuge tube. The blood was centrifuged at 6000 rpm for 10 min, following which 100 μL of plasma were separated and stored in a refrigerator at −20 °C until analysis.
Preparation of the B striata Extracts
B striata was refluxed 3to 4 times the amount of 95% ethanol for 2 h/time and then filtered. The filtrate was combined, concentrated under reduced pressure, and then the D101 macroporous adsorption bar was eluted with 80% ethanol. The eluate was collected, concentrated under reduced pressure, and the residue was dried under a microwave vacuum to give an effective fraction. The yield of the obtained paste was 6.82%.24,34,35
Preparation of Plasma Samples
A volume of 100 μL of rat plasma was placed in a 1.5 mL plastic centrifuge tube and mixed with 100 μL of 1.0 μg/mL internal standard solution. The mixture was vortexed for 1 min, followed by the addition of 400 μL of methanol. Subsequently, the mixture was vortexed for 3 min, sonicated for 10 min, and then centrifuged at 12 000 rpm for 10 min. The supernatant was placed in a centrifuge tube, air dried at 45 °C, dissolved in 400 μL of 50% methanol solution, and then centrifuged at 12 000 rpm for 10 min. The supernatant was subsequently subjected to ultra performance liquid chromatography–tandem mass spectrometer (UPLC–MS) analysis.
UPLC–MS Analysis
Samples were analyzed by UPLC–MS. Chromatographic separation was performed using a Waters BEH C18 (2.1 mm × 50 mm, 1.7 μm) column with a Waters Van Guard BEH C18 (2.1mm × 5 mm, 1.7 μm) as a guard column. The separation flow rate and column temperature were 0.35 mL/min and 45 °C. The mobile phase was treated with 0.1% formic acid acetonitrile (A) to 0.1% aqueous formic acid (B), and the injection volume was 6 μL. The gradient elution procedure was as follows: 90% B (0 min), 35% B (3 min), 10% B (3.5 min), 90% B (4.5 min). A Waters TQD Quantum triple-quadrupole mass spectrometer (Waters Co., Ltd) equipped with an electrospray ionization source was used for mass analysis and detection.
The mass spectrometer was operated in positive or negative mode, with the main working parameters set as follows: the capillary voltage, 3 kV; the ion source temperature, 120 °C; the desolvation gas temperature, 50 °C; the dissolvent gas, N2 (650 L/h); the backflush gas, N2 (50 L/h); the collision gas, Ar (0.16 mL/min). The mass spectrometry data acquisition and processing software used was the MassLynx V4.1 workstation, and the selected single ion recording (SIR) mode was chosen for quantification of the probes. Mass spectrometry conditions were shown in Table 7.
Specific Mass Spectrometric Parameters of Single Ion Recording of the Probe Drugs and Puerarin (IS).

Statistical Analysis
The main pharmacokinetic parameters of six probes, including AUCs, MRTs, t1/2z, CL z , and Vz, were calculated using the noncompartment model of the DAS 2.0 software (Chinese Pharmacological Society). The results were expressed as a mean ± SD. Statistical analysis was performed using the SPSS software (IBM SPSS Statistics 22 Developer, IBM Corp.). Factorial analysis of variance was used for comparison between groups and Dunnett's test was used for pairwise comparison. P < .05 was considered statistically significant, and P < .01 indicated a very significant difference.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Key R&D Program of China (grant numbers 2018YFC1708100); the National Natural Science Foundation of China (grant numbers U1812403/81460630); Guizhou Science and Technology Department (grant numbers [2019]5641, [2018]4006); Guiyang Science and Technology Bureau (grant number [2017]30-29); and Guizhou Education Department (grant number KY [2018]050]).
