Abstract
Presently, South Africa is one of the world’s largest producers and exporters of high-quality cannabis and day by day is becoming the main African regional hub for cannabis cultivation and trading.
5
The South African government and medical research community are still waiting for the scientific validation of the medicinal claims of the plant prior to approving legalization of medical marijuana in the country.
8
-10
Therefore, scientific validation and chemical characterization of the African variety of
In the present study, the African variety of
Three extracts were generated from authenticated
The
The peaks which correspond to the molecular formula and exact masses of the three compounds in the DCM extract in negative ESI mode are shown in Figure 1(a). The peaks with retention times (minutes) of 11.31,

UPLC chromatogram of the DCM extract: (a) ESI negative mode; (b) ESI positive mode.
In the UPLC chromatogram of the methanol extract, one peak with a specific retention time of 5.99 minutes (
3-(4,5-Dimethylthiazol-2-yl)-−2,5-diphenyltetrazolium bromide (MTT) assay was performed to determine the cytotoxicity to MDA-231 cells of the DCM extract after 24 and 48 hours of exposure to the extract. The extract did not show any major effect toward the MDA cancer cell line (5 × 104 cell densities) after 24 hours of exposure, although a concentration-dependent reduction of cell survival was observed (10-100 µg/mL). An IC50 value of 76.6 ± 15.2 µg/mL was determined. After 48 hours of exposure, the DCM extract remarkably inhibited the breast cancer cell growth in a concentration-dependent manner and an IC50 value of 27.8 ± 5.0 µg/mL was determined (positive control, doxorubicin, IC50 at 24 hours 50.3 ± 6.1 and at 48 hours 19.7 ± 4.7 µg/mL).
The DCM extract of
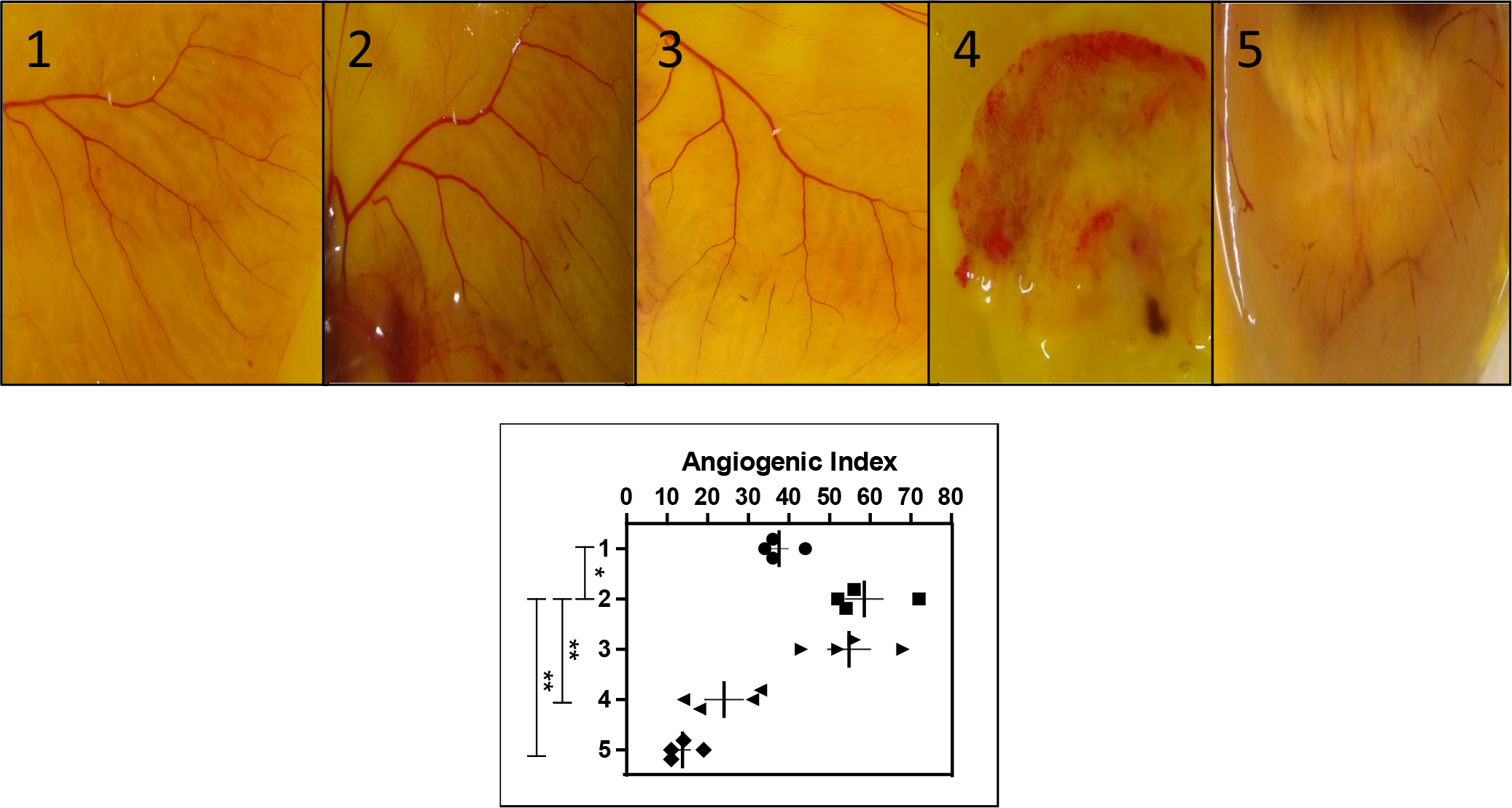
(a) Representative figures of CAM assay (
The identification of active lead molecules is an important requirement for quality control and dose determination of plant-related drugs.
11
LC and MS have been applied widely for the quality control of traditional medicine.
12
The present study aimed to characterize the African “Dagga” variety of
UPLC-MS analysis was performed using electro spray ionization; data recorded included retention times and iFit% confirmations in negative and positive modes. Both the DCM and methanol extracts showed the presence of the three marker compounds (CBD, THC, and THCA), although CBD and THC have the same accurate mass and hence are non-distinguishable through MS analysis. In the DCM extract, two similar peaks appeared at two different retention times with the same accurate mass as THCA, which may confirm the presence of a similar THCA isotope. The percentage of the marker compounds was higher in the DCM extract than the methanol extract. The DCM extract therefore was selected for further studies on breast cancer cell survival and progression assay.
Breast tumors are one of the most common human neoplasms and one of the leading causes of death among women.
13
Although the antiproliferative effect of cannabinoids on different tumor cells has been extensively confirmed both in vitro and in vivo,
14
a recent report indicates that THC may enhance breast cancer cell growth under certain circumstances.
15
The African “Dagga” variety of
Cancer cells need ample blood supply to enable metastatic processes and cell progression and, therefore, angiogenesis is the key regulator for extreme metastatic cancers like breast cancer.
16
To check the efficacy of
Tumor cells always establish angiogenesis by secreting various angiogenic factors like VEGF, MMPs, NO, and IL-8. 17 Inhibition of vasculature rather than inhibiting the tumor cells directly is considered as the most reliable approach to antitumor research these days. 18 In vitro and in vivo experiments showed that cannabinoids directly inhibit vascular endothelial cell migration and survival, as well as decrease the expression of proangiogenic factors like VEGF and MMPs. 19
Experimental
Collection, Authentication and Extraction
UPLC-MS Analysis
Compound separation and detection was performed using a Waters UPLC system with a Waters Synapt G2 QTOF instrument. Only the DCM and MeOH extracts were analyzed through UPLC-MS. The DCM extract was dissolved in 80% acetone (ACN) and 20% water, while the methanol extract was dissolved in 80% MeOH and 20% water. Approximately 1 mg of each extract was analyzed.
The analysis was accomplished using an Acquity UPLC BEH C18 1.7dµm (2.1 × 100 mm column). Gradient mobile phases were used: A, water: 0.1% formic acid; and B, ACN + 0.1% HCO2H. The elution rate was 0.400 mL/min. Gradient elution was used for the separation of the extracts. Elution started with 97% A and 3% B; this remained linear until 14 minutes, then from 14 to 16 minutes, elution was kept constant with 0% A and 100% B; a linear gradient of 97% A and 3% B was then used to reach completion after 20 minutes. Both negative and positive modes were used in the MS analysis. Data were accumulated using Software Masslynx V4.1.
Cytotoxicity in MDA-231 Breast Cancer Cells
The strongly metastatic breast cancer cell line MDA-MB-231 was grown in Dulbecco’s modified Eagle’s medium (ATCC® 30-2002™) supplemented with 10% fetal bovine serum (Hyclone, Thermo Fisher Scientific Inc, USA). The cells were maintained at 37°C in an atmosphere containing 5% CO2. Cell growth was measured under a light microscope and 70% to 80% confluence of the cells was used in all experiments. MDA-231 breast cancer cells at cell densities of 5 × 104 were seeded in 96/48 well tissue culture plates and the cells were allowed to adhere for 24 hours at 37°C in the CO2 incubator. On the next day, the cells were exposed to different concentrations of extracts (0–100 µg/mL) and doxorubicin (0–100 µg/mL) for 24 and 48 hours, respectively. A cell viability assay was then performed using MTT assay. 20 Data were analyzed by plotting % cell survival vs concentration, and IC50 values were calculated in the plot of % inhibition vs concentration using Graph Pad Prism software version 5.
VEGF-Induced Angiogenesis Assay in Chick Chorioallantoic Membrane
Twenty fertile chicken eggs were obtained from a local farmer in Bloemfontein, South Africa, and incubated (“Prolab” Model PL-0001) at 37ᵒC ± 1ᵒC. Sixty percent humidity was maintained by keeping a tray full of sterile distilled water in circulating air condition. On day 8, approximately a 1 cm2 hole was prepared on the surface of the egg shells in aseptic conditions and 1 cm2 (diameter) Whatman filter paper (Sigma-Aldrich, USA) shocked with VEGF (0.2 nM/CAM)/L-Arg(10 µg/CAM)/extract (20–40 µg) as per grouping, inserted and sealed with adhesive tape. Tinzaparin, at 20 µg/CAM, was used as a positive angiogenesis inhibitor in the experiment. All the eggs (
Statistical Analysis
Statistical significance (
Footnotes
Acknowledgement
Authors are thankful to NRF and DST-IKS Based Technology, South Africa (Research Development and Innovation on Medical Cannabis Project Funding agreement Number: DST/CON 0162/2015), for financial support and University of the Free State for institutional support. Dr Asis Bala sincerely acknowledges the University of the Free State, Bloemfontein, South Africa for a Post-Doctoral Research Fellowship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NRF and DST-IKS Based Technology, South Africa (Research Development and Innovation on Medical Cannabis Project Funding agreement Number: DST/CON 0162/2015).
