Abstract
A simple, rapid, and selective ultra-performance liquid chromatography-mass spectrometry (MS)/MS method was established to investigate tissue distribution of 6 polyphenolic compounds of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat tissues, including isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid. An Agilent Eclipse Plus C18 column was used. The mobile phase consisted of acetonitrile and water, both with 0.1% formic acid. Quantification was performed in negative ion multiple reaction monitoring mode. All the analysts had good linearity with r ≥ 0.9912. Accuracy ranged from 12.49% to −13.98% for the 6 compounds; within-day variation (precision) was ≤9.98% and interday precision was ≤11.88%. Extraction recovery of the analysts ranged from 80.55% to 99.92%; the matrix was 81.00%–98.73%. The analyst preparations were stable throughout. The 6 compounds were rapidly distributed in various tissues after oral administration, without accumulation over 12 hours. Compared with normal rats, distributions of 6 compounds in the heart, liver, spleen, lung, kidney, brain, stomach, and intestine in MI model rats were different from those in the normal group. The study provides an insight for further research of P. orientale L.
Introduction
Polygonum orientale L. is a member of the Polygonaceae family and is widely distributed in the southwest and southeast subtropical regions of China. As a traditional medicine, it has been described as an effective treatment for rheumatoid arthritis and coronary heart disease. 1 -3 Previous pharmacological studies have found that P. orientale L. extract (POE) reduced myocardial infarction (MI)-related damage and increased the vascular transport of nutrients to cardiac muscle. 4,5 The Hongye Xintong Soft Gel, which is prepared from P. orientale L., is widely used in clinical practice. 6 The pharmacological effects of P. orientale L. are mediated by polyphenols, such as isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid. 7 -9 These components possess multiple biological activities, including antioxidative, anti-inflammatory, antitumor, and neuroprotective effects. 10,11
Currently, most studies focus on the pharmacological effects and content determination of POE. 2,7 In addition, some studies have revealed the pharmacokinetic characteristics of POE in normal animals. 1 Several analytical methods, including spectrophotometric methods and high-performance liquid chromatography (HPLC) with ultraviolet detection, have been developed in order to quantify isoorientin, orientin, vitexin, and quercitrin, in biological samples. 1 -3 However, few reports were related to tissue distribution of isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid in normal rats and myocardial ischemia (MI) condition rats after oral administration of POE. Pathological state affected absorption, tissue distribution, and metabolism of drugs in vivo. 12,13 The pharmacokinetic characteristics in disease condition are different from those in the normal condition, which are directly related to the efficacy and adverse reactions of drugs. 14,15 It is thus necessary to compare the tissue distribution characteristics of drugs under normal and pathological conditions.
In the present study, an ultra-performance liquid chromatography–tandem mass spectrometry (UPLC–MS/MS) was established and validated. The newly developed UPLC–MS/MS method was successfully applied to compare the content of 6 POE components in normal and MI model rat tissues after oral administration. The results might provide insight for further research on P. orientale L.
Experimental Methods
Reagents
Puerarin was purchased from Shanghai Yuanye Bio-Technology Co., Ltd. (Shanghai, China). Isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid were provided by Sichuan Victory Biological Technology Co., Ltd. (Sichuan, China). HPLC-grade formic acid and acetonitrile were obtained from Merck & Co. (Darmstadt, Germany). HPLC-grade methanol was provided by Tianjin Kemiou Chemical Reagent Co. (Tianjin, China). Isoproterenol was purchased from Aladdin Industrial Corporation (Shanghai, China).
Polygonum orientale L. was obtained from Panzhou (Guizhou, China) in 2017 and identified by Professor Chun-hua Liu in Guizhou Medical University. A voucher specimen (No. 20171101) was deposited in Guizhou Provincial Key Laboratory of Pharmaceutics (Guizhou Medical University, Guiyang, China). Polygonum orientale L. was stored in dry conditions at room temperature and protected from light.
Preparation of POE
POE was prepared as previously described by Huang et al. 16 Briefly, the dried medicinal parts of P. orientale L. (10 kg) were extracted 3 times with water (100 L each time) under reflux for 1 hour. The solutions were combined, filtered, and vacuum concentrated to form a crude extract with a concentration of 1 g/mL. Then, 95% ethanol was added with stirring to obtain a final concentration of 65%. After 12 hours, the solution was filtered and vacuum concentrated to form a crude extract with a concentration of 1 g/mL. The filtrate was then extracted 4 times with 5 L of water-saturated n-butanol. The combined solutions were vacuum evaporated. The residue was dissolved in 80% ethanol and transferred to a polyamide column for chromatography analysis involving successive elutions with water and 80% ethanol. The ethanol eluate was vacuum dried to obtain a flavonoid-enriched extract (500 g). The contents of isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid in POE were 3.550%, 3.906%, 0.490%, 1.696%, 0.0570%, and 0.509%, respectively. The POE powder was dissolved in 0.5% carboxymethyl cellulose-sodium solution to form a 0.266 g/mL suspension.
Animals and MI Model Development
Sixty male Sprague-Dawley rats weighing 250 ± 20 g were provided by Changsha Tianqin Biotechnology (SCXK [Xiang] 2014–0011). The rats were housed in an environmentally controlled breeding room with free access to standard rat feed and water. The study protocol and procedures were approved by the Animal Ethics Committee of Guizhou Medical University (permit 1702077). The rats were randomly assigned in equal numbers to a MI model and a control group. MI model rats were injected subcutaneously with 50 mg/kg of isoproterenol (ISO) once daily for 2 consecutive days. Control rats were given the same volume of saline. To assess whether the MI model was successful, the rats were anesthetized with isoflurane vapor (1%-2%) 24 hours after the last ISO injection. Following confirmation of ST-segment elevation in the electrocardiogram (ECG), the rats were sacrificed and the hearts were collected to assay the content of adenosine triphosphate (ATP). Myocardial damage was determined in tissue stained with hematoxylin and eosin (HE). 17,18
Instrumentation and Chromatographic Conditions
A Waters Acquity UPLC system (Waters Corp., Milford, MA, USA) was interfaced with a Waters Xevo TQ/MS (Waters) equipped with Turbo Ion Spray electrospray ionization source. Separation of the analyte was performed on a 2.1 × 50 mm, 1.8 µm Agilent Eclipse Plus C18 column. The column temperature was maintained at 40°C during the analysis. The mobile phase consisted of acetonitrile (solvent A) and water (solvent B), both of which contained 0.1% formic acid. The flow rate was 0.3 mL/min. The gradient elution procedure was used according to the following profile: 10% A, 0.0-0.5 minutes; 10%-19% A, 0.5-1.0 minutes; 19% A, 1.0-3.0 minutes; 19%-90% A, 3.0- 3.1 minutes; 90% A, 3.1-3.5 minutes; and 90%-10% A, 3.5-4.0 minutes. The injection volume was 1 µL. MS analysis was performed in negative ion mode using multiple reaction monitoring (MRM). The optimized MS ionization source settings included a temperature of 150°C, capillary voltage of 3 kV, collision energy, and cone voltage are shown in Table 1. The raw data were processed with a MassLynx V4.1 workstation (Waters).
Mass Spectrometry Settings.
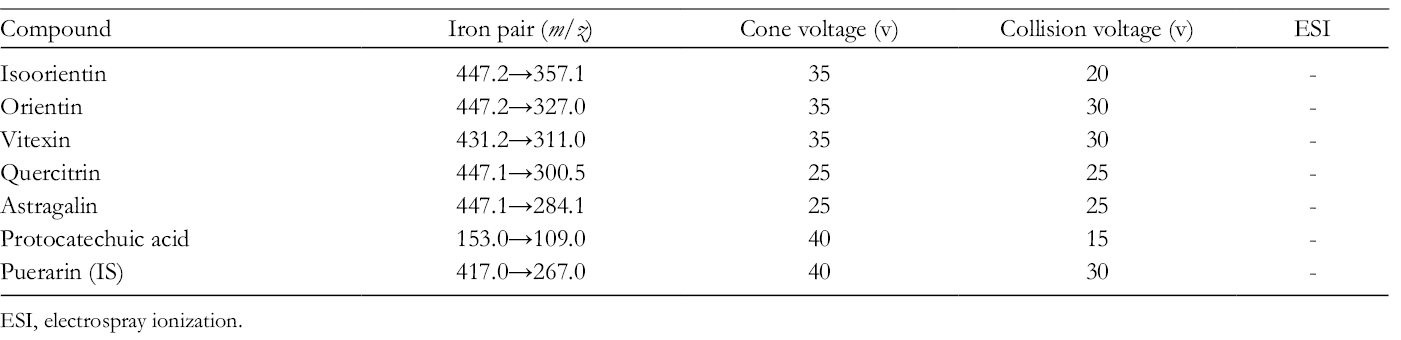
ESI, electrospray ionization.
AUC of Isoorientin, Orientin, and Vitexin After Intragastric Administration of POE in Normal and MI Rats (N = 6).
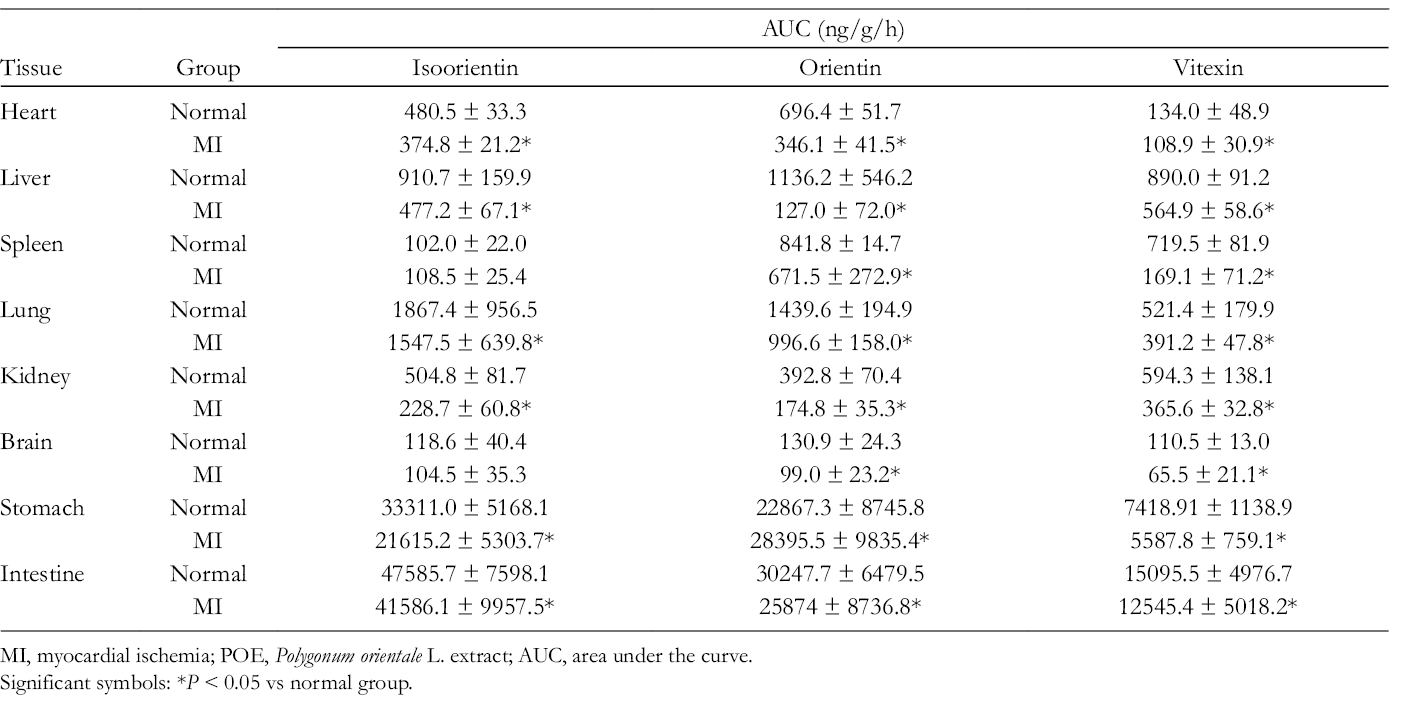
MI, myocardial ischemia; POE, Polygonum orientale L. extract; AUC, area under the curve.
Significant symbols: *P < 0.05 vs normal group.
AUC of Quercitrin, Astragalin, and Protocatechuic Acid After Intragastric Administration of POE in Normal and MI Rats (N = 6).
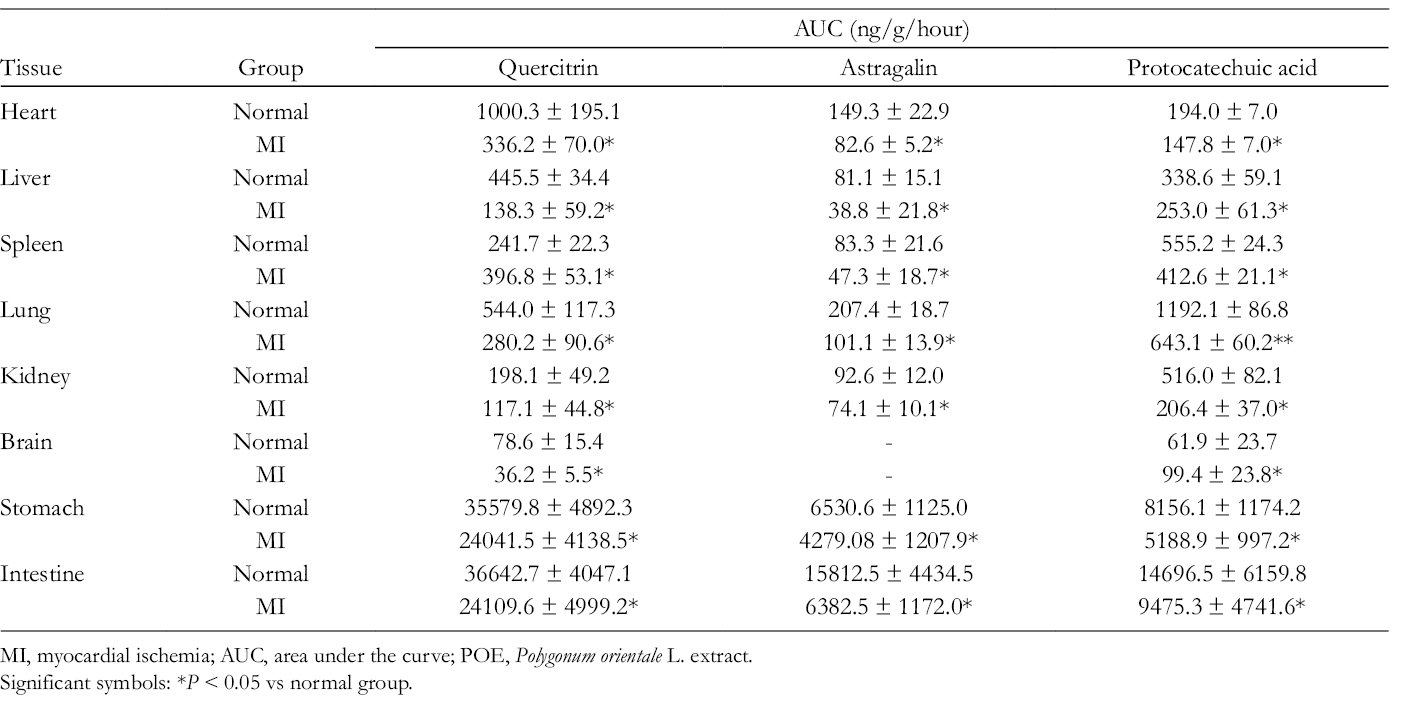
MI, myocardial ischemia; AUC, area under the curve; POE, Polygonum orientale L. extract.
Significant symbols: *P < 0.05 vs normal group.
Sample Preparation
Heart, liver, spleen, lung, kidney, brain, stomach, and intestine tissue samples (1 g) were homogenized in 2 mL saline containing 10% methanol and centrifuged for 10 minutes at 6738g. The supernatant (300 µL) was combined with 10 µL of formic acid in water (1:10, v:v) and 10 µL of puerarin(10 µg/mL) was added. The mixture was vortexed for 5 minutes and 1200 µL of acetonitrile was added. The sample homogenate was centrifuged at 18 000g for 10 minutes. The supernatant was transferred to a clean Eppendorf tube and evaporated under a nitrogen stream at 40°C. The residue was dissolved in 150 µL of methanol:water (50:50, v:v) solution and vortexed for 5 minutes. The samples were centrifuged at 18 000g for 10 minutes. A 1 µL aliquot was injected into the LC–MS/MS system for analysis.
Preparation of Standard Solutions, Calibration Standards, and Quality Control Samples
Stock solutions of isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid were prepared with methanol at a concentration of 1 mg/mL. Mixed stock solutions of 6 polyphenols were serially diluted with methanol to obtain working standard solutions of desired concentrations (shown in Supplemental Table S1). Stock solution of internal standard (IS) was prepared by dissolving puerarin (10.04 mg) in methanol to obtain a concentration of 1.04 mg/mL. Then, the stock solution of IS was diluted by methanol to obtain a concentration of 10 µg/mL. All of the solutions were stored at −20°C until used. Calibration standards were prepared by combining 50 µL of each standard series concentration, 10 µL of IS, and 1200 µL of acetonitrile with 300 µL of blank tissue homogenate. The mixture was processed as described in the section Sample Preparation to obtain the calibration curves. Eight concentrations were included in each calibration curve. Quality control samples were prepared in the same way to obtain low, middle, and high concentrations of each component. All of the samples were stored at −20°C until used.
Method Validation
The proposed method was validated by determining specificity, precision, accuracy, matrix effect, extraction recovery, and stability in accordance with the “Guidance for Industry Bioanalytical Method Validation” of the US-Food and Drug Administration. 19
Specificity
The specificity of the analysis method was determined by analyzing homogenates of drug-free tissue, homogenates of drug-free tissue containing standard solutions and IS, and homogenates obtained after oral administration of POE. The procedure also verified the absence of endogenous substances that interfered with the assay within the range of retention time of each analyte and IS.
Linearity and lower limit of quantification
The linearity of the calibration curve was demonstrated using 8 tissue concentrations. Briefly, peak area analyte/IS ratios (Y) were tested against the theoretical concentration of each analyte (X) using 1/X 2 weighting of the linear regression. The lower limit of quantification (LLOQ) was defined as the lowest concentration in the calibration curve.
Accuracy and precision
Five low-concentration, middle-concentration, and high-concentration samples were evaluated in 3 successive days to determine intraday and interday accuracy and precision. Precision was reported as the relative standard deviation (RSD) and accuracy was expressed as a relative error (RE, %).
Recovery and matrix effect
The extraction recovery of the 6 compounds was assessed by comparing the peak areas of every analyst extracted from QC tissue samples at low, middle, and high concentrations with those extracted from drug-free tissue samples post-spiked with mixed standard solutions at the same concentrations. The matrix effect was reported as the ratio of the peak area of analysts spiked with drug-free tissue samples to those of pure standard solutions at low, middle, and high concentrations of analysts in methanol.
Stability
The stability was determined by analyzing QC samples at 3 concentrations (n = 5 assays for each concentration) under the following conditions: (1) stored samples in auto-sampler at 6°C for 6 hours; (2) 3 freeze-thaw cycles from –20°C to room temperature.
Tissue Distribution
For tissue distribution study, POE (3.2 g/kg) was orally administered to each rat. After oral administration, heart, liver, spleen, lung, kidney, brain, stomach, and intestine tissues were harvested at 0.16, 1, 2, 6, and 12 hours (6 rats per each time point in the normal and MI model groups). The tissue samples were washed with physiological saline and dried with filter paper. The tissue samples were stored at −80°C until used.
The drug concentration–time curve was prepared with GraphPad Prism 5 (https://www.graphpad.com/scientific-software/prism/) and the maximum drug concentration (C max) and time to C max (T max) were obtained directly from the mean concentration–time curve. The AUC0–t (area under the drug concentration-time curve from 0 to the last measurable concentration point) was calculated using WinNonlin 6.4 software program. Data analysis was performed with SPSS 22.0 (IBM Corp., Armonk, NY, USA). Data were expressed as the means ± standard deviation. P < 0.05 was considered to be statistically significant.
Results and Discussion
Preparation of the MI Model
The MI model was induced by a large dose of ISO. ISO is a β-adrenoceptor agonist that can generate cytotoxic free radicals, which may cause severe myocardial damage by peroxidation of membrane phospholipids. 17,18 It has many advantages, including simple modeling, minimal trauma, and high survival. 20 To determine the best condition for establishing the MI model, rats were injected subcutaneously with 50 mg/kg ISO on 2 consecutive days. Changes in the ECG, HE staining, and the content of ATP were evaluated at 24 and 72 hours and 1 week after the last injection of ISO. The results are shown in Supplemental Figures S1-3. Compared with the normal rats, the ST segment of the ECG in the MI model was elevated at 24 and 72 hours and 1 week (Supplemental Figure S1). The content of ATP decreased from 0.608 ± 0.112 mg/g at 24 hours to 0.453 ± 0.274 mg/g at 1 week and was significantly lower than those in the normal rats (2.047 ± 0.0441 mg/g, Supplemental Figure S2). HE staining revealed swelling and necrosis of myocardial cells in MI model rats at 24 and 72 hours and 1 week. Tissue infiltration and myocardial hemorrhage were also evident at 24 hours (Supplemental Figure S3). Three determination methods all revealed severe myocardial damage in MI model rats at 24 hours after the last injection of ISO.
UPLC-MS/MS
All of the analysts and IS had stronger signals in negative ion mode compared with positive ion mode. The optimized mass transition ion pairs (m/z), collision energy, and cone voltage are shown in Table 1. To optimize the chromatography results and shorten retention time, mobile phase type, ratio, and pH value were evaluated in a preliminary experiment. An acetonitrile/water system including 0.1% formic acid resulted in the best separation, highest stability, and lowest background noise.
Method Validation
Specificity
The chromatograms of blank tissue homogenates, blank tissue homogenates spiked with the 6 mixed standard solutions, IS, and homogenates after oral administration of POE are shown in Supplemental Figures S4-11. The results indicated that the separation of the 6 compounds and IS were achieved without significant endogenous interference.
Linearity and LLOQ
The standard calibration curves, correlation coefficients, LLOQs, and linear ranges of the 6 components are shown in Supplemental Tables S2-9. All analysts showed good linearity in the tested concentration ranges with r ≥ 0.9912. The LLOQs are shown in Supplemental Tables S1-8 and were adequate to assay the tissue distribution of the 6 components.
Accuracy and precision
The accuracy and precision are summarized in Supplemental Tables S10-17. The accuracy (RE) of low, middle, and high QC samples ranged from 12.49% to −13.98%. The RSDs of intraday was below 9.98%, and interday was below 11.88%. The values were all within 15% of nominal ones. The method was sufficiently accurate and precise for the analysis of biological samples.
Recovery and matrix effect
Supplemental Tables S18-25 show the extraction recovery and matrix of the 6 POE analysts at 3 concentrations in blank tissue homogenates. The mean extraction recovery of the 6 analysts ranged from 80.55% to 99.92% with RSDs <12.18% and matrix effect ranged from 81.00% to 98.73% with RSDs <12.71%. The results indicated that the extraction recovery of these analysts was reliable and there was no obvious matrix effect.
Stability
The stability of the 6 analysts in blank tissue homogenates at low, middle, and high concentrations are shown in Supplemental Tables S26-33. The accuracy (RE) ranged from 11.66% to 12.01% with RSDs <13.25%, which indicated that 6 analysts were stable in blank tissue homogenate samples after 3 repeated freeze-thaw cycles from −20°C to room temperature or in an auto-sampler for 6 hours.
Tissue Distribution
Multiple researchers have confirmed that the blood concentration of drugs and their pharmacokinetic behaviors are affected by disease state. 14,15 Polygonum orientale L. has been officially listed in the local standard of traditional materia medica in Guizhou Province, China.2-3 POE has been reported to possess a broad range of pharmacological properties, including antioxidative, anti-inflammatory, anti-MI, and vasorelaxant activities. 2,3 Based on the potential value of POE, it is necessary to investigate the distribution profiles of POE in normal and pathological states. In the present study, a MI model was selected and the data obtained for the tissue distribution of 6 POE analysts demonstrated similarities and significant differences between normal and MI model rats.
In the present study, the tissue distribution of 6 analysts in normal and MI model rats was investigated after the oral administration of POE (0.32 g/kg). The tissue distribution curves for isoorientin, orientin, vitexin, quercitrin, astragalin, and protocatechuic acid in heart, liver, spleen, lung, kidney, stomach, intestine, and brain tissues were determined at 0.16, 1, 2, 6, and 12 hours (Figures 1 -6). The maximum drug concentration (C max) of all components was observed at 0.16 or 1 hour after oral administration of POE, which indicated that 6 components were rapidly and widely distributed in both normal and MI model rats (Figures 1 -6). At 6 and 12 hours, the concentrations of 6 components in all tissues declined, indicating that all analysts were excreted fast in all tissues without a long-term accumulation phenomenon. All the compounds were detected in each tissue, but astragalin could not be quantified in the brain. This result indicated that astragalin might be difficult to cross the blood–brain barrier. Researchers have found that most flavonoid glycosides are converted by the intestinal flora to flavonoid aglycones, taken up into the circulation, and transported to other tissues. 21 A previous study has also reported that hepatic or gastric first-pass effects can affect the oral bioavailability of isoorientin. 22

Distribution profiles of isoorientin after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rats (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) brain, (g) stomach, and (h) intestine (n = 6).

Distribution profiles of orientin after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) brain, (g) stomach, and (h) intestine (n = 6).

Distribution profiles of vitexin after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) brain, (g) stomach, and (h) intestine (n = 6).

Distribution profiles of quercitrin after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) brain, (g) stomach, and (h) intestine (n = 6).

Distribution profiles of astragalin after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) stomach, and (g) intestine (n = 6).

Distribution profiles of protocatechuic acid after intragastric administration of Polygonum orientale L. extract in normal and myocardial ischemia (MI) model rat (a) heart, (b) liver, (c) spleen, (d) lung, (e) kidney, (f) brain, (g) stomach, and (h) intestine (n = 6).
The area under the drug concentration–time curve from 0 to the last measurable concentration point (AUC0–t) is shown in Tables 2–3. In the present study, the AUC0−t of 6 analysts in the stomach and intestine was higher than in other tissues, which might reflect the absorptive and metabolic functions of the digestive tract. The drug’s ability to reach the target organ is a prerequisite for its effectiveness. Isoorientin, orientin, quercitrin, and vitexin are the main components of POE against MI. Many researchers have reported that isoorientin, orientin, and quercitrin possess anti-inflammatory and antioxidative activities. 23 -26 Isoorientin can act against H9C2 myoblast cell damage by releasing free radical DPPH (1,1-diphenyl-2-picrylhydrazyl). 23,24 Quercitrin can inhibit lipid peroxidation by inhibiting xanthine oxidase activity, chelating iron, and directly scavenging hydroxyl and superoxide anion radicals. 25,26 However, the AUC0–t of these components in the heart was low, which indicated that they might be metabolized in vivo. It is possible that their metabolites play an important role in anti-MI, but their metabolic pathways and metabolites need further study.
In addition, the distributions of isoorientin (AUC0−t = 910.73 ± 159.86 ng/g/hour in normal rats and AUC0−t = 477.18 ± 67.14 ng/g/hour MI model rats), orientin (AUC0−t = 1136.15 ± 546.19 ng/g/hour in normal rats and AUC0–t = 127.80 ± 71.98 ng/g/hour in MI model rats), vitexin (AUC0 −t = 889.96 ± 91.20 ng/g/hour in normal rats and AUC0−T = 564.93 ± 58.61 ng/g/hour in MI model rats), and quercitrin (AUC0–t = 445.52 ± 34.45 ng/g/hour in normal rats and AUC0−t = 138.28 ± 59.18 ng/g/hour in MI model rats) were high in liver tissue, indicating excretion by the liver. Astragalin (AUC0−t = 92.58 ± 11.99 ng/g/hour in normal rats and AUC0−t = 74.1 ± 10.08 ng/g/hour in MI model rats) and protocatechuic acid (AUC0-t = 516.01 ± 82.12 ng/g/hour in normal rats and AUC0−t = 206.38 ± 37.01 ng/g/hour in MI model rats) were high in kidney tissue, indicating excretion by the kidney.
Although the tissue distribution trend of 6 POE components in normal and MI model rats was similar, there were significant differences between the groups. The distribution of flavonoids, such as orientin and vitexin, depends on blood flow and organ perfusion. 20,27,28 However, in this ISO-induced MI model, the blood flow, blood perfusion, and oxygen supply decreased because of coronary vessel blocking. 16,18 However, C max of the 6 compounds in the rat tissues of the MI model was lower than that of normal rats at 0.16 and 1 hour, except for spleen and brain, which might be affected by blood flow and blood perfusion. Another reason for the C max decrease in 6 analysts in the MI model group tissues might be related to the gastrointestinal microbiotal dysbiosis in disease state. 12 Multiple researchers have indicated that gastrointestinal microbiota diversity, composition, and counts are altered in pathological conditions. 29 -31 Most of the flavonoid glycosides in traditional Chinese medicine can be transformed to flavonoid aglycons by intestinal bacteria before they are absorbed into circulation. 30,31 For example, baicalin and wogonoside are completely or partially hydrolyzed to baicalein and wogonin, respectively, and by β-glucuronidase in the stomach and intestine. 32,33 Studies have shown that bifidobacteria and lactobacillus in the intestine of patients with cardiovascular disease are significantly downregulated, while Escherichia coli and Streptococci are significantly increased. Intestinal flora disorders can cause metabolic disorders of the body’s sugars, lipids, and other substances and strengthen the inflammatory response and oxidative stress in the body, promoting the occurrence or deterioration of the disease. 34,35 Therefore, the imbalance of gastrointestinal microbiota in MI model rats might lead to the decreased absorption of these components in blood circulation.
In addition, 2 and 6 hours after POE administration, the content of 6 components decreased significantly in both normal and MI model rats. However, concentrations in some organs were higher in the MI model than in normal rats, for example, the content of quercitrin in the heart, liver, spleen, lung, and kidney tissues at 6 hours. This indicated that the elimination rate in the MI model rats was slower than that of normal rats.
Conclusion
In this study, a simple, rapid, and sensitive UPLC-MS/MS method for simultaneous determination of 6 components in the tissue of the MI model and normal rats after oral administration of POE was established and validated. The tissue distribution characteristics of 6 components in normal and MI model rats after oral administration of POE were significantly different, but the overall trend was similar. The results might provide a reference for further research on P. orientale L.
Supplemental Material
Supplementary Material 1. - Supplemental material for Comparative Tissue Distribution of 6 Major Polyphenolic Compounds in Normal and Myocardial Ischemia Model Rats After Oral Administration of the Polygonum orientale L. Extract
Supplemental material, Supplementary Material 1., for Comparative Tissue Distribution of 6 Major Polyphenolic Compounds in Normal and Myocardial Ischemia Model Rats After Oral Administration of the Polygonum orientale L. Extract by Na Li, Ting Lv, Jie Pan, Chunhua Liu, Jia Sun, Yanyu Lan, Aimin Wang, Yongjun Li, Yonglin Wang and Yuan Lu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was financially supported by the National Natural Science Foundation of China (No. U1812403, No. 81660712, No. 81803700), the Team Project of Guizhou Provincial Science and Technology Department ([2016]5613\5677), the Central Guidance for Local Science and Technology Projects ([2018]4006), Scientific Program of Guizhou Province ([2017]5718) and Guizhou Provincial Administration of Traditional Chinese Medicine (QZYY-2018-108).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
