Abstract
The purpose of this article is to establish a method to use ultra-high performance liquid chromatography (UPLC)-mass spectrometry (MS)/MS to simultaneously determine 9 main components of Lonicera japonica Thunb. in negative-ion scanning mode, and the main components were analyzed by chemometrics. The chromatographic separation uses the Thermo Hypersil GOLD column (100 mm × 2.1 mm, 1.9 µm) with a constant temperature of 45 °C. The mobile phase consists of methanol and water containing 0.2% formic acid. The results show that 9 compounds had a good linear relationship (R² > 0.9991), and both intraday and interday precisions and stability have the eligible ranges of relative SDs (RSDs; 0.96%-2.26%, 0.52%-3.04%, and 0.85%-2.15%, respectively). The recovery rates were between 75.90% and 110.58%. The results of chemometrics including hierarchical cluster analysis and principal component analysis showed that there were obvious differences in the content of active components in L. japonica from different regions, and the compounds with the highest contribution to the drug were identified. Through the UPLC-MS/MS combined chemometrics analysis of L. japonica, this experiment can provide a reference for further research on the modernization and innovation of L. japonica and the application research of a high level and multidirection.
Traditional Chinese medicine (TCM) has a long history in China, its excellent safety and effectiveness to ensure people’s health and life. Different from the western medicine of chemical synthesis, TCM is usually the root and stem of plants that can cure diseases. Therefore, TCM that grown through cultivation is cheaper and easier to obtain, and TCM usually has fewer side effects. However, due to the complex components of TCM, a single type of TCM contains a large number of different compounds. Only 1 or 2 indicators are difficult to roundly reflect the quality of TCM. Hence, we should simultaneously determine a variety of effective ingredients in TCM. 1 At present, high-performance liquid chromatography (HPLC) combined with ultraviolet and other similar methods have been used for the simultaneous determination of compounds in TCM, 2 -5 and the quality of TCM can be well controlled. However, the advent of ultra-high performance liquid chromatography (UPLC)-mass spectrometry (MS)/MS has made the quantitative analysis technology of Chinese medicine more mature. 6 -8 The UPLC-MS/MS method embodies the complementary advantages of chromatography and MS, combining the high separation power of chromatography for complex samples with high selectivity, sensitivity, and relative molecular mass of MS/MS. This technology has been increasingly valued and used.
Lonicera japonica Thunb., known as Jin Yin Hua or Ren Dong, was originated in China. Around 1500 years ago, it was planted largely in Henan province, and the flowers were used as the local and traditional medicine in clinical practice for the treatment of some diseases. 9 It was commonly used in carbuncle swelling boils, throat numbness, erysipelas, blood poisoning dysentery, wind-heat cold, and warm fever. Modern pharmacological research shows that it has anti-inflammatory, 10 antioxidant, 11 hepatoprotective, cytoprotective, antimicrobial, 12 antidiabetic, 13 and anti-hepatitis B virus activities. 14 Phytochemical studies show that L. japonica contains a large number of active compounds, such as flavonoids, iridoids, triterpenoid saponin, alkaloids, and cerebrosides. 15,16 At present, L. japonica cultivation areas are mainly concentrated in Shandong, Shanxi, Henan, Hebei, Hubei, Jiangxi, Guangdong, and other places. And due to geographical conditions, climate, and other reasons, the contents of the main active ingredients from each place of production are also different, and the efficacy will be different. Pharmacology research has shown that among them, the main antibacterial and anti-inflammatory effects are made by the flavonoids and phenolic acids. 17,18 The Pharmacopeia also specifies that the index components in L. japonica are 3-O-caffeoylquinic acid and luteolin. Therefore, it is necessary to use phenolic acids and flavonoids as indicators to conduct quality control of L. japonica.
In this article, a UPLC-MS/MS method was established to determine the content of L. japonica from 10 different regions. In the negative-ion scanning mode, 9 main components of antibacterial and anti-inflammatory were determined, including 7 phenolic acids and 2 flavonoids. Then, 10 batches of samples were analyzed by chemometrics, including hierarchical cluster analysis (HCA) and principal component analysis (PCA). 19 The results showed that there were obvious differences in the content of the components from different areas. 3-O-caffeoylquinic acid and 3,5-dicaffeoylquinic acid were the main components with a cumulative contribution rate of 99%.
Materials and Methods
Chemicals, Reagents, and Materials
HPLC-grade methanol was purchased from Fisher Scientific (Pittsburgh, PA, USA). HPLC-grade formic acid was purchased from Dikma Co. (USA). Distilled water was purchased from the Hangzhou Wahaha Group (Hangzhou, China). All other reagents are of analytical grade. Ten batches of L. japonica samples (S1-S10) were purchased from Guangdong, Hebei, Henan, Hubei, Hunan, Jiangxi, Shandong, and Yunnan, respectively. L. japonica identified by Su Lianjie, Heilongjiang University of Traditional Chinese Medicine. The standards of 3,4-dihydroxybenzoic acid, 5-O-caffeoylquinic acid, 3-O-caffeoylquinic acid, caffeic acid, 4-O-caffeoylquinic acid, 3,5-dicaffeoylquinic acid, luteoloside, rutin, 3,5-dicaffeoylquinic acid, and chloramphenicol (IS) were acquired from Chengdu Must Biotechnology (Chengdu, China).
Instrument
Chromatographic separation using a UPLC system consisting of an autosampler and a binary pump (Thermo Scientific, Vanquish, Waltham, MA, USA). Sample analysis was performed using Thermo TSQ QUANTIS triple quadrupole MS connected with an electrospray ionization (ESI) interface.
Chromatographic Conditions
The chromatographic separation uses the Thermo Hypersil GOLD column (100 mm × 2.1 mm, 1.9 µm) with a constant temperature of 45 °C. The mobile phase consists of methanol (A) and water (B) containing 0.2% formic acid (v/v). Gradient elution: 0-6 minutes, 90%-83% B; 6-7 minutes, 83%-60% B, 7-12 minutes, 60%-50% B; 12-13 minutes, 50%-90% B; and 13-15 minutes, 90% B. The flow rate is constant at 0.3 mL/min, and the injection volume is 2 μL.
Mass Spectrometric Conditions
All samples were analyzed in negative-ion mode. The analysis method uses the selective reaction mode (SRM) of multiple reaction monitoring. The optimized parameter settings are as follows: Sheath Gas: 30 Arb, Aux Gas: 10 Arb, Sweep Gas: 0 Arb, ion transfer tube temperature: 325 °C, vaporizer temperature: 350 °C.
Preparation of Sample Solutions
Lonicera japonica materials were first ground and passed through the 60-mesh sieve. An aliquot of 0.05 g dried sample powder was extracted with 10 mL of methanol. Then, ultrasonic extraction was performed at 35 Hz for 30 minutes. Then the samples were centrifuged at 5000 rpm for 15 minutes, and the supernatant was taken, filtered with 0.22 μm membrane, and stored in a refrigerator at 4 °C for analysis. All samples were prepared with the above method.
Preparation of Standard Solutions
Appropriate amounts of 9 standards were dissolved in methanol to prepare stock solutions and diluted with methanol by chromatographic analysis. The concentration of the 9 standard solutions was 1.00 mg/mL.
Method Validation
The calibration curves, limits of detection (LODs), limits of quantification (LOQs), precision (including intraday precision and interday precision), stability, and recovery were investigated. The calibration curves of 9 compounds were prepared by diluting the stock solution into a series of appropriate concentrations with methanol-water. LOD and LOQ were determined by a signal-to-noise (S/N) the wire of 3 and 10, respectively. Intraday precision was verified by RSD% of the data obtained from 6 injections in 1 day. Interday was verified by RSD% with an average of 6 injections per day for 3 days. The stability was verified by RSD% injected 6 times at 0, 2, 4, 8, 12, 24, and 48 hours, respectively. The accuracy of this method with 3 levels (80%, 100%, 120%) of the sample. The recovery rate formula is:
Chemometrics Analysis
Hierarchical Cluster Analysis
Dendrogram originated from the traditional taxonomy created and developed by Swedish naturalist Carl Linnaeus. In the classical systematic Cluster analysis, the individual biological samples and the samples with the most similar characteristics and attributes are first combined into clusters, the basic unit, to form higher classification levels such as species, genus, family, and order by gradual aggregation. As a general method in many fields, HCA has been widely used in the analysis of TCM. In this study, this method was used to verify the differences among different batches of samples. The ChemPattern software (Chemmind Technologies. Co., Ltd) is used to obtain the HCA.
Principal Component Analysis
PCA was proposed by British statistician Karl Pearson in 1901. PCA is the most classically and widely used multivariate statistical method based on eigenvector analysis, which is widely used in various fields. Generally, the cumulative contribution rate of the first few principal components to be retained should reach 85%-95% when measured by the cumulative variance. In this study, this method was used to identify the most influential components in the active components of L. japonica. The ChemPattern software (Chemmind Technologies. Co., Ltd) is used to obtain the PCA.
Results and Discussion
Optimization of Extraction Conditions
To obtain a high-content sample solution, we performed an extraction optimization experiment. First, the extraction efficiency of 9 compounds in different solvents was investigated to maximize the content of the compounds. An aliquot of 0.05 g dried sample powder was extracted with 4 different ratios of methanol solution (methanol, 85% methanol/water, 70% methanol/water, 50% methanol/water) and 4 different ratios of ethanol solution (ethanol, 85% ethanol/water, 70% ethanol/water, 50% ethanol/water); the volume of the solvent was 10 mL and then ultrasonically extracted at 35 kHz for 60 minutes. The results after injection are shown in Figure 1A. Under 8 different solvents, methanol has the highest extraction efficiency. Second, to obtain a better extraction effect, the effect of extraction time on the extraction efficiency of 9 compounds was investigated. An aliquot of 0.05 g dried sample powder was extracted with 10 mL of methanol. And ultrasonic extraction was performed at 35 Hz for 30 minutes, 45 minutes, 60 minutes, and 90 minutes. The results after injection are shown in Figure 1B. The total content of the compounds was not different at 4 different extraction times. By the t-test, there was no obvious difference in the total content of the compounds at the 4 extraction times. Therefore, the shortest extraction time of 30 minutes was selected. To sum up, we choose to put the 0.05 g sample into 10 mL methanol. The optimal extraction method was 35 kHz frequency ultrasonic extraction for 30 minutes.
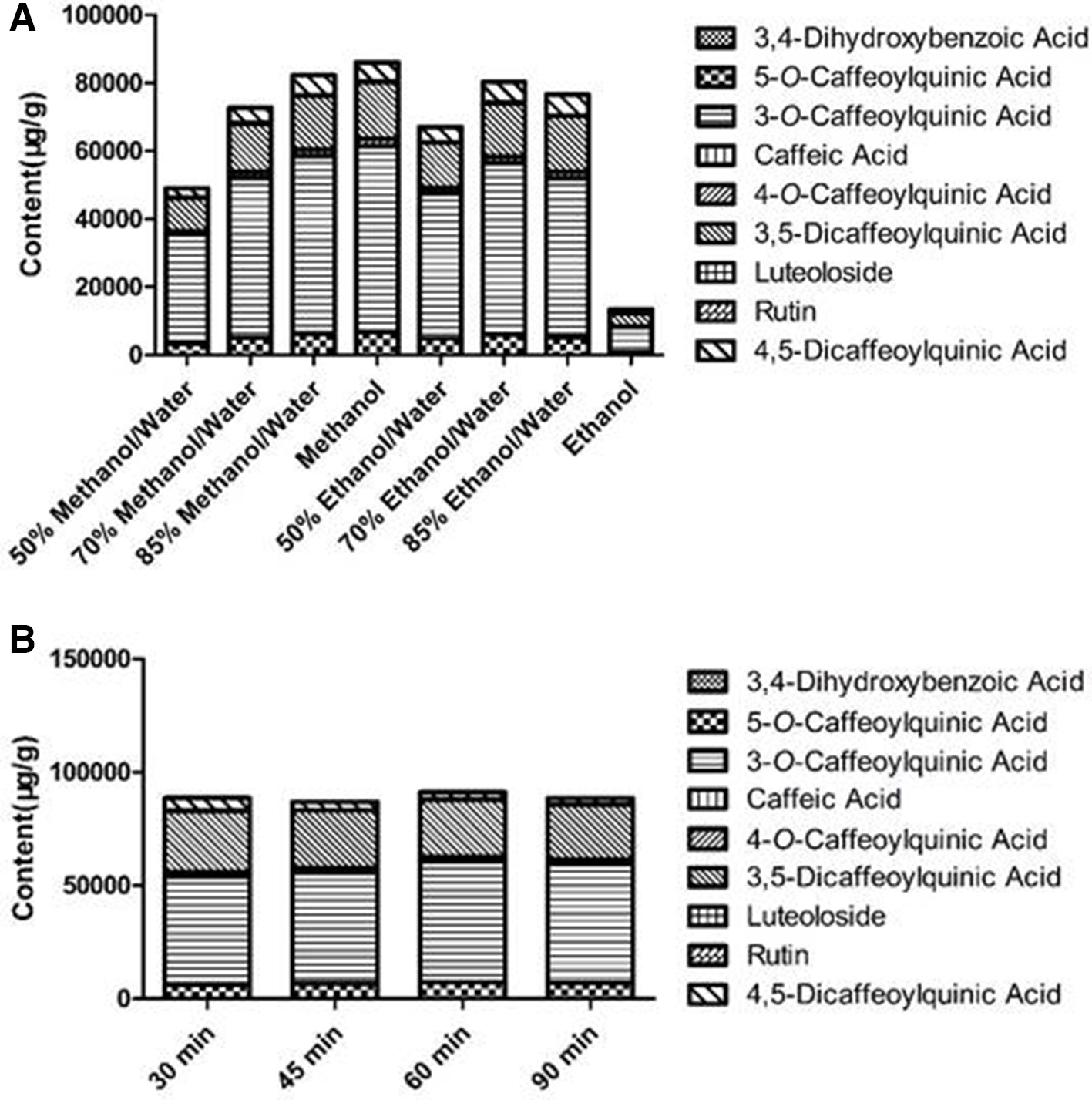
The extraction efficiency of different (A) solvent combinations and (B) extraction times.
Optimization of Chromatographic Conditions
To improve the resolution and peak shape of the sample, we chose the Thermo Hypersil GOLD column (100 mm × 2.1 mm, 1.9 µm) and the Waters Acquity UHPLC HSS T3 column (50 mm × 2.1 mm, 1.8 µm). Under the same column temperature, mobile phase and gradient elution conditions, the Thermo Hypersil GOLD column (100 mm × 2.1 mm, 1.9 µm) has a better separation effect, better peak shape, and shorter retention time. Therefore, we chose a Thermo Hypersil GOLD column (100 mm × 2.1 mm, 1.9 µm) for separation.
The precursor and product of each compound were optimized using UPLC-MS/MS, and then the quantitative analysis is conducted under SRM. Each compound has been optimized to have 2 pairs of precursors and a product, 1 for qualitative and the other for quantitative purposes. The advantage of this method is that each compound can be accurately quantified from the mixture without the need for baseline separation, which saves time and solvents.
Optimization of Mass Spectrometric Conditions
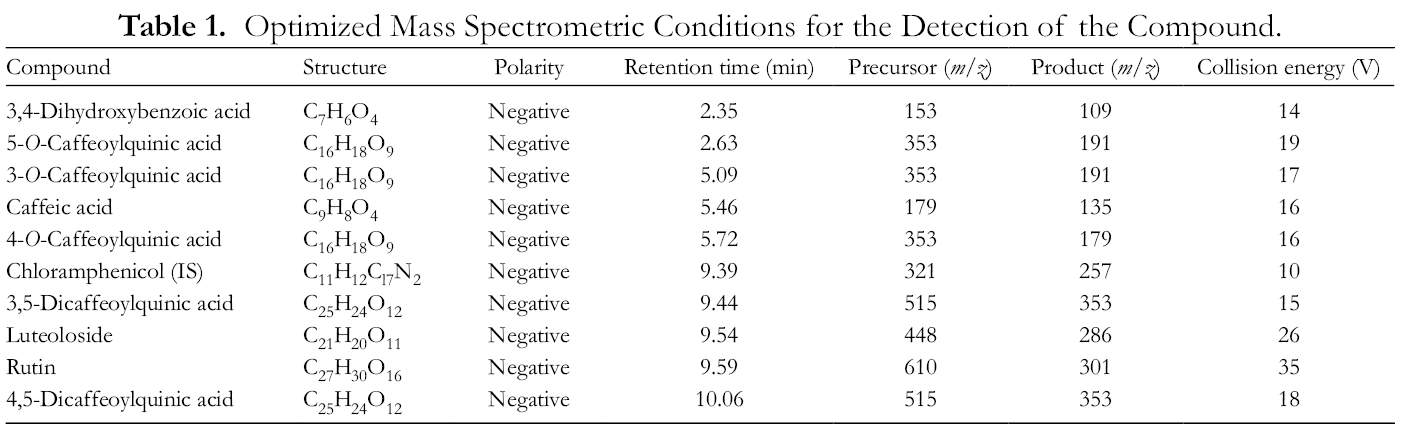
This study optimized mass spectrometry conditions, including Precursor, Product, and Collision, which obtained higher signals. The results of mass spectrometry optimization are shown in Table 1.
Optimized Mass Spectrometric Conditions for the Detection of the Compound.
Method Validation
As shown in Table 2, the calibration curves of 9 compounds in a certain concentration range were obtained by the above method in this experiment, and the results showed that the linear relationship and acceptable correlation coefficient were good (R 2 > 0.9991). LOD and LOQ are obtained by diluting the lowest point of the standard curve to the signal-to-noise ratio of 3 and 10, and their ranges are 0.00001-0.00318 µg/mL and 0.00003-0.01061 µg/mL, respectively. The intraday and interday precisions and stability within 48 hours were all good. The RSD% ranges were 0.96%-2.26%, 0.52%-3.04%, and 0.85%-2.15%, respectively. As shown in Table 3, the recovery rates were between 75.90 and 110.58%.
Calibration Curves, Linear Range, Limit of Detection (LOD), Limit of Quantification (LOQ), Precision, and Repeatability for 9 Active Compounds.

Recovery Rates and the Relative SD (RSD) of the 9 Active Compounds With Different Concentrations (80%, 100%, and 120%).

Quantitative Analysis
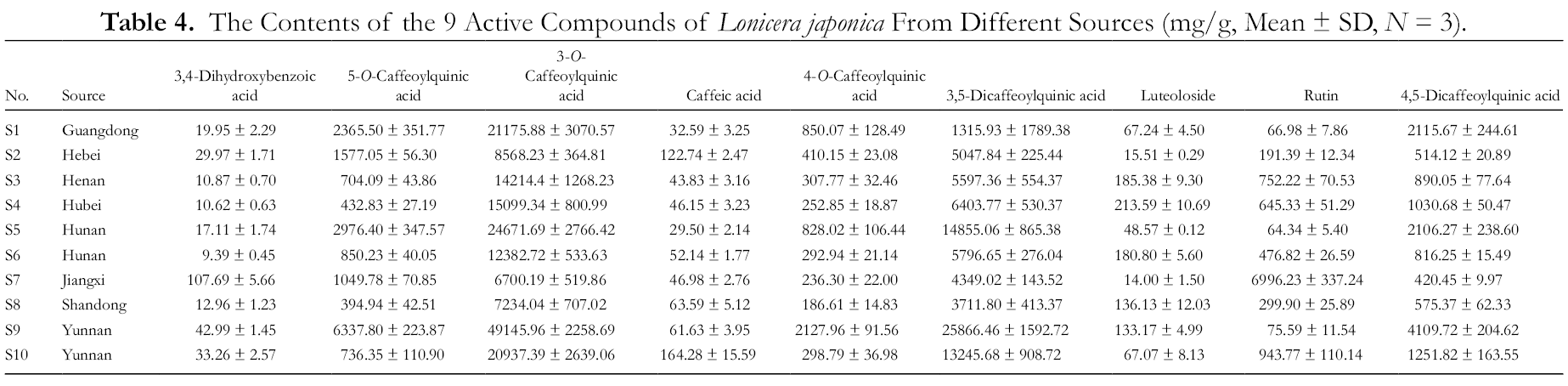
Each batch of samples was prepared in triplicate, and the content of compounds in each batch of samples was calculated by the calibration curves. The results are shown in Table 4. It can be found from the preliminary observation that the content of 3-O-caffeoylquinic acid in all samples is very high, which consistent with the indicators in the Pharmacopeia. And even from the same province, such as S5 and S6, S9, and S10, the differences in content between them are not small. It can be seen that the content of phenolic acid in S9 is higher and the content of flavonoids in S7 is higher.
The Contents of the 9 Active Compounds of Lonicera japonica From Different Sources (mg/g, Mean ± SD, N = 3).
Chemometrics analysis
Hierarchical Cluster Analysis
Cluster analysis with heat map analysis was performed using 9 compounds as index components, and the results are shown in Figure 2. The results show that 10 batches of samples are divided into many clusters, indicating that even the same TCM has a large difference in content between different origins. Heat maps can turn huge data into easy-to-understand, visual strip charts. In the heat map, it can be observed that 3-O-caffeoylquinic acid and 3,5-dicaffeoylquinic acid are the main components of samples.

The results of hierarchical cluster analysis combined with the heat map.
Principal Component Analysis
As shown in Figure 3 A,B, the S7 and S9 samples are significantly different from other batches, which is consistent with the results of higher S7 flavonoid content and higher S9 phenolic acid content in the quantitative analysis. Besides, the contribution rate of 3-O-caffeoylquinic acid is 97%, the contribution rate of 3,5-dicaffeoylquinic acid is 2%, and the cumulative contribution rate of 3-O-caffeoylquinic acid and 3,5-dicaffeoylquinic acid reaches 99%, indicating that 3-O-caffeoylquinic acid and 3,5-dicaffeoylquinic acid are the main components of L. japonica. The results are consistent with the results of the heat map.

The results of principal component analysis: (A) 2-dimension and (B) 3-dimension.
Conclusions
In this study, a UPLC-MS/MS method was established for the rapid and simultaneous determination of 9 compounds in L. japonica. The method was verified to have good precision, accuracy, repeatability, and stability. The results of chemometrics showed that L. japonica varied widely from region to region, and the cumulative contribution rate of 3-O-caffeoylquinic acid and 3,5-dicaffeoylquinic acid reached 99% of the 9 compounds. To sum up, UPLC-MS /MS combined with chemometrics is a simple and effective method to control the quality of L. japonica, which lays a theoretical foundation for the sustainable development of honeysuckle and provides some suggestions for the comprehensive development and utilization of TCM.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this experiment was supported by the School of Pharmacy, Jiangxi University of Traditional Chinese Medicine and Key Laboratory of Chinese Materia Medica, Heilongjiang University of Chinese Medicine, Ministry of Education.

