Abstract
Leishmaniases are a spectrum of poverty-linked neglected parasitic diseases that are endemic in 88 countries around the globe and affect millions of people every year. Currently available chemotherapeutic options are inadequate due to side effects, high cost, prolonged treatment, and parasite resistance. Thus, there is an existing need to develop new potent and safer leishmanicidal drugs. Considering the folkloric antiulcer and leishmanicidal use of the genus Berberis and its alkaloids, 5 reported alkaloids, namely berberine (
Keywords
Introduction
Leishmaniasis is an important public health and social problem in tropical, subtropical, and Mediterranean regions of the world including Pakistan. It is one of the world's most neglected and poverty-related diseases affecting the poorest of people in developing countries. 1 Leishmaniasis is caused by obligate intramacrophagic protozoans of the genus Leishmania with 30 different species (10 in Old World and 20 in New World). Twenty-one species out of 30 have been identified to be pathogenic to humans. 2 The species are widespread in all continents except Antarctica. 3 In the absence of any effective vaccine the only means to treat and control leishmaniasis is affordable chemotherapy. The first-line drugs used to treat both visceral and cutaneous leishmaniasis (CL) are miltefosine, pentavalent antimonials, amphotericin B, and pentamidine. Alternative drugs include paromomycin, allopurinol, triazoles, azithromycin, stimaquin, rifampicin, and dapsone. 4 The currently available drugs like miltefosine, amphotericin B, and pentamidine suffer from some limitations like high toxicity, high cost, prolong treatment duration, and resistance by the leishmaniasis parasites. 5 Therefore, new antileishmanial therapeutic agents that are safe, more effective, less cytotoxic, and economically feasible are needed to supplement the currently available therapies.
Over the past 2 decades, several institutions in South America and India have undertaken investigations in an endemic area of cutaneous leishmania to use medicinal plants as medicines for the treatment of leishmaniasis. Recently, the use of medicinal plants and traditional medicines for the treatment of leishmaniasis has been considered a priority by the Tropical Diseases program of WHO. 6 Bioactive natural products are small chemical entities with pharmacological properties. Historically, natural products have always played a key role in fighting various kinds of diseases. 7
Plant-derived natural products like isoquinoline, bisbenzylisoquinoline alkaloids, and flavonoids have shown good antileishmanial potential already.8,9 The leishmanicidal potentials of compounds
This study describes the antileishmanial activity and in-silico molecular docking with Leishmania protein targets of 5 (

Berberine alkaloids are discussed in this work.
Results
Structure Elucidation of Alkaloids
Structures of the isolated berberine alkaloids (Figure 1) were elucidated from their 1D and 2D nuclear magnetic resonance (NMR) spectroscopic data and were then confirmed by comparison with literature.
Antileishmanial Effects of Berberine Alkaloids
All the isolated alkaloids displayed good leishmanicidal effects against the promastigotes stage of Leishmania tropica clinical field isolates at all the used concentrations (100, 50, 25, 12.5, 6.25, and 3.125 µM). 8-Trichloromethyldihydroberberine was found to be the most potent of the compounds tested (Figure 2, Table 1). Furthermore, the alkaloids exhibited minimal cytotoxic activity against THP-1 monocytic leukemia cells.

Antileishmanial activity of the isolated alkaloids at different concentrations. Results presented here are the mean value of n = 3 ± SEM. The data were analyzed by one-way ANOVA followed by Dunnett's test. *P < .05, **P < .01, and ***P < .001 were considered as statistically significant differences.
Cytotoxic and Antileishmanial Activities for Berberine Alkaloids and Amphotericin-B.

Values expressed here are mean ±SEM of 3 individual experiments.
Molecular Docking of Berberine Alkaloids
Several Leishmania enzymes have been suggested to be targets of isoquinoline alkaloids, in particular, N-myristoyl transferase (NMT)14,15 and mitogen-activated protein kinase. 16 The berberine alkaloids may also serve as topoisomerase 1B poisons, 17 presumably via intercalation of DNA. Planar alkaloids are known to be intercalators of DNA.18,19 In order to help identify possible molecular targets of Leishmania, a molecular docking analysis was carried out with the berberine alkaloids, berberine, jatrorrhizine, palmatine, columbamine, 8R-trichloromethyldihydroberberine (8R-TCMDHB), and 8S-trichloromethyldihydroberberine (8S-TCMDHB) with Leishmania protein targets for which X-ray crystal structures are available (a total of 85 protein structures). 20 The most exothermic (best docking energies) from the molecular docking analysis are summarized in Table 2 along with key intermolecular interactions. A complete listing of docking energies of the berberine alkaloids with Leishmania target proteins is available in Supplemental material (Table S1).
Molecular Docking Energies (Edock, kJ/mol) for the Interactions of Berberine Ligands with Leishmania Protein Targets.

In addition, molecular docking of the berberine alkaloids with structures of DNA was carried out. A total of 9 structures available from the PDB were used to examine potential intercalation of the alkaloids (Table 3). A complete listing of docking energies of the berberine alkaloids with DNA structures is available in Supplemental material (Table S2).
Lowest Molecular Docking Energies (Edock, kJ/mol) for the Intercalation Interactions of Berberine Ligands with DNA.

Discussion
In the absence of novel therapeutics against leishmaniasis, natural and herbal products could be very important in the development of new drugs. 21 The perspectives for the cure of leishmaniasis are tentative and there is a strong need to find low-cost therapeutic agents, especially in underdeveloped and developing countries. The use of medicinal plants for the treatment of parasitic diseases is well known and has been in practice since ancient times. 22 In recent years, advances in chemical and pharmacological techniques have contributed to the knowledge of new therapeutically active compounds obtained from natural sources. Alkaloids represent the largest single class of plant secondary metabolites and many alkaloids are used in therapeutics and pharmacological procedures. 23 Plants can be a source of active, safe, and inexpensive antimicrobial agents therefore, it becomes necessary to explore the endemic plants for their antileishmanial properties.24,25
The exact antileishmanial mechanism of action of miltefosine, amphotericin-B, antimonials, and pentamidine are still not clearly known. However, pentamidine causes antileishmanial potential by inhibiting DNA minor groove binding, polyamine biosynthesis and affecting inner mitochondrial membrane Leishmania parasites. 26 The antileishmanial mechanism of amphotericin-B also includes significant changes in plasma membrane sterol profile and replacement of ergosterol by cholesta-5,7,24-trien-3β-ol. 27 Similarly, the antileishmanial mechanism of action of miltefosine includes DNA fragmentation, cell shrinkage, phosphatidyl serine exposure, and DNA condensation. 28
Isoquinoline alkaloids are usually good antileishmanial agents. Although the exact mechanism of action of the tested alkaloids is not known, it may possibly include any one or a combination of the above-stated pathways. Overall, the results obtained, in our case, for the antileishmanial potential of the tested alkaloids were very similar and closely related to each other. The obvious reason for that is the fact the all 5 tested alkaloids possess the same pharmacophores. However, 8-trichloromethyldihydroberbeine and berberine were slightly more potent than the remaining 3 alkaloids because of the fact that their structures are slightly different from the other 3. The berberine alkaloids may have interacted with receptors of the promastigotes. The selectivity index for 8-trichloromethyldihydroberberine was the lowest of the 5. It may be due to the fact that it has a trichloromethyl attached to it, which could have caused more cytotoxic effects.
Enzyme inhibition studies is a very important area in the field of pharmaceutical research as it has led to the discovery of many drugs for the treatment of many diseases. Molecular docking of the isoquinoline alkaloids from Berberis glaucocarpa root extract has revealed potential Leishmania enzymes that may be targeted by the alkaloids. The most targeted Leishmania proteins based on this in-silico investigation were NMT, methionyl-tRNA synthetase (MetRS), pteridine reductase 1 (PTR1), oligopeptidase B (OPB), tyrosyl-tRNA synthetase (TyrRS), and glycerol-3-phosphate dehydrogenase (GPDH).
N-Myristoyltransferase (NMT) catalyzes the covalent attachment of myristic acid (tetradecanoic acid) to the N-terminal glycine residue of proteins, which facilitates the association of substrate proteins with membranes or activation and stabilization of the substrate protein. 29 The protein has been verified as a target for antileishmanial drug development.30,31 Columbamine showed excellent docking to L donovani and L major NMT with docking energies of −118.7 and −125.2 kJ/mol, respectively. Not surprisingly, the columbamine ligand adopts the same lowest-energy docked pose in both L donovani NMT and L major NMT (Figure 3). The docked ligand occupies the peptide-binding site of NMT 32 with key intermolecular interactions between the columbamine ligand and NMT are Val378 (hydrogen bonding), Met420 (van der Waals), Tyr217 (hydrogen bonding), Leu421 (van der Waals), Thr203 (hydrogen bonding), and Tyr80 (hydrogen bonding) (Figure 3). Interestingly, the docking energy of the co-crystallized ligand, 4-bromo-2,6-dichloro-N-(1,3,5-trimethyl-1H-pyrazol-4-yl)benzenesulfonamide, was only −83.5 kJ/mol, and occupies a site within the active site, but in a different location (a hydrophobic pocket formed by Tyr217, Phe90, and Phe88).
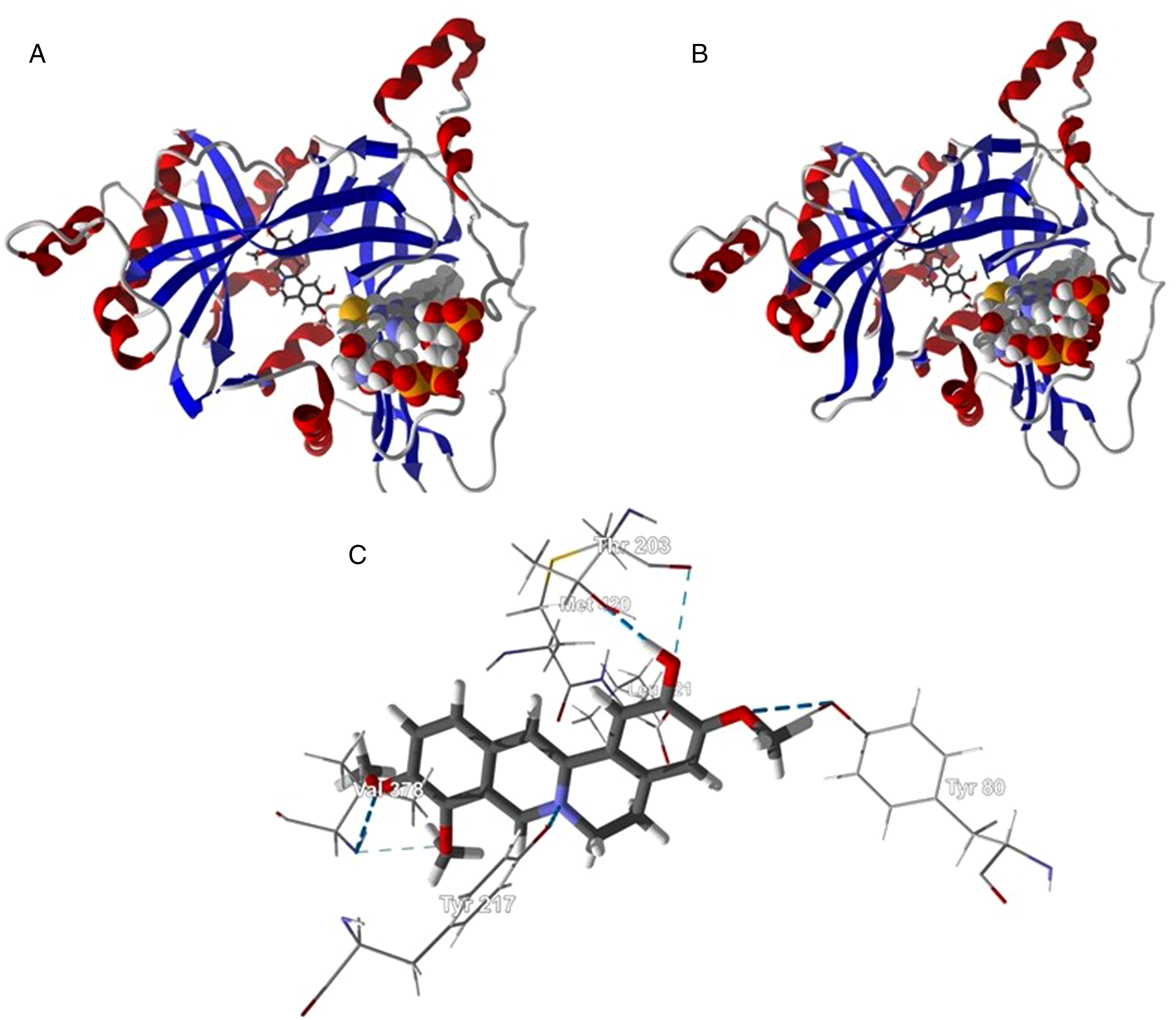
Lowest-energy docked poses of columbamine with Leishmania N-myristoyltransferase (NMT). (A) Ribbon structure of L donovani NMT (PDB 2WUU) with columbamine. (B) Ribbon structure of L major NMT (PDB 4A30) with columbamine. (C) Key intermolecular interactions between columbamine and L major NMT (PDB 4A30). Hydrogen-bonding interactions are indicated with a blue dashed line.
Leishmania PTR1 catalyzes the reduction of folates and pterins, for example, reduction of biopterin to tetrahydrobiopterin. 33 Leishmania are able to overcome inhibition of dihydrofolate reductase (DHFR) by overexpression of PTR1. Therefore, inhibition of both Leishmania PTR1 and DHFR could represent a viable antileishmanial chemotherapeutic option. 34 Berberine, jatrorrhizine, palmatine, and 8S-TCMDHB showed selective docking to L major PTR1. The lowest-energy docked poses of the ligands with L major PTR1 placed the ligand into close proximity to the NADP+ cofactor (Figure 4). In the case of berberine, the ligand is sandwiched (π–π) between the adenine ring of the cofactor and Phe113 (Figure 4). Additional key intermolecular interactions are hydrophobic interactions of the ligand with Leu188 and Met183, and hydrogen-bonding of the ligand with Tyr194. The co-crystallized ligand, 7,8-dihydrobiopterin, had a docking energy of −97.5 kJ/mol and occupies the same location in the active site (ie, sandwiched between adenine and Phe113, hydrophobic interactions with Leu188, and hydrogen-bonding with Tyr194).

Lowest-energy docked pose of berberine and L major pteridine reductase 1 (PDB 1E92) showing key intermolecular contacts. The NADP+ cofactor is shown as a space-filling model, hydrogen-bonding is indicated with a blue dashed line.
All 6 berberine structures showed good docking properties with L major MetRS. Berberine, palmatine, and jatrorrhizine, in particular, demonstrated excellent docking energies of −115.8, −113.6, and −110.9 kJ/mol, respectively. These 3 ligands adopted the same orientation in the active site of the enzyme (Figure 5). Key intermolecular contacts between the docked ligands and the protein were Tyr218 (edge-to-face π–π interaction), Tyr219 (hydrogen bonding), His228 (edge-to-face π-π interaction), Gly227 (van der Waals interaction), Trp515 (van der Waals interaction), Asp486 (electrostatic interaction between the carboxylate of the residue and the nitrogen of the ligand), and Trp516 (hydrogen bonding) (Figure 5). The co-crystallized ligand, methionyladenylate, did have a lower docking energy (−197.0 kJ/mol).

Lowest-energy docked pose of berberine alkaloids and L major methionyl-tRNA synthetase (PDB 3KFL). (A) Ribbon structure showing the orientation of the berberine (aqua), jatrorrhizine (yellow), and palmatine (magenta) in the active site. The co-crystallized ligand (methionyladenylate) is shown as a green stick figure. (B) Lowest-energy docked pose of berberine showing key intermolecular contacts. Hydrogen-bonding interactions are indicated with a blue dashed line.
Columbamine interacted with L major MetRS and L major TyrRS during docking studies. These 2 enzymes are members of the aminoacyl-tRNA synthetase family of enzymes and serve to guarantee that the amino acid properly attaches to its cognate tRNA. 35 There are significant differences in the structures of Leishmania MetRS 36 and TyrRS 37 and the human homologs, suggesting that Leishmania MetRS and TyrRS are promising targets for antileishmanial drug discovery. Columbamine preferentially docked into the active site of each enzyme. Important intermolecular contacts between the ligand and L major MetRS (Figure 6) are a hydrophobic pocket formed by Trp515, His228, Gly227, and Asp486; an edge-to-face π–π interaction between Tyr218 and the ligand; and hydrogen bonding of the ligand with Tyr219, Ile217, Trp516, and Lys522.
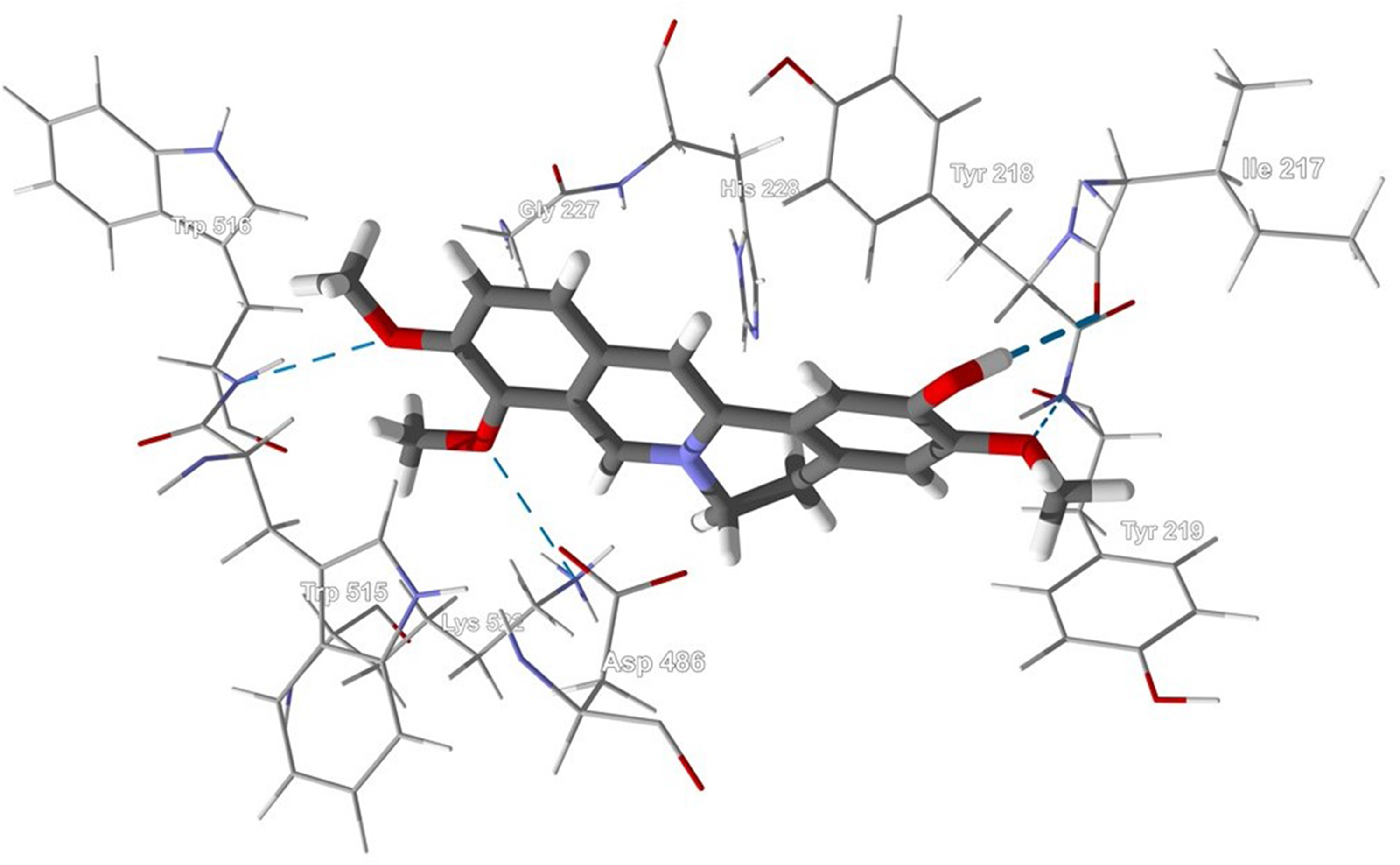
Lowest-energy docked pose of columbamine with Leishmania major methionyl-tRNA synthetase (PDB 3KFL). Hydrogen-bonding interactions are indicated with a blue dashed line.
Similarly, columbamine docked into the active site of L major TyrRS in a hydrophobic pocket formed by Glu40, Gln167, Met166, Tyr163, and Phe75. There are hydrogen-bonding interactions between the ligand and the protein with residues Gln185, Asp170, Tyr126, Trp70, and Ser115 (Figure 7). The tyrosinol co-crystallized ligand had a much more modest docking energy of −70.8 kJ/mol.
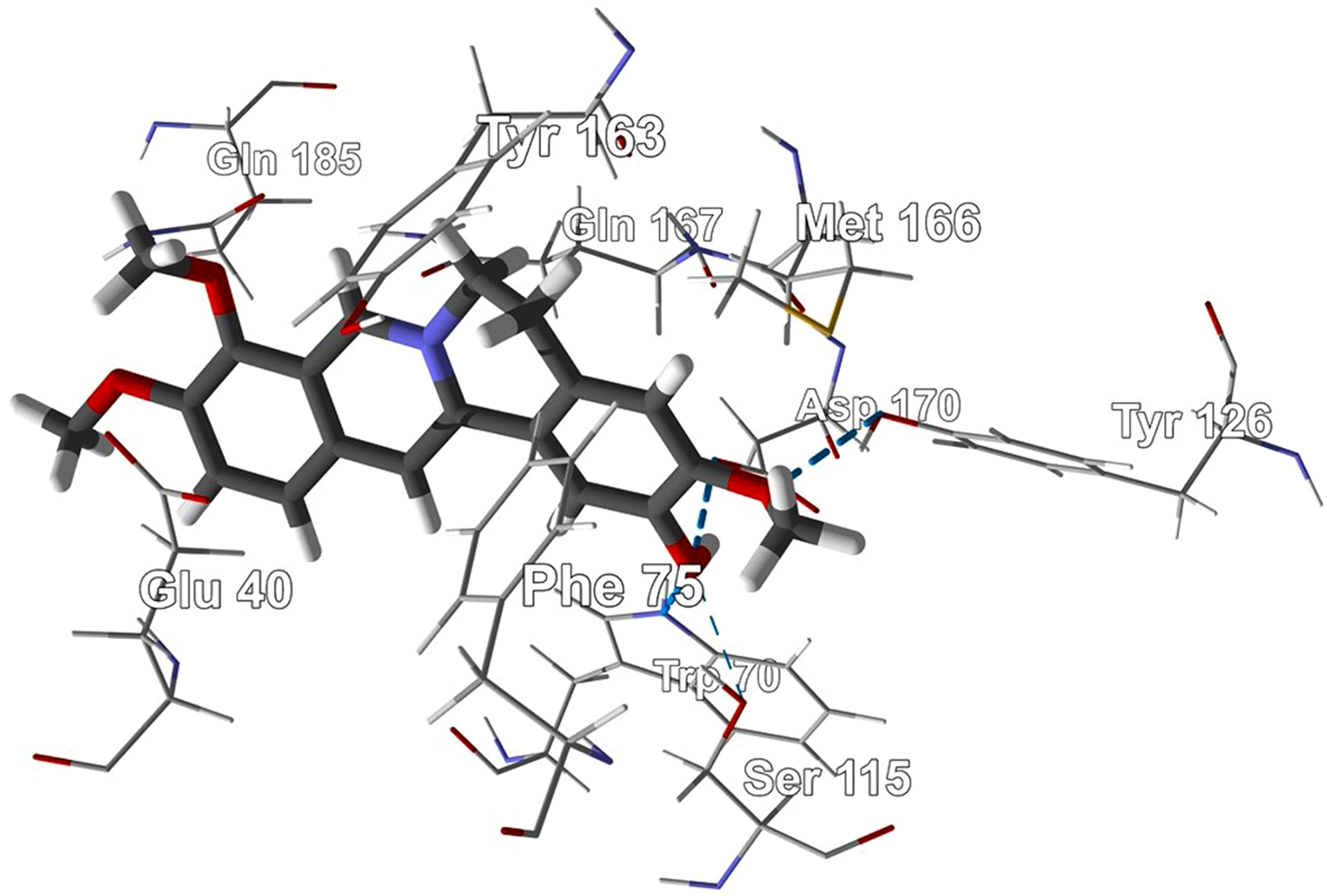
Lowest-energy docked pose of columbamine with Leishmania major tyrosyl-tRNA synthetase (PDB 3P0J). Hydrogen-bonding interactions are indicated with a blue dashed line.
Both the (R)- and (S)-enantiomers of 8-trichloromethyldihydroberberine were used in the docking analysis. 8R-TCMDHB showed preferential docking to L major MetRS (Edock = −102.7 kJ/mol) and L major OPB (Edock = −100.6 kJ/mol). The (S)-enantiomer (8S-TCMDHB) selectively docked to L major MetRS (Edock = −105.4 kJ/mol), L major PTR1 (Edock = −107.4 kJ/mol), L major deoxyuridine triphosphate nucleotidohydrolase (dUTPase) (Edock = −102.2 kJ/mol), and L infantum sterol 14-demethylase (CYP51) (Edock = −101.2 kJ/mol). The 8R-TCMDHB and 8S-TCMDHB ligands both occupy the active site of L major MetRS, but with very different orientations of the lowest-energy docked poses (Figure 8). The lowest-energy docked pose of 8S-TCMDHB with L major PTR1 placed the ligand into close proximity to the NADP+ cofactor (Figure 9) and with a lower docking energy than the co-crystallized ligand, 7,8-dihydrobiopterin (Edock = −95.5 kJ/mol). The ligand is sandwiched between the adenine ring of the cofactor and Phe113 (Figure 9). Additional key intermolecular interactions are hydrophobic interactions of the ligand with Tyr194, Asp181, and Thr184, and hydrogen-bonding interactions of the ligand with the NADP+ cofactor, Leu188, Thr195, and Arg17.

Lowest-energy docked poses of 8R-trichloromethyldihydroberberine (yellow stick figure) and 8S-trichloromethyldihydroberberine (green stick figure) with Leishmania major methionyl-tRNA synthetase (PDB 3KFL).
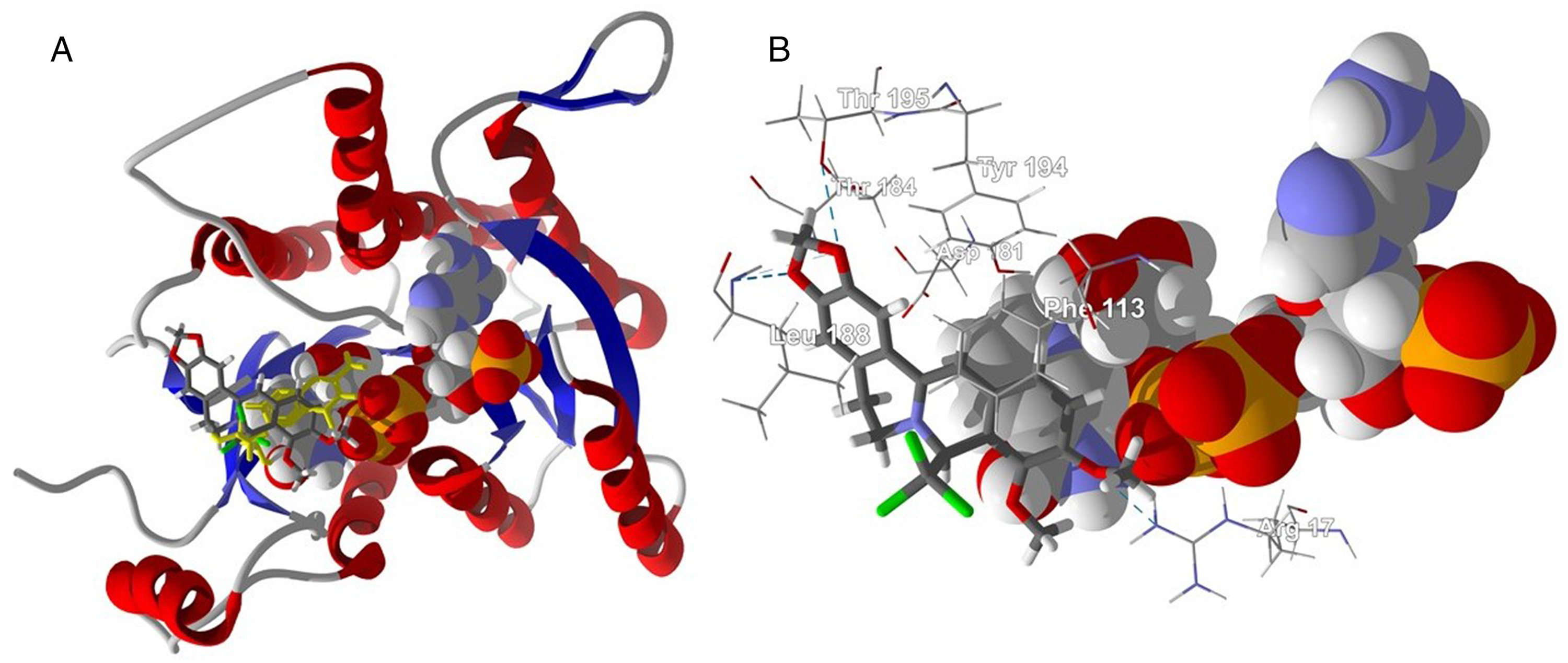
Lowest-energy docked pose of 8S-trichloromethyldihydroberberine and L major pteridine reductase 1 (PDB 2BF7). (A) Ribbon structure showing the orientation of the ligand (stick figure), the co-crystallized ligand (7,8-dihydrobiopterin, yellow stick figure), and the NADP+ cofactor (space-filling structure). (B) Docking site of the ligand showing key intermolecular contacts. Hydrogen-bonding interactions are indicated with a blue dashed line.
Promising docking interactions were also observed for 8S-TCMDHB with L major deoxyuridine triphosphate nucleotidohydrolase (dUTPase) (Edock = −102.2 kJ/mol) and with L infantum sterol 14-demethylase (CYP51) (Edock = −101.2 kJ/mol). The binding site for the ligand with L major dUTPase is a pocket formed by Phe83, Asn183, Asp79, Glu48, His82, Tyr191, and Arg186 (Figure 10), the same site as the co-crystallized ligand, 2′-deoxyuridine-5′-α,β-imido triphosphate (Edock = −118.6 kJ/mol). There is a face-to-face π–π interaction between the ligand and Phe83. Additional interactions include hydrogen bonding of the ligand with Lys179, Asn206, and Gln21. Docking of 8S-TCMDHB with L infantum CYP51 placed the ligand in close proximity to the heme cofactor with key interactions with residues Leu355, Tyr102, Met459, Phe104, and Pro209 (Figure 11). There do not seem to be any hydrogen-bonding interactions between 8S-TCMDHB and L infantum CYP51. The co-crystallized ligand, fluconazole, had a docking energy of −88.1 kJ/mol.

Lowest-energy docked pose of 8S-trichloromethyldihydroberberine and L major deoxyuridine triphosphate nucleotidohydrolase (PDB 2YAY) showing key intermolecular contacts. Hydrogen-bonding interactions are indicated with a blue dashed line.

Lowest-energy docked pose of 8S-trichloromethyldihydroberberine and L infantum sterol 14-demethylase (PDB 3L4D). (A) Ribbon structure showing the orientation of the ligand (stick figure), the co-crystallized ligand (fluconazole, yellow stick figure), and the heme cofactor (space-filling structure). (B) Docking site of the ligand showing key intermolecular contacts. The ligand is shown as a stick figure and the heme cofactor is shown as a space-filling structure.
There have been several reports in the literature regarding molecular docking of antiparasitic alkaloids with Leishmania protein targets. Ogungbe et al 15 carried out in-silico screening of 209 antiparasitic alkaloids with 24 Leishmania protein targets. The strongest docking alkaloid ligands were the indole alkaloids flinderole A and flinderol B and the bis(alkylpiperidinyl)indolizidine alkaloid juliflorine with L major MetRS; the bis(alkylpiperidinyl)indolizidine alkaloids juliflorine, juliprosine, prosopilosidine, and prosopilosine with Leishmania mexicana GPDH; and the naphthylisoquinoline ancistrogriffithine A with L major NMT. More recently, Lorenzo et al 14 used molecular docking to screen a library of 215 isoquinoline alkaloids on L donovani NMT. This study revealed linderegatine, lindoldhamine, ocoteamine, demerarine, and lancifoliaine to be the best docking alkaloids with L donovani NMT. Azadbakht et al 38 carried out a molecular docking study of tropolone alkaloids from Colchicum kurdicum and found that colchicoside showed a good docking score with tubulin as a potential molecular target (Colchicum alkaloids are known to be effective antitubulin agents). Wadanambi 39 has demonstrated, based on molecular docking, that the naphthylisoquinoline alkaloid ancistrotanzanine B and the steroidal alkaloid holamine may be effective inhibitors of L donovani squalene synthase.
All of the berberine alkaloids exhibited strong docking intercalation interactions with DNA with docking energies less than −130 kJ/mol (Table 3, Figure 12). The relatively exothermic docking energies of the alkaloids with DNA suggest that these compounds may also be cytotoxic to normal mammalian cells. However, the experimental selectivity indices (Table 1) do not support this. Previous investigations using molecular docking have shown that planar or nearly planar alkaloids such as skimmianine, 18 sanguinarine, 19 or coptisine 40 can readily intercalate DNA, presumably via favorable π–π interactions, and therefore serve as topoisomerase inhibitors. Nonplanar alkaloids such as vinblastine41,42 or carbolines, 43 on the other hand, interact with DNA via groove binding rather than intercalation.

Lowest-energy docked pose of berberine with DNA (PDB 465D). The DNA is shown as a stick figure and the intercalated berberine is shown as a space-filling structure.
Materials and Methods
Chemistry Procedures
Merck Kieselgel silica gel 60 PF254 (70-230 mesh ASTM, Merck) and Sephadex LH-20 (Amersham Biosciences) were used for column chromatography (CC) and thin-layer chromatography (TLC). Melting points were determined on a Stuart digital melting point apparatus (SMP 10, Cole-Parmer) and are uncorrected. Ultraviolet spectra were recorded on a Thermo Spectronic Unicam UV-300 (Thermo Scientific). Infrared spectra were recorded on JASCO FTIR-4200A (JASCO Deutschland GmbH) and Thermo Scientific FTIR Nicolet-380 (Thermo Scientific). Electron impact ionization mass spectrometry (EIMS) was carried out with the JEOL MS-route instrument (JEOL USA, Inc.). High-resolution electrospray ionization mass spectrometry (HR-ESIMS) was performed on a Thermo scientific exactive LCQ fleet instrument (Thermo Scientific). 1HNMR, 13CNMR, and 2D NMR were recorded on Bruker Avance DRX-400 and 500 MHz (Agilent) and JEOL 400 MHz (JEOL USA, Inc.) instruments.
Plant Material
Berberis glaucocarpa Stapf was collected from Azad Kashmir, Pakistan, during the flowering period and was identified by Professor Dr Tanveer Akhtar (chairperson, Botany Department, University of Azad Jammu and Kashmir). Voucher specimen number 9615-A was deposited in the herbarium of Botany Department, University of Peshawar.
Extraction and Isolation
Root bark (2 kg) was first dried at room temperature, pulverized and then extracted with commercial-grade methanol. This, upon concentration with a rotary evaporator, yielded a dark brownish-black residue (172 g). The residue was then treated with 5% aqueous HCl solution to afford fraction
Parasite Source
The strain KWH23 was provided by the London School of Hygiene and Tropical medicine. They acquired it, in 2010, from the lesion of an 8-year-old boy suffering from CL who was from Jamrud, Khyber Agency Khyber-Pakhtunkhwa, Pakistan.
Parasite Culture
Culture of L tropica clinical field isolate KWH23 was established from already cryo-preserved collection in the Leishmania molecular and culture laboratory at the Department of Zoology, University of Peshawar. The initial culture was raised in 5 mL M-199 medium supplemented with 10% heat-inactivated fetal bovine serum (HI-FBS), 100 µg/mL penicillin, 100 µg/mL streptomycin, 50 µg/mL kanamycin, and 5 µg/mL of Hemin in a 15-mL culture flask. The culture flask was kept in an incubator at 26°C and the medium was changed every third day. The cultures were given enough time (10-14 days) to fully develop and promastigotes to reach the metacyclic stage. The culture was then sub-passaged by pouring the contents of the flask containing the promastigotes into a centrifuge tube. The tubes were then spun using a Sigma centrifuge at 2000 r/min for 12 min. After spinning, the medium was carefully poured off leaving the pellet only. This pellet was re-suspended in a 2 to 4 mL growth medium and new cultures were established. The primary culture was used for sensitivity assays and additional cultures were cryopreserved in liquid nitrogen at −196°C for future use.
Berberine Type Alkaloids and Standard Drug Concentrations
Six standard concentrations of amphotericin B (Bristol), used as a standard antileishmanial drug, and the 5 isolated alkaloids (
Alkaloids Assay Against Leishmania tropica KWH23 Promastigotes
Antileishmanial assay of the isolated alkaloids was performed by using a modified Jamal et al
47
procedure. Briefly, the preserved isolates of L tropica KWH23 were cultured in growth medium (M199), which was supplemented, with 10% heat-inactivated fetal calf serum (HI-FCS), for 10 to 14 days at 24 to 26°C under anaerobic conditions. For the alkaloid sensitivity assay, 1 × 106 promastigotes/well in 200 μL fresh M199 medium in 96-well plates was treated with each concentration (100, 50, 25, 12.5, 6.25, and 3.125 µM) of berberine type alkaloids (
Cytotoxicity of Berberine Alkaloids Against THP-1 Cells
The cytotoxic effects of berberine type alkaloids and the standard drug (amphotericin B) were determined by using an MTT-based [3-(4.5-dimethylthiazol-2-yl)-2.5-diphenyl tetrazolium bromide] in-vitro toxicology assay kit (Sigma Aldrich) by following the manufacturer's instructions. All the concentrations (100, 50, 25, 12.5, 6.25, and 3.125 µM) of the berberine-type alkaloids (
Cytotoxicity of Alkaloids
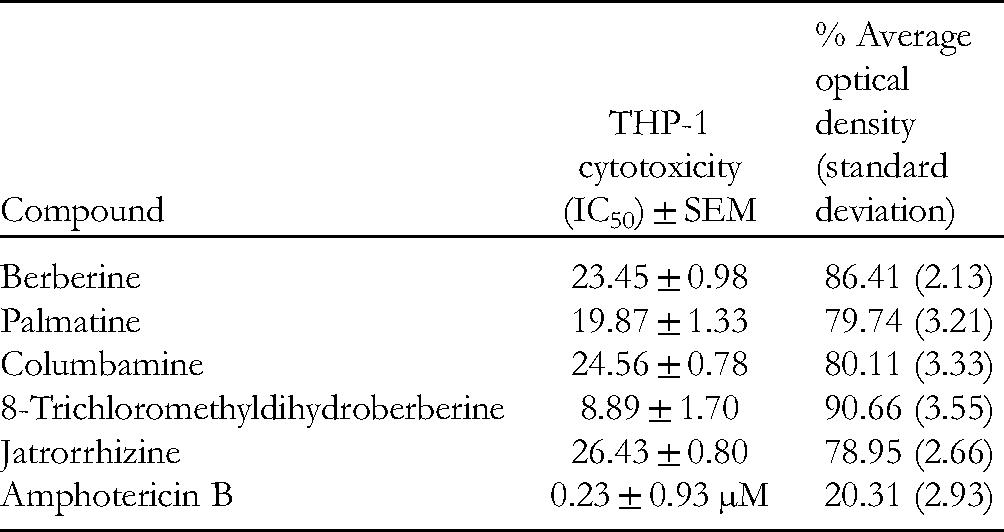
The median cytotoxic concentration (CC50) of berberine alkaloids was defined as the concentration (µM) of an alkaloid to reduce cell viability by 50% and was calculated by regression analysis.
Selectivity Index (SI) Calculation
SI was calculated by using the following formula:
Molecular Docking
Structures of the berberine alkaloids were prepared using Spartan 18 for Windows, v 1.4.4 (Wavefunction, Inc.). Molecular docking was carried out as previously described 49 using Molegro Virtual Docker v 6.0.1 (Molegro ApS). 50 A total of 85 Leishmania protein targets and 9 DNA structures from the Protein Data Bank (PDB) were used in the molecular docking. If a co-crystallized inhibitor or substrate was present in the structure, then that site was chosen as the binding site. If no co-crystallized ligand was present, then suitably sized cavities were used as potential binding sites. A 15-Å radius docking sphere was located on the binding sites of each biomolecular target structure. The biomolecular target and the ligand structures were prepared using the default parameters of the Molegro Virtual Docker program. Standard protonation states of the proteins based on neutral pH were used in the docking studies. Assignments of charges on each protein and the alkaloid ligands were based on standard templates as part of the Molegro Virtual Docker program; no other charges were set. The biomolecular target structures were used as rigid models without relaxation, but a flexible-ligand model was used in the docking optimizations. The root-mean-square deviation threshold for multiple cluster poses was set at <1.00 Å. The docking algorithm was set at maximum iterations of 1500 with a simplex evolution population size of 50 and a minimum of 100 runs for each ligand. The orientations of the ligands with the targets were ranked based on “rerank” energy values.
Conclusions
The results of our study indicated that the berberine alkaloids berberine, columbamine, jatrorrhizine, palmatine, and 8-trichloromethyldihydroberberine possess strong leishmanicidal properties against L tropica with low cytotoxicity on mammalian cells. Furthermore, the likely protein targets of the alkaloids are Leishmania NMT, MetRS, PTR1, OPB, TyrRS, and/or GPDH. Molecular docking has also indicated that intercalation of the alkaloids with Leishmania DNA may be important. Note, however, that there may be additional Leishmania biochemical targets that berberine alkaloids may affect. In addition to targeting Leishmania enzymes or DNA, berberine alkaloids may disrupt immunomodulatory signaling molecules 51 or may interact with the parasite mitochondria. 11 Note also that the molecular docking method itself suffers from inherent limitations: The protein is modeled as a rigid structure without flexibility, solvation of the active site and the ligand is excluded, and free-energy estimation of protein–ligand complexes is largely ignored. Nevertheless, this work confirms the general antileishmanial potential of berberine alkaloids and suggests promise for this class of compounds for further development as antileishmanial drugs.
Future studies on antiparasitic berberine alkaloids can be envisioned. This present work involved screening only against L tropica promastigotes; additional screening against the intracellular amastigotes needs to be carried out. Additional screening against other parasitic protozoa such as Trypanosoma spp. or other Leishmania spp. should be carried out to expand the scope of antiparasitic activity of these alkaloids. In addition, enzyme inhibition assays are needed to confirm the docking studies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211031148 - Supplemental material for Antileishmanial Potential of Berberine Alkaloids From Berberis glaucocarpa Roots: Molecular Docking Suggests Relevant Leishmania Protein Targets
Supplemental material, sj-docx-1-npx-10.1177_1934578X211031148 for Antileishmanial Potential of Berberine Alkaloids From Berberis glaucocarpa Roots: Molecular Docking Suggests Relevant Leishmania Protein Targets by Muhammad Alamzeb, Saqib Ali, Mamoon-Ur-Rashid, Behramand Khan, Ihsanullah, Adnan, Muhammad Omer, Asad Ullah, Javed Ali, William N. Setzer, Syed M. Salman, Ajmal Khan and Akram Shah in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X211031148 - Supplemental material for Antileishmanial Potential of Berberine Alkaloids From Berberis glaucocarpa Roots: Molecular Docking Suggests Relevant Leishmania Protein Targets
Supplemental material, sj-docx-2-npx-10.1177_1934578X211031148 for Antileishmanial Potential of Berberine Alkaloids From Berberis glaucocarpa Roots: Molecular Docking Suggests Relevant Leishmania Protein Targets by Muhammad Alamzeb, Saqib Ali, Mamoon-Ur-Rashid, Behramand Khan, Ihsanullah, Adnan, Muhammad Omer, Asad Ullah, Javed Ali, William N. Setzer, Syed M. Salman, Ajmal Khan and Akram Shah in Natural Product Communications
Footnotes
Acknowledgments
Higher Education Commission of Pakistan is acknowledged for its support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
