Abstract
Drug repurposing is commonly employed in the search for potential therapeutic agents.
As the coronavirus disease (COVID-19) pandemic worsens, with over 2 million related deaths by early 2021, the need for an effective treatment has become even more urgent.
6
Several vaccines have received conditional approval for emergency use, while others are still being investigated with aims of slowing down transmission and preventing severe cases of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.
7
-10
Although community immunization programs have been rolled out in several countries, the timely accessibility to vaccines, especially in middle and low-income countries, remains challenging. Supply capacity, cost, and logistics issues such as the maintenance of cold-chain transport and storage are significant hurdles for poorer countries and rural areas.
11
As one of the popular herbs used for the common cold, cough, and flu,
In general, preclinical studies have reported mostly antibacterial
14
-16
and anti-inflammatory effects
17
-19
for
In January 2021, Thailand announced its pilot program for administering and investigating the effectiveness of
Results and Discussion
Study Inclusion
A total of 139 records were identified through keyword searches on online databases of published journals and gray literature. Of the 139 records, 12 articles are included in this review as presented in the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) flow chart (Figure 1).

Preferred reporting items for systematic review and meta-analysis (PRISMA) flow chart of articles inclusion.
Study Demographics
All 12 articles identified and included based on this review’s inclusion and exclusion criteria are on preclinical studies. From the 12 studies, more than half (58.3%) were reported by Indian scientists. All of the Indian studies were computational investigations and analysis. In fact, a majority (83.3%) of the total papers identified were exclusively in silico papers reporting molecular docking and 3D simulation of the interaction of phytochemical compounds to potential therapeutic targets to prevent attachment and replication of SARS-CoV-2. There were only 2 articles reporting in vitro results. No articles covered clinical and in vivo preclinical studies. Although the search was conducted to identify studies investigating both
Study Demographics.

Results: Antiviral Evidence Against SARS-CoV-2
In silico
In silico docking and simulation studies revealed andrographolide’s potential to bind with several therapeutic targets that are important for viral attachment to host cells, replication, and production (Table 2). In several studies, various docking methods and software consistently predicted negative binding energy (ie, indicating potential for good binding affinity) for andrographolides and its derivatives when docked against SARS-CoV-2 spike protein-angiotensin converting enzyme (ACE)−2 complex, spike protein, ACE-2 receptor, 3-chymotrypsin-like protease (3CLpro, previously known as SARS-CoV-2 main protease (Mpro)), and RNA dependent RNA polymerase (RdRp). 29 -39 3CLpro was the most commonly investigated target in 9 out of 11 in silico studies (81.8%). 30 -32,34 -39 Negative binding energy was also predicted for andrographolide and its derivatives when docked against SARS-CoV-2 papain-like protease (PLpro) 30 and nucleocapsid protein binding domain 37 by single studies. The overall potential multi-modal mechanisms of action of andrographolide and its analogs are illustrated in Figure 2.
Predicted Ligand-Receptor Interactions of Andrographolide and Its Analogs Against Potential SARS-CoV-2 Antiviral Therapeutic Targets Based on Molecular Modeling.

Abbreviations: NA = not applicable; NS = not specified; PDB = protein data bank.
1Not obtained from database, but was modified from the SARS coronavirus proteins instead to mimic the corresponding SARS-CoV-2 protein, hence a PDB ID is not available.
2Details on amino acid residues bond interactions were not extractable from paper.
3Targets may have additional functions in modulating host-pathogen interactions.
4Negative binding energy denotes potential for binding. A more negative value represents better predicted binding affinity than a less negative value in the same study.

Potential SARS-CoV-2 antiviral target sites and mechanisms of actions predicted for andrographolide and its derivatives as indicated by the red boxes: (1) spike protein-ACE-2 complex, (2) spike protein, (3) host cell ACE-2 receptor, (4) PLpro, (5) 3CLpro, (6) RdRp, and (7) nucleotide protein binding domain. The diagram illustrates the overall suggested life cycle of SARS-CoV-2 infecting a human lung cell, including (
Eight studies (72.7%) compared the binding affinities and energies of andrographolide with standard reference drugs
29,31,33
-37,39
while 9 studies (81.8%) compared these parameters with other phytochemicals, including analogs of andrographolide and other bioactive compounds of
Comparison Summary of Antiviral Receptor-Ligand Activity of Andrographolide With Reference Drugs/ Compounds and Other Phytochemicals Against SARS-CoV-2. 29,31 -37,39
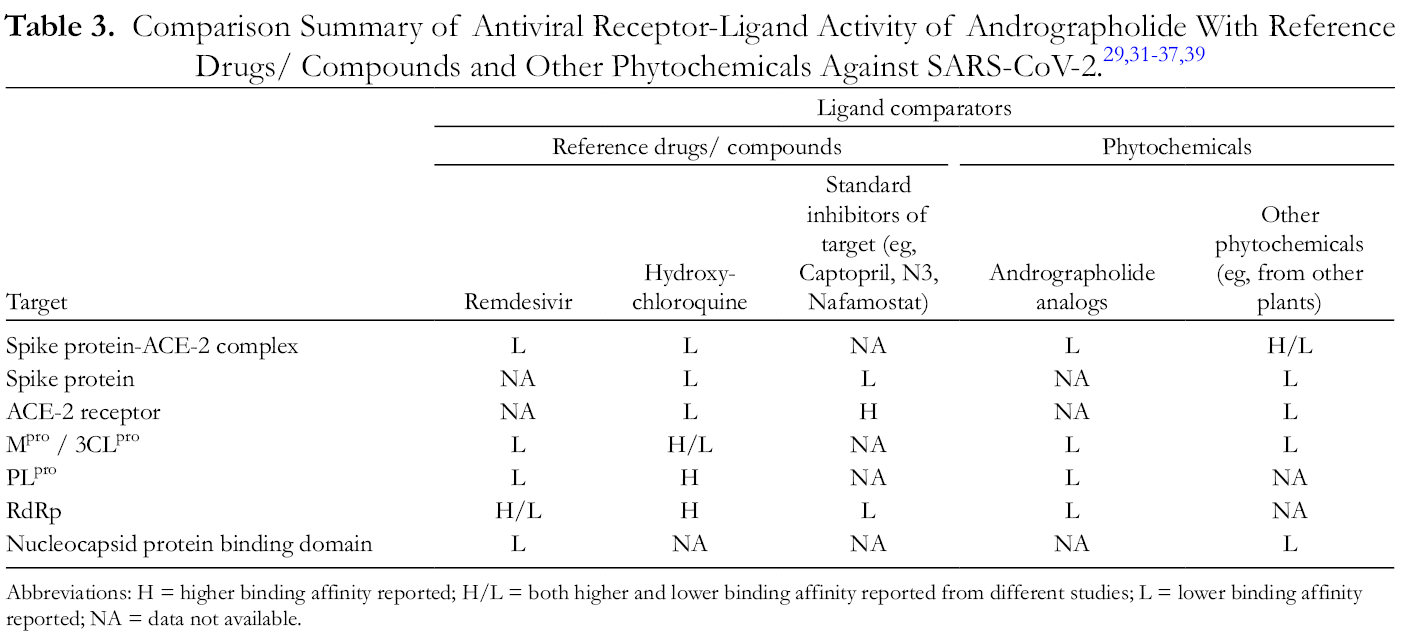
Abbreviations: H = higher binding affinity reported; H/L = both higher and lower binding affinity reported from different studies; L = lower binding affinity reported; NA = data not available.
In vitro
Two in vitro studies that investigated the antiviral efficacy of
Discussion: Antiviral Evidence Against SARS-CoV-2
Based on media coverage, andrographolide, a constituent of
The versatility of andrographolide as a SARS-CoV-2 antiviral is demonstrated by its potential to bind to several important targets at various stages of viral attachment, replication, and host-pathogen interactions. 29 -34,36 -39 This property may be an important advantage to any potential therapeutic agent being developed. Viral life cycle modeling studies have suggested that effective infection attenuation of repurposed drugs is more likely to be achieved when multiple segments of the viral life cycle are targeted, especially when timely administration of an antiviral in the early phases of infection is challenging in real-life settings. 52,53
Molecular dynamics simulation suggests that andrographolide can bind to interfacial regions of both the SARS-CoV-2 spike protein and human ACE-2 receptors. 29,33 The SARS-CoV-2 spike protein-ACE-2 attachment is one of the most well studied and recognized interactions that constitute the first step of viral invasion into human lung cells. 54 This step is thought to initiate viral membrane fusion, entry, and initiation of downstream inflammatory and immunogenic effects. 55 Another study predicted that andrographolide and its derivatives bind allosterically to the spike protein S1 subunit of the spike protein-ACE-2 complex, instead of interfacial regions. Therefore, it was suggested that this allosteric binding may alter the conformation of the complex and subsequently interfere with the binding of SARS-CoV-2 to the ACE-2 receptors. 34 This study did not investigate further the dynamics of allosteric modulation and its effects on virus spike protein and human ACE-2 receptor interaction. As the spike protein-ACE-2 interaction is thought to be one of the key targets of anti-SARS-CoV-2 therapeutic candidates, 56 there are existing concerns that administration of potent ACE-2 inhibitors may eventually lead to upregulation of ACE-2 receptors at lung epithelial cell surfaces. Ironically, ACE-2 receptors at lung cell surfaces may have a protective role, especially at severe stages of the infection with overt inflammation. 57,58 Therefore, the benefits of ACE-2 blocking are still debated. As the involvement of the ACE-2 receptor in multiple physiological functions further complicates its position as a potential therapeutic target, it has been suggested that antivirals targeting multiple pathways of the viral life-cycle may be more effective. 59
Two papers predicted good binding of andrographolide and its derivatives with RdRp.
34,37
At present, the first and only antiviral currently approved by the U.S. FDA for the treatment of severe COVID-19, Remdesivir, is an antiviral that is thought to act through inhibition of RdRp, though its benefit in lowering mortality in COVID-19 patients remains to be investigated.
60
Remdesivir is a repurposed broad-spectrum antiviral against RNA viruses, originally developed for Ebola.
61
It is a prodrug, which after a series of transformations, forms a nucleoside triphosphate to be uptaken by the virus’s RdRp enzyme, a crucial enzyme responsible for the RNA replication of coronavirus. This intentional but faulty incorporation is thought to be responsible for the viral replication inhibitory action of Remdesivir.
62
Unlike Remdesivir’s mechanism of action as a prodrug,
Several in silico studies collectively indicated the potential of andrographolide binding to SARS-CoV-2 3CLpro,
30
-32,34
-39
supported by a target-based in vitro study.
38
3CLpro is an important protease enzyme involved in viral genome replication, transcription, translation, and other cellular processes in coronaviruses,
63,64
though details of its molecular processes in SARS-CoV-2 are yet to be fully elucidated.
65
Molecular modeling suggested that andrographolide behaves as an electrophilic Michael acceptor inhibitor and binds to the catalytic pocket of SARS-CoV-2, similar to N3, a computer designed potent coronavirus and SARS-CoV-2 3CLpro inhibitor.
38
Compared to N3, the andrographolide analog 3–
Andrographolide and its derivatives were also predicted to bind to 2 lesser investigated therapeutic targets of SARS-CoV-2, PLpro and nucleocapsid protein N-terminal RNA binding domain. 34,36 Both PLpro and nucleocapsid protein (N-protein) are thought to play multiple roles in viral replication and host-pathogen interactions. 68 In addition to attenuating viral replication, PLpro inhibition reportedly reduced the virus-induced pathogenic effects in host cells. 68,69 The structure of the binding domain for the RNA terminal on the SARS-CoV-2 N-protein is one of the most recently elucidated structures. 70 In SARS-CoV-2, the N-protein is suggested to promote viral transcription via RNA-induced liquid-liquid phase separation to form condensates with RNA and RdRp. 71 The structure of the SARS-CoV-2 N-protein has been reported to be similar to previously reported coronaviruses, with some differences in surface electrostatic potential, while details of its molecular immunogenic pathways in SARS-CoV-2 infection remain to be established. 72
As single compounds, andrographolide and its analogs were generally not predicted to have the best binding affinity when compared to standard drugs like Remdesivir or other phytochemicals such as curcumin (Table 3 and Supporting information: Supplemental Table S1). However, andrographolide and related compounds were predicted to have binding affinity to several key components of the SARS-CoV-2 life cycle and pathogenicity. 29,31 -39 This potential ability to target multiple pathways of viral attachment, replication, and function may provide a unique advantage for andrographolide to be investigated as a versatile antiviral against SARS-CoV-2. When comparison is made between andrographolide and its analogs, the analogs, especially neoandrographolide and bisandrographolide, have better predicted binding affinities regardless of target receptor. 32,34,36,37 Future research to verify these andrographolide analogs as being better SARS-CoV-2 antivirals may be warranted.
There is rising interest in repurposing known small drug molecules as potential therapeutic candidates, especially in emergency situations like the present COVID-19 pandemic. The use of in silico methods and the application of molecular modeling provide a rapid way to screen available compound databases, explore the activities of potential therapeutic candidates through specific algorithms, as well as predict a ligand’s potential to be developed as a medicinal drug.
73
In addition to general screening of databases that encompass tens of thousands, and at times up to millions of available chemical structures, targeted screening for potential therapeutic candidates based on structural similarities with side chains or structures of known drugs for a particular indication can be performed. In this review, it was observed that andrographolide was identified as a potential ligand that can bind to various SARS-CoV-2 viral targets from different screening methods. General screening on more than ten thousand compounds and targeted screening on selected groups of potential medicinal plants predicted the binding affinities of andrographolide and its analogs, highlighting their potential to be investigated further in vivo. Studies applying structure-based screening which successfully identified andrographolide and its analogs as potential antivirals against SARS-CoV-2 were not found. However, molecular simulation suggested minor similarities in hydrogen bonding of 3–
Regardless of the prediction accuracy of an in silico approach, the integration of phenotypic and target-based assays can further substantiate the role of a potential therapeutic candidate in managing a disease.
74
Despite the high number of in silico articles collectively predicting the anti-SARS-CoV-2 activity of andrographolide, in vivo data to support such predictions are still limited. Preclinical data on efficacy and safety from in vivo studies are generally required to conduct further clinical trials.
75
Specifically for SARS-CoV-2, it has been shown that in vitro antiviral efficacy demonstrated using Vero-cells (similar to the cell models used in one of the in vitro studies included in this review
43
needs to be validated in primary human airway epithelial cells and in vivo studies.
76
Therefore, to support these findings, a recent preprint on SARS-CoV-2 antiviral evidence of
In addition to their multiple targets on the SARS-CoV-2 viral life cycle, andrographolide and the major bioactive compounds of
The looming safety concern over andrographolide is primarily due to the premature halt of a phase 1 dose-escalating clinical trial among HIV patients. In that study, one out of 13 patients reported an anaphylactic event, which was not fatal.
92
That being said,
Review Limitations
This rapid review has a few limitations, mainly the lack of in vivo data to support all of the in silico predictions, which, therefore, does not enable us to draw strong conclusions on the SARS-CoV-2 antiviral effects of andrographolide. As only one phenotypic-based in vitro study briefly investigated
Conclusions
In conclusion, in silico modeling has predicted the potential of andrographolide and its analogs to exert SARS-CoV-2 antiviral effects by binding to several key targets in SARS-CoV-2 infection, including the spike protein-ACE-2 receptor complex, spike protein, ACE-2 receptor, RdRp, 3CLpro, PLpro, and N-protein RNA-binding domain. The potential SARS-CoV-2 antiviral activities of
Materials and Methods
Research Question
This review aimed to address the main research question “What is the available scientific evidence on the antiviral effects and mechanisms of
Population, Intervention, Comparator, Outcome (PICO) Elements of the Research Question.

Search Strategy and Articles Inclusion
A search using predetermined keywords and synonyms of
Data Extraction and Analysis
Data extraction was carried out by 2 independent investigators using a predesigned data extraction table (Supporting information: Supplemental Table S3). Disagreements were reviewed by a third investigator. The main data extracted include study characteristics (author, year, title, country, type of study, objective), methodology (study model, in silico model and program), intervention details (description, dose, frequency, duration), comparator (description, dose, frequency, duration), and outcome (quantitative values, qualitative description, comparison with comparator).
In line with the nature of rapid reviews, in addition to our findings that most were in silico data with different study models, programs, and simulation settings applied; the data obtained were numerically and descriptively analyzed. Numerical descriptive analysis was carried out for type of study, country, and methods leading to the discovery of
Supplemental Material
Table S1 - Supplemental material for Andrographis paniculata (Burm. F.) Wall. Ex Nees, Andrographolide, and Andrographolide Analogues as SARS-CoV-2 Antivirals? A Rapid Review
Supplemental material, Table S1, for
Supplemental Material
Table S2 - Supplemental material for Andrographis paniculata (Burm. F.) Wall. Ex Nees, Andrographolide, and Andrographolide Analogues as SARS-CoV-2 Antivirals? A Rapid Review
Supplemental material, Table S2, for
Supplemental Material
Table S3 - Supplemental material for Andrographis paniculata (Burm. F.) Wall. Ex Nees, Andrographolide, and Andrographolide Analogues as SARS-CoV-2 Antivirals? A Rapid Review
Supplemental material, Table S3, for
Supplemental Material
Online supplementary file 1 - Supplemental material for Andrographis paniculata (Burm. F.) Wall. Ex Nees, Andrographolide, and Andrographolide Analogues as SARS-CoV-2 Antivirals? A Rapid Review
Supplemental material, Online supplementary file 1, for
Footnotes
Acknowledgments
We would like to thank the Director General of Health Malaysia, Deputy Director General of Health Malaysia (Research & Technical Support), Director of Institute for Medical Research, and Head Centre of Herbal Medicine Research Centre for their support and permission to publish this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
