Abstract
The literature on coronavirus disease 2019 (COVID-19) treatments, including herbal medicine, has expanded rapidly due to intense global interest. We conducted this scoping review to map the characteristics and assess the quality of systematically conducted reviews of herbal medicine and COVID-19 published during the pandemic era. Electronic databases were searched to identify published systematic reviews and other research synthesis articles relating to herbal medicine vis-a-vis COVID-19 from 2020 to 2022. Forty-eight articles were included. Most were systematic reviews (SR) and/or meta-analyses (MA) on Chinese herbal medicine for clinical treatment during the acute phases of COVID-19 (44%). Regarding adherence to a measurement tool to assess systematic reviews (AMSTAR-2) critical items, only one-third (n = 15) reported an a priori protocol, while 21% (n = 10) presented a comprehensive literature search strategy. When analysed by type of review, SR/MA (n = 23) of clinical interventions had better than the overall average adherence rate in having a pre-registered protocol (87%, n = 20) and comprehensive literature search strategy (45%, n = 9). Exploratory analysis showed that most articles reported search results from at least two established electronic databases. Risk of bias assessment was performed in almost all SR/MA though there was limited exploration of any potential sources of heterogeneity. Overall, there is a need to improve the methodological design and implementation of systematically conducted reviews of herbal medicine in general to enhance the robustness and reliability of findings, especially given the rapid expansion of literature during unique circumstances such as the COVID-19 pandemic.
Introduction
Since the start of the coronavirus disease 2019 (COVID-19) pandemic in 2020, there has been a proliferation of primary and secondary literature surrounding the topic. Strongly driven by the need for information vital for responding to this new disease, research on various topics, including treatment, prevention, diagnosis, and public health measures, have been conducted and published rapidly. The cumulative amount of synthesis research (ie, reviews on COVID-19) produced over the last two years is fairly large.1‐3
One rapidly growing area of interest largely boosted by the COVID-19 pandemic is the role of alternative medicine, including herbal medicine. 4 Accordingly, there has been a surge in herbal medicine review publications, which raises concerns about the quality of those reviews. 5 To date, several scoping and systematic reviews assessing the quality of COVID-19 reviews have been published,5‐9 of which three were about or related to herbal medicine.7‐9 These were interventional reviews performed to collate and assess systematic reviews, with the common primary objective of summarising the effectiveness of herbal medicine for COVID-19 while considering the quality of the evaluated systematic reviews.7‐9 Based on the general distribution of types of literature, it can be anticipated that findings from these overviews of systematic reviews represent only a small portion of the body of published reviews on herbal medicine. It is expected that the bulk of evidence for herbal medicine research is more likely to be preliminary and exploratory. These articles may not satisfy the strict requirements of systematic reviews but may be more appropriately evaluated under other types of synthesis research, such as scoping or narrative reviews. 10
Therefore, to better map the characteristics and overall quality of COVID-19 associated herbal medicine reviews, we have conducted a scoping review of systematically conducted reviews of herbal medicine concerning COVID-19 published during the pandemic era. For this review, systematically conducted reviews refer to those conducted with a robust, reproducible, and clearly-reported search, screen, and article inclusion strategy.
Materials and Methods
This scoping review follows the preferred reporting items for systematic reviews and meta-analyses extension for scoping reviews (PRISMA-ScR) checklist (Supplementary File 1). 11 An a priori protocol was developed before the commencement of this study but could not be registered with PROSPERO as it currently does not accept scoping review protocols for registration. 12 This research is registered with the Malaysian National Medical Research Register (NMRR ID-22-00950-VH5).
Objectives
To assess, collate, and present an overview of the characteristics and quality of published reviews on herbal medicine and COVID-19, conducted systematically.
Search Strategy
We searched three online journal databases: PubMed, Web of Science, and Cochrane CENTRAL, using the keywords (covid-19 OR SARS-CoV-2 OR covid19 OR 2019-nCoV OR coronavirus) AND (herbal OR herb OR phytotherapy OR plant OR plants OR phytomedicine OR botanical) for relevant review papers that were published from January 2020 to April 2022. The complete search strategy for all three databases is presented in Supplementary File 2.
Article Inclusion
Articles found were deduplicated using EndNote X8, followed by title and abstract screening using Rayyan AI,
13
and then full-text screening based on pre-determined inclusion and exclusion criteria. A pair of independent investigators performed all screening processes, with disparities addressed by a third.
Article type and synthesis quality: Topic: Language:
Review articles that reported clear, robust, and reproducible systematic search strategies were included. This included articles with clear reporting of keywords and databases (at least two). Studies that did not have explicit research question(s) based on the essential components of population, intervention, control, outcome (PICO) or population (or participants), concept, and context (PCC), where relevant, were excluded.
Articles that reported direct evidence on the use of herbal medicine for COVID-19. Direct evidence on COVID-19 was defined as evidence obtained from COVID-19 patients or preclinical in vivo/in vitro/in silico models of COVID-19. Articles presenting inferred benefits of herbal medicine in COVID-19 with no explicit COVID-19 findings were excluded.
English articles only.
These inclusion and exclusion criteria were considered important in ensuring that only synthesis research of sufficient minimal quality were included, based on the recommendations for critical components of a systematic review as stated by the a measurement tool to assess systematic reviews (AMSTAR-2) tool. 14 Only review articles with direct evidence on COVID-19 were included as many herbal medicine reviews inferred and hypothesised the roles and benefits of herbal medicine in COVID-19 without direct objective evidence.
Data Extraction and Analysis
Data extraction was performed by a pair of independent investigators using a pre-designed Google Form (Supplementary File 3). Before the commencement of full-text data extraction, pilot testing was performed among all investigators on 10% of the articles eligible for full-text screening to achieve a consistent understanding of the usage of the Google Form. Collected data were generated and downloaded as Microsoft Excel files for individual investigators and then matched using Microsoft Excel. The same pair of investigators further verified the computer-assisted matched results by performing manual verification. Disagreements were discussed with a third investigator.
The following data were extracted and analysed.
Review Characteristics
The following data were extracted for numerical and descriptive analysis to produce an overview of the characteristics of included reviews.
Name of intervention Review type: Systematic review, meta-analyses, systematic review and meta-analyses, scoping review, umbrella review. For this study, systematic and scoping reviews were differentiated by the broadness of the topic reviewed, that is, systematic reviews have precise research questions with narrow topics, for example, effectiveness or efficacy of treatment, while scoping reviews usually provide an overview or overall map of evidence on a broader topic.
10
An umbrella review is defined as a review of reviews.
15
Type of studies included in the original review Topic of research: Treatment, prevention, pattern of use, guidelines, and others. Modality of treatment: Traditional (treatment principles based on traditional medicine principles/use) or botanical (ie, treatment principles based on modern medicine principles)
Quality of Reviews
Sixteen items as per the AMSTAR-2 tool were assessed. 14 As the AMSTAR-2 tool is designed specifically for systematic reviews of clinical intervention, an additional choice of N/A (not applicable) was added for items to cater for studies that are not clinical interventional systematic reviews. The AMSTAR-2 tool and its applicability to different types of reviews are presented in Supplementary File 4. Individual items with inadequate ratings were identified and descriptively analysed. Although it is not recommended to combine ratings from individual items to generate an overall score, for the purpose of comparing the quality of herbal medicine reviews against a previously published review on the quality of all COVID-19-related systematic synthesis research, the same scoring system as previously reported was employed. 5 Briefly, reviews were downgraded 1.0 for full (Response: No) or 0.5 for partial (Response: Partial yes) failure of adherence to an item on the AMSTAR-2 tool, wherever applicable. No downgrades were employed in cases of full adherence (Response: Yes) or non-applicability (Response: N/A). An overall rating of low (≥3.5 downgrades), medium (>1 and ≤3.0 downgrades), and high (≤1.0 downgrades) was then applied.
Sub-Analysis of Databases Searched in Included Articles
For included articles, we additionally analysed the type of databases searched in each article. In general, there are no fixed guidelines for assessing the suitability and adequacy of searched databases. However, the JBI-evidence synthesis manual suggests an initial search of at least two databases. 16 Therefore, this sub-analysis is exploratory, and the concept is based on the general recommendation of searching at least 2 or 3 major credible databases. 17 Due to the exploratory nature of this analysis, information on grey literature search was not included.
Results and Discussions
Of 1333 records identified, 118 articles were eligible for full-text screening, of which 48 articles7,9,18‐63 were included in this review. Of the 70 articles which were excluded during full-text screening, 66 did not have a clear, robust, and reproducible search strategy and/or findings with direct and objective evidence on the use of herbal medicine among COVID-19 patients or in COVID-19 preclinical models. Article screening and selection are presented in Figure 1, while a list of excluded articles (n = 70) with their corresponding reasons for exclusion is available in Supplementary File 5.
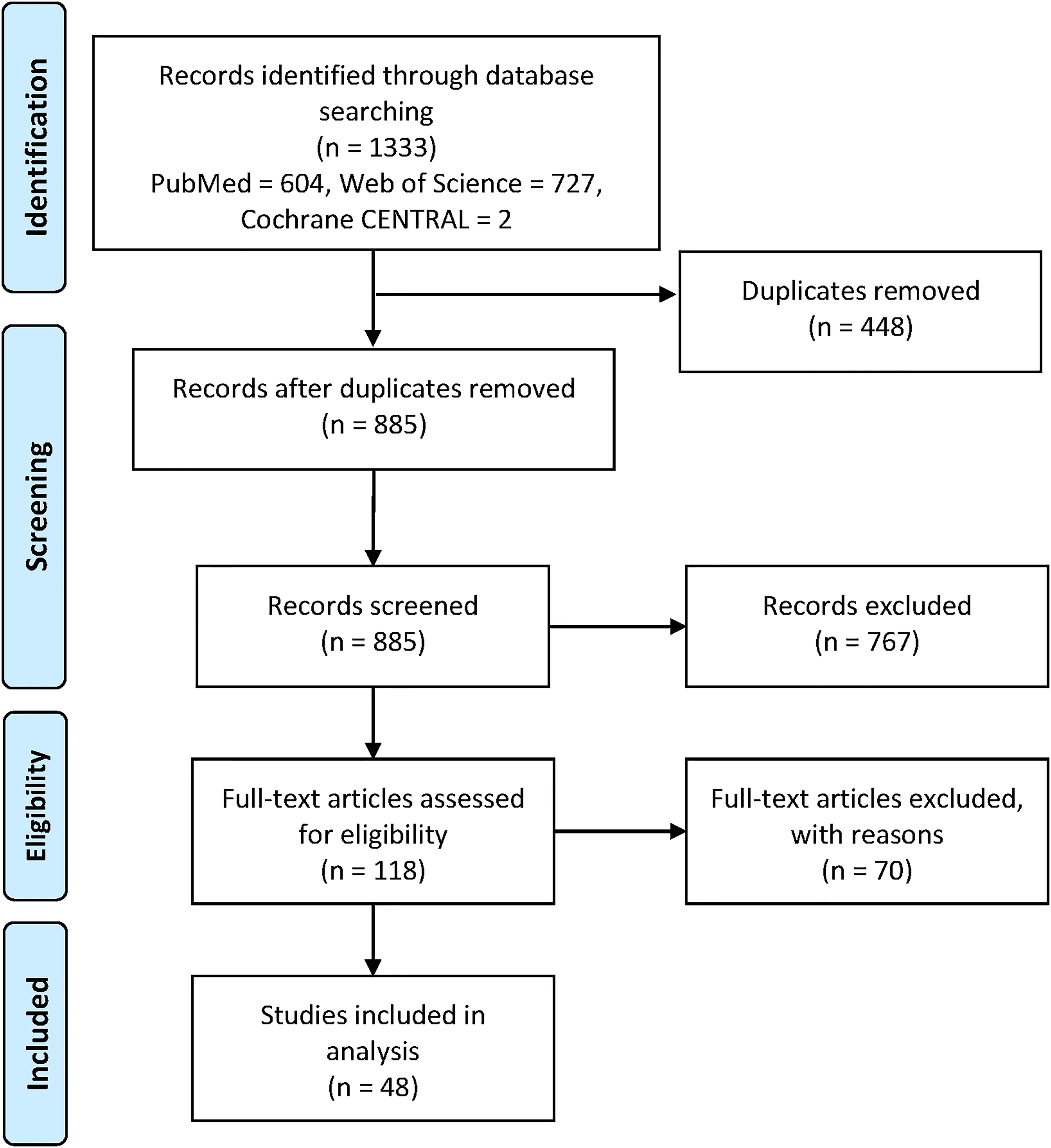
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flow diagram of study inclusion.
Review Characteristics
Review Types
Most included reviews were scoping reviews (47.9%) and systematic reviews (SR) and/or meta-analyses (MA) (45.8%), while the remainders were umbrella reviews and a rapid review (Figure 2a).
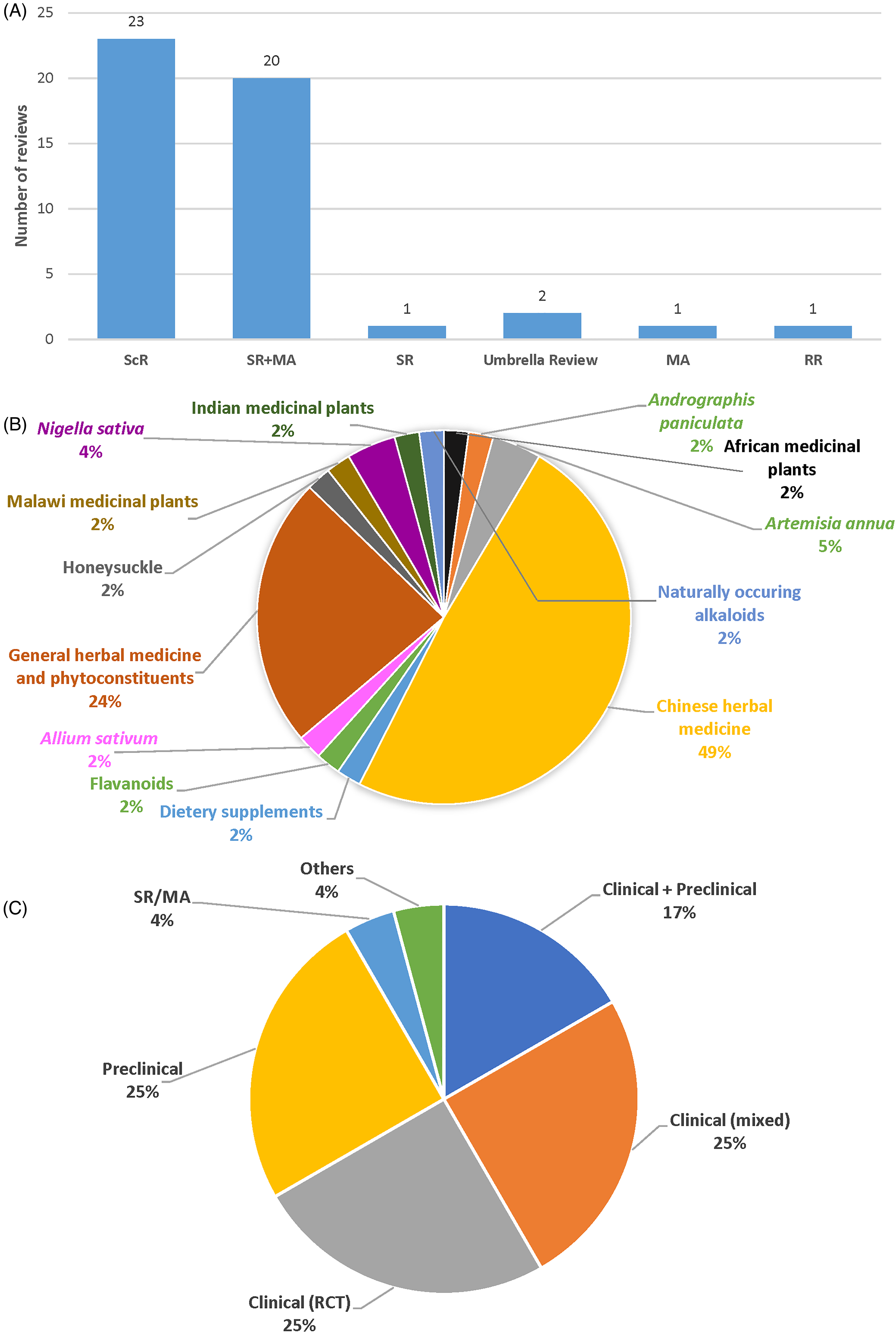
Characteristics of included reviews. (A) Review types. (B) Review topic. (C) Types of articles included. Clinical (mixed) refers to a mixture of clinical studies, including RCT, NRSI, cohort, case-control, observational, case reports, and case series. MA: meta-analyses; NRSI: non-randomised studies of intervention; RCT: randomised controlled trials; RR: rapid review; ScR: scoping reviews; SR: systematic reviews.
Review Topic
Almost all of the included articles were intervention-based reviews (97.9%, n = 47), while one was a wide-ranging qualitative review of various COVID-19 topics, such as treatments with an herbal medicine component as one of its findings. Of the 47 intervention-based reviews, Chinese herbal medicine was the most frequently reviewed (49%), followed by reviews that provided an overview of the effects of herbal medicines and associated general phytoconstituents in COVID-19 (24%). Few studies focused on single herbs such as Allium sativum L. (n = 1), Nigella sativa L. (n = 2), Artemisia annua L. (n = 2), and Andrographis paniculata (Burm.f.) Nees (n = 1). Apart from Chinese medicine, other traditional treatment modalities reviewed were Indian and Malawian traditional medicinal plants (n = 1 for each) (Figure 2b). All intervention-based reviews focused on herbal medicine's role in treating acute phases of COVID-19, while none provided direct evidence of post-COVID-19 complications.
Type of Articles Included in Published Reviews
The types of articles included in the published reviews identified in this study are shown in Figure 2c. Half of the reviews were of clinical studies. A quarter was performed solely on preclinical studies, while 17% of reviews included a mixture of clinical and preclinical studies. Other study types (n = 2) included in the reviews were perspective and opinion-based articles.
Quality of Reviews
Table 1 shows the overall percentage adherence of included reviews to items (Item No. 1, 2, 4, 5, 6, 7, 8, 16) on the AMSTAR-2 tool with regard to all types of reviews. Table 2 shows the overall percentage adherence of included SR/MA for all items on the AMSTAR-2 tool. Overall, most of the included studies did not adhere to most items on the AMSTAR-2 tool. When narrowed down to SR/MA of human interventional trials, the adherence was better compared to results from the overall assessment across all types of reviews. Most studies (∼60%), except for SR/MA of human interventional trials, did not explicitly state the availability or registration of an a priori protocol. This was similarly observed for other AMSTAR-2 items on the adequacy of inclusion and exclusion criteria to support the research questions, a comprehensive literature search strategy, and performing article screening and data extraction in duplicates. One item frequently not addressed by most of the included reviews, including SR/MA, was listing or citing excluded studies with their corresponding reasons for exclusion. Only about 10% of the included reviews reported this item adequately. We also found that most studies (∼97%) did not sufficiently describe the study characteristics of comparator, settings, and/or time-frame for follow-up. Specifically for SR/MA, there was limited exploration of potential sources of heterogeneity (which was reportedly high in pooled estimates of individual reviews) as well as inadequacy in adjusting for confounding factors when pooling non-randomised studies of intervention (NRSI), although risk of bias was assessed using appropriate tools for both randomised controlled trials (RCT) and NRSI (Cochrane Risk of Bias tool 1.0 and 2.064,65; and New-Castle Ottawa Scale 66 ). Pooled scoring shows that the quality of only one study was considered moderate, while the rest were assessed as low. Detailed assessment and scoring of each review and item are presented in Supplementary File 6.
Percentage adherence to AMSTAR-2 tool (item No. 1, 2, 4, 5, 6, 7, 8, 16) of all included reviews (n = 48).

PCC: Population (or Participants), Concept, and Context; PICO: Population, Intervention, Comparator, and Outcome.
Percentage adherence to AMSTAR-2 tool of all included systematic reviews and/or meta-analyses of human interventional trials.

a Both overall (combined assessment on analysis of RCT and NRSI in the same review) and individual results (separate analysis for RCT and NRSI in the same review) are presented.
NRSI: non-randomised studies of intervention; PCC: population (or participants), concept, and context; PICO: population, intervention, comparator, and outcome; RCT: randomised controlled trials; RoB: risk of bias.
Exploratory Analysis: Databases Searched
Of the 48 reviews included, 93.8% searched at least two while 79.2% searched at least three electronic databases for published articles. In contrast, two reviews searched the database of a single publisher in addition to one electronic database, and one only searched a single electronic database. PubMed/Medline was the most searched database, given that the authors of all included articles reported performing keyword searches on it, followed by China National Knowledge Infrastructure (CNKI), EMBASE, and Wanfeng databases. CNKI and Wanfeng databases are both Chinese language databases. Further exploratory analysis shows that 23 (47.9%) articles included search results from at least one non-English database. Of those 23 articles, all involved Chinese language databases, while two reported results from Korean and Japanese databases, respectively, in addition to the Chinese language databases. This finding corresponds with the conclusion that Chinese herbal medicine is the most frequently reported modality in the included review papers (49%, Figure 2b). Six articles reported search results from databases of preprint articles. The details of the individual review articles and their corresponding searched electronic databases can be found in Supplementary File 7.
Discussion
This scoping review found that most systematically conducted reviews on herbal medicine for COVID-19 were interventional reviews for acute phases of the infection. Chinese herbal medicine was found to be most frequently reviewed through SR/MA, potentially due to the abundance of clinical studies available, while the rest were scoping reviews on herbal medicine in general, phytoconstituents, and naturally occurring compounds, as well as other modalities of traditional medicinal plants. Based on the AMSTAR-2 tool, the overall quality of reviews was considered low due to limited clear reporting on multiple items such as the establishment of a prior protocol, completeness of literature search, comprehensive reporting of study characteristics, identification of plausible sources of heterogeneity, adjustment for confounding when pooling NRSI, and consideration of risk of bias in the analysis of results. When comparing the findings for SR/MA against the general results, SR/MA had better adherence to some critical items of the AMSTAR-2 tool, including protocol establishment and complete literature search.
This review paints a clear picture of the current landscape of herbal medicine research. Although herbal medicine is utilised globally, with much hype for its use before and during the COVID-19 pandemic, it is evident that only Chinese medicine, almost exclusively, was studied in high-quality randomised controlled human trials on a much larger scale compared to other modalities. This may be driven by a few factors, including the long, documented, and well-accepted use of Chinese herbal medicine integrated with conventional medicine in China. 67 On the other hand, apart from traditional Chinese herbal decoctions or Chinese patent medicines which have a long history of use and are governed within their home country, it is challenging to develop a new, previously unregistered, botanical drug with sufficient quality, safety, and efficacy data to meet the clinical trial and registration requirements of current international standards.. 67 This is mainly because botanical drugs comprise complex combinations of potentially bioactive phytoconstituents which can lead to challenges in ensuring batch-to-batch consistency and elucidation of mechanisms of actions and herb-drug interactions.67,68 Due to these challenges, the progress of herbal medicine into large-scale randomised clinical trials is slow. For example, to date, the United States Food and Drug Administration (U.S. FDA) has only approved two botanical products for therapeutic claims since establishing its Botanical Review Team in 1984. Records from the U.S. FDA office have shown that the success rate of a commercial herbal product (ie, botanical drug) which has entered into early-phase trials in proceeding to a phase III clinical trial is much lower than investigational new drug applications in general (5% vs ∼14%). 68 Possibly due to the lack of readily available herbal product candidates for clinical trials, despite much interest in herbal medicines for COVID-19, most of the published studies were still confined to preliminary studies, which were better suited to be included in scoping reviews, as reflected by our findings in this review.
Compared to previously published overviews of reviews, our study accounted for a larger number of included articles and provided broader coverage of the topic, as previous reviews only included systematic reviews with more focus on Chinese Herbal Medicine.7,9 Regarding the quality of reviews, our findings contrast with the study on complementary and alternative medicine interventions by Jeon et al, 9 with our study assessing more reviews as low in quality. However, as the approach to judging the level of quality is different, head-to-head comparisons should not be made. As our study was not limited to systematic reviews only and had a different aim, our approach to analysis was more similar to another scoping review on the quality of systematic reviews and other synthesis research during the COVID-19 era. 5 Compared to the overall quality of general reviews on COVID-19 assessed by Baumeister et al, 5 the herbal medicine reviews included here were found to be of lower quality. In our study, one of the most evident reasons for rating down the quality of reviews is the lack of a prior protocol, an exclusion criterion for the previous evaluation by Baumeister et al, as these were deemed too low quality to be included in their review. 5 The limited availability of a prior protocol could be partly due to the lack of an internationally recognised registration platform for scoping reviews, which comprise about half of the included articles in this review. PROSPERO, one of the most well-recognised protocol registration platforms, does not currently accept the registration of scoping review protocols. 12 Improved awareness of the importance of declaring an a priori protocol and utilising other registration platforms, such as the Open Science Framework, 69 may help raise the quality of future scoping reviews.
Apart from assessing the quality of reviews based on the listed criteria of the AMSTAR-2 checklist, the additional exploratory analysis revealed that most reviews on herbal medicine and COVID-19 covered an adequate range of databases (ie, at least three reputable databases). Furthermore, two of the four top databases searched were non-English language databases representing comprehensive coverage. Preprint databases were also covered in a few included articles, reflecting the unique need for rapid acquisition of information due to the urgency of the scenario, that is, the virulence of the pandemic.
Challenges and Limitations
Our team faced several challenges when using the AMSTAR-2 tool to assess the quality of included reviews. Among these was the lack of an option for ‘unclear reporting’ for items on the AMSTAR-2 tool and the strictness of the tool, which requires transparent reporting of all points listed to award a full or partial adherence towards individual items. For example, for item no. 4 (complete literature search strategy), in cases where there was no mention of search restrictions, such circumstances did not necessarily warrant a ‘no restrictions applied’ classification, and an option of ‘unclear reporting’ may be more appropriate. In such cases, if non-adherence is judged on account of ‘no justified search restrictions due to no clear reporting’, the item is to be awarded a ‘No’ and a full downgrade. Nevertheless, it was the intention of the AMSTAR-2 development team to apply such strict criteria to force users to make a choice when assessing the quality of reviews. 14
Among the main limitations of this review were the inclusion of articles in the English language only and using the AMSTAR-2 tool to assess the quality of non-systematic reviews. In addition, we could only search three major databases due to resource restrictions. It is generally recommended that a comprehensive search should cover a minimum of two to three large reputable databases. Our inability to cover other non-US-based databases may result in this review missing out on some reviews from non-US sources. 17 However, to properly assess the quality of included reviews, we felt that it was necessary to restrict inclusion to articles that could be adequately accessed and understood by our investigators. Another essential point to consider is the applicability of the AMSTAR-2 checklist to non-systematic review type articles. Currently, there is still no systematic tool to assess the quality of scoping reviews. Therefore, we could only attempt to apply the AMSTAR-2 tool to other reviews by evaluating only certain applicable items. Our findings may be updated if a more suitable tool is developed in the future.
A cross-sectional study by Gupta et al, 70 published during the early phases of the COVID-19 pandemic, highlighted the issues of information and misinformation, which are thought to be driven by the urgent need for information. This study highlights the need for stringent peer-reviewing, editing, and publishing of accurate information in the face of a pandemic. As our review is among the first available studies that systematically searched, identified, and assessed the quality of reviews on herbal medicine and COVID-19, we believe that, despite the limitations, it still adds valuable insights into the quality of literature on phytomedicine research.
Conclusions
In conclusion, this overview of reviews showed that clinical research, especially interventional clinical trials, on herbal medicine for COVID-19 was primarily conducted on Chinese herbal medicine. Overall, current herbal medicine reviews focus on treatment for acute phases of the infection though this may soon change depending on global health demands. Most reviews have low or partial adherence towards critical items of the AMSTAR-2 tool though the overall quality of SR/MA is generally better than other systematically conducted reviews. There is still a need to improve the methodological designs, implementation, and reporting of herbal medicine reviews to address the growing concern of misinformation in scientific literature.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-docx-1-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-docx-2-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-docx-3-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-docx-4-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-xlsx-5-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-xlsx-5-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-xlsx-6-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-xlsx-6-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Supplemental Material
sj-xlsx-7-npx-10.1177_1934578X231192160 - Supplemental material for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews
Supplemental material, sj-xlsx-7-npx-10.1177_1934578X231192160 for Herbal Medicine and COVID-19: Characteristics and Methodological Quality of Published Reviews by Xin Yi Lim, Muhammad Nor Farhan Sa’at, Nur Salsabeela Mohd Rahim and Terence Yew Chin Tan in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
