Abstract
A new neoflavonoid, named as (7R)-(-)-3′,5-dihydroxy-4′,2,4-trimethoxy-dalbergiquinol (
Myocardial hypoxia/reoxygenation (H/R) injury can lead to significant morbidity and mortality. 1 In previous reports, latifolin isolated from Dalbergia genus exhibited cardioprotective effects against doxorubicin-induced cardiotoxicity by macrophage polarization in mice. 2
Dalbergia melanoxylon, belonging to the genus Dalbergia (Fabaceae family), is widely cultivated in tropical and subtropical African areas. 3 Dalbergia melanoxylon called by African people as “African Blackwood” or “Mpingo” 4 have been commonly used for the treatment of abdominal pain, joint pain, headache, hernia, gonorrhea, and bronchitis. 5 Dalbergia melanoxylon has a rich source of biologically active constituents, including neoflavones, 6 flavanones, 5 benzofurans, 7 N-cinnamoyl, 8 quinoid 9 derivatives, and a phenanthrenedione. 10 However, the pharmacological activities and mechanisms of D. melanoxylon about the protective effect on H/R injury are still unknown.
In a continuing phytochemical study of the dichloromethane extract, a new neoflavonoid (
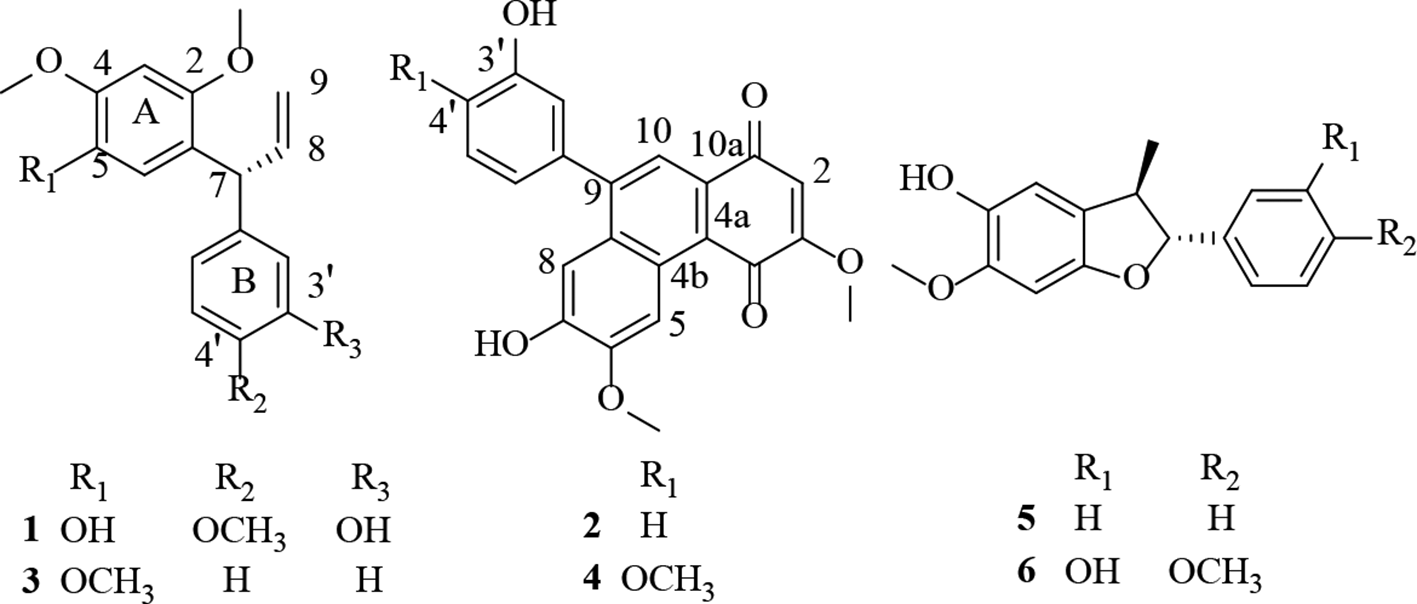
Chemical structures of compounds 1-
Materials and Methods
General Experimental Procedures
Ultraviolet (UV) spectra were obtained on a 210A double-beam spectrophotometer (Shimadzu, Japan). Mass spectrometry (MS) data were obtained on a Triple TOF 5600 + MS (AB SCIEX, USA). One-dimensional (1D) and 2D nuclear magnetic resonance (NMR) spectra were recorded on a Bruker AV 600 spectrometer (Bruker Corporation, Fallanden, Switzerland). Circular dichroism (CD) spectra were recorded using a JASCO J-1500 spectropolarimeter (CA, USA). Preparative high-performance liquid chromatography (HPLC) was performed on an LC 3000 (Beijing Tong Heng Innovation Technology Co., Ltd., China) with a semipreparative C18 column (250 × 10 mM, 10 µM, Phenomenex, USA). Optical rotations were measured using a JASCO P-1020 polarimeter (JASCO Corporation, Tokyo, Japan). Sephadex LH-20 (25, 100 mm, Pharmacia Fine Chemical Co Ltd, Uppsala, Sweden) was used for column chromatography. Analytical thin-layer chromatography plates (GF 254 Silica gel) and column chromatography silica gel (100-200 mesh, 200‐300 mesh) were purchased from Qingdao Haiyang Chemical Co, Ltd. (Qingdao, China). All solvents were of analytical grade. The H9c2 culture supernatants were tested with lactate dehydrogenase (LDH) assay kits (Jiancheng Bioengineering Institute, Nanjing, China). The homogenates of H9c2 were tested with malondialdehyde (MDA) and superoxide dismutase (SOD) assay kits (Jiancheng Bioengineering Institute, Nanjing, China). The value of optical density was measured by Absorbance Microplate Reader (SpectraMax 190, Molecular Devices Corporation, USA).
Plant Material
The heartwoods of D. melanoxylon were purchased from Fang Cheng Gang market, Guangxi Province, China, in July 2014 and identified by Professor Feng Xu at the product quality inspection center of Guangxi University. A voucher specimen (No.Liu-20140702) was deposited in the Key Laboratory of Innovation Drug and Efficient Energy-saving Pharmaceutical Equipment, Jiangxi University of Traditional Chinese Medicine.
Extraction and Isolation
The air-dried pieces of the heartwood of D. melanoxylon (50.0 kg) were powdered, passed through a 40-mesh sieve, and was extracted with 70% ethanol under refluxing 3 times. After removing the solvent under reduced pressure, the residue (13.9 kg) was suspended in water and partitioned with dichloromethane (CH2Cl2; 5 L × 15), ethyl acetate (EtOAc; 5 L × 10), and n-butanol (n-BuOH; 5 L × 5) to afford CH2Cl2 (8.5 kg), EtOAc (1.8 kg) and n-BuOH (0.8 kg) fractions, respectively.
The CH2Cl2 extract (8.5 kg) was chromatographed over silica gel eluting with a gradient of petroleum ether-EtOAc (100:1 to 10:1, v/v) to yield 22 subfractions (A1-A22). Subfraction A1 (14.8 g) was purified by silica gel column chromatography eluting with petroleum ether-acetone to (50:1 to 2:1, v/v) yield 11 subfractions (A1a-A1k). Subfraction A1a (2.2 g) was separated by silica gel eluting with petroleum ether-EtOAc (20:1 to 2:1, v/v) to yield 3 subfractions (A1a1-A1a3). Subfraction A1a3 (1.0 g) was separately performed on a semipreparative C18 column (250 × 10 mM, 10 µM, Phenomenex, USA) eluting with methanol (CH3OH or MeOH)/water |(H2O) (v/v, 60:40, flow rate: 4 mL/min) to afford compound
(7R)-(-)-3',5-Dihydroxy-4',2,4-Trimethoxydalbergiquinol (1)
Yellow amorphous powder;
13C and 1H Nuclear Magnetic Resonance Data for Compounds 1 and

Abbreviation: DMSO, dimethyl sulfoxide.
3′,7-Dihydroxy-3,6-Dimethoxy-9-Phenyl-1,4-Phenanthrenedione (2)
Reddish amorphous powder; UV (MeOH) λmax 248, 307 nm; IR (KBr) λmax 3201 1725 1600 1504 cm−1; 1H NMR (DMSO-d 6, 600 MHz) and 13C NMR (DMSO-d 6, 151 MHz) data, see Table 1; HRESIMS (postive) m/z 377.0848 [M + H]+ (calcd. for C22H17O6 377.0846).
Activity Assay
H9c2 cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). The cells were cultured in Dulbecco’s Modified Eagle’s Medium (Invitrogen, USA) supplemented with 10% fetal bovine serum at 37 °C with carbon dioxide incubation. Cells were split when a confluence of 80% was achieved using trypsin-ethylenediaminetetraacetic acid (EDTA), seeded onto 96-well plates at a density of 5.0 × 104/mL (100 µL/well), incubated for 24 hours before treatment.
H9c2 cells were randomly divided into 3 groups
11
: (1) the control group was cultured under normal medium; (2) the model group was cultured under oxygen and glucose deprivation (OGD) for 20 hours, and then the cells were maintained in normal culture medium for 4 hours (reoxygenation); (3) the drugs groups, which were pretreated with compounds
The H9c2 culture supernatant was used for the estimation of LDH. H9c2 cells were washed with phosphate-buffered saline (PBS), enzymatically dissociated with the use of trypsin/EDTA, and finally collected in PBS buffer. H9c2 were lysated by ultrasound in 3 times, and the lysates of H9c2 were centrifuged for 10 minutes at 1000 r/min at 4 ℃. The homogenate of H9c2 was used to estimate the activities of SOD and MDA.
Statistical Analysis
All data are presented as mean ± SD, and analyses were performed with SPSS 20.0 software. The multigroup comparisons were conducted by a one-way analysis. Values of P < 0.05 were considered to indicate a statistically significant difference.
Results and Discussion
Structural Elucidation of Compounds 1-6
Compound

Circular dichroism spectrum of compounds 1 and 3 in methanol.
Compound

Selected heteronuclear multiple bond correlations of compounds 1 and 2.
The 4 known compounds were identified as 5-O-methyldalbergiphenol (
Up to now, the structures of 7-hydroxy-3,6-dimethoxy-9-phenyl-1,4-phenanthrene-diones10,14 and (2S,3S)-3-methyl-2-phenyl-2,3-dihydrobenzo[b]furan18,19 were only isolated from the Fabaceae family. Moreover, the 7-hydroxy-3,6-dimethoxy-9-phenyl-1,4-phenanthrenediones were uncommon in natural products; there were only 2 derivatives that had been discovered.10,14 So, the 2 skeletons of compounds
Pharmacological Activities of Compounds 1-6
The cytotoxicities of all compounds (
The protective effect of compounds
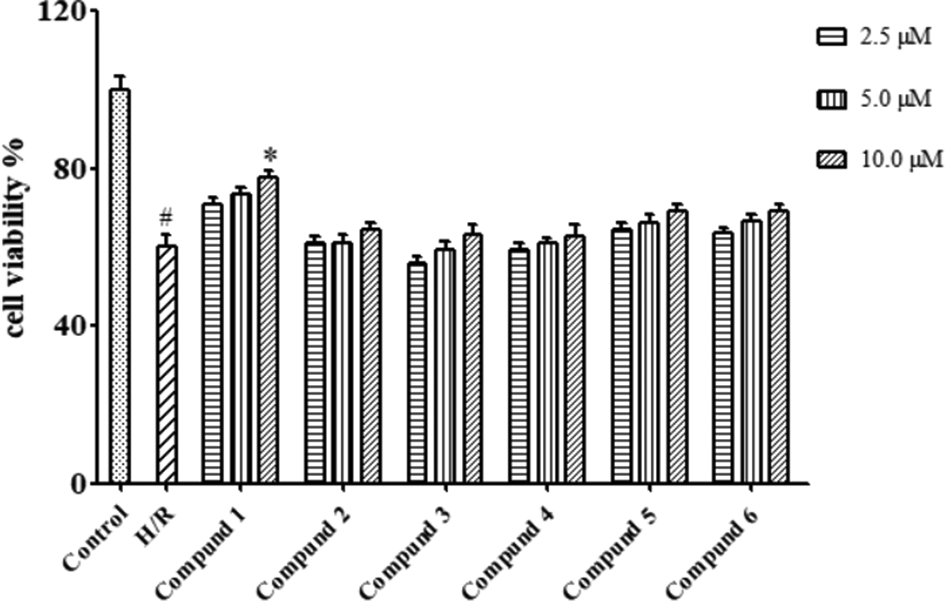
Protective effects of compounds 1-
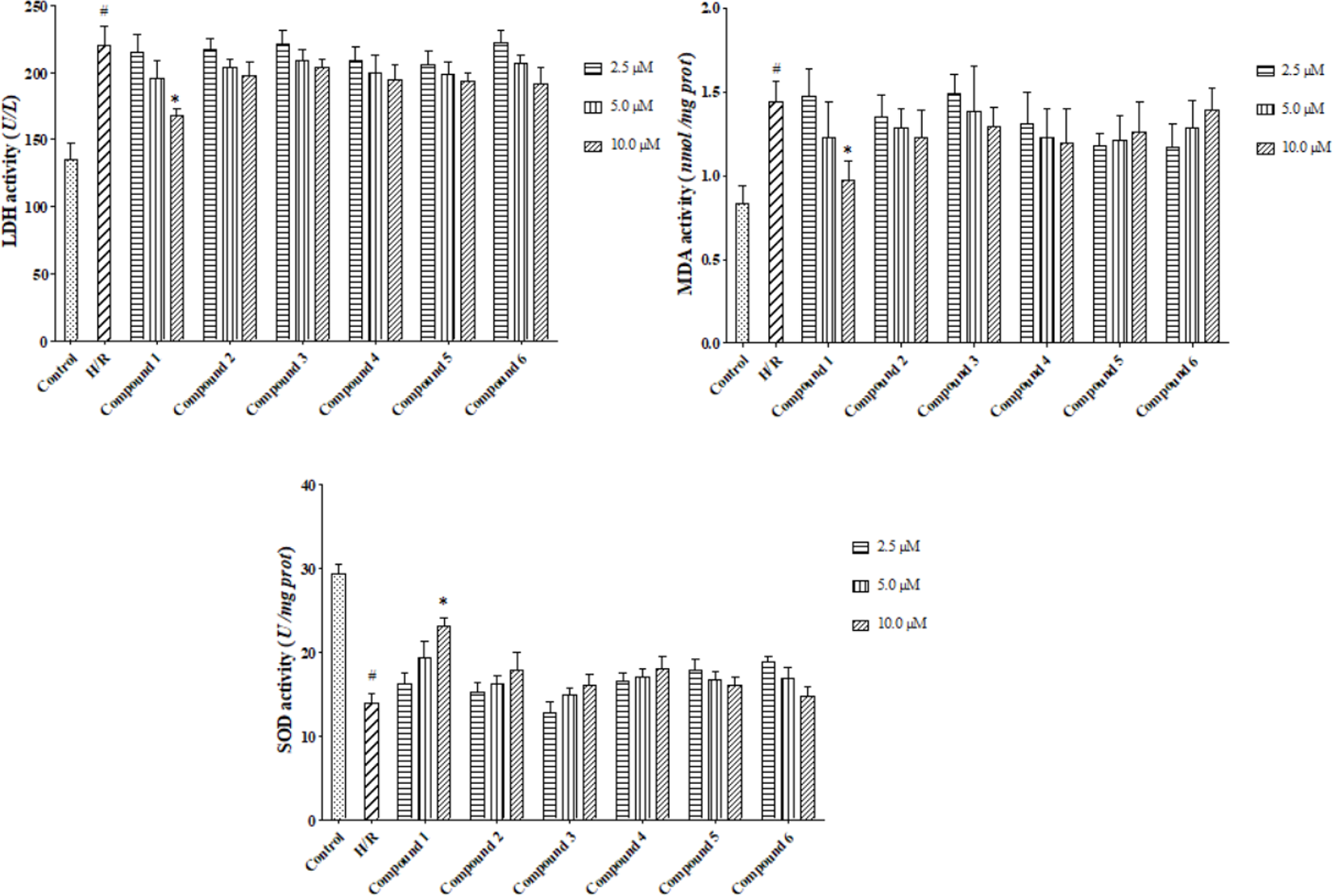
Lactate dehydrogenase, malondialdehyde, and superoxide dismutase level of compounds 1-
Based on the above evidence, we suggested that neoflavonoids with a protective effect on H/R-induced injury are the main bioactive constituents of D. melanoxylon.
Conclusions
In summary, 1 new neoflavonoid (
Supplemental Material
Supplementary Figures - Supplemental material for Two New Compounds From the Heartwood of Dalbergia melanoxylon and Their Protective Effect on Hypoxia/Reoxygenation Injury in H9c2
Supplemental material, Supplementary Figures, for Two New Compounds From the Heartwood of Dalbergia melanoxylon and Their Protective Effect on Hypoxia/Reoxygenation Injury in H9c2 by Yang Liu, Ni Zhang, Jun-wei He, Lan-ying Chen, Li Yang, Xiao-wei Meng, Feng Shao and Rong-hua Liu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Key R&D Program of China (2018YFC1706102), the National Natural Science Foundation of China (NSFC) (No. 81360629 and 81660676), the Natural Science Foundation of Jiangxi Province (No. 20171BAB205096 and 20202BABL216074), and the Postgraduate Innovative Foundation of Jiangxi University of traditional Chinese medicine (No. JZYC17S042).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
