Abstract
Orthosiphon stamineus Benth. (Lamiaceae) is commonly used for the treatment of kidney diseases, but its role in intestinal barrier function remains unknown. The present study investigated the protective effects of O. stamineus extract (OE) against oxidative stress-induced injury to the small intestinal epithelium and the possible mechanism. High-performance liquid chromatography fingerprinting was used to analyze OE. Oxidative stress was induced by hydrogen peroxide (1 mM for 1 hour) in an IPEC-J2 cell monolayer model and a high-fat diet in C57BL/6 mice (8 weeks). The malondialdehyde (MDA) content was tested in both models. To evaluate permeability, transepithelial electrical resistance (TEER) was tested in a cell model. Serum diamine oxidase (DAO) and endotoxin contents were determined in a mouse model, and histological sections were analyzed. The messenger ribonucleic acid expression of tight junction proteins was measured by quantitative real-time polymerase chain reaction. Pretreatment with OE (50 µg/mL) increased the IPEC-J2 cell monolayer TEER (12.4%) and decreased MDA (from 6.1 to 4.7 mmol/mg prot). Oral administration of OE (100 mg/kg) decreased serum DAO (34.2%), endotoxin (13.4%), and MDA (from 21.3 to 11.0 mmol/mL) in mice. OE upregulated ZO-1 (42.8% in the cell model and 125.0% in mice) and occluding (127.0% in the cell model and 120.3% in mice) gene expression. These results confirmed the protective effect of OE on the intestinal barrier, which was associated with the antioxidant effect of OE; thus, OE is suitable for the prevention and treatment of intestinal barrier injury.
The epithelium of the small intestine not only digests and absorbs nutrients but also serves as a barrier against noxious antigens and pathogens. 1 The intestinal barrier is formed from intestinal epithelial cells and tight junctions (TJ) between intestinal epithelial cells, which are largely composed of tight junction proteins (TJP), including junctional adhesion molecule, the claudin family, occludin, etc., 2 which are linked to the cytoskeleton by scaffold proteins, such as zona occludens (ZO).
Oxidative stress is a universal stressor that causes intestinal epithelial cell injury or apoptosis, 3 as well as downregulation of TJP, 4 given that the intestinal barrier is formed from TJP and intestinal epithelial cells. Oxidative stress injures the intestinal barrier.
Damage to the intestinal barrier leads to increased concentrations of endotoxins in the blood, bacterial translocation, and diarrhea, which further exacerbates some diseases, 5 including irritable bowel disease, autism, and obesity. 6
Orthosiphon stamineus Benth (Lamiaceae), more commonly known as “Java tea,” is widely cultivated throughout Southeast Asia, Australia, and southern China, where it is not only a popular drink but also is used as an herbal medicine, owing to its antiangiogenetic, antitumor, anti-inflammatory, antihyperglycemic, and antihypertensive properties. 7 However, few reports have described the protective effects of O. stamineus extract (OE) on the intestinal epithelium. A previous study demonstrated the protective effects of water-ethanol extracts of O. stamineus on the intestinal epithelial cells, 8 thus indicating the potential protective effects of OE on the intestinal barrier.
Most researchers have obtained OE via methanol extraction. However, for food or feeds, some consumers will worry about the negative effects of methanol residues, whereas ethanol will be more acceptable for most consumers.
The present study investigated the protective effect of OE (ethanol/water, 50:50) on the small intestinal epithelium both in vitro and in vivo. First, the high-performance liquid chromatography (HPLC) fingerprints of the major phenolic acids and flavones in the main production areas in China were determined to characterize the extract. Then, to test the protective effect of OE, an in vitro model with IPEC-J2 cells 9,10 and an in vivo model in mice were used. The transepithelial electrical resistance (TEER) of an IPEC-J2 cell monolayer was measured and messenger ribonucleic acid (RNA) expression levels of ZO-1, occludin, and claudin-1 were determined in IPEC-J2 cells and mouse intestinal epithelial cells.
Materials and Methods
Plant Materials and Extraction
The plant material used in animal model and cell model was collected from Yulin, Guangxi, China, in November 2016. Other samples for HPLC fingerprinting analysis were representative samples from Guangxi and Yunnan, China. The identity of the plant material was confirmed by Dr Haibo Hu (Gannan Medical University), and voucher specimens have been retained in our laboratory for future reference. Dry powdered OE (20 g) was extracted with 50% aqueous ethanol by using a rotary shaker at room temperature for 24 hours. Then, the OEs were filtered and concentrated by spray-drying. The resulting powders were stored at −20 °C until use.
Spectrophotometric Analysis of Samples
The major antioxidant compounds in the samples were detected with an Agilent 1260 HPLC system (Santa Clara, CA, USA). The HPLC method applied was a modification of that reported by Akowuah et al. 11 Briefly, HPLC analyses were performed with a Waters Symmetry C18 column (4.6 × 250 mm, 5 µm) at 30 °C with a flow rate of 1.0 mL/min. Samples and standards were dissolved in methanol, and the effluent was monitored by ultraviolet (UV) detection at 340 nm. The mobile phase was 0.2% formic acid (A), acetonitrile (B), and methanol (C) in a gradient elution system as follows: 5%-15% B, 15%-20% C in 0-5 minutes; 15%-45% B, 20%-10% C in 5-15 minutes; 45%-60% B, 10%-0% C in 15-20 minutes; 60%-95% B in 20-25 minutes; 95%-85% B in 25-35 minutes; 85%-5% B in 35-36 minutes; 5% B, 15% C in 36-40 minutes. Caffeic acid, rosamarinic acid, 3′-hydroxy-5,6,7,4′- tetramethoxyflavone, eupatorine, and sinensetin were used as reference markers, as in previous studies. 8,11,12 The HPLC data were analyzed with the “Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine” software (Version 2012). The correlation coefficients of the samples were calculated according to the method of Xie et al. 13
IPEC-J2 Cell Culture and Treatment
The IPEC-J2 cell line was a gift from Dr Zhanyong Wei (Henan Agricultural University). The cells were grown in 1:1 Dulbecco’s modified Eagle’s medium (DMEM)/Ham’s F-12 mixture (HyClone, South Logan, UT, USA) supplemented with 5% fetal bovine serum (Biological Industries Israel Beit Haemek Ltd., Beit Haemek, Israel), 5 µg/mL of insulin, 5 ng/mL of epidermal growth factor (Sigma-Aldrich Corporation), 100 U/mL of penicillin, and 100 µg/mL of streptomycin (Gibco, Carlsbad, CA, USA) at 37 °C under a humidified atmosphere of 5% carbon dioxide (CO2), as described elsewhere. 10
IPEC-J2 cells were seeded on Transwell-COL collagen-coated membranes (1.12 cm2, 0.4 µm; Corning, MA, USA) at a density of 6 × 105 cells/well. 14 The medium was refreshed every other day. When the IPEC-J2 cell monolayer had reached confluence (>1000 Ω·cm2), 15 the cells were incubated with/without OE for 12 hours and then exposed to either 1 mM hydrogen peroxide (H2O2) or phosphate-buffered saline (PBS) for 1 hour, as described previously. 9 For each group, TEER was determined using a voltameter (Millicell-Electrical Resistance System; Merck KgaA). The TEER results are expressed as the ratio of the value of the test cells to that of the control group.
Animals and Treatment
Forty-eight male C57BL/6 mice, weighing 14-16 g, were purchased from Shanghai Laboratory Animal Center Co., Ltd. (Shanghai, China) and housed in standard cages under controlled temperature conditions (22 ± 2 °C) with a 12-hour light/dark cycle and ad libitum access to food and water. The animal study was approved by the Ethics Committee for Research using Laboratory Animals of Shanghai Academy of Agricultural Sciences and conducted in accordance with local guidelines for the care and use of laboratory animals. At the beginning of the experiment, the mice were randomly assigned to 1 of 4 groups of 12 mice each as follows: a normal control (NC) group, a normal diet with OE supplement (NO) group, a high-fat diet control (FC) group, and a high-fat diet with OE supplement (FO) group.
Mice in the NC group were fed a normal diet containing 10 kcal% fat (D12450B, Research Diet Inc., New Brunswick, NJ, USA) and gavaged daily with PBS. Mice in the NO group were fed a normal diet containing 10 kcal% fat (D12450B) and gavaged daily with 100 mg/kg body weight of OEs. Mice in the FC group were fed a high-fat diet containing 60 kcal% fat (D12492) and gavaged daily with PBS. Mice in the FO group were fed a high-fat diet containing 60 kcal% fat (D12492) and gavaged daily with 100 mg/kg body weight of OEs. The body weight was recorded weekly.
At the end of the 8-week feeding period, blood was collected from the eyeballs, and then the mice were sacrificed by cervical dislocation. The jejunum was dissected and placed on a chilled stainless steel tray. Approximately 1 cm segments were cut and immediately immersed in either glutaraldehyde (2.5%) or formalin (4%) for histopathology study; a 0.5cm segment was stored in RNAhold reagent (TransGen Biotech Co., Ltd., Beijing, China) at −20 °C for RNA extraction; and the rest was frozen at −20 °C for redox state analyses.
Determination of Redox State Index
For determination of malondialdehyde (MDA) content, IPEC-J2 cells were washed twice with cold PBS and harvested with a cell scraper. The harvested cells were lysed by sonication (300 W, 10 seconds with 20 seconds intervals for 30 cycles) on ice, and the supernatant was collected by centrifugation, as described by Zou et al. 16
The MDA content of mouse serum and IPEC-J2 cells was measured using the thiobarbituric acid method at a wavelength of 532 nm, according to the manufacturer’s instructions (Shanghai Congyi Technology Co., Ltd, Shanghai, China). The protein content of homogenates was measured by the Coomassie Brilliant Blue G-250 method (Shanghai Congyi Technology Co., Ltd).
Other antioxidant activity indexes (superoxide dismutase, catalase, glutathione peroxidase activity, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) radical scavenging activity, and 8-iso-PG content) of mouse serum were analyzed according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Histopathology
For light microscopy, a single 2-5 mm specimen was cut from each jejunum section, fixed in 4% phosphate-buffered parafor-maldehyde, dehydrated with increasing concentrations (70, 80, 95, and 100%) of ethanol, cleared with xylene, and embedded in polyfin wax. Tissue sections (5 µm) were cut and placed onto slides, which were stained with hematoxylin and eosin (HE), and then photographed under a light microscope (Olympus Corporation, Tokyo, Japan). The heights of villus and the crypt depths of crypts were calculated as previously described. 12
For electron microscopy, a single 2-4 mm sample was cut from each jejunum section, fixed in 2.5% glutaraldehyde overnight at 4 °C, post-fixed with 1% osmium tetroxide, dehydrated with a graded ethanol series, and embedded in Epon 812 embedding medium (Sigma-Aldrich, St. Louis, MO, USA). Thin sections were cut, stained with uranyl acetate and lead citrate, and examined under a transmission electron microscope (H-600; Hitachi Medical Corporation, Tokyo, Japan) operated at 80 kV.
Serum Diamine Oxidase and Endotoxin Content
Serum diamine oxidase (DAO) concentrations were quantified with an enzyme-linked immunosorbent assay kit (Cusabio Biotech Co., Ltd., Wuhan, China) using the quantitative sandwich enzyme immunoassay technique. Endotoxin content was measured with Tachypleus Amebocyte Lysate (Xiamen Bioendo Technology, Xiamen, China) according to the operation manual.
Measurement of TJP mRNA Expression by qRT-PCR
Total RNA of IPEC-J2 cells and mouse jejuna were isolated with Transzol UP reagent (TransGen Biotech Co., Ltd.), and then immediately reverse transcribed to complementary deoxyribonucleic acid (DNA) with a reverse transcription kit (TaKaRa Biotechnology Co., Ltd., Dalian, China) according to the manufacturer’s instructions. The quality of total RNA was assessed using both absorbance, as measured with a NanoDrop Lite Spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) (1.8 ≤ A260/280 ≤ 2.0 indicates high quality), and electrophoresis on an 0.8% agarose gel. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed with the primer sequences shown in Table 1 to quantify gene expression. All reactions were performed in triplicate and each reaction mixture (20 µL in total) contained complementary DNA (1 µL), forward primer (0.4 µL), reverse primer (0.4 µL), double distilled water (8.2 µL), and SYBR Premix Ex Taq polymerase (10 µL; TaKaRa Biotechnology Co., Ltd.). Reactions were incubated in an ABI 7500 instrument (Applied Biosystems, Carlsbad, CA, USA) at 95 °C for 30 seconds, followed by 40 cycles of denaturation at 95 °C for 5 seconds and annealing and extension at 60 °C for 30 seconds.
Primers Used for Quantitative Real-Time Polymerase Chain Reaction.

aFor the gene names, no * indicates used in IPEC-J2 cell model (pig) and with * used mouse model.
Gene expression was quantified using analytical software. The relative expression of mRNA species was determined by the comparative 2-ΔΔCt method.
Statistical Analysis
All data are expressed as the mean ± SEM. Data were subjected to one-way analysis of variance followed by Duncan’s multiple range tests in SPSS version 17.0 software (SPSS, Inc., Chicago, IL, USA). A probability (P) value of <0.05 was considered statistically significant.
Results
HPLC Analysis
HPLC chromatographic profiles of the water-ethanol extracts of samples from different locations and different seasons are shown in Figure 1. Caffeic acid, rosmarinic acid, 3'-hydroxy-5,6,7,4'-tetramethoxyflavone, sinensetin, and eupatorin were the main phenolic acids and flavonoids in OE. A similarity evaluation based on the main phenolic acid and flavonoids was performed (Table 2), and samples from different locations and different seasons showed very high similarities.

High-performance liquid chromatogram of major marker components of Orthosiphon stamineus extract. Ultraviolet detection at 340 nm. 1: the sample used in this study and collected in Yulin, Guangxi in 11/2016; 2‐6: samples collected in Yulin, Yulin, Nanning, Xishuangbanna, and Xishuangbanna in 05/2017, 11/2017, 05/2017, 11/2017 and 05/2017, respectively. a: caffeic acid; b: rosmarinic acid; c: 3'-hydroxy-5,6,7,4'-tetramethoxyflavone; d: sinensetin; e: eupatorin.
Similarities of Chromatograms of Orthosiphon stamineus Extract Samples. a

aThe reference fingerprint was developed with the median of all chromatograms.
bThe information for samples 1-6 is shown in Figure 1.
OE Supplementation Did Not Alter Mouse Body Weight
Figure 2 shows that feeding mice a high-fat diet significantly increased (P < 0.05) the body weight from the third week (FC and FO vs NC and NO), but OE supplementation did not alter (P > 0.05) the body weight (FO vs FC; NO vs NC).
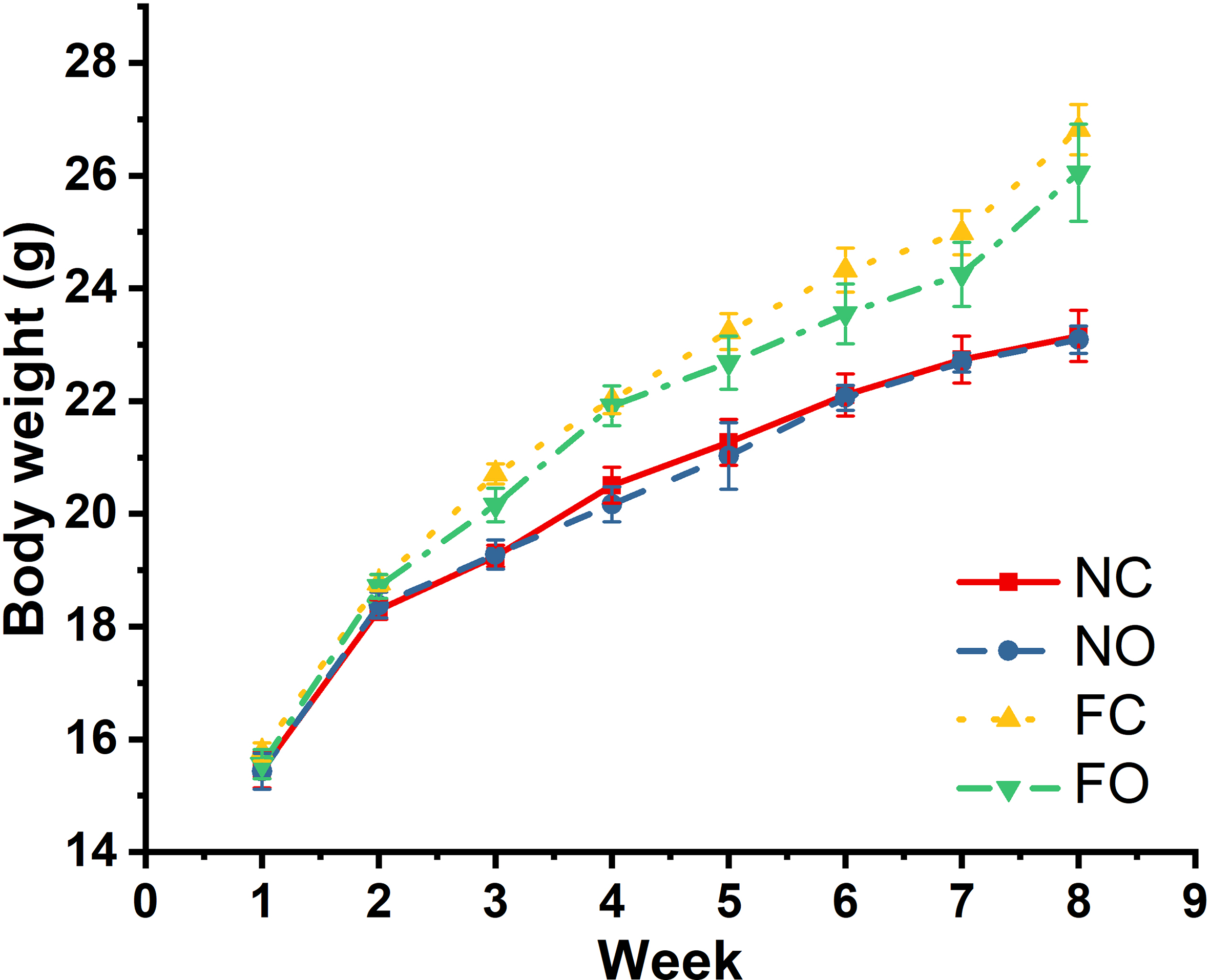
The body weights of mice in this study. FC, high-fat diet control; FO, high-fat diet; NC, normal control; NO, normal diet.
OE Increased TEER in an H2O2-Induced Oxidative Stress Cell Model And Decreased the DAO Content in the Serum of Mice Fed a High-Fat Diet
A monolayer cell model was used to determine whether OE might protect the intestinal epithelial barrier from oxidative stress. As shown in Figure 3(A), treatment with H2O2 (1 mM) for 1 hour caused a significant decrease in the TEER of the IPEC-J2 cell monolayer. Pretreatment with OE altered the TEER, and moreover, when the cells were undergoing oxidative stress, pretreatment with OE (50 µg/mL) for 24 hours significantly increased the TEER of the cell monolayer (from 71.5% of control to 84.0% of control, P < 0.05).
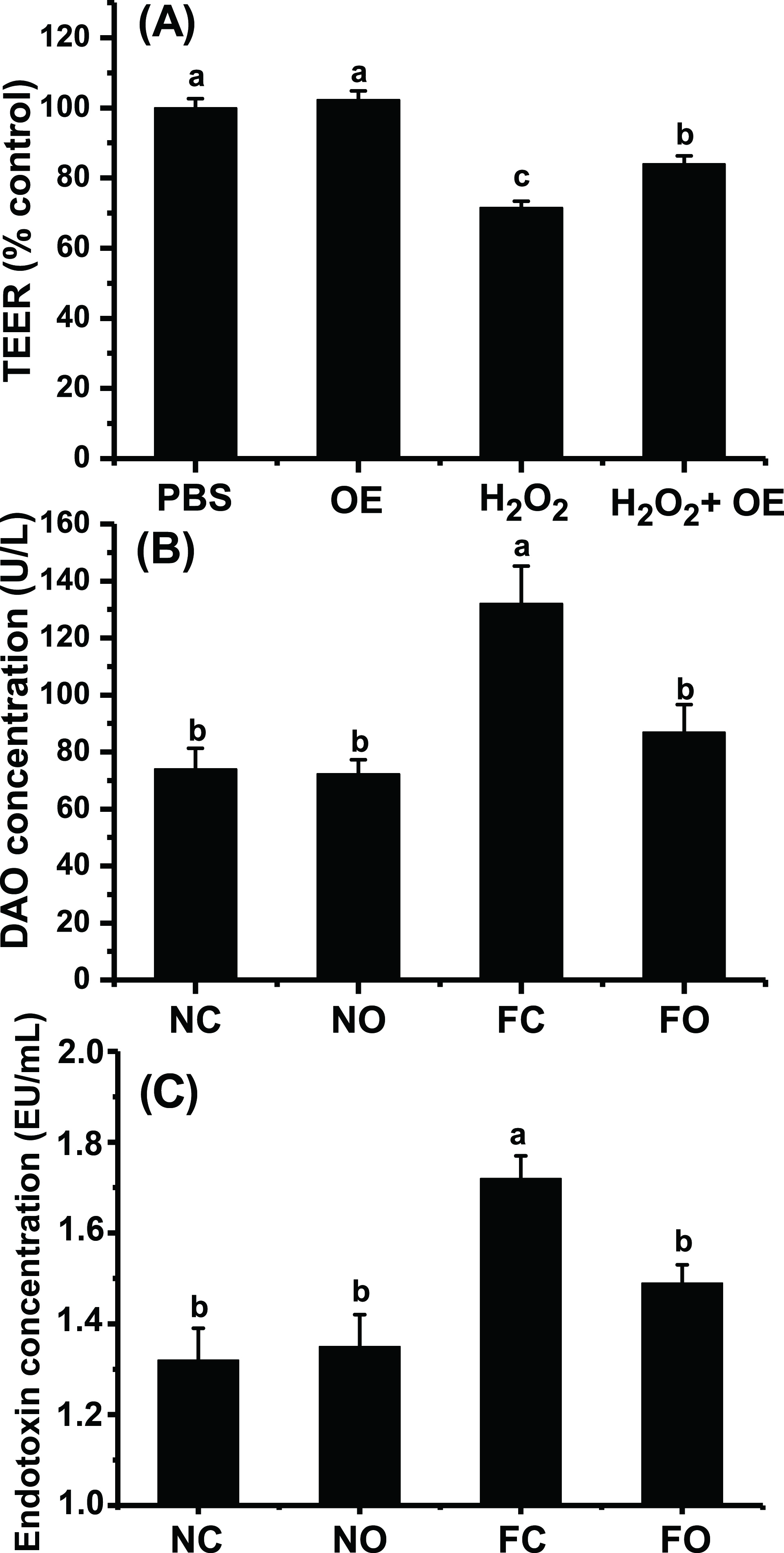
Effect of OE on intestinal permeability. (a) Effect of OE on the TEER of a monolayer of IPEC-J2 cells. The PBS group was used as a control, and data for other groups are expressed as a percentage of the control. (b) Effect of OE on mouse serum DAO. (c) Effect of OE on mouse serum endotoxin content. Different letters indicate significant differences (P < 0.05). DAO, diamine oxidase; FC, high-fat diet control; FO, high-fat diet; H2O2, hydrogen peroxide; NC, normal control; NO, normal diet; OE, Orthosiphon stamineus extract; PBS, phosphate-buffered saline; TEER, transepithelial electrical resistance.
Serum DAO and endotoxin are considered important indices of intestinal barrier function. An increase in either serum DAO or endotoxin level indicates the presence of intestinal barrier damage. As shown in Figure 3, a high-fat diet significantly increased serum DAO levels in mice (78.4%, P < 0.05), whereas OE supplementation significantly decreased DAO levels in mice fed a high-fat diet (34.2%, P < 0.05). There was no significant difference in serum DAO levels between mice fed a normal diet and supplemented with OE versus those in the normal control group (P > 0.05). The serum endotoxin levels showed the same trend as DAO (Figure 3(C)): OE supplementation decreased the endotoxin levels in mice fed a high-fat diet (13.4%, P < 0.05).
OE Altered Villus Morphology
As shown in Figure 4(A), the intestinal mucosa in mice fed a normal diet was intact, and the villi were long and integrated. In contrast, the histological appearance of the intestine in mice fed a high-fat diet showed marked shortening, clubbing, and blunting of the villi. The villi of mice fed a high-fat diet and supplemented with OE exhibited morphological changes, as compared with the control mice fed a normal diet, but still not as those fed high-fat diets and treated with PBS. Table 3 shows that OE protected the intestinal villus from high-fat diet induced villus shortening (P < 0.05) and also increased the villus height/crypt ratios.
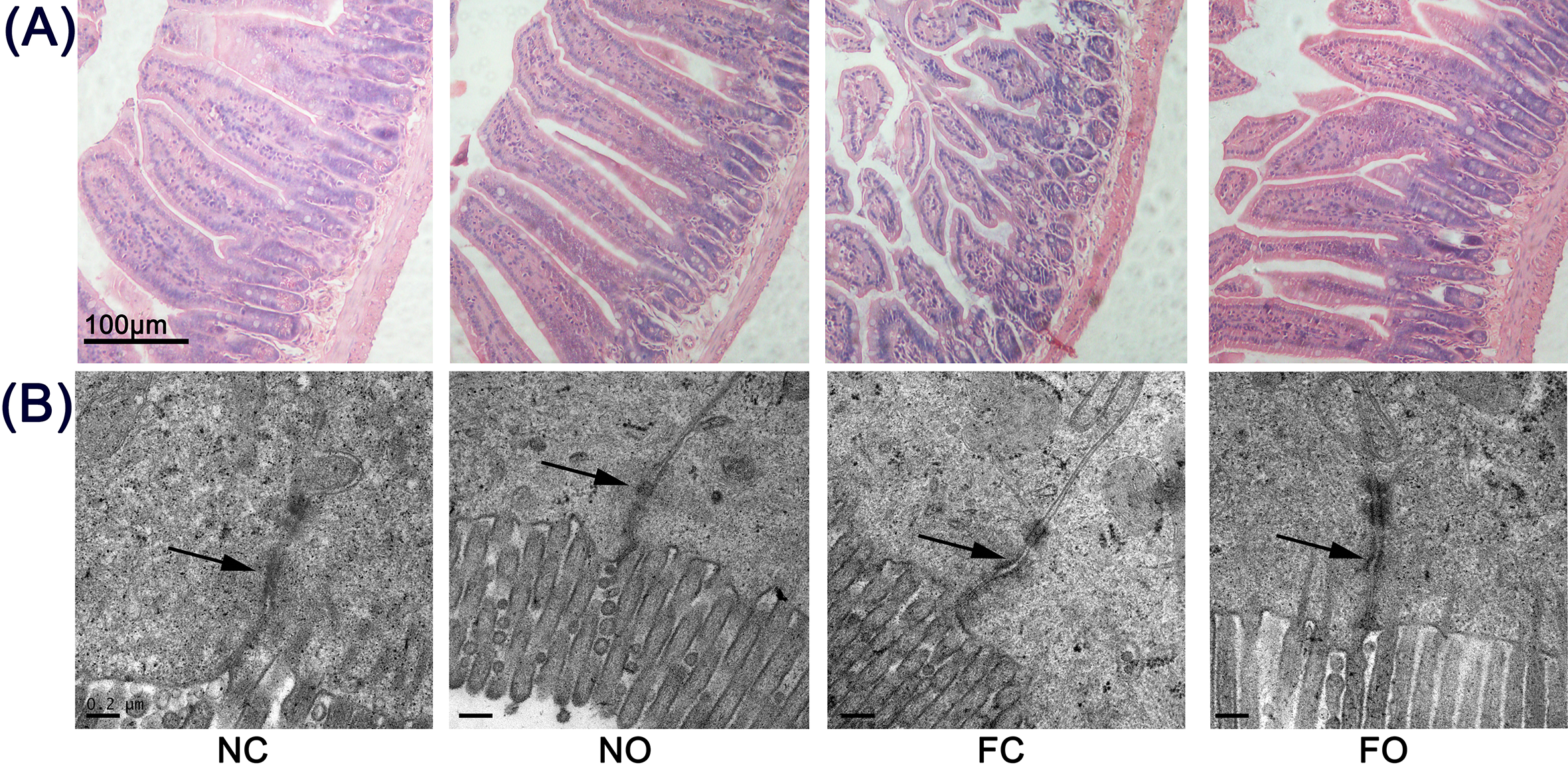
(A) Representative microphotographs of hematoxylin and eosin-stained sections of the jejunum. The pictures were captured with a light microscope. The villi of the mice gavaged with Orthosiphon stamineus extract (OE) and fed a high-fat diet (FO) exhibited shortening, clubbing, and blunting, as compared with those in mice fed a normal diet (NC and NO groups), but still not as those of mice treated with phosphate-buffered saline (FC group). (B) Ultrastructure of tight junction (TJ) in the jejunum. In the transmission electron microscopy images, the arrows indicate the desmosomes and the locations of TJ (scale bar = 0.2 µm). A high-fat diet significantly disrupted the TJ of the jejunum (arrow). OE supplementation significantly decreased the effect of a high-fat diet and protected the TJ in the jejunum.
Effect of Orthosiphon stamineus Extract (OE) Supplementation on Jejunum Morphology in Mice.

Abbreviations: FC, high-fat diet control; FO, high-fat diet with OE supplement; NC, normal control; NO, normal diet with OE supplement.
Data expressed as means ± SEM, and means within a column with different superscripts are different at P < 0.05.
Increased intestinal permeability along with histological evidence of epithelial damage suggests a TJ defect. We therefore examined enterocyte ultrastructure by transmission electron microscopy analysis. As shown in Figure 4(B), the NC and NO groups showed typical intact TJ structures. However, the mice fed a high-fat diet (FC and FO groups) showed loss of membrane fusion and a decrease in electron-dense materials between the adjoining cells, thus suggesting that the high-fat diet disrupted the architecture of the TJ of the jejunum. The addition of OE to the diets alleviated the distortion of the TJ in mice fed a high-fat diet because the abundance of electron-dense materials was increased between the adjoining cells, but obvious gaps between the adjoining cells remained. In mice fed a normal diet, the addition of OE had no impact on the ultrastructure of the TJ in mice treated with PBS.
OE Supplementation Prevented High-Fat Diet-Induced Intestinal Barrier Injury via TJP
Occludin and claudin-1 are the most important transmembrane proteins in the intestinal epithelium, and ZO-1 links the cytoskeleton with transmembrane proteins. As shown in Figure 5(A), oxidative stress significantly downregulated the mRNA expression levels of occludin, claudin-1, and ZO-1 (P < 0.05) in IPEC-J2 cells. As compared with the oxidative stress condition, pretreatment with OE (50 µg/mL) significantly upregulated mRNA expression of occludin (127.0%) and ZO-1 (42.8%) but had no effect on claudin-1 (P > 0.1). As compared with the control group levels, the mRNA levels of occludin and claudin-1 were unchanged in the OE group, whereas the ZO-1 mRNA expression was upregulated.
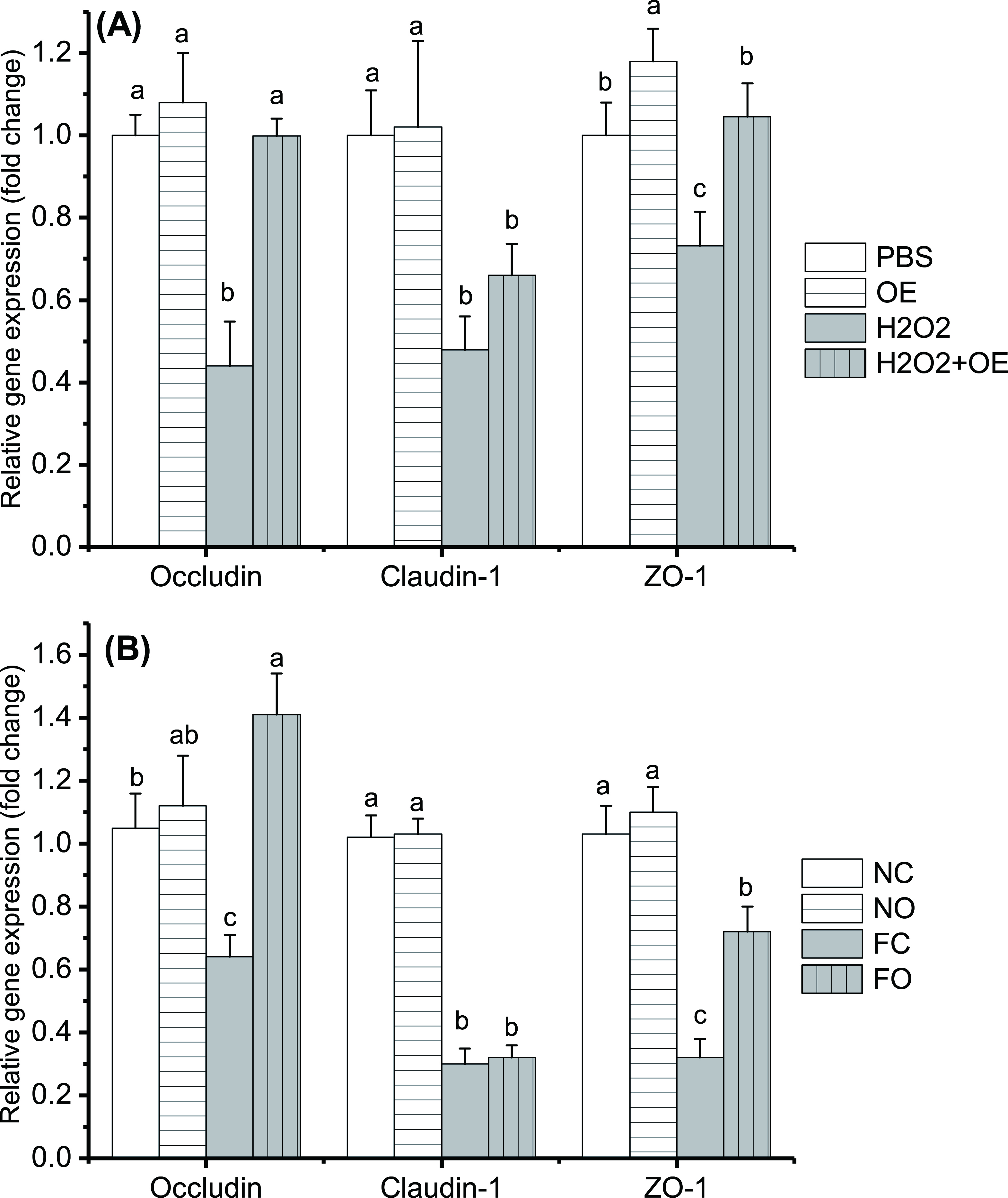
OE supplementation altered mRNA expression of occludin, claudin-1, and ZO-1. (A) IPEC-J2 cell model. (b) Mouse model. Either the β-actin gene (IPEC-J2 model) or GAPDH gene (mouse model) was used as an internal reference gene in this study. The PBS only group (a) and the PBS gavage with normal diet group (B) were considered as control groups. Relative mRNA expression levels were determined with the comparative Ct method. Different letters indicate significant differences (P < 0.05). FC, high-fat diet control; FO, high-fat diet; H2O2, hydrogen peroxide; NC, normal control; NO, normal diet; OE, Orthosiphon stamineus extract; PBS, phosphate-buffered saline; mRNA, messenger ribonucleic acid.
Similarly, feeding mice a high-fat diet decreased the mRNA expression of occludin, claudin-1, and ZO-1 in the jejunum, whereas OE supplementation (FO group) increased the mRNA expression of occludin and ZO-1, but not claudin-1, as compared with that in mice fed a high-fat diet and supplemented with PBS (FC group) (P < 0.05, Figure 5(B)). OE supplementation had no effect on the mRNA levels of occludin, claudin-1, and ZO-1 in the jejunum (P > 0.05) in mice fed a normal diet.
OE Supplementation Alleviated Lipid Peroxidation Both in Mice Fed a High-Fat Diet and in H2O2-Induced IPEC-J2 Cells
MDA is a secondary oxidation product of lipid peroxidation and is often used as a biomarker of oxidative stress. 17 Figure 6 shows the MDA content of the mouse jejunum and IPEC-J2 cells after different treatment regimens. In both the cell and mouse models, oxidative stress (H2O2 treated or high-fat diet fed) significantly (P < 0.05) increased the MDA concentrations in jejunum cells and serum, as compared with the control levels. In oxidative stress treatment, OE supplementation significantly (P < 0.05) decreased the MDA concentrations in jejunum cells (from 6.13 to 4.71 mmol/mg prot, P < 0.05) and serum (from 21.3 to 11.0 mmol/mL, P < 0.05). Interestingly, in the mouse model, serum MDA levels were significantly (P < 0.01) decreased (by 41.3%), and the concentration of MDA in mice fed a high-fat diet and supplemented with OEs did not differ from that in mice fed a normal diet (P > 0.1).

MDA concentrations in IPEC-J2 cell homogenates and mouse serum. The left Y-axis and black bar show the MDA content in IPEC-J2 cell homogenate, and the right Y-axis and gray bar show the MDA content in mouse serum. Different lowercase letters indicate significant differences in the cell model and different capital letters indicate significant differences in the mouse model (P < 0.05). H2O2, hydrogen peroxide; MDA, malondialdehyde; OE, Orthosiphon stamineus extract.
Other antioxidant indices also showed that OE decreased oxidative stress in mice fed a high-fat diet (data summarized in Supplemental Materials).
Discussion
In recent years, the quality control of O. stamineus has attracted increasing attention. Akowuah et al 11 studied 12 O. stamineus samples from various locations in Malaysia and found that the UV and HPLC chromatographic fingerprints of the major phenolics were qualitatively similar. 11 Then Hernadi et al 18 developed an HPLC fingerprinting method to detect O. stamineus from other similar plant extracts. Hernadi et al found that O. stamineus in different locations in Indonesia was similar in terms of bioactive marker compounds. To facilitate research and avoid the risk of 1 sample being insufficiently representative, this study also used an HPLC fingerprinting method to analyze O. stamineus in different seasons in China. On the basis of similarity analysis, we found that the main bioactive marker compounds of OE in different seasons in China showed very high similarities to those reported by Akowuah et al 11 and Hernadi et al. 18 A typical sample (sample 1) was selected for subsequent studies. This sample was also selected because Yulin (in Guangxi province) has the highest output of O. stamineus in China. 19
The intestinal epithelial barrier is important for animal health. 1 Our previous study confirmed that OE is beneficial for intestinal epithelial cells, 12 thus, implying that it might protect the intestinal epithelial barrier. Therefore, this study was performed. This study used IPEC-J2 cells, a nontumorigenic epithelial cell line isolated from the mid-jejunum in neonatal piglets, which has successfully been used in studies of oxidative stress-induced injury to the intestinal epithelium. 9,10 Western diets containing excess energy from fat and low concentrations of fiber, calcium, and cholecalciferol have been shown to cause oxidative stress to animal cells. 20 Thus, an H2O2-induced IPEC-J2 cell model and high-fat diet-induced mouse model were used in the present study to evaluate the protective effects of O. stamineus water-ethanol extracts on the intestinal barrier.
In the cell model, the permeability of the intestinal epithelium was measured with an electrical resistivity meter. As expected, the results showed that H2O2 treatment significantly decreased TEER. In the mouse model, the permeability of the intestinal barrier was measured with an indirect method. In response to oxidative stress, intestinal mucosa cells become necrotic, and DAO leaves the serum and enters the mucosal space between cells in the intestinal mucosa, thus, resulting in markedly elevated levels. 21 Serum endotoxin produced by bacteria has also been used as a molecular marker of intestinal barrier integrity. The results of the present study showed that a high-fat diet significantly increased intestinal epithelium permeability, in agreement with the findings of Suzuki et al, 22 who have reported that a high-fat diet increases small intestinal permeability in rats. In both the cell and mouse models, the treatment course caused significant injury to the intestinal barrier and the intestinal epithelial permeability increased.
Both paracellular and transcellular pathways participate in nutrient transport across the intestinal epithelium. The intestinal epithelial barrier is composed of intestinal epithelial cells and intercellular junctions. 2 Given that our previous study has reported that OE treatment increases the viability 8 and decreases the apoptosis 12 of intestinal epithelial cells in response to oxidative stress, the focus of the present study was on the paracellular pathway.
A major determinant of intestinal permeability through the paracellular pathway is intercellular TJ, which are positioned around the apical end of the lateral cell membrane of intestinal epithelial cells and consist of TJP, such as occludin, claudins, and scaffold protein ZOs. 2 More than 10 claudins and at least 3 ZOs have been identified. 2 This study showed that in both the cell and mouse models, OE supplementation increased mRNA levels of occludin and ZO-1 in the intestinal epithelium. Interestingly, in the cell and mouse models, OE supplementation had no effect on claudin-1 mRNA expression, thus, indicating that OE specifically altered TJP expression. Although animal cells are likely to contain more than 10 claudins, the effect of OE on other claudins remains unknown, and thus further systematic research is needed.
Many studies have reported the antioxidant properties of water-ethanol extracts of O. stamineus. 7,23 The results of our previous study 8 and the present one also confirmed this finding, but whether the antioxidant ability of OE is induced by a decrease in the permeability of the intestinal epithelium remains uncertain.
As demonstrated by the results of Pierre’s relevant analysis presented in Table 4, MDA concentrations were negatively and very significantly correlated with TEER, as well as mRNA levels of occludin, claudin-1, and ZO-1 in the cell model (P < 0.01). Similarly, in the mouse model, MDA concentrations were negatively correlated with mRNA levels of occludin, claudin-1, and ZO-1, and positively correlated with serum DAO concentrations (P < 0.01). MDA is often used as a biomarker of oxidative stress, and a higher MDA content indicates greater lipid oxidization and more severe oxidative stress, thus, implying that OE protects against intestinal barrier injury mainly through its antioxidant properties.
Correlations Between MDA Concentrations and TEER values/DAO Concentrations and mRNA Levels of Occludin, Claudin-1, and ZO-1. a

Abbreviations: DAO, diamine oxidase; MDA, malondialdehyde; mRNA, messenger ribonucleic acid; TEER, transepithelial electrical resistance.
*P< 0.01.
aThe data were calculated via Pierre’s relevant analysis using SPSS software.
bIn the cell model, the data are correlations between the MDA content and TEER value. In the mouse model, the data are correlations between the MDA content and DAO concentration.
Conclusion
As a traditional Chinese medicine, O. stamineus is usually used for the treatment of kidney diseases. However, to the best of our knowledge, this is the first study on the protective effect of O. stamineus against injury to the intestinal barrier. These results showed that OE from different locations and picking times has similar chemical components. The water-ethanol extract of O. stamineus decreased intestinal permeability in response to oxidative stress-mediated injury both in vitro and in vivo. This effect may be due to the antioxidant effect of OE in the up-regulation of the scaffold protein ZO-1 and the TJP occludin, but not claudin-1, which repairs the TJ between intestinal epithelial cells. The results of this study also confirmed that under normal conditions, O. stamineus had no negative effect on intestinal health and thus is safe for daily use. Given that oxidative stress is very common and causes great harm to animal health, O. stamineus extracts can be used as a functional food or feed additive for the prevention and treatment of intestinal barrier injury.
Supplemental Material
Supplementary Material 1 - Supplemental material for The Protective Effects of Orthosiphon stamineus Extract Against Intestinal Barrier Injury in High-Fat Diet-Induced Mouse and Oxidative Stress Cell Models
Supplemental material, Supplementary Material 1, for The Protective Effects of Orthosiphon stamineus Extract Against Intestinal Barrier Injury in High-Fat Diet-Induced Mouse and Oxidative Stress Cell Models by Xuan Cai, Lihui Zhu, Xiaofeng Yin, Huiqin Xue, Changfeng Xiao, Yiqiong Hang, Jianxiong Xu and Yonghong Lu in Natural Product Communications
Footnotes
Acknowledgments
Statement of Human and Animal Rights
All procedures involving animals in this study were conducted in accordance with the Ethics Committee for Research using Laboratory Animals of Shanghai Academy of Agricultural Sciences guidelines for the care and use of laboratory animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful for the financial support from the National Key R&D Program of China (Grant number 2018YFD0500600); the Shanghai Sailing Program (grant number 17YF1413600).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
