Abstract
This study investigated the protective effects of guaiane-type sesquiterpenoids isolated from Dendranthema morifolium (Ramat.) S. Kitam flowers on lipopolysaccharide (LPS)-induced injury in H9c2 cardiomyocyte. Cell viability was determined by thiazolyl blue tetrazolium bromide (MTT). The content of released tumor necrosis factor alpha (TNF-α) and interleukin 6 (IL-6) was evaluated by enzyme-linked immunosorbent assay. The levels of lactate dehydrogenase (LDH) and creatine phosphate kinase (CK) were measured by using commercial available kits. The protein expression levels of pelF2α, GRP78, Bax, caspase-3, caspase-9, Bcl-2, LC3-II, and p62 were measured by in-cell Western. Flow cytometry was used to detect H9c2 cardiomyocyte apoptosis. Compounds
Keywords
Graphical abstract
Guaiane-type sesquiterpenoid compounds were isolated from Dendranthema morifolium (Ramat.) S. Kitam flowers. They could prevent LPS-induced injury in cardiomyocyte by decreasing ER stress, apoptosis, and autophagy as well as downregulating the inflammatory mediators involved in the inflammatory responses. In addition, the active groups might be the angelate at C-8 and the exocyclic double bond at C-11.
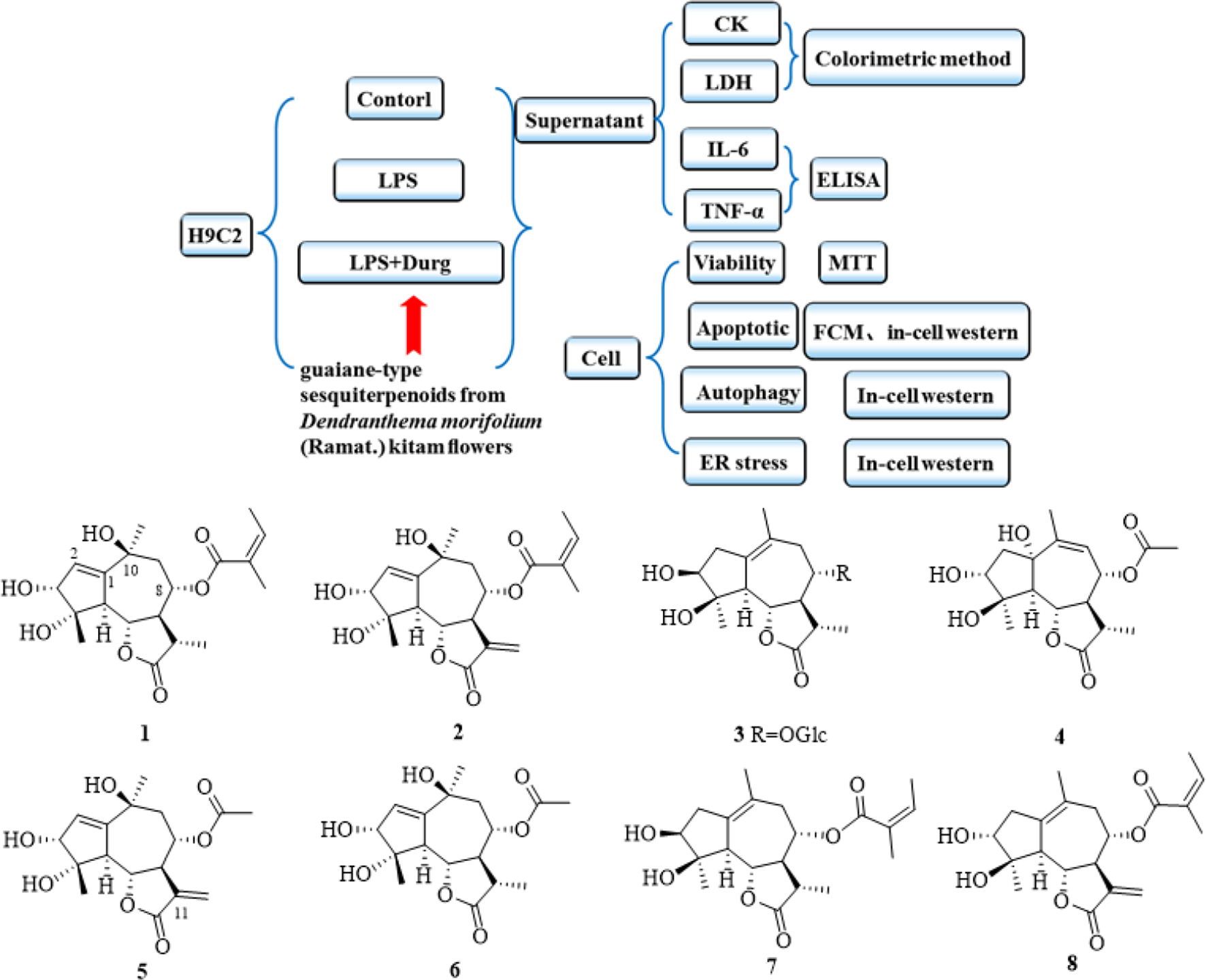
Cardiomyocyte injury is the leading cause of morbidity and mortality in cardiomyopathy patients, such as sepsis, 1 diabetes, 2 and hypertension. 3 Finding effective drugs to protect cardiomyocyte injury will ultimately cure many diseases. A growing body of evidence suggests that cardiomyocyte injury induced by bacterial endotoxin LPS is mediated by cardiomyocyte ER stress, apoptosis, autophagy, and a variety of proinflammatory cytokines such as tumor necrosis factor alpha (TNF-α) 4 and interleukin 6 (IL-6). 5 Regulating cardiomyocyte ER stress, apoptosis, autophagy, and systemic inflammatory response can improve myocardial function. 6 In our study, we investigated the protective effects of guaiane-type sesquiterpenoids from Dendranthema morifolium (Ramat.) S. Kitam flowers on LPS-induced injury in H9c2 cardiomyocyte in order to find the effective drugs to protect cardiomyocyte injury.
Dendranthema morifolium (Ramat.) S. Kitam (Flos chrysanthemum) is one of the well-known daily beverage in China which exhibits antibacterial,
7
antioxidant,
8
anti-inflammatory,
9,10
and heart-protective
11
characteristics. Flos chrysanthemum was recorded in the Shen Nong’s herbal classic and mainly contains flavonoids, triterpenoids, glycosides, and alkaloids.
12,13
In our previous study, 4 new sesquiterpenoids chrysanthguaianolactones C to F (compounds

Structures of compounds 1 to
Results
Activity Assay
To investigate whether compounds
Activity Assay of Compounds 1 to
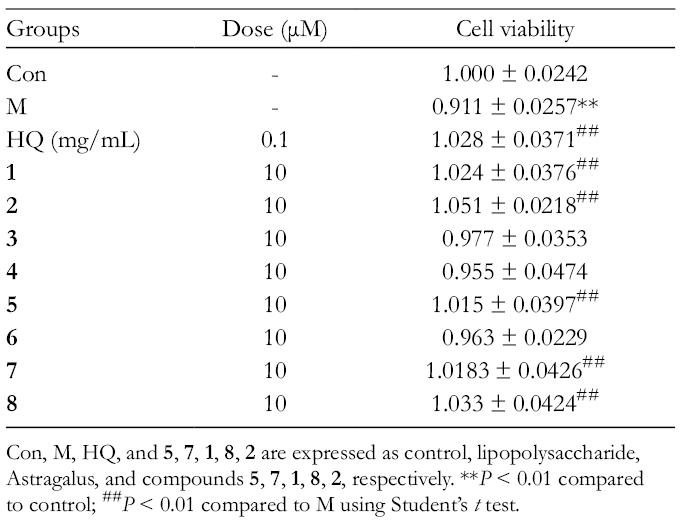
Con, M, HQ, and
Effects of 5 Sesquiterpenoids on LPS-Induced Changes on Proinflammatory Cytokines, Lactate Dehydrogenase, and Creatine Phosphate Kinase
To detect the effect of inflammation on 5 sesquiterpenoids cardioprotection against LPS, the levels of the lactate dehydrogenase (LDH), creatine phosphate kinase (CK), and proinflammatory cytokines such as IL-6 and TNF-α were evaluated. As shown in Table 2, LPS significantly upregulated the expression of LDH, CK, IL-6, and TNF-α. Five sesquiterpenoids mitigated LPS-induced changes in these cytokines. As a positive control, HQ could downregulate the expression of LDH, CK, IL-6, and TNF-α.
Effect on the Levels of Lactate Dehydrogenase, Creatine Phosphate Kinase, Interleukin 6, and Tumor Necrosis Factor Alpha (x ± SD, n = 4).

CK, creatine phosphate kinase; IL-6, interleukin 6; LDH, lactate dehydrogenase; TNF-α, tumor necrosis factor alpha.
Con, M, HQ, and
Effects of 5 Sesquiterpenoids on LPS-Induced Changes on ER Stress Response
As depicted in Figure 2, in-cell Western analysis demonstrated that LPS could significantly increase the levels of ER stress protein markers including pelF2α and GRP78 (P < 0.01), but the effects were ablated by 5 sesquiterpenoids. In addition, HQ could downregulate the expression of pelF2α and GRP78.

Effects of 5 sesquiterpenoids on lipopolysaccharide-induced endoplasmic reticulum stress. Con, M, HQ, and 5, 7, 1, 8, 2 are expressed as control, lipopolysachharide, Astragalus, and compounds 5, 7, 1, 8, 2, respectively. *P < 0.05 and **P< 0.01 compared to control; # P < 0.05 and ## P < 0.01 compared to M using Student’s t test, n = 3.
Effects of 5 Sesquiterpenoids on LPS-Induced Changes on Cell Apoptosis
To translate the effect of 5 sesquiterpenoids on LPS-induced changes on cell apoptosis, we used flow cytometry to examine the cell death of H9c2 cardiomyocyte. Cell apparently underwent 1 type of cell death, apoptosis (38.3%), after 24 hours of LPS exposure as shown by flow cytometry. The compounds treatment significantly inhibited apoptosis (decreased to 20.6%, 16.4%, 15.8%, 12.8%, and 11.8%, respectively). As a positive control, HQ could significantly inhibit apoptosis (decreased to 14.9%) and the results are shown in Figure 3.
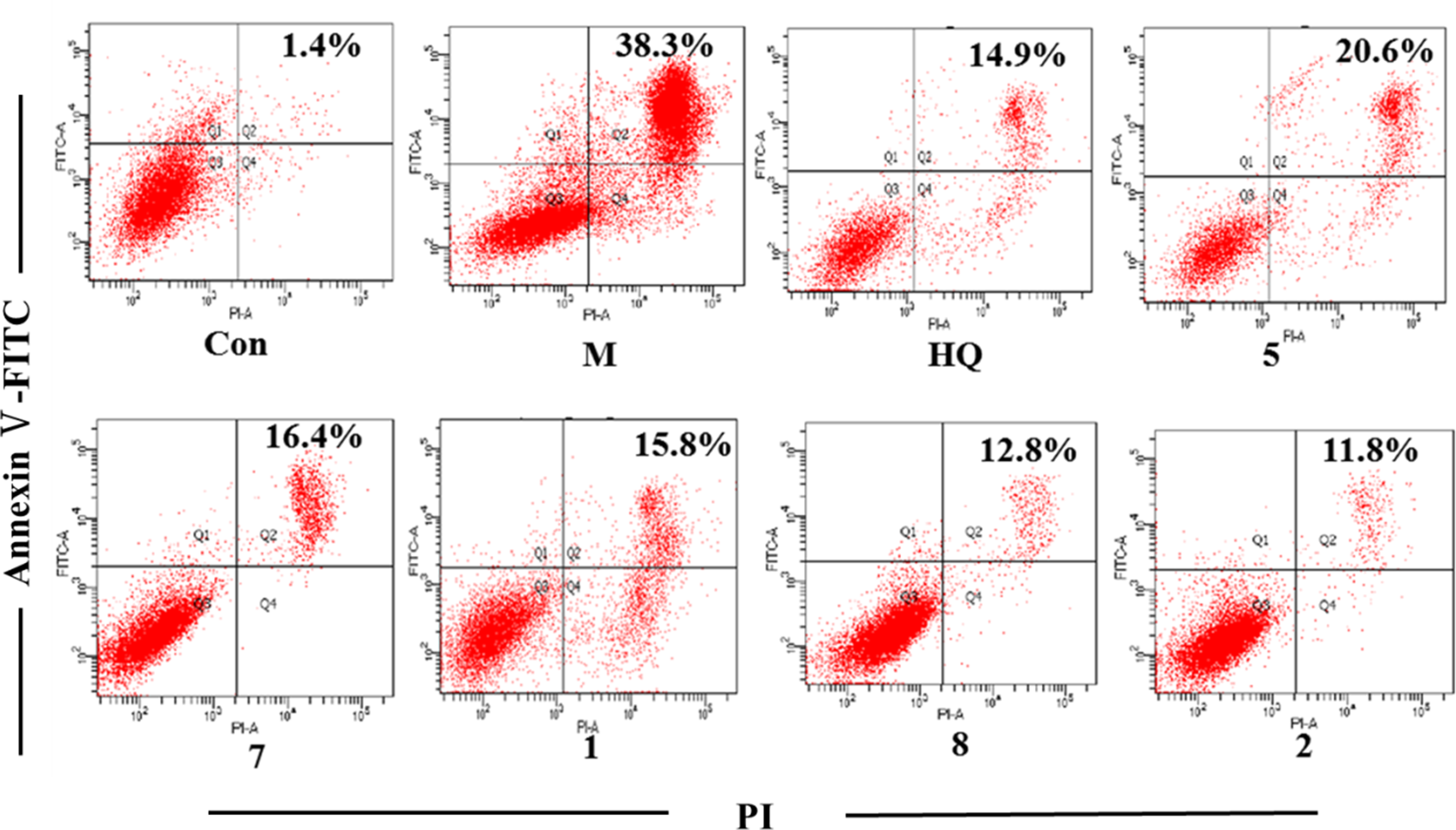
Five sesquiterpenoids on lipopolysaccharide-induced changes on cell apoptosis. H9c2 cardiomyocyte was exposed to lipopolysaccharide for 24 h in the presence or absence of 10 µM 5 sesquiterpenoids. Fluorescence activated cell sorting (FACs) analysis via annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining was utilized to observe the induction and inhibition of apoptosis. Con, M, HQ, and 5, 7, 1, 8, 2 are expressed as control, lipopolysaccharide, Astragalus, and compounds 5, 7, 1, 8, 2, respectively. The number of events that the Con, M, HQ, and compounds 5, 7, 1, 8, 2 use for the analysis are 8901, 10 000, 7097, and 8004, 9612, 9158, 10 000, 10 000, respectively. The lower left quadrants contain the viable cells (annexin V−/propidium iodide−), the lower right quadrants contain the necrotic cells (annexin V−/propidium iodide+), the upper right quadrants contain the apoptotic cells (annexin V+/propidium iodide+), and the upper left quadrants contain the early apoptotic cells (annexin V+/propidium iodide−).
Effects of 5 Sesquiterpenoids on LPS-Induced Changes in Apoptotic Markers
To evaluate the possible role of apoptosis in 5 sesquiterpenoids-offered cardioprotection against LPS treatment, the apoptotic proteins such as Bax, caspase-3, caspase-9, and Bcl-2 were examined. As shown in Figure 4, LPS challenge significantly upregulated the expression of Bax, caspase-3, and caspase-9, while reducing the level of Bcl-2, the effects of which were ablated by 5 sesquiterpenoids. As a positive control, HQ could significantly downregulate the expression of Bax, caspase-3, and caspase-9, while upregulate the expression of Bcl-2.

Effects of 5 sesquiterpenoids on lipopolysaccharide-induced changes on apoptotic markers. Con, M, HQ, and 5, 7, 1, 8, 2 are expressed as control, lipopolysaccharide, Astragalus, and compounds 5, 7, 1, 8, 2, respectively. *P < 0.05 and **P < 0.01 compared to control; # P<0.05 and ## P<0.01 compared to M using Student’s t test, n=3.
Effects of 5 Sesquiterpenoids on LPS-Induced Autophagic Response
To explore if autophagy contributes to 5 sesquiterpenoids-offered cardioprotection against LPS treatment, the levels of autophagic markers including LC3-II and p62 were evaluated. Results shown in Figure 5 depicted that LPS significantly increased the level of p62 and decreased the level of LC3-II, the effects of which were reversed by 5 sesquiterpenoids compared to the M group. In addition, HQ could reverse the expression of LC3-II and p62.

Effects of 5 sesquiterpenoids on lipopolysaccharide-induced autophagic response. Con, M, HQ, and 5, 7, 1, 8, 2 are expressed as control, lipopolysaccharide, Astragalus, and compounds 5, 7, 1, 8, 2, respectively. **P < 0.01 compared to control; ## P < 0.01 compared to M using Student’s t test, n = 3.
Discussion
In our study, the guaiane-type sesquiterpenoid compounds were isolated from D. morifolium (Ramat.) S. Kitam flowers. And pharmacological investigations showed that this kind of compounds exhibited mainly cytotoxicity and anti-inflammatory activities
15
-17
; however, there has been no research about the mechanism. We investigated the protective effects of guaiane-type sesquiterpenoids on LPS-induced injury in H9c2 cardiomyocyte in order to find the mechanism and the effective drugs to protect cardiomyocyte injury in our study. The results of MTT analysis indicated that compounds
Experimental results also showed that the levels of ER stress, apoptosis, and autophagy of LPS group were increased, and the levels of inflammatory factor (TNF-α and IL-6) of the supernatant were also increased. It has been suggested that LPS can lead to H9c2 cardiomyocyte ER stress, apoptosis, autophagy, and release of inflammatory cytokines, eventually resulting in cardiomyocyte injury. The finding of our study showed that 5 sesquiterpenoids can obviously reduce the myocardial cell ER stress, apoptosis, and autophagy as well as downregulate proinflammatory cytokines following LPS challenge. In addition, the compounds could decrease the levels of CK and LDH. We speculated that 5 sesquiterpenoids might have the effects of cardioprotection.
Huang Qi is a traditional Chinese medicine that has the effect of supplementing qi, mainly contained astragalussaponins, sucrose, astragalus polysaccharides, amino acids, selenium, zinc, copper, and so on. 18 -20 Many research have reported that HQ has many kinds of pharmacological activities, such as strong heart, blood vessels, diuresis, antithrombotic, fight free radical damage, protect myocardium, and enhance the immune function. 21 -23 Huang Qi is used for the treatment of myocardial injury, such as sepsis, diabetes, and hypertension. 24 -26 In our study, HQ is a positive control drug that could obviously reduce the myocardial cell ER stress, apoptosis, and autophagy as well as downregulate LDH, CK, and proinflammatory cytokines following LPS challenge. The mechanism of 5 sesquiterpenoids from Flos chrysanthemum protects H9c2 cardiomyoblasts from LPS-induced injury which might be the same as HQ.
The imbalance between the delivery of unfolded proteins to the ER and the unfolded protein response (UPR) is called ER stress.
27
Endoplasmic reticulum stress plays an important role in obesity, neurodegenerative diseases, trauma, diabetes mellitus, and ischemic heart diseases.
28
-30
The apoptosis pathway initiated by ER stress is a new apoptosis pathway found in recent years. The activation of UPR triggered the adaptive response of ER function and promoted cell survival. On the other hand, severe and sustained stress can result in cell apoptosis. Therefore, ER stress is not only the physiological adaptive protection mechanism of cell resistance injury but also an important pathologic mechanism of cell injury and apoptosis. In addition, this study reported that the ER stress also contributes to cardiac dysfunction.
31
In our in vitro study, the ER stress markers including GRP78 and peIF2α were markedly elevated in response to LPS challenge in H9c2 cardiomyoblasts, the effects of which were attenuated or obliterated by compounds
Apoptosis is a programmed cell death and its mechanism is clear. Apoptosis plays an important role in a wide variety of cardiovascular diseases such as ischemia injury, atherosclerosis, alcoholic cardiomyopathy, diabetic cardiomyopathy, and sepsis-associated cardiac dysfunction. Our results demonstrated that 5 sesquiterpenoids ablated LPS-induced apoptosis (Bax, Bcl-2, caspase-3, and caspase-9). This is consistent with the notion that endotoxin is capable of inducing proapoptotic markers such as Bax and caspase-3 in the heart to contribute to heart defects. 32,33 In our study, we also found that LPS upregulated the levels of Bax, caspase-3, and caspase-9, while downregulated the antiapoptotic protein Bcl-2. Furthermore, 5 sesquiterpenoids ablated LPS-induced changes in these apoptosis markers.
Autophagy is a conserved cellular mechanism through which mammalian cells degrade and recycle damaged macromolecules and organelles. 34,35 Low levels of autophagy will lead to myocardial cells that cannot be self-renewed and myocardial function decreased. High levels of autophagy will lead to normal protein, and the organelle of cardiomyocyte degraded and myocardial function is also reduced or even died. LC3 involved in the formation of the mammalian cell autophagy 36 are mainly distributed in the surface of autophagy bubble membrane, besides, the content of LC3-II can reflect positive autophagy bubble number. 37,38 Sequestosome-1 (SQSTM, also known as p62) is an adaptor for autophagosome formation. The higher expression of p62 usually means hindering of fusion of autophagosome and lysosome. 39 Our study suggested that LPS facilitates the levels of p62, as well as decreases LC3-IIin H9c2 cardiomyocyte. Besides, 5 sesquiterpenoids reverse LPS-induced responses in autophagy signaling molecules, suggesting a higher role of autophagy in 5 sesquiterpenoids-induced cardioprotection.
Autophagy and apoptosis are programmed cell death which play an important role in the process of disease pathological progress, including neural degenerative diseases, aging, cancer, and autoimmune diseases. But autophagy is the protector or the executioner of cell death which still has a lot of controversy. Many data suggest that autophagy can change the way of cells die, not just whether they die. 40 So, how do autophagy and apoptosis intersect? Do they cross? Some study showed that severe and sustained stress can result in cell apoptosis and autophagy. Data from our study suggest that 5 sesquiterpenoids ablated LPS-induced changes in ER stress, apoptosis markers, and autophagy signaling molecules. However, the multiple relationships between apoptosis and autophagy are still not demonstrated. A deeper understanding of the relationship between autophagy and apoptosis (either directly or indirectly) is an important research area in the field of cell death in the next few years.
Compounds
In summary, 5 guaiane-type sesquiterpenoids could decrease ER stress, apoptosis, and autophagy, as well as downregulate the inflammatory mediators involved in the inflammatory responses. Results demonstrated that 5 guaiane-type sesquiterpenoids could prevent LPS-induced injury in cardiomyocyte; in addition, the active groups might be the angelate at C-8 and the exocyclic double bond at C-11; furthermore, the activity of C=C bonds C-1(2) is better than that of C-1(10).
Materials and Methods
Plant Materials
Dendranthema morifolium (Ramat.) S. Kitam was collected from Jiaozuo, Henan Province, China, in 2015, identified by Prof Sui-qing Chen (Henan University of Chinese Medicine). A voucher specimen (No. 20150715A) was deposited in the Research Department of Natural Medicinal Chemistry, School of Pharmacy, Henan University of Chinese Medicine. Compounds
Cell Culture and Treatment
The H9c2 were grown in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Pittsburgh, United States) supplemented with 2 mM l-glutamine, 50 U/mL penicillin, 50 µg/mL streptomycin, and 10% heat-inactivated fetal calf serum (FBS, HyClone, Logan, United States) at 37°C in a water-saturated 5.0% CO2 incubator. H9c2 were seeded in 96-well plates in DMEM with 10% (V/V) FBS. The density of the cells in each plate was 2 × 104 cells/mL. Then, the cells were divided into the control group, model group (LPS 20 µg/mL was dissolved in DMEM with 2% dimethyl sulfoxide (DMSO); Sigma, Louis, United States), positive control group (LPS 20 µg/mL, HQ 1 mg/mL) and different sesquiterpenoids group (LPS 20 µg/mL and compounds
MTT Assay
We added 20 µL MTT solution (12.08 × 10−3 mmol/L) to each well and cultured the cells for 4 hours in the incubator. After removing the medium from each well, we added 150 µL DMSO (Amresco, Seattle, United States) and gave shock to the cells for 10 minutes to dissolve the crystals. The absorbance was measured at a wavelength of 490 nm using a microplate reader (Bio-Rad, Hercules, United States).
Enzyme-Linked Immunosorbent Assay for the Detection of IL-6 and TNF-α
For the enzyme-linked immunosorbent assay, a 100 mm × 20 mm Petri dish was used. Cell treatment was done as mentioned earlier; 24 hours later, the culture medium was collected from each petri dish and centrifuged. The supernatants were collected and used to detect the levels of IL-6 (R&D, Minneapolis, United States) and TNF-α (R&D), according to the manufacturer’s instructions.
Measurement of the Levels of LDH and CK
Cell treatment was done as mentioned earlier; 24 hours later, the culture supernatant was collected for the subsequent measurement of CK and LDH levels. The release of the cytosolic enzymes, CK and LDH, indicators of cytotoxicity, reflected a loss of membrane integrity in the damaged cells and was detected by colorimetric assay. Creatine phosphate kinase and LDH activity was measured using commercially available kits (Nanjing Jiancheng Bioengineering Institute, Jiangsu, China) according to the manufacturer’s instructions.
In-Cell Western
In-Cell Western was performed in 96-well plate (165305, Thermo Scientific, Waltham, United States). Cell culture and treatment were done as mentioned earlier; 24 hours later, the cells were fixed with 4% paraformaldehyde for 20 minutes at room temperature (RT) and permeabilized by 5 washes in 0.1% Triton-X 100 in phosphate buffered saline (PBS) with 5 minutes for each wash. Cell monolayers were blocked for 90 minutes and then incubated with primary antibodies (pelF2α ab32157, GRP78 ab181499, Bax ab32503, caspase-3 ab13847, caspase-9 ab32539, Bcl-2 ab59348, LC3-II ab48394, and p62 ab56416; Abcam, MA, United States) diluted in blocking buffer (1:200, 50 µL) overnight at 4°C. After washing with phosphate buffered saline with Tween-20 (PBST) buffer, the cell layers were stained in IRDyeIgG (1:500, 50 µL) for 1 hour at RT in dark, rinsed, and scanned in Odyssey Infra-red Imager (LI-COR Biosciences, Lincoln, United States). Relative protein expression level was normalized against Image Studio.
Apoptosis Assay
The assessment of apoptosis was carried out by double staining the cells with Annexin V (AV)-FITC and PI as recommended by the manufacturer (BD Biosciences 556547, United States). After 24-hour incubation with LPS and sesquiterpenoids, the cells were centrifuged twice in cold PBS, resuspended in 1× binding buffer, and incubated with 5 µL FITC-conjugated AV and 5 µL PI for 15 minutes at RT in the dark. The cells were then analyzed on a flow cytometer (BD FACS AriaIII, United States). Three independent experiments were performed.
Statistical Analysis
The results were expressed as the mean ± SD (standard error of the mean). The significant differences between the negative control and the test fractions were assessed by analysis of variance followed by Student’s t test for multiple comparisons. For comparisons with the negative control groups, P-values less than 0.05 were considered significant.
Footnotes
*
The authors Beibei Zhang, Mengnan Zeng and Meng Li contributed equally to the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Central Government Guide Local Science and Technology Development Funds (14104349) and the Doctoral Research Fund of Henan University of Chinese Medicine (BSJJ2018-08).
