Abstract
Cardiovascular disease has become one of the main diseases that endanger humans, and oxidative damage plays an important role in this. Shenxiong glucose injection (SGI) is a common clinical treatment in China for the treatment of this condition. To understand further the protective effects and related mechanisms of SGI on cardiovascular diseases, H9c2 cells were treated with SGI at different concentrations (0.5%, 1%, 2% [v/v]) before hypoxic damage was induced by treatment with CoCl2). In CoCl2-induced H9c2 cells, SGI treatment increased cell viability and the activity of superoxide dismutase, glutathione peroxidase, catalase, elevated mitochondrial membrane potential, and decreased the rate of cellular apoptosis, lactic dehydrogenase release, and the content of malondialdehyde and reactive oxygen species, while also upregulating Bcl-2 expression and downregulating Bax, Cyt-c, and cleaved caspase-3 expression. Together, these results suggested that SGI has a protective effect on CoCl2-induced damage, and its mechanism may be related to increased antioxidant and antiapoptosis capacity in H9c2 cells. This study provides the basis for further research and potential practical applications of SGI.
Cardio-cerebrovascular diseases, such as coronary heart disease and angina pectoris, are common and frequently occurring diseases that seriously threaten human life and health. Currently, the number of people suffering from cardio-cerebrovascular diseases in China has exceeded 13 million. Current medical research has focused on attempting to prevent effectively and treat cardio-cerebrovascular diseases. 1 Oxidative stress-induced cellular damage plays an important role in these diseases, 2 and anti-apoptosis therapies that scavenge oxygen free radicals are hypothesized to repair and protect myocardial cells from free radical injury. 3,4
It has been well documented that cobalt chloride (CoCl2) is a chemical hypoxia mimetic agent. 5 Under normal physiological conditions, the generating and eliminating of free radicals remain in an equilibrium state. After the cells are treated with CoCl2, Co2+ enters the cells, and the balance is disrupted, resulting in a large accumulation of reactive oxygen species (ROS), 6 altered mitochondrial permeability, and reduced membrane potential. Cyt-c sheds from the inner membrane and is released into the cytoplasm, affecting the expression of the Bcl family and activating the caspase cascade, inducing cell apoptosis. 7,8 Thus, CoCl2 is commonly used to establish hypoxia models and has been widely used to explore the mechanism of cardiac hypoxia.
Shenxiong glucose injection (SGI) is a treatment that utilizes a mixture of Salvia miltiorrhiza Bge extract and ligustrazine hydrochloride and is a common clinical treatment in China for cardio-cerebrovascular diseases, 9 such as coronary heart disease, 10 angina pectoris, 11,12 and cerebral infarction. 13,14 Salvia miltiorrhiza is also known as red sage and has been demonstrated to activate circulation in order to remove blood stasis, relieve pain, treat heartburn, calm the mind, and diminish restlessness. 15,16 Ligustrazine (2,3,5,6-tetramethylpyrazine) is the main component of the traditional Chinese medicine Chuanxiong, which functions by promoting blood circulation, alleviating abdominal stress, and relieving pain and is widely used to treat cardio-cerebrovascular diseases. Studies have shown that both S. miltiorrhiza and ligustrazine have free radical scavenging and antiapoptosis effects. 17,18 It is not clearly known if SGI also treats cardio-cerebrovascular diseases through antioxidative and antiapoptotic pathways. Therefore, this study used a CoCl2-induced H9c2 cell model to establish an in vitro oxidative damage model to explore the protective effect and mechanism of SGI pretreatment on oxidative damage to cells, providing a basis for further development and research on SGI.
Results
CoCl2 Optimum Modeling Conditions
H9c2 cells were treated with different concentrations of CoCl2 (100, 200, 400, 600, 800, and 1000 μmol/L) for different amounts of time (6, 12, and 24 hours). Cell viability was decreased in a dose-dependent manner, while the cell viability after 24 hours of treatment with 600 μmol/L CoCl2 was 54.6 ± 5.2% (Figure 1). Therefore, this concentration and length of time were employed for subsequent experiments.

Screening for the optimum modeling conditions for CoCl2. H9c2 cells were incubated with different concentrations of CoCl2 for different lengths of times. Cell viability was measured by MTS assay. Data are presented as mean values ± standard deviation, n = 3, * P < 0.5, ** P < 0.01, *** P < 0.001 versus control.
SGI Promoted Cell Viability in CoCl2-Induced H9c2 Cells
To explore the protective activity of SGI against CoCl2-induced oxidative damage, H9c2 cells were pretreated with different concentrations of SGI prior to CoCl2 treatment. Cell viability was diminished after adding CoCl2 (P < 0.001), while a cytoprotective effect against CoCl2-induced oxidation damage was exerted by SGI concentrations between 0.5% and 8% (P < 0.05, P < 0.01, P < 0.001, Figure 2). Low, medium, and high stimulant concentrations (0.5%, 1%, 2% [v/v], respectively) were chosen for downstream experiments due to the concentration-dependent effect that SGI exerted when the concentration was between 0.5% and 4%.
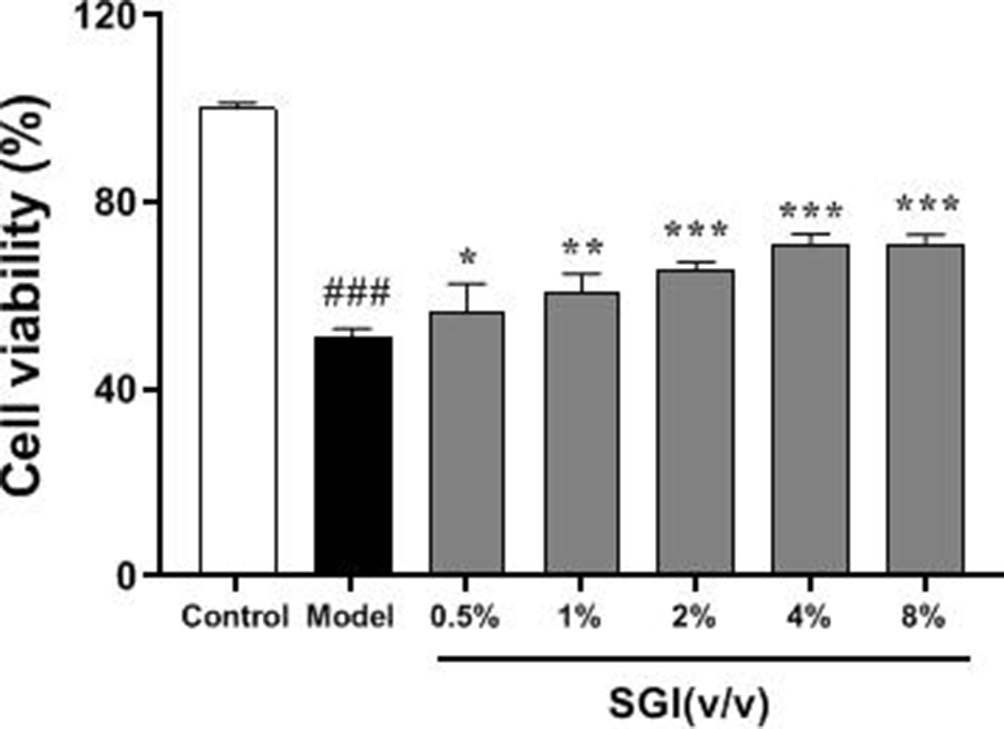
Shenxiong glucose injection (SGI) attenuated the decline in cell viability that was induced by oxidation damage. Cell viability was measured using an MTS method. H9c2 cells were treated with different concentrations of SGI (0.5%, 1%, 2%, 4%, 8% [v/v]) for 24 hours prior to oxidation inducement. data are presented as mean values ± standard devaition, n = 3, ### P < 0.001 versus control, * P < 0.5, ** P < 0.01, *** P < 0.001 versus model.
SGI Protected H9c2 Cells From CoCl2-Induced Oxidative Stress
Oxidative stress has been implicated in the pathogenesis of myocardial injuries. In order to explore the putative protective role of SGI for H9c2 cells during CoCl2-induced oxidative stress injury, the activities of oxidative and antioxidant enzymes, such as lactate dehydrogenase (LDH), malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), and intracellular ROS levels were measured in H9c2 cell lysates. LDH and MDA activity, as well as ROS levels, were significantly decreased in the SGI pretreatment group relative to the CoCl2 group (P < 0.5, P < 0.01, P < 0.001) (Figure 3a, b and f), whereas SOD, GSP-Px, and CAT activities were increased (P < 0.05, P < 0.01) (Figure 3c, d and e).
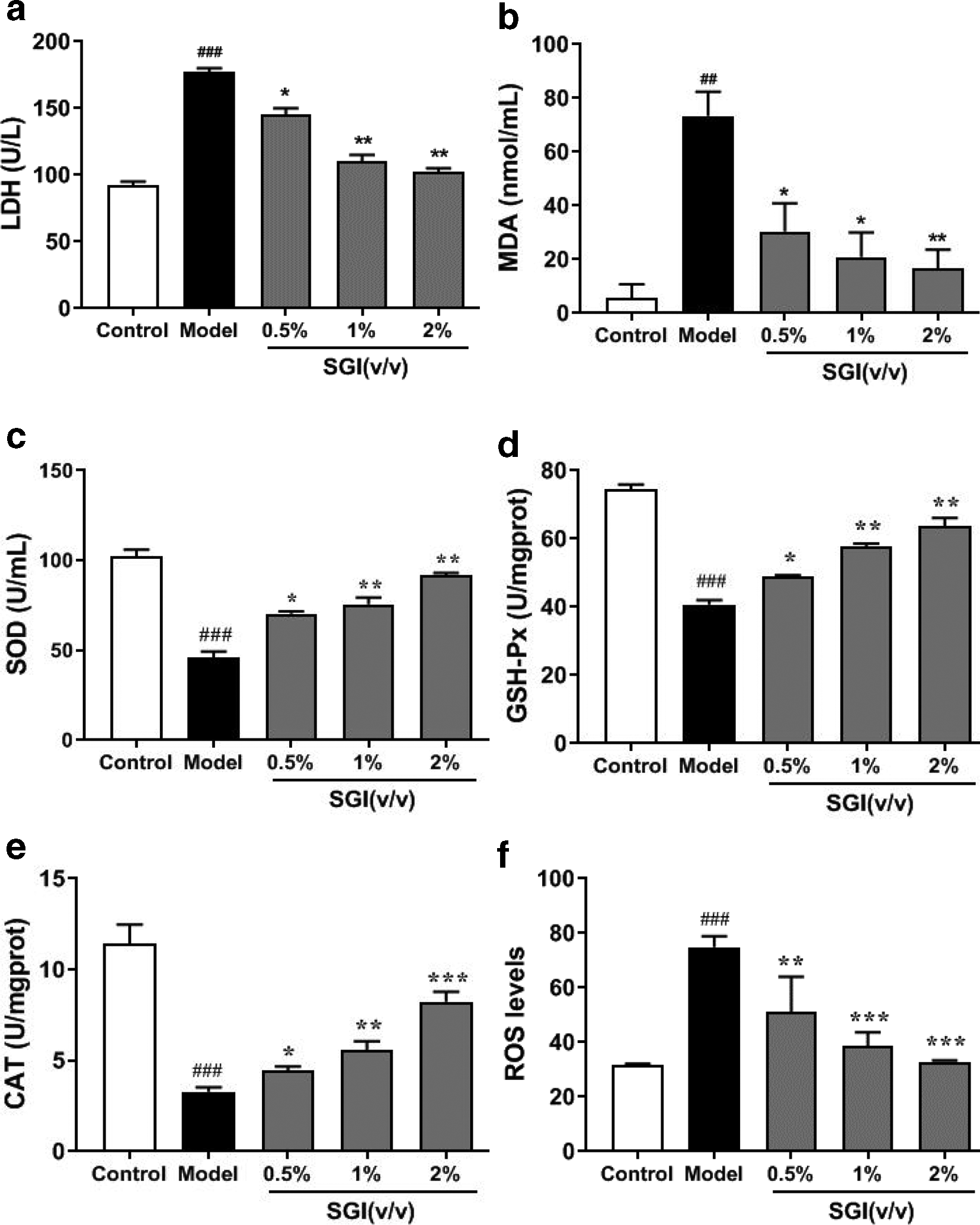
Protective effects of Shenxiong glucose injection (SGI) by enhancing antioxidant function examined by measuring the activity of lactate dehydrogenase (LDH), malondialdehyde (MDA), superoxidedismutase (SOD), glutathioneperoxidase (GSP-Px), and catalase (CAT), as well as reactive oxygen species (ROS) levels. H9c2 cells were treated with SGI at low, medium, and high SGI concentrations (0.5%, 1%, 2% [v/v]) for 24 hours prior to cobalt chloride treatment. Data are presented as mean values ± standard deviation, n = 3, ### P < 0.001 versus control; * P < 0.5, ** P < 0.01, *** P < 0.001 versus model.
SGI Protected H9c2 Cells From CoCl2-Induced Apoptosis
The effect of SGI on CoCl2-induced H9c2 apoptosis was measured using Annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI) staining. The proportion of apoptotic cells was significantly increased after CoCl2 treatment compared with the control group (25.3% ± 3.5% and 7.1% ± 2%, P < 0.001) (Figure 4a,b), whereas 24-hour pretreatment with low, medium, and high SGI reduced the percentage of apoptotic cells to 18.7%± 3.4%, 18.1% ± 2.2%, 14.2% ± 0.9%, respectively (P < 0.05, P < 0.01, (Figure 4c,d,e).

Shenxiong glucose injection (SGI) protected H9c2 cells from cobalt chloride (CoCl2)-induced apoptosis. The percentage of apoptotic cells was determined by Annexin V-fluorescein isothiocyanate and propidium iodide staining to quantify the effects of SGI on CoCl2-induced H9c2 apoptosis. (a) H9c2 cells without SGI or CoCl2 (control); (b) H9c2 cells exposed to 600 μmol/L CoCl2 (model); (c-e) H9c2 cells pretreated with 0.5%, 1%, 2% (v/v) SGI, followed by treatment with 600 μmol/L CoCl2; (f) apoptosis histogram. ### P < 0.001 versus control; * P < 0.05, ** P < 0.01 versus model.
SGI Rescued Loss of Mitochondrial Membrane Potential in CoCl2-Induced H9c2 Cells
Mitochondrial membrane potential (MMP) rapidly decreases during the apoptotic process. JC-1 assay was used to determine the extent to which mitochondrial membrane potential participates in CoCl2-induced apoptosis. The MMP decreased (green fluorescence) in CoCl2-treated H9c2 cells, while SGI pretreatment significantly reduced this loss (Figure 5).

Shenxiong glucose injection (SGI) reduced loss of mitochondrial membrane potential in cobalt chloride (CoCl2)-induced H9c2 cells, as assessed by JC-1 staining. (a) H9c2 cells without SGI or CoCl2 as control, H9c2 cells exposed to 600 μmol/L CoCl2 as model, H9c2 cells pretreated with 0.5%, 1%, 2% (v/v) SGI followed by treatment with 600 μmol/L CoCl2 . (b) Mitochondrial membrane potential histogram. ### P < 0.001 versus control; * P < 0.5, ** P < 0.01 versus model.
SGI Attenuates CoCl2-Induced Apoptosis via the Mitochondrial Pathway
Bcl-2, Bax, Cyt-c, and cleaved caspase-3 are proteins that regulate apoptosis via the mitochondrial pathway. The expression of these proteins was examined by Western blot analysis to investigate if SGI protects H9c2 cells from CoCl2-induced damage via the mitochondrial pathway. Treatment with CoCl2 for 24 hours markedly increased Bax and Cyt-c, and cleaved caspase-3 expression in H9c2 cells, while Bcl-2 was significantly decreased compared with the control (P < 0.001). However, pretreatment with SGI for 24 hours significantly attenuated these effects, bringing Bcl-2, Bax, Cyt-c, and cleaved caspase-3 expression closer to control levels (P < 0.05, P < 0.01, P < 0.001), suggesting that SGI could ameliorate CoCl2-induced apoptosis (Figure 6).

Shenxiong glucose injection (SGI) attenuates cobalt chloride-induced apoptosis via the mitochondrial pathway. (a) Detection of Bcl-2, Bax, Cyt-c, and cleaved caspase-3 proteins by Western blot analysis. (b) Protein expression histogram. ### P < 0.001 versus control; * P < 0.5, ** P < 0.01 versus model.
Discussion
SGI is a traditional Chinese medicine injection consisting of danshen water extract and ligustrazine hydrochloride. Salvia miltiorrhiza is widely used in the treatment of cardio-cerebrovascular diseases, hyperlipidemia, and acute ischemic stroke. 19 Its effective active ingredients are mainly two categories of water-soluble phenolic compounds and fat-soluble diterpene quinones. 20 SGI’s Salvia components are water-soluble phenolic compounds, which mainly include danshensu, protocatechuic aldehyde, salvianolic acid A, B, C, D, I/H, rosmarinic acid, and shikonic acid. 21 Studies have shown that salvianolic acid has antimyocardial ischemia effects and increases coronary blood flow 22 . Danshensu is a strong antioxidant that can scavenge major free radicals in the body such as superoxide anion and hydroxyl radicals. 23 Salvia miltiorrhiza is equivalent to an oxygen free radical scavenger, which can inhibit the expression of inflammatory mediators during atherosclerosis by inhibiting lipid peroxidation during reperfusion of myocardial cell membranes. 24 Significant inhibition of lipid peroxidation in the heart, liver, kidney, and testis of animals was also reported by Gao et al. 25 Ligustrazine hydrochloride has oxygen free radical and antiapoptotic effects. 26 Therefore, we speculate that SGI prevention of cardiovascular disease may be related to antioxidation and antiapoptosis.
The underlying mechanisms by which myocardial infarction causes hypoxia injury are complicated, although oxidative stress is currently considered to be the main cause of many cardiovascular diseases. 27 Under physiological conditions, endogenous antioxidant enzymes—such as SOD, GSH-Px, and CAT—play essential protective roles in myocardial ischemia and hypoxia injury by either indirectly or directly scavenging oxygen free radicals, while also maintaining the balance of generating and eliminating these radicals. 28 -30 However, this balance is disrupted under hypoxic/ischemic conditions, causing a series of redox reactions that can aggravate the pathological condition. This study revealed that SGI pretreatment could increase SOD, GSH-Px, and CAT activity, reduce MDA and ROS levels, diminish LDH leakage, and increase the cell survival rate.
It has been reported in the literature that apoptosis is related to the death receptor (DR) pathway, mitochondrial pathway, endoplasmic reticulum (ER) stress, and autophagy. 31 The death receptor pathway, also known as the exogenous pathway, is initiated by the production of death ligands in natural killer cells or macrophages. After the death ligands have bound to specific DRs, they activate Caspase-8 and cause apoptosis. 32 Mitochondria are the control center of cell life activity, which regulate cellular respiration, oxidative phosphorylation, and apoptosis. Studies have shown that the release of Cyt-c from mitochondria is a key step in apoptosis, and the entry of Cyt-c into the cytoplasm can lead to Caspase-9 activation; activated Caspase-9 further activates Caspase-3, 6 and, etc., to induce apoptosis. 33 ER stress is an adaptive or protective response that can increase the protein folding ability of the ER by activating the unfolded protein response (UPR), eliminating unfolded or misfolded proteins, 34 but, if the stress cannot be tolerated, ER stress upregulates GRP78, CHOP, and caspase-12 proteins, leading to apoptosis. Autophagy is an important cellular process involved in the recovery of long-lived proteins and organelles and is essential for maintaining the phenotype and function of normal cells. 35 When autophagy is disrupted, the overexpression of the autophagy protein Atg5 leads to apoptosis. In addition, there are reports in the literature that there are interactions in the above pathways that co-regulate apoptosis. 36
Recent studies have demonstrated that CoCl2-induced apoptosis in H9c2 cells is associated with the mitochondrial pathway. 37 Mitochondrial transmembrane potential dissipates before nuclease activation and phosphatidylserine exposure at the cell surface, and apoptosis is irreversible once this event occurs. 38 Under hypoxic/ischemic conditions, the imbalance between endogenous antioxidant enzymes and ROS production becomes exacerbated, increasing ROS levels causing decreased MMP and Bcl-2/Bax expression, while also releasing Cyt-c. 39 As Cyt-c enters the cytoplasm, caspase-3 is activated and leads to cell apoptosis. 40,41 In this study, CoCl2 treatment increased ROS production, Bax, Cyt-c, and cleaved caspase-3, and also the apoptosis rate, while MMP and Bcl-2 expression were decreased, which was consistent with a previous report. 42 However, pretreatment with SGI reversed the above phenomena, suggesting that the mitochondrial pathway may be one of the mechanisms by which CoCl2 induces apoptosis in H9c2 cells. As apoptosis is a complex process, the potential methods by which SGI inhibits apoptosis through the death receptor pathway, endoplasmic reticulum stress, and autophagy pathways remain to be elucidated.
SGI contains danshensu, protocatechuic aldehyde, salvianolic acid A, B, C, D, I/H, rosmarinic acid, shikonic acid, other danshen water-soluble phenolic compounds, and ligustrazine hydrochloride, and danshensu, salvianolic acid, and ligustrazine hydrochloride have antimyocardial ischemic properties. Therefore, we speculated that danshensu, salvianolic acid, and ligustrazine hydrochloride may be the active ingredients of SGI to antagonize CoCl2-induced H9c2 cell hypoxia injury, but subsequent experiments are needed to verify this. In addition, due to the characteristics of traditional Chinese medicines, such as multiple active components, multiple targets, and multiple pathways, further research is needed on their main active components and pathways.
This study revealed that SGI could inhibit oxidative cellular injury by increasing SOD, GSH-Px, and CAT activity, reducing LDH leakage, and MDA and ROS production. Simultaneously, SGI also inhibited apoptosis by upregulating Bcl-2 expression and decreasing the expression of Bax, Cyt-c, and cleaved caspase-3. These results indicated that SGI can protect H9c2 cells from CoCl2-induced hypoxic injury through both antioxidative and antiapoptosis pathways, which provide a basis for further research regarding practical applications of SGI.
Materials and Methods
Cell Culture and Treatments
H9c2 embryonic rat cardiac cells (Type Culture Collection of the Chinese Academy of Sciences, Shanghai, China) were cultured in Dulbecco’s modified Eagle’s medium supplemented with 100 U/mL penicillin, 100 µg/mL streptomycin, and 10% fetal bovine serum at 37°C under an atmosphere of 5% CO2 and 95% air.
A chemical hypoxia model was achieved by pretreating H9c2 cells with SGI for 24 hours, then adding 600 μmol/L CoCl2 for 24 hours. Control cells were maintained with no treatment.
MTS Assay
The MTS (Promega Corporation, Madison, WI, USA) assay was performed using a CellTiter 96 AQueous One solution Reagent according to the manufacturer’s instructions. Briefly, H9c2 cells were seeded into 96-well plates at a density of 9000 cells per well. After incubating overnight, the cells were treated with different doses of SGI (0.5, 1.0, 2.0, 8.0%) for 24 hours, followed by the addition of SGI and CoCl2 as described above. Subsequently, 5 µL of MTS solution (90 mg/mL) was added to each well, followed by incubation at 37°C for 2 hours. The absorbance at 490 nm of each culture well was measured with a microplate reader (Bio-Rad, Hercules, CA, USA).
Detection of Intracellular MDA, SOD, CAT, GSH-Px, and LDH in Culture Supernatant
After the treatments described above, culture supernatant and digested cells were collected. The concentrations of intracellular MDA, SOD, CAT, and GSH-Px were measured as described by the assay kit protocol. The release of LDH from H9c2 cells in the supernatant was measured using an LDH assay kit.
Detection of Intracellular ROS
Intracellular ROS was measured using a dichloro-dihydro-fluorescein diacetate (DCFH-DA) probe. Suspensions of digested H9c2 cells were incubated with DCFH-DA for 20 minutes, washed 3 times with phosphate buffer saline (PBS) to remove the residual probe, and resuspended in 500 µL of PBS. ROS was detected using a C6 Plus flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA).
Apoptosis Detection Using Annexin V-PI
H9c2 cells were harvested with trypsin, washed twice with cold PBS at 4°C, and resuspended in 1× binding buffer at a concentration of 1 × 106 cells/mL. A total of 100 µL of the cell resuspension was transferred to a 5 mL culture tube. Cells were incubated with 5 µL of FITC Annexin V and 5 µL of PI for 5 minutes in the dark at room temperature, followed by addition of 400 of μL 1× binding buffer. Fluorescence was analyzed using a C6 Plus flow cytometer (BD Biosciences).
Measurement of MMP
H9c2 cells were harvested with trypsin, washed twice with cold PBS at 4°C, and centrifuged at 1000 rpm for 5 minutes. JC-1 (5 µM) and 1 mL of medium were added to the cell pellet and incubated for 20 minutes at 37°C. The cells were washed twice with dye buffer, followed by adding 2 mL of medium and detection with a fluorescence microscope (Nikon, Toyko, Japan).
Western Blot Analysis
After the treatments described above, H9c2 cells were harvested and lysed with RIPA buffer (Beijing Solarbio, Beijing, China). Total protein content was measured by the bicinchoninic acid assay and Bradford protein assay (Beijing Solarbio). Sample buffer was added to cytosolic protein extracts and then boiled for 5 minutes. Equal amounts of supernatant from each sample were fractionated by electrophoresis through a 15% sodium dodecyl sulfate–polyacrylamide gel and transferred onto a polyvinylidene difluoride membrane (Millipore, MA, USA). Membranes were blocked for 2 hours at 4°C in fresh blocking buffer (5% Tween 20 in Tris-buffered saline [TBS-T] containing 5% bovine serum albumin [BSA]) and then incubated with anti-Bcl-2, anti-Bax, anti-Cyt-c, anti-cleaved caspase-3, or anti-β-actin antibodies (1:1000 dilution; Abcam, Cambridgeshire, UK) in 5% BSA TBS-T overnight with gentle agitation at 4°C. Following 5 washes with TBS-T, the membranes were incubated with horseradish peroxidase-conjugated goat antirabbit secondary antibodies (1:2000; Abcam) in TBS-T with 5% BSA for 2 hours at room temperature. Membranes were washed 5 times with TBS-T and then treated with an enhanced chemiluminescence agent (Beyotime, Shanghai, China). The signals were captured using a SYNGENE G:BOX Chemi XL 1.4 (SYNGENE, Cambridge, UK) system and normalized for quantification using Image Lab software (Bio-Rad).
Statistical Analysis
All data are presented as the mean ± standard deviation. Differences between groups were analyzed by one-way analysis of variance using SPSS 13.0 (SPSS, Chicago, IL, USA). P < 0.05 was considered to be statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China (81760699, 81803827 and U1812403), Guizhou Provincial Natural Science Foundation ([2019]1280), and Innovation Talent Team Project of Guizhou Province (20165613, 20165677).
