Abstract
An ultra-performance liquid chromatography-electrospray ionization-mass spectrometry targeted metabolomics strategy was applied to analyze protocatechuate, syringin, eleutheroside E, isofraxidin, hyperoside, kaempferol, and oleanolic acid, the active compounds in 3-year-old, 5-year-old, and 9-year-old Acanthopanax senticosus. Then, targeted metabolomics was conducted with 3 growth year plants to identify 19 phenolic metabolites related to the above-mentioned active compounds, including 9 C6C3C6-type, 6 C6C3-type, and 4 C6C1-type. Multivariate statistical analysis was applied to the bioactive metabolite data, and targeted metabolic profiling was used for marker compound classification and characterization. The results showed that 7 active compounds in the roots and stems in the 3 growth year plants differed. The principal component “Q” values showed that the total contents of 7 active compounds in 5-year-old roots and stems were higher than in other growth years. Results of targeted metabolomics profiling of 19 phenolic metabolites showed that the C6C1-type compounds accumulated in 9-year-old plants, the C6C3-type in 3-year-old plants, and the C6C3C6-type in 5-year-old plants. The stems had the greatest accumulations of the phenolic metabolites. C6C1 and C6C3-type metabolites are the most abundant in both roots and stems. In conclusion, the active compounds and pharmacological effects of A. senticosus in different growth years are different. The best harvest age for A. senticosus roots and stems was 5 years. The accumulation of 19 phenolic metabolites in different growth years also showed significant differences.
Acanthopanax senticosus (Rupr. et Maxim.) Harms (A. senticosus thereafter), family Araliaceae, 1 is a tonic and sedative Chinese herb useful in treating various conditions, including stress-induced pathophysiological changes and inflammation. 2,3 The species has been shown to contain many active constituents, including flavonoids, 4 simple phenylpropanoids, 5 organic acids, 6 coumarins, 7 triterpenoid saponins, 8 and amino acids. Previous studies have shown that syringin, eleutheroside E, and isofraxidin are the major pharmacologically active components of A. senticosus. 1 Syringin has anti-fatigue, immunomodulation, increasing insulin secretion, and anti-tumor activities. 9 -11 Eleutheroside E has antidepressant and anti-fatigue effects, 12,13 and isofraxidin has anti-tumor, antibacterial, and anti-inflammatory activities. 14,15 Rutin, hyperoside, and kaempferol are common flavonoids, mainly present in A. senticosus leaves. 16 They have anti-oxidant and anti-infective effects. 17 -19 In addition, protocatechuate is also an active compound found in A. senticosus. 20
Over the past 20 years, A. senticosus on the market has been derived from wild plants, which grow only at high altitudes in north-eastern China. At present, wild A. senticosus resources are nearing depletion. Thus, it is necessary to cultivate the species to guarantee supplies. Plants have different levels of bioactivities dependent on the number of years of growth. The optimal harvest time of a plant coincides with when it exhibits its highest level of bioactive compounds. So, determining the best harvest time for A. senticosus is of considerable significance, which can guide the large-scale cultivation of this species.
Liquid chromatography (LC)–mass spectrometry (MS) technology was developed in the 1990s. Because of its high selectivity and low detection limits, LC–MS has become a promising analytical method. 21,22 Thus far, several high-performance liquid chromatography (HPLC)–ultraviolet (UV) methods have been used for the quality control of A. senticosus at different growth years. These studies analyzed only a few active compounds and used a long runtime; the results showed that the content of active compounds in the 5-year-old plants was higher than that in other growth years. 23 -25 Although some ultra-performance liquid chromatography (UPLC)-time-of-flight mass spectrometry (TOF)-MS methods have been developed to determine the bioactive constituents of A. senticosus, the difference in different growth years of the plant has not been reported. 26,27 At present, A. senticosus of different ages is sold in the market, but these plants vary in their contents of active ingredients and pharmacological actions. Thus, it is necessary to establish a multi-index method to control the quality of the different growth years of A. senticosus.
Because plants produce diverse compounds, the metabolomics approach is required to estimate the therapeutic effects of natural products and ensure their quality control. Currently, LC–MS has been widely used for the study of plant metabolites. 28,29 As multiple reaction monitoring (MRM) has the advantages of sensitivity, accuracy, and specificity, UPLC-electrospray ionization (ESI)-MS and MRM are often used for the quantitative analysis of targeted metabolites. 30 Our previous studies compared the metabolic profiles of 7 active compounds and 19 phenolic metabolites between A. senticosus and Acanthopanax sessiliflorus. The results showed that A. senticosus possess a higher abundance of compounds, particularly in the roots and stems. The 19 phenolic metabolites could be clearly grouped into 5 clusters based on their tissue-specific accumulation patterns. Roots are the tissue with the most abundant accumulations, and the C6C3C6-type metabolites are the most widely existent type in both plants. 31
Up to date, there has been little research about the metabolomics profiling of cultivated A. senticosus of different ages. We hypothesize that the content of the 7 active compounds and the accumulation of phenolic metabolites in different growth years is different and that the active compounds in 5-year-old A. senticosus are higher than in other growth years. Therefore, metabolomics strategies and correlation analysis were used to determine the growth year properties of 7 active compounds and 19 phenolic metabolites. We hope that the consequences of this research can provide a reference for the cultivation of A. senticosus.
Materials and Methods
Chemicals and Reagents
UHPLC-grade acetonitrile and methanol were bought from Fisher Scientific (Pittsburgh, PA, USA). All other reagents were of analytical purity. Ultrapure water was obtained using a Milli-Q (Millipore, Bedford, MA, USA) water purification system. The reference compounds needed were all purchased from Chroma Dex Inc (Santa Ana, CA, USA). Eleutheroside E, rutin, kaempferol, isofraxidin, protocatechuate, syringin, and hyperoside were used to determine the contents of the active compounds. Syringic acid, vanillic acid, salicylic acid, cinnamic acid, gallic acid, ferulic acid, chlorogenic acid, caffeic acid, p-hydroxycinnamic acid, p-coumaric acid, petunidin, luteolin, apigenin, naringenin, genistein, quercetin-3-O-rhamnoside, quercetin, myricitrin, and catechin were used for the targeted metabolomics study. The purity of these standards was higher than 98%.
Sample Preparation
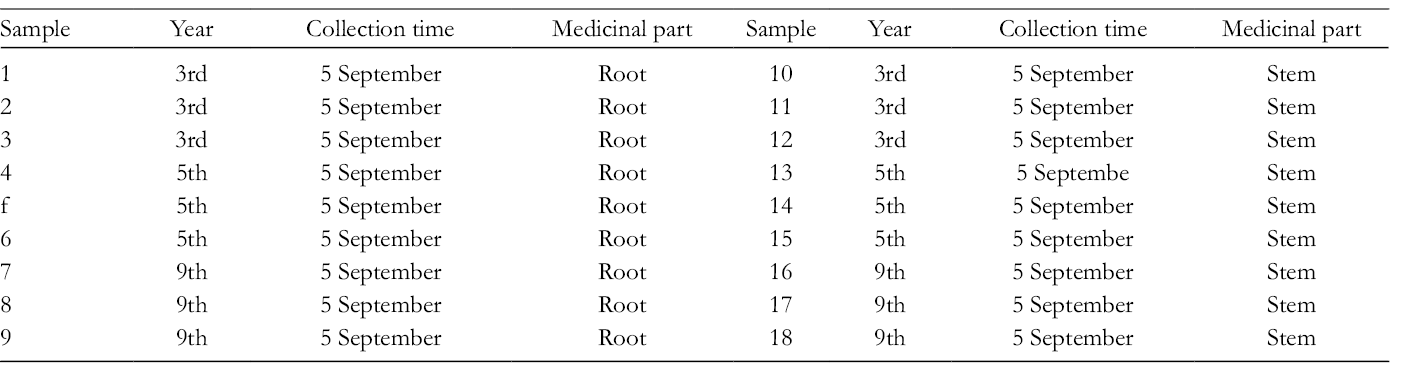
For eliminating the interference of biotic and abiotic stress factors on the results, 18 samples of A. senticosus, including the roots and stems from plants (3 years, 5 years, and 9 years), were collected from the same producing areas in Qitaihe of Heilongjiang Province in northeast China (45′95N, 131′05E) in September 2017. Because 3-year-old and 5-year-old A. senticosus plants are currently sold as commodities in China, and the 9-year-old A. senticosus is the oldest sample collected, the above 3 growth years were chosen for the research. Six independent biological samples were collected per growth year, randomly selected in our experimental plot (Table 1). The whole plant was dissected to separate roots and stems (ASR and ASS thereafter).
Detailed Information of Acanthopanax senticosus Samples.
The raw samples were dried at 60°C for 48 hours. Then, the dried samples were powdered in a mill and sieved through a 20 µm mesh. Powdered samples (0.5 g) were extracted with 20 mL methanol for 30 minutes in an ultrasonic bath (100 kHz, 40°C). After filtration, 20 mL solvent was added to the residue and subjected to supersonic extraction for 30 minutes; the 2 filtrates were merged. All samples were filtered through a 0.45 µm Millipore filter before being analyzed by UPLC. The experiment was performed in triplicate.
LC–MS Conditions
The methods used for the quantitative analysis of the 7 active compounds were the same as in our previous studies. 31 UPLC-MS analysis was performed with an ACQUITY UPLC system (Waters Corporation, Japan) coupled with an LC-20AD pump, SIL-20A autosampler (Waters Corporation, Japan). The ACQUITY UPLC BEH C18 column (1.7 µm, 2.1 mm × 50 mm) used for UPLC analysis was held at 25°C; injection volume was 10.0 µL, and the flow rate was 0.5 mL/minute. Mobile phase A consisted of methanol, while mobile phase B comprised water. The chromatographic column was eluted with a linear gradient of 25% A for 0-1.5 minutes, 25-50% A for 1.5-2.0 minutes, 50% A for 2.0-4.0 minutes, 50%-90% A for 4.0-4.5 minutes, 90% A for 4.0-4.5 minutes, 90%-25% A for 5.5-6.0 minutes, and 25% A for 6.0-7.0 minutes. Mass spectrometric detection was performed using a QTRAP 5500 (AB SCIEX, USA) equipped with an ESI source. Operation parameters were as follows: cone voltage 3 kV and ion source atomizing gas temperature 500°C; 25 psi atomizing gas and 20 psi air curtain gas. The retention time, ion pair, declustering potential,collision energy, and collision chamber injection voltage of the 7 active compounds are shown in Table 2. Both MS and MS/MS data were determined in the positive mode, and data were used for multiple reaction monitoring (MRM).
tR, MS/MS Fragment Ions, DP, CE, and CXP of the 10 Active Metabolites.
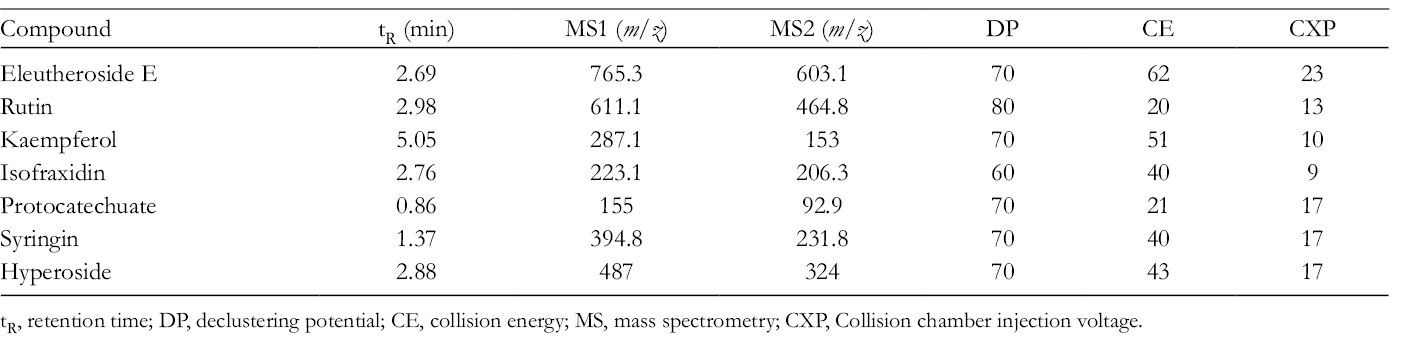
tR, retention time; DP, declustering potential; CE, collision energy; MS, mass spectrometry; CXP, Collision chamber injection voltage.
The conditions for targeted metabolomics analysis of the 19 phenolic metabolites were the same as in our previous studies. 21 Samples were analyzed using an LC system coupled to a QTOF tandem-mass spectrometer via an ESI interface (Agilent 6520) (Agilent Technologies, Santa Clara, CA, USA). Sample extracts were separated on a Shimpack LC column (VP-ODS C18 pore size 5.0 µm, 2 × 150 mm), the mobile phase consisting of solvent A and solvent B. Solvent A contained 0.04% acetic acid in water, and solvent B 0.04% acetic acid in acetonitrile. The following gradient was adopted at a flow rate of 0.5 mL/minute: 0-20 minutes, 5% B-95% B; 20-22.1 minutes: 95% B-5% B; 22.1-28 minutes: 5% B-5% B. A blank measurement with the initial solvent was made after each HPLC run. The injection volume and column temperature were adjusted to 5 µL and 40°C, respectively. Optimal MS conditions for positive ion electrospray were: capillary temperature 350°C; curtain gas pressure 40 psi; capillary voltage 3500 V, and fragmentation voltage 135 V. The instrument was tuned prior to each batch run. A full-scan ranging between 50 and 1000 m/z was conducted with a scan time of 1 second and an interscan delay of 0.1 seconds in centered mode.
Statistical Analysis
Analysis of variance of data was performed using SPSS 22.0 software (SPSS, Inc., Chicago, IL USA). The results for 7 active compounds were shown as mean values (mean ± standard error), and the differences between the samples were compared by using a one-way analysis of variance at P < 0.05 probability level (Duncan’s test). The relative peak intensities of 19 phenolic metabolites were normalized and imported into R Project (https://www.r-project.org/) for cluster analysis and heat map drawing. Metabolic pathway enrichment was performed using MBROLE 2.0, and the metabolic network diagram was drawn using Cytoscape 3.7.
Results
Method Validation
As shown in Figure 1, we obtained the total ion current (TIC) spectra for 7 active compounds detected within 6 minutes. The retention times of protocatechuate, syringin, eleutheroside E, isofraxidin, hyperoside, rutin, and kaempferol were found to be almost 0.86, 1.37, 2.69, 2.76, 2.88, 2.98 and 5.05 minutes, respectively. The correlation coefficients (r) of all the calibration curves were higher than 0.998. The limit of detection (LOD) and the limit of quantification (LOQ) values of the protocatechuate, hyperoside, isofraxidin, and kaempferol were 0.030 ng/mL and 0.1 ng/mL, respectively, and values for LOD and LOQ of rutin, syringin, and eleutheroside E were 0.6 ng/mL and 1 ng/mL, respectively. These results displayed good linear relationships between peak area and concentration. The results gave precision (RSD) variations of 0.44%-2.25% and stability ranged from 0.05% to 1.13%, respectively. The repeatability (RSD) ranged between 0.23% and 2.09%. Thus, the developed method was reproducible and accurate. Based on these results, a reliable and straightforward UPLC-MS method for the quantification of the target compounds in A. senticosus has been developed and validated for its linearity, accuracy, precision, LOD, and LOQ.

Ultra-performance liquid chromatography-mass spectrometry chromatograms of 7 standard target compounds.
Accumulation of Active Compounds in Different Growth Years
All A. senticosus samples from different growth years were collected from the same planting area, and their growing environment was the same, so the influence of environmental factors on their active compounds can be ignored. The principal component “Q” is an indicator of a comprehensive, scientific evaluation of objective phenomenon. 32 As shown in Figure 2.H, the Q value reflecting accumulation abundance of the 7 active compounds in ASR and ASS at different growth years was obtained. The most extensive accumulation of the active compounds was in the 5-year-old plants. The stems possessed a higher content of the active compounds, especially those of the 3-year-old and 5-year-old plants.

The contents of 7 bioactive compounds in different parts of different growth year (a-g) and the corresponding Q value (h). The letters a, b, c indicates significant differences between different growth year of the same part (P < 0.05); ** indicates significant differences between different tissues of same growth year (P < 0.01) ; * indicates significant differences between different tissues of same growth years (P < 0.01).
Figure 2 shows that the concentrations of the 7 active compounds are different in the 3-year-old, 5-year-old, and 9- year-old plants, being higher in stems than in roots, except for eleutheroside E, which seems almost constant in both organs. The 3-year-old and 5-year-old plants are a clue for the better production of the active compounds than the 9-year-old plants. In the roots of A. senticosus, the content of isofraxidin was 100% higher in the 3-year-old than 5-year-old plants and 33.3% higher than in the 9-year-old. The content of rutin was 1101% higher in the 5-year-old plants than in the 3-year-old ones and 71.4% higher than in the 9-year-old. The content of hyperoside was 333% higher in the 5-year-old plants than in the 3-year-old and 160% higher than in the 9-year-old. The content of eleutheroside E in the 5-year-old plants was 11.0% higher than that in the 3-year-old and 66.0% higher than in the 9-year-old. The content of syringin was 41.7% higher in the 9-year-old plants than in the 5-year-old and 112.5% higher than in the 3-year-old. The content of kaempferol was 14.3% higher in the 9-year-old plants than in the 5-year-old and 166.7% higher than in the 3-year-old. The content of protocatechuate was 25.0% higher in the 9-year-old plants than in the 5-year-old and 66.7% higher than in the 3-year-old. In the stem of A. senticosus, the content of hyperoside was 28.5% higher in the 3-year-old plants than in the 5-year-old and 283.1% higher than in the 9-year-old. The content of eleutheroside E was 57.9% higher in the 3-year-old plants than in the 5-year-old and 87.5% higher than in the 9-year-old. The content of isofraxidin was 283% higher in the 3-year-old than in the 5-year-old and 143% higher than in the 9-year-old. The content of rutin was 265.7% higher in the 5-year-old plants than in the 3-year-old and 905% higher than in the 9-year-old. The content of kaempferol was 199.6% higher in the 5-year-old plants than in the 3-year-old and 211.6% higher than in the 9-year-old. The content of syringin was 133.3% higher in the 5-year-old plants than in the 3-year-old and 218.2% higher than in the 9-year-old. The content of protocatechuate was 300 times higher in the 5-year-old than in the 3-year-old and 666% higher in the 9-year-old.
Targeted Metabolomics Analysis of Phenolic Metabolites
The results of the hierarchical cluster analysis of 19 phenolic metabolites are shown in Figure 3. Accumulation of phenolic metabolites showed an apparent variation in their abundance affected by the different growth years. Based on their accumulation patterns, the 19 phenolic metabolites detected could be grouped into 3 clusters. Cluster 1 showed the highest accumulation in 3-year-old plants; 2 types of compounds accumulated in this cluster: C6C3-type (chlorogenic acid, p-coumaric acid, caffeic acid, p-hydroxycinnamic acid, cinnamic acid, ferulic acid) and C6C3C6-type (naringenin). Two types of compounds also occurred in cluster 2: C6C1-type (vanillic acid, syringic acid, salicylic acid) and C6C3C6-type (quercetin), with high accumulation detected in the 9-year-old plants. In cluster 3, there were 2 types of compounds: the C6C1-type (gallic acid) and C6C3C6 type (catechin, myricitrin, quercetin-3-O-rhamnoside, luteolin, petunidin, genistein, apigenin). The compounds of cluster 3 were accumulated in the 5-year-old plants. As shown in Figure 3, the accumulation of phenolic metabolites in ASR and ASS displayed significant organ-specific mode. ASS contained the most abundant phenolics grouped in clusters 1, 2, and 3, whereas ASR contained cluster 1 compounds.

Heat map visualization of phenolics in the different growth year of ASR and ASS. The color ranges from red to blue indicates relative abundance from high to low (color key scale right the heat map). Each sample was repeated three times. Y, year-old; r, roots; s, stems.
Differences in Target Metabolic Pathways of Above Ground and Underground Organs
To understand the organ specificity of these metabolites in 5-year-old plants, we enriched the metabolic pathways with 26 metabolites. The results revealed that 15 metabolites were involved in 4 metabolic pathways: flavonoid, flavone, flavonol, and phenylpropanoids. We visualized the network map of the metabolomics pathways based on their cumulative situation in the underground and above-ground tissues (Figure 4). We observed a difference in the 2 organs, with most compounds displaying different trends. Syringin takes part in the phenylpropanoid biosynthesis pathway and is mainly accumulated in the above-ground part. Some phenolic metabolites, such as p-hydroxycoumaric acid, protocatechuate, gallic acid, caffeic acid, salicylic acid, chlorogenic acid, apigenin, genistein, and quercetin, also had the same accumulation pattern. However, in underground tissues, abundantly accumulated compounds mainly involved the active ingredients kaempferol and rutin, and the phenolic metabolites naringenin, ferulic acid, and ferulic acid, and they separately take part in the biosynthesis pathways of flavonoid, flavone, flavanol, and phenylpropanoids.

The associated network map of metabolites in ASR and ASS. The circles and diamonds nodes represent different metabolites and related metabolic pathways. Red indicates high abundance, whereas low relative phenolic compounds are green. Phenylpropanoids biosynthesis, biosynthesis of phenylpropanoids, flavonoid biosynthesis, and flavone and flavanol biosynthesis are mutually different .
Correlation Analysis of the Active Compounds and Phenolic Metabolites
To study further the relationship between the detected metabolites, 26 metabolites were subjected to correlation assay. As shown in Figure 5, syringin showed a positive correlation within the C6C3C6-type compounds (naringenin, genistein, and apigenin; P < 0.05). In ASR, the C6C1-type compounds, gallic acid and syringic acid, displayed a negative correlation with the C6C3-type compounds, chlorogenic acid, caffeic acid, and p-coumaric acid, but, in ASS, a positive correlation with the C6C3-type compounds (P < 0.05). In ASR, isofraxidin displayed a positive correlation with p-coumaric acid and caffeic acid (C6C3-type compounds) and a negative correlation with the C6C3C6-type compounds genistein, apigenin, and luteolin; the tendency in ASS was the opposite. Eleutheroside E showed a negative correlation with the C6C1-type compounds, salicylic acid (P < 0.05) and syringic acid (P < 0.05), but in ASS, a negative correlation within the C6C3C6-type compounds myricetin, luteolin, and quercetin-3-O-rhamnoside (P < 0.05). Kaempferol displayed a negative correlation with chlorogenic acid (P < 0.05) and a positive correlation with gallic acid. Rutin and hyperoside positively correlated with genistein (P < 0.01), apigenin (P < 0.05), and gallic acid (P < 0.05), but a negative correlation with chlorogenic acid and caffeic acid. Protocatechuate displayed a negative correlation with C6C1-type compounds and positively correlated with the C6C1-type compounds syringic acid and salicylic acid, and the C6C3C6-type compound quercetin. These results were according to the information available in the network map of metabolites (Figure 5).
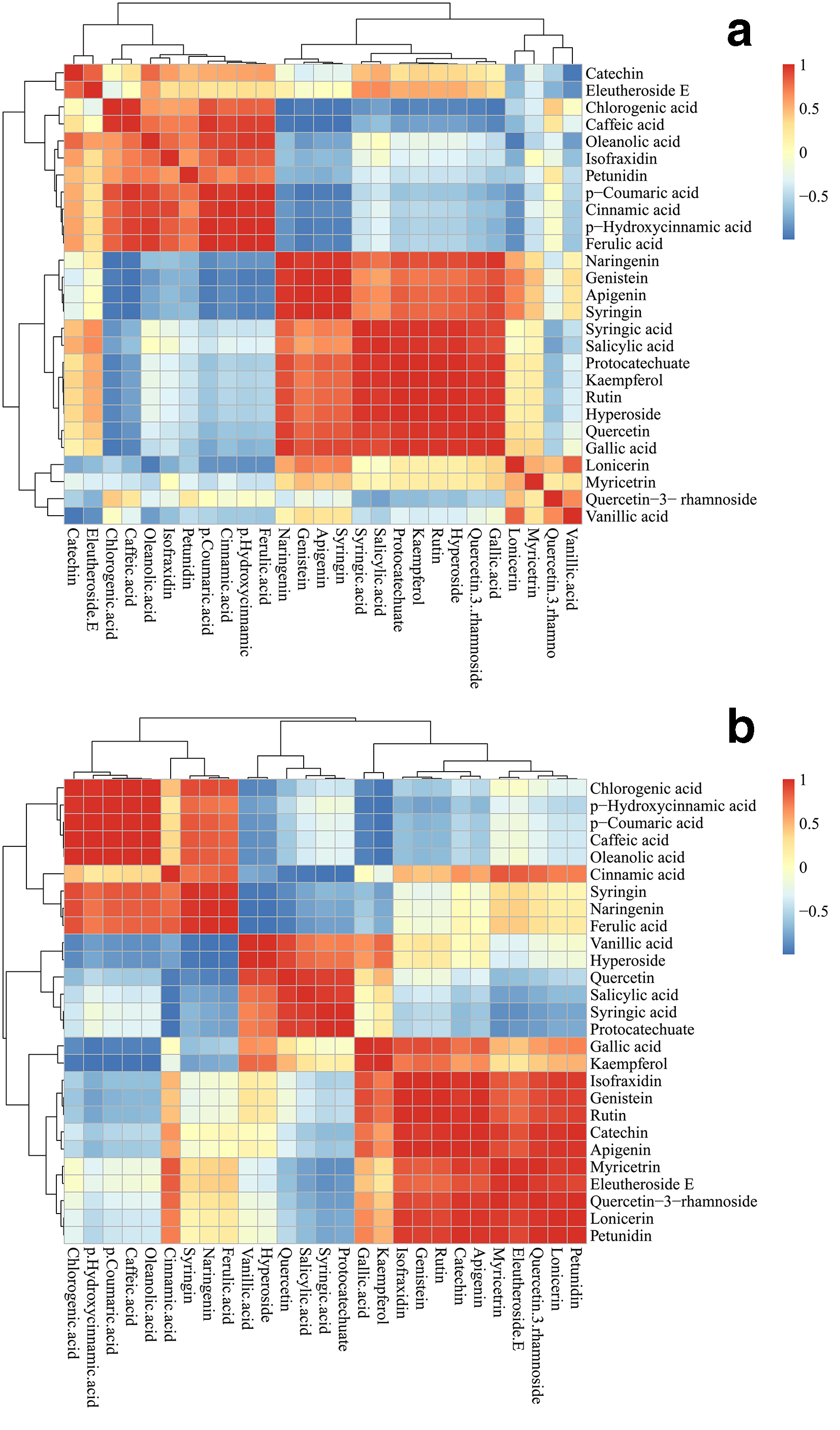
Correlation coefficients of the metabolites analyzed with 7 bioactive compounds and 19 phenolic compounds in a different part. Each square indicates r (Pearson’s correlation coefficient value for pairs of metabolites). The red color represents a positive correlation and the blue color represents a negative correlation. (a) ASR and (b) ASS.
Discussion
The influence of the growth year on the secondary metabolites of medicinal plants has always been a concern. The analysis of constituents of A. senticosus by UPLC-QTOF-MS was developed, and 131 compounds were identified in leaf extracts, but the growth year of the plant was not reported. 16 The metabolomics strategy was not only applied to distinguish the sexual type of A. senticosus but also was not used for the distinction between growth year. 27 In this study, we used a UPLC-MS method for the quantitative analysis of 7 active compounds in different growth years. The MS ion mode, MS parameters, and mobile phase of all analyses were optimized for higher MS sensitivity and better peak shape. After repetitive testing, we found that the positive ion mode and the methanol–water mobile phase gave the optimal conditions. Using these, we improved the speed and quality of detection (Figure 1).
In the root, the contents of rutin, kaempferol, hyperoside, and protocatechuate are meager, so they may not exert any pharmacological effect in this organ. The content of isofraxidin is higher in the 3-year-old plants than in the other growth years. We conclude that the roots of 3-year-old plants have anti-infection and anti-tumor effects. 14,33 The content of eleutheroside E is higher in 5-year-old roots. Therefore, we conclude that the 5-year-old roots have anti-infection, anti-depressant, anti-fatigue, and anti-diabetic effects. 13,34 The content of syringin is higher in the 9-year-old stems. Therefore, we conclude that the 9-year-old roots have immunoregulation, anti-diabetic, and anti-tumor effects. 9,12,35 In the stems of A. senticosus, the contents of hyperoside, isofraxidin, and eleutheroside E are higher in the 3-year-old plants. We conclude that the 3-year-old stems have anti-infection, anti-tumor, anti-oxidation, antidepressant, anti-fatigue, and anti-diabetic effects. 13,14,17,34,36 Rutin, kaempferol, syringin, and protocatechuate are the highest in 5-year-old stems, and we conclude that 5-year-old stems have anti-infection, anti-oxidation, immunoregulation, anti-tumor, and anti-diabetic effects. 9,18,19,37 The contents of the 7 active compounds in the 9-year-old stems were low, so from this, we conclude that the 9-year-old stems have some pharmacological effects, but the effects are weak. Our results showed that we could target these active marker compounds in different growth years during cultivation and extract them.
Phenolics are important plant secondary metabolites, displaying a tissue and growth year-specific distribution in the content and composition. 38 -40 Targeted analyses were performed to recognize 19 phenolic metabolites in our study. According to Figure 3, the C6C3C6-type metabolites were the most accumulated compounds, and they are mainly involved in the biosynthesis of flavonoids. 41 The results of the determination of 19 phenolic metabolites showed growth year-specific properties, with the C6C3-type metabolites mainly accumulated in 3-year-old plants, the C6C1-type in 9-year-old plants, and the C6C3C6-type in 5-year-old plants. p-Coumaric acid is said to have antioxidant, 42 anti-inflammatory, 43 and neuroprotective effects. 44 The results showed that p-coumaric acid accumulated mainly in the 3-year-old plants. A previous study found lonicerin, apigenin, and quercetin-3-O-rhamnoside to have high anti-cancer properties. 45 -47 Therefore, we speculate that 5-year-old A. senticosus has the above effects. Vanillic acid and syringic acid reduce the risk of stroke and myocardial infarction but are only present in 9-year-old plants. 48 When treating such diseases, 9-year-old A. senticosus can be selected as pharmaceutical material. However, this study only uses relative MS intensity for quantification, and further determination of the above metabolites is needed to confirm these findings.
During the creation of related network maps of metabolites in ASR and ASS, the accumulation patterns of 15 metabolites involved in the 4 metabolomics pathways in different growth years were obtained (Figure 4). The biosynthetic pathway of the C6C3C6-type metabolites results in the production of flavonoids and isoflavones. 21,49 However, C6C1-type metabolites may play essential roles, mainly as signaling molecules, such as benzoic acid derivatives and salicylic acid. 50 C6C3-type metabolites may originate in the phenylpropanoid biosynthesis pathway. 51 All compounds have different patterns of accumulation in ASR and ASS. According to this differentiation, we can provide an alternative way to identify ASR and ASS, by the accumulation of target metabolites between the aboveground and underground organs of 5-year-old plants. In this study, only targeted metabolomics research strategies were used to analyze A. senticosus in different growth years, and the study has certain limitations. In the future, non-targeted metabolomics can be used for further research.
Combined with the metabolomics network map and metabolites correlation analysis, we can conclude that the correlation between compounds may affect the accumulation of them in different medicinal organs. 50 The C6C3-type metabolites, namely chlorogenic acid, caffeic acid, p-coumaric acid, and p-hydroxycinnamic acid, are reported to be grouped with salicylic acid. 21 Syringin showed a negative correlation with them in the roots, and they had a significant correlation; syringin had a significant positive correlation with the C6C3-type metabolites in the aboveground organs (Figure 5). We speculate that the C6C3-type metabolites have different regulatory mechanisms for the accumulation of syringin in the aboveground and underground organs in the different growth years, which needs further study. Kaempferol, rutin hyperoside, and protocatechuate showed a positive correlation with C6C1 and C6C3C6- type metabolites in 2 medicinal organs in different growth years. These phenolic metabolites, as reactive oxygen scavengers, have important functions in plant development and defense. 52,53 These results show that the crucial components might be different in different plant organs. These results indicate that the target secondary metabolic pathways affecting the accumulation of active components are regulated differently in different plant organs.
Conclusion
In summary, the active compounds in A. senticosus have higher accumulation in the roots and stems of 5-year-old plants than in other growth years. The accumulation of phenolic metabolites was significantly different in different growth years and organs. Five-year-old stems are the most abundant resource for the accumulation of phenolic metabolites. We propose that ASR and ASS can be more efficiently used separately for different objectives considering the difference in abundance of their specific active compounds.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are thankful for the financial support of the National Key Research and Development Program of China (No. 2016YFC0500303); Heilongjiang Province Foundation for the National Key Research and Development Program of China (No. GX17C006); Post-Doctoral Foundation of Heilongjiang Province of China (No. LBH-Z17208); Key Project of Heilongjiang Provincial Administration of Traditional Chinese Medicine (No. 2018-009).
