Abstract
Presented is a series of narrative reviews that summarize published information regarding the effect or potential effect of interfering substances on the accuracy of continuous glucose monitoring (CGM) devices. While drawing together what is currently known regarding this topic, the future direction in this field and clinical implications posed by polypharmacy on CGM performance are considered. This second in a series of review articles introduces the glucose-sensing designs of commercially available continuous glucose monitors (CGMs) and describes those interfering substances currently or historically identified within manufacturer’s labeling for a given CGM model. Where known, an overview of the mechanistic effects of labeled interfering substances on the corresponding CGM designs is also described, as are sensor design approaches introduced by leading manufacturers to reduce interference effects. The review also considers: the effect of interferents on market-leading minimally invasive CGMs; design changes made to limit the flux of interfering substances to the sensor’s glucose-sensing element; and an appraisal of the minimally invasive implantable Senseonics Eversense system, which employs a unique synthetic glucose-recognition ligand coupled to optical signal generation, which correspondingly gives rise to a unique interference profile.
Keywords
Introduction
Manufacturers continuously strive to improve continuous glucose monitoring (CGM) design in terms of lower manufacturing costs, promoting sustainability and improving the user experience, this evolution being driven by more stringent regulatory requirements, stakeholder feedback and a competitive marketplace. Correspondingly, newer generations of CGMs have incorporated additional design features to reduce the effect of interfering substances and further improve system accuracy. For example, the Dexcom G6 (G6) and Dexcom G7 (G7) CGMs incorporate design improvements aimed at reducing the interfering effects of acetaminophen, while the recently approved FreeStyle Libre (FSL) 2 Plus and FSL3 Plus systems are less susceptible to ascorbic acid compared to earlier models. Table 1 summarizes interfering substance labelling for the more widely distributed CGMs, as identified from user guides and manufacturer’s online resources. The challenges in determining levels of interfering substances in interstitial fluid (ISF) are recognized by manufacturers, with some labeling acknowledging that potential interferent levels in ISF are unknown compared to circulating blood. 1
Labeling and Claims Information for Current Widely Distributed CGMs.
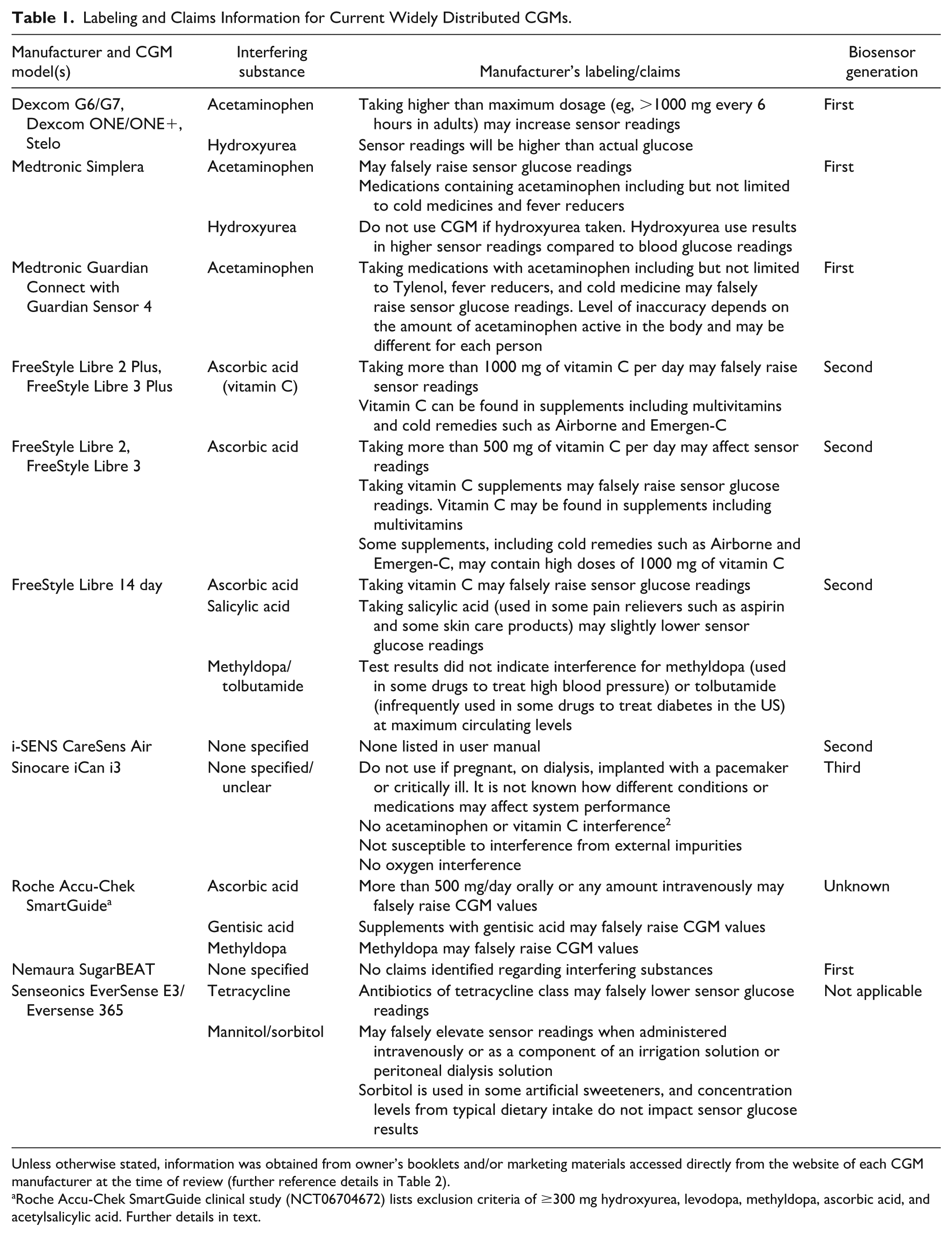
Unless otherwise stated, information was obtained from owner’s booklets and/or marketing materials accessed directly from the website of each CGM manufacturer at the time of review (further reference details in Table 2).
Roche Accu-Chek SmartGuide clinical study (NCT06704672) lists exclusion criteria of ≥300 mg hydroxyurea, levodopa, methyldopa, ascorbic acid, and acetylsalicylic acid. Further details in text.
An uncritical review of this table may suggest that certain CGM devices are less influenced by interfering substances than others. However, published literature has, by definition, greater focus on those systems that have been most commercially successful and have been in market longest, albeit with modifications to the glucose-sensing design. By default, less information has been published regarding newer systems, such as Sinocare’s CGM design, which correspondingly may appear less prone to interfering substances to the casual observer. This again reinforces the need for regulatory guidance to ensure adequate and appropriate interfering substance evaluations are performed and the results made publicly available.
A Note on Electrochemical Biosensor Classification
A commonly used convention is to classify electrochemical biosensor designs into one of the four generational types. First-generation systems employ oxygen, naturally dissolved within the test sample that effectively shuttles electrons between the active site of the enzyme, used as the glucose-recognition agent, and electrode surface. Example CGM designs include those manufactured by Dexcom Inc. (San Diego, California) and Medtronic Inc (Minneapolis, Minnesota). Second-generation systems employ an artificial mediator species in place of oxygen, allowing reduced operating potentials to be applied. Example CGM designs include the FSL CGMs manufactured by Abbott Diabetes Care (Alameda, California). Third-generation systems are engineered to facilitate direct electron transfer from the embedded enzyme cofactor directly to the electrode surface. The Sinocare iCan i3 CGM system (Changsha Sinocare Inc, Changsha, China) is an example of this design. There are currently no examples of CGM designs based on the fourth-generation electrochemical biosensor principle, in which the system is engineered to measure the direct interaction between the analyte and electrode. This classification approach is described in more detail in Part 1 of this review series.
Perhaps confusingly, CGM manufacturers may refer to their own CGM models as being of different generational types. For example, Dexcom’s G7 system is sometimes referred to as Dexcom’s seventh-generation CGM model. Correspondingly, the articles in this review series refer to “generational” systems in terms of electrochemical biosensor classification, while referring to CGM systems of multi-generational design by their marketed name only (eg, G7 or FSL2).
Interfering Substances and Continuous Glucose Monitors of First-Generation Electrochemical Biosensor Design
Dexcom Continuous Glucose Monitoring Designs (First-Generation Electrochemical Biosensor)
Dexcom has released a series of CGM models to market, the latest being the G6 and G7 models. These are understood to have the same fundamental glucose-sensing design but have differing form-factors and differentiated claims. Other recently launched models from Dexcom include the Dexcom ONE device, a derivative of the G6 system, and the Dexcom ONE+ and Stelo devices, both derivatives of the G7 system. 3
Dexcom designs incorporate a wire working electrode containing platinum, around which is coiled a silver/silver chloride (Ag/AgCl) reference/counter electrode, separated by an intervening insulation layer. Device design evolution has resulted in a series of membranes (which Dexcom frequently refers to as “domains”) formed around the working electrode and which may include: (i) an electrode/electrolyte membrane to provide optimal measurement conditions at the working electrode surface; (ii) an interference membrane to reduce the passage of interfering species to the working electrode; (iii) an enzyme-containing membrane that localizes GOx enzyme; (iv) a diffusion resistance membrane to control the flux of glucose relative to oxygen; and (v) a bioprotective membrane, which, in addition to its biocompatibility and anti-biofouling properties, may also influence the passage of potentially interfering species into the underlying sensing assembly. 4 While each of these membranes may influence the passage of species from the surrounding ISF to the electrode surface, it is reasonable to suppose that the interference and bioprotective membranes are more specifically configured to reduce the effect of interfering species.
Given that all Dexcom models have the same sensing principle, this review will focus on the newer G6 and G7 systems. The sensing elements of these designs differ from earlier Dexcom CGM models through introduction of a permselective membrane, designed to reduce the effect of certain interfering substances that may be present in the sample matrix. 5 Figure 1 provides a general schematic of a first-generation electrochemical CGM design.
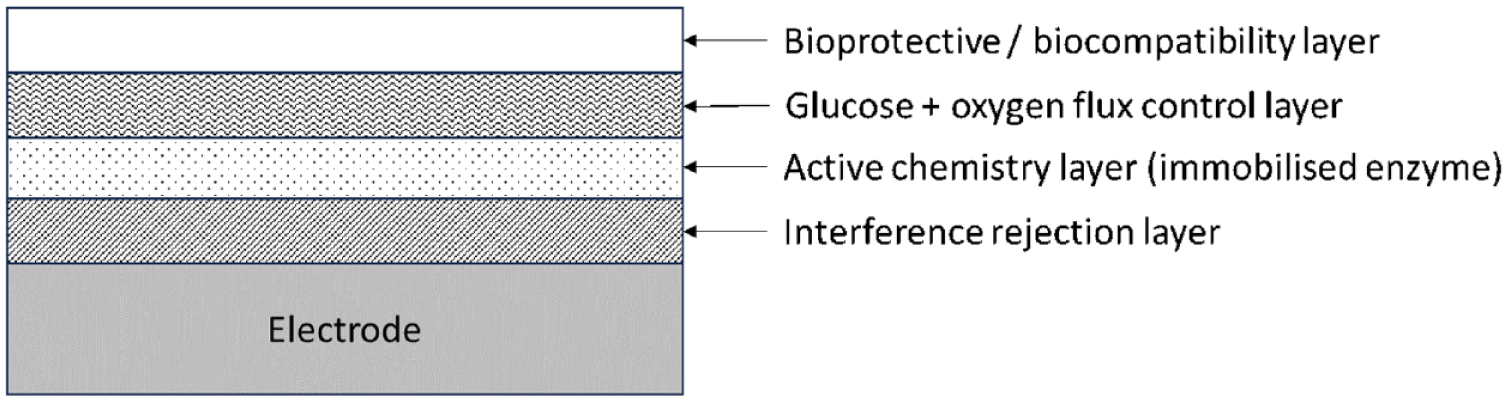
Generalized schematic of a first-generation CGM system. Typically, such systems will include an interference rejection layer between the active chemistry layer and electrode. The flux of glucose and oxygen is controlled by a further layer, that, in turn, is covered by a bioprotective layer. Other layers may be present, depending on the specific CGM model design.
Medtronic Continuous Glucose Monitoring Designs (First-Generation Electrochemical Biosensor)
The Medtronic series of CGMs include the more recently introduced models incorporating the Guardian Sensor 3, Guardian Sensor 4, or Simplera (DS5) sensor. These sensors incorporate a three-electrode system composed of separate working and counter electrodes formed from a platinum derivative and an Ag/AgCl reference electrode, deposited onto a suitable planar substrate. Similarly to Dexcom’s family of CGMs, Medtronic sensor models have evolved to incorporate multiple membrane layers, 4 which may include (i) an interference rejection membrane formulated to reduce the passage of low molecular weight charged species to the working electrode; (ii) an analyte sensing layer incorporating a cross-linked GOx enzyme; (iii) an adhesion promoting layer; (iv) a diffusion layer (“analyte modulating layer”) that promotes oxygen permeability while limiting glucose flux; and (v) a protective layer that provides a suitably porous biocompatible interface. The adhesion promoting layer supports integration of the analyte sensing layer to the analyte modulating layer. Medtronic patent application US2022/0047193A1 describes interference rejection membranes based on poly(vinyl alcohol) crosslinked with (di)carboxylic acid functionalities, 6 while patent application US2011/0319734A1 describes membranes formed from crosslinked primary amine and crosslinked polymethacrylate polymers. 7
Acetaminophen Interference in First-Generation Continuous Glucose Monitoring Models
The commonly taken analgesic, acetaminophen (variously paracetamol, N-acetyl-para-aminophenol, APAP), consists of a hydroxylated six-carbon aromatic ring (Figure 2). It readily undergoes electrooxidation at moderate potentials in aqueous solutions at physiological pH where it exists in a largely non-ionized state.8,9 Acetaminophen causes interference in first-generation electrochemical CGMs due to the relatively high operating potentials required to oxidize hydrogen peroxide, thus establishing a need for mitigating strategies to reduce the non-glucose signal. 10
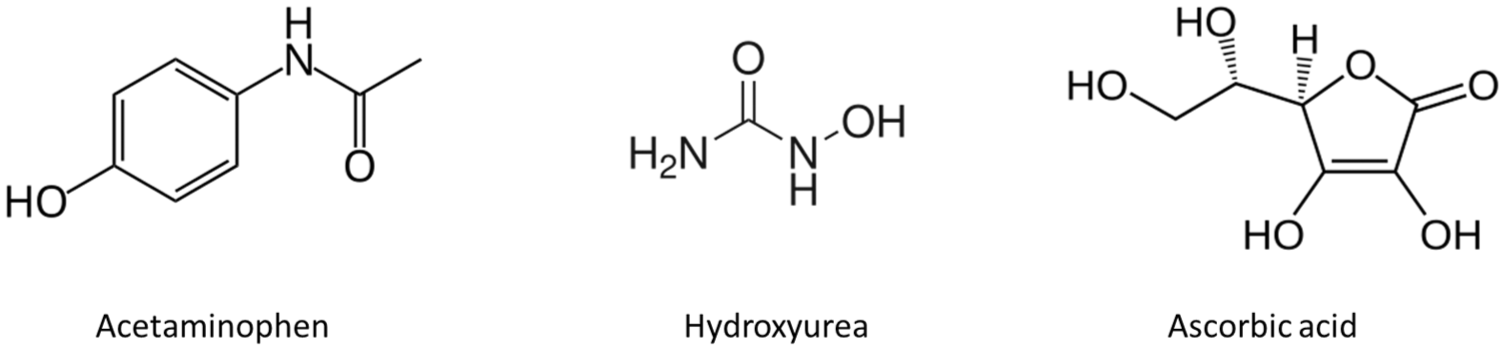
Chemical structures of acetaminophen and hydroxyurea, interferents within CGMs of first-generation biosensor design and ascorbic acid, a notable interferent in a commercial CGM of the second-generation biosensor design.
The in vivo studies of Basu et al. on the Dexcom Seven Plus and G4 systems and the Medtronic Guardian REAL-Time and Sof-Sensor CGMs found interfering effects in all systems following oral acetaminophen administration.11,12 Similar results were found by Maahs et al. in a 40-subject outpatient study using the G4 system. 13 Medtronic’s recent CGM device, the Simplera DS5 sensor, also contains a warning that users should consult a health care professional before using glucose readings to make treatment decisions when taking medications containing acetaminophen, due to risk of falsely elevated readings. 14 Heinemann references a statement made at the 2021 annual meeting of the American Diabetes Association (ADA) in which the Guardian 4 CGM sensor was described as not being compatible with acetaminophen. 15 While earlier generation Dexcom CGMs were contraindicated against acetaminophen, G6 and G7 labeling defines the maximum dosage that can be tolerated by the systems within a fixed time-period. 16
Dexcom patents refer to “interference domains” and “bioprotective domains,” membranes designed to limit the flux of acetaminophen to the sensing element, along with other potential interfering substances. Dexcom patent US 9572523 describes several membrane configurations, including those containing polyurethane-based polymers with hydrophilic segments which, when challenged in vitro with acetaminophen and other potential interfering substances, yielded a moderated interfering effect. 17 An approximate 4-fold reduction in the current response was obtained from the sensor incorporating the test membrane design compared to the non-modified control sensor when challenged with a 1000 mg oral acetaminophen dose. Dexcom patent US9549692 also describes interference membrane formulations designed to reduce effects from acetaminophen and other non-glucose substances based on polyelectrolyte layers containing polycationic and polyanionic motifs. 18
The Food and Drug Administration’s (FDA) decision summary response to Dexcom’s De Novo request for the G6 device includes a description of a design aimed at reducing interference from acetaminophen and also a 65-subject study in which the accuracy of the device, as measured against a laboratory comparator method, was assessed 60 minutes either side of a single oral dose of 1000 mg acetaminophen. 19 This dosage was not considered to introduce significant interference to the device, since only a 3% decrease in the percentage of CGM readings within ±15 mg/dL or ±15% of reference and a 1% increase in mean absolute relative difference (MARD) was observed between the pre- and post-dose values. The mean maximum interfering effect of 5.2 mg/dL was not considered statistically significant. However, in vitro tests, in which 60 mg/dL glucose test solutions were spiked with 6.5 mg/dL and 20 mg/dL acetaminophen yielded glucose biases of 22 mg/dL and up to 70 mg/dL, respectively. It was concluded that, while “standard dosing of acetaminophen with physiologic concentrations up to ~2 mg/dL does not appear to cause significant bias, higher supra-therapeutic levels appear to lead to clinically significant bias. Therefore, although the contraindication to acetaminophen may be acceptable for standard doses of acetaminophen, higher supra-therapeutic (> ~2-3 times therapeutic) levels showed significant bias.”
These observations are consistent with Calhoun et al., who reported a significant reduction in interference for subjects consuming 1000 mg of acetaminophen in a Dexcom-supported clinical study incorporating a then-investigational G6 system featuring a newly introduced permselective membrane. 20 In an Abbott-funded response to this study, Denham proposed that G6 effectiveness should be assessed against repeated doses of acetaminophen and conducted a clinical study in which 1000 mg of acetaminophen was administered on a 4-hour basis for three doses to 14 subjects wearing a G6 system. A progressively greater bias from baseline was observed, leading to a conclusion that membranes may fail to effectively block acetaminophen over repeated doses. While the magnitude of the deviation would have a minimal effect on insulin dosing, biases were of sufficient magnitude to potentially miss clinically significant hypoglycemia. The G7 clinical accuracy study of Garg et al. states that the G6 and G7 systems are not susceptible to interference by acetaminophen, from which one may presume that these systems meet FDA requirements regarding the acceptance criteria that define a non-interfering effect. 21 It may be noted that certain acetaminophen metabolites exhibit a catechol moiety and thus are readily electro-oxidized, as will be described in Part 3 of this series.
Hydroxyurea Interference in First-Generation Continuous Glucose Monitoring Models
Hydroxyurea (hydroxycarbamide; Figure 2) is an antineoplastic agent used to treat several health conditions, including cancer and sickle-cell disease. Both the Dexcom and Medtronic CGM systems list hydroxyurea as an interferent. The electrochemistry of hydroxyurea under physiological pH conditions at unmodified electrodes elicits significant electroactive responses at operating potentials lower than those employed by the first-generation electrochemical CGMs.22-24 Hydroxyurea’s interfering behavior was identified through post-market surveillance by Szmuilowicz and Aleppo, based on observations of higher-than-expected glucose readings in a patient wearing both a Dexcom G5 and a G6 CGM, behavior that was not seen in a concurrently worn FSL2 CGM. 25
The in vitro interfering substance screening studies of Pfützner recorded an expected interfering effect from hydroxyurea on the G6 and G7 devices.26-28 No such interfering behavior was observed on the FSL2 and FSL3 systems, 29 this initial in vitro observation being consistent with the in vivo findings of Szmuilowicz and Aleppo.
Ascorbic Acid Interference in First-Generation Continuous Glucose Monitoring Models
While not a labeled interferent for first-generation electrochemical CGM systems, ascorbic acid (Figure 2) is found in many foods and is a strong reducing agent with a ubiquitous presence in blood and ISF, where it exists in the form of the ascorbate anion at physiological pH. 30 The electrooxidation of ascorbic acid at platinum and other electrodes may occur at potentials significantly below those employed in first-generation biosensors and can foul electrode surfaces.31,32
Membranes aimed at reducing acetaminophen flux to the working electrode may similarly account for the relative insensitivity of the G6 and G7 systems to ascorbic acid. Dexcom patent US9572523 describes a membrane coated onto test sensors and challenged in vitro with ascorbic acid, in which a calibrated “enzyme-electrode” featuring a novel membrane formulation was immersed in a solution containing a test concentration of 6 mg/dL ascorbic acid. 17 The resulting signal was monitored and used to calculate an equivalent estimated glucose signal of 17 mg/dL. Dexcom patent US7651596 similarly describes reagent-coated enzyme electrodes featuring semi-permeable membranes where only small glucose equivalent signals were obtained when challenged with higher than therapeutic levels of ascorbic acid. 33
While the direct electrooxidation of ascorbic acid can lead to a non-glucose-specific increase in signal in first-generation electrochemical CGMs, Doherty observed that ascorbic acid detection at conventional electrode surfaces can result in rapid and severe electrode passivation, leading to a deterioration in electrode sensitivity. 34 This phenomenon may have particular importance in the performance of CGM devices, given they are intended for multi-day use, and so may be prone to cumulative effects caused by repeat exposure to such substances. The inclusion of membranes designed to inhibit the transport of ascorbic acid and similar potentially passivating species to the electrode interface is therefore a beneficial design feature.
In addition to the electrode-mediated oxidation of ascorbic acid, there is also evidence that ascorbic acid may also undergo oxidation in the presence of hydrogen peroxide, the main signal-generating agent within first-generation CGM devices. This reaction has been much studied and is of considerable complexity.35-37 While the effect of this interaction on first-generation electrochemical CGM accuracy is undetermined, it is to be appreciated that hydrogen peroxide is generated within the active chemistry region of first-generation CGM designs. However, only a portion of this product will diffuse through the adjacent electrode/electrolyte membrane to the working electrode surface, since this membrane has been configured to support the flux of low molecular weight hydrogen peroxide while limiting the flux of larger molecular weight species, including potential interferents. Therefore, given the physical distance of the interference rejection membrane from both the zone of hydrogen peroxide evolution and the electrode interface, the extent of the interaction between hydrogen peroxide and ascorbic acid in the sample matrix may be minimal.
Interfering Substances and Continuous Glucose Monitors of Second-Generation Electrochemical Biosensor Design
FreeStyle Libre Continuous Glucose Monitoring Designs (Second-Generation Electrochemical Biosensor)
The most commercially successful second-generation electrochemical CGMs to date have been the FSL series from Abbott. Models include the FSL 14-day, FSL2, FSL3, FSL2 Plus, FSL3 Plus, and FSL Select, all intended for people with diabetes who use insulin; FSL Rio, intended for people with diabetes who are not using insulin; and FSL Lingo, for people without a diabetes diagnosis for general health and wellness purposes and not intended for treatment of medical conditions.
The FSL system sensing filament is formed from a plastic substrate overlaid with a screen-printed carbon three-electrode assembly and an Ag/AgCl reference electrode. 4 The working electrode is coated with a membrane containing a polymeric mediator and GOx enzyme complex.38,39 The mediator contains an osmium redox center (Os2+/Os3+) within a surrounding biimidazole, bipyridyl, or pyridylimidazole complex tethered to a polyvinyl-based backbone and further formulated within a polymer hydrogel containing the cross-linked enzyme.4,38-41 This “wired-enzyme” formulation serves to constrain the mediator and enzyme within the vicinity of the electrode interface while facilitating the shuttling of electrons from the active site of the enzyme via the flexibly tethered osmium redox centers to the electrode surface without the need for oxygen as a co-reactant.42-46
Hoss and Budiman 47 report the operating potential of the FSL working electrode to be +40 mV vs the reference (Ag/AgCl) electrode, compared to ~600 mV as reported for the first-generation CGM systems.48,49 Das has commented that this low operating potential has eliminated the requirement for a complex interferent rejection membrane within the FSL designs. 4 Hoss states that the most critical elements requiring control during the manufacturing process are the enzyme-sensing layer, which is located adjacent to the sensing electrode, and the overlying glucose-limiting membrane. Given that the wired-enzyme technology removes the oxygen dependency inherent in first-generation CGMs, there is no requirement for membranes that promote oxygen flux to the sensing chemistry. A key design requirement of the glucose flux membrane is to reproducibly control the transfer of glucose from the surrounding ISF to the immobilized GOx enzyme while achieving optimum system linearity and dynamic range.50,51 This outer membrane has also been formulated with hydrophilic moieties to provide a suitably biocompatible interface with the surrounding tissue.4,40
Current labeling for FSL systems acknowledges that ascorbic acid can falsely raise sensor glucose readings; thus, labeling for FSL2/FSL3 and FSL2 Plus/FSL3 Plus include limitations regarding maximum doses of ascorbic acid per day (Table 1). 1 Labeling also states that salicylic acid may slightly lower readings, with the level of inaccuracy being a function of the quantity consumed. Specific reference is also made to the medications methyldopa and tolbutamide, but with neither meeting the criteria required for listing as interferents, when present at maximum therapeutic levels. The FSL labeling also recognizes that concentrations of many potential interferents in ISF remain unknown in comparison to circulating blood. Figure 3 provides a general schematic of a second-generation electrochemical CGM design.
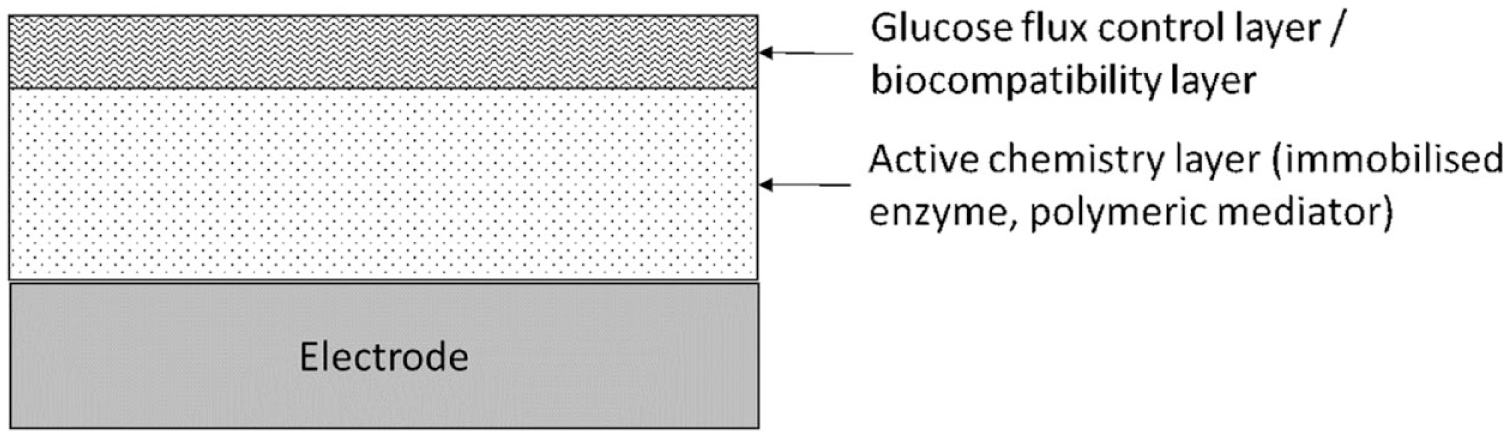
Generalized schematic of a second-generation CGM system. Such systems will include an active chemistry layer in which enzyme and mediator are co-immobilized to facilitate electron transfer from the active site of the enzyme to the electrode surface. The flux of glucose is controlled by a further layer that also serves a biocompatibility role.
Ascorbic acid interference in FreeStyle Libre models
In an online discussion at the 2022 Diabetes Technology Society meeting, senior Abbott development personnel stated that improved membrane formulations were being used to overcome ascorbic acid interference in their continuous ketone device, which is reported as being based on the FSL system design. 52 Shortly thereafter, in March 2023, Abbott received US FDA clearance for FSL2 Plus and FSL3 Plus (modified versions of FSL2 and FSL3 with the removal of the automated insulin delivery contraindication that had applied to earlier FSL systems and an increased wear time from 14 to 15 days). One may speculate that this claimed improvement in performance was achieved through use of reformulated membrane designs.
Despite these improvements, an underlying sensitivity to ascorbic acid remains in the FSL design. The FDA decision summary K222447 describes a clinical study in which the performance of a modified FSL2 system was assessed in subjects with diabetes while ingesting 1000 mg doses of ascorbic acid. 53 A maximum average interference bias of 5.1 mg/dL was recorded following a single ascorbic acid dose, rising to 9.2 mg/dL and 9.0 mg/dL after a second and third dose, respectively, given at 4-hour intervals. This modified system is believed to be that deployed within the FSL2 Plus and FSL3 Plus systems, based on the purpose for submission listed in the K222447 document described as “modification to the sensor tail to reduce interference from ascorbic acid and to remove the contraindication against the use within automated insulin dosing (AID systems).”
Evidence suggests that this interfering effect is linked to the FSL mediator design. Contemporaneously to Heller and co-workers45,54-56 who were developing the FSL osmium-based wired-enzyme technology in the mid-1990s, Doherty et al. were investigating the electrocatalytic behavior of ascorbic acid at glassy carbon electrodes coated with osmium redox polymers similar to those used in the FSL design (an osmium redox center complexed to biimidazole, bipyridyl, or pyridylimidazole ligands and tethered to a derivatized polyvinyl backbone). 34 Doherty et al. found osmium redox films to be efficient electrocatalysts for ascorbic acid, readily facilitating the transfer of electrons from the compound, via the mediator, to the electrode at relatively modest potentials (Figure 4). A pH-dependent ascorbic acid oxidation current was recorded at a half-wave potential of +250 mV vs a saturated calomel electrode reference, significantly lower than the corresponding half-wave potential of ~+800 mV obtained on non-osmium modified control electrodes. The researchers successfully constructed and characterized an ascorbic acid sensor based on this design.
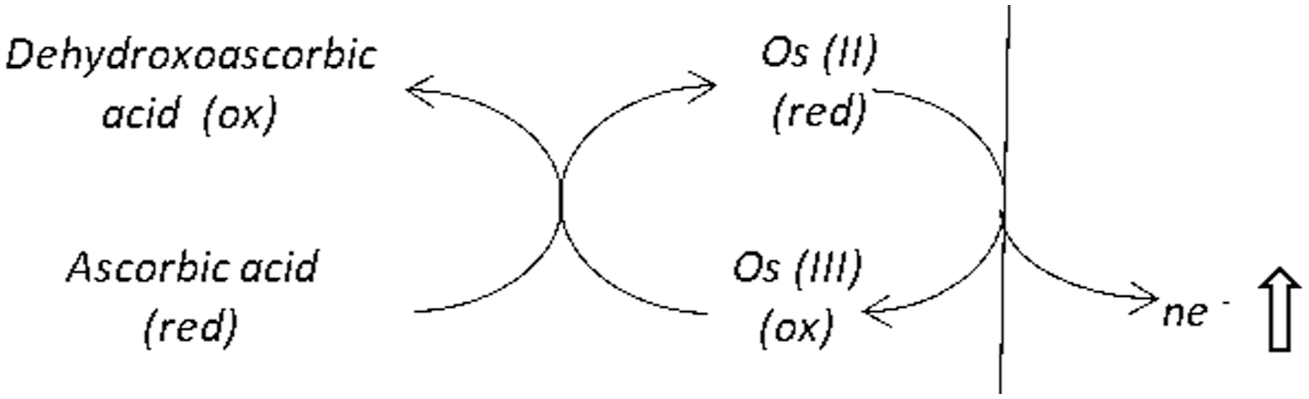
Schematic of direct electron transfer from ascorbic acid to an electrode via an osmium mediator. The reduced form of osmium mediator undergoes oxidation at a suitably poised electrode to yield an additional, non-glucose-specific current.
Doherty acknowledged that analytical sensor designs featuring osmium redox polymers as mediators coupled to analyte-specific enzymes offered benefits including low redox potentials, rapid charge transfer characteristics, synthetic methodologies, and stability, but cautioned that sensor performance may be potentially compromised by the interfering effect of ascorbic acid. The authors considered the use of permselective membranes to be an inadequate design solution due to the risk of incomplete exclusion of interfering substances and reduced mass-transport rates, leading to extended response times. They proposed a means of differentiating an ascorbic acid interferent response from a second analyte-specific response by means of a dual working electrode design coupled to an examination of differences in kinetic reaction rates at the working electrodes. While current FSL systems operate at a fixed potential of +40 mV (vs Ag/AgCl reference electrode), lower than the osmium complexes studied by Doherty, all FSL models to date include labeling regarding the maximum permissible intake of ascorbic acid per day.
In addition to ascorbic acid, the early work of Ohara and Heller suggests that other reducing agents, including reduced glutathione, L-cysteine, and uric acid, may also contribute to the electrochemical responses of osmium mediator electrodes, although they considered that these effects would be negligible at physiologic levels. 45 However, Fei and co-workers57,58 have reported strong electrocatalytic activity toward both uric acid and epinephrine, a catecholamine, for electrodes modified with osmium redox polymers of a similar design to Ohara and Heller.
Interfering Substances and Continuous Glucose Monitors of Third-Generation Electrochemical Biosensor Design
Sinocare iCan i3 (Third-Generation Electrochemical Biosensor)
There is little information publicly available regarding the effect of common interfering substances on the third-generation Sinocare iCan i3 system. Since the transduction methodology does not require a mediator, whether oxygen or artificial, mediator-related interference mechanisms are not a consideration. However, electrochemical interference effects may remain a factor given that an operating potential is still required to facilitate electron transfer from the enzyme active site to the electrode interface.
The manufacturer claims their product to be free of acetaminophen and ascorbic acid interference, with supporting data on file. They further claim that the system is “not susceptible to interference from external impurities.” However, the user guide states that the system should not be used if pregnant, on dialysis, implanted with a pacemaker or critically ill, since “It is not known how different conditions or medications common to these populations may affect performance of the system. iCan i3 CGM results may be inaccurate in these populations.” A clinical assessment of the iCan i3 system by Brazg included ascorbic acid, acetaminophen, and exercise challenges, with no interfering effects being reported and a claimed MARD of 10.5%. 59
Interfering Substances and Implantable Continuous Glucose Monitors
The sensing mechanism behind the Eversense CGM models (Senseonics Inc, Germantown, Maryland) is based on fluorescence, with glucose recognition achieved through selective, reversible binding of glucose to boronic acid derivatives within the hydrogel encasing the sensing unit. Conformational changes due to glucose binding are recorded as proportional fluorescent signals upon excitation by an LED light source within the sensor capsule (Figure 5). The hydrogel layer is covered with a proprietary porous membrane that serves to enhance biocompatibility within the adjacent subcutaneous tissue. 60 The sensor further incorporates a silicone collar that contains the anti-inflammatory steroid drug dexamethasone acetate that elutes locally to reduce tissue inflammation around the sensor. The device is described as operating within ISF. 61
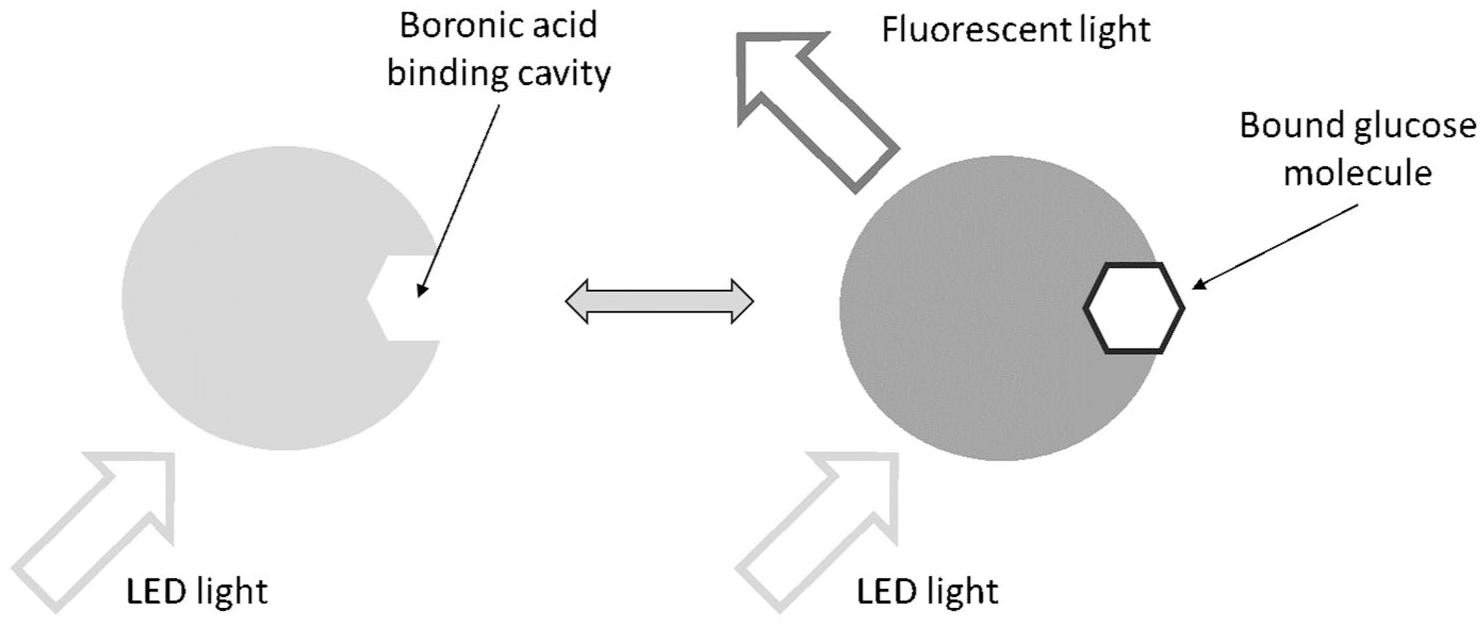
Glucose-sensing principle of the Senseonics Eversense CGM models.
The Eversense systems carry statements regarding the potential for the antibiotic, tetracycline, and the sugar alcohols, mannitol, and sorbitol (Figure 6), to interfere with sensor glucose readings, with in vitro data reported in the Senseonics-funded laboratory study of Lorenz et al. 61
Chemical structures of tetracycline, mannitol, and sorbitol as labeled for the Senseonics Eversense CGM models.
Tetracycline, by virtue of its fluorescent properties, interferes with the optical transduction methodology employed within the Eversense systems. Lorenz et al. performed laboratory testing using surrogate ISF solutions spiked with tetracycline across a therapeutically relevant range and conducted a regression analysis to confirm a tetracycline interfering effect, as defined by ISO 15197:2013 interfering substance acceptance criteria for blood glucose monitor (BGM) systems.61,62
The polyols mannitol and sorbitol, reduction products of the aldohexoses mannose and glucose, respectively, have medical applications but are also added to foods as alternatives to sugars to impart a sweet taste. Based on in vitro data, Lorenz considered mannitol to be non-interfering if used as a sweetener, but with the potential to significantly increase glucose readings when administered at higher concentrations to subjects undergoing intravenous administration, irrigation or peritoneal dialysis. 63 Similarly, while sorbitol was not found to interfere under the test conditions employed, it was again considered that elevated glucose readings could be recorded in patients receiving medical treatments containing this substance. 64
Lorenz also observed a notable negative bias (−24%) for ribose, an endogenous aldopentose sugar, although testing was conducted at a concentration ~4-fold greater than the maximum physiologic concentration. This decreased bias was considered indicative of ribose competition with glucose for boronic acid binding sites. Regression analysis demonstrated this interfering effect to be non-clinically significant under physiological concentrations. Decreased biases were also recorded for other sugar species, including xylose, also an aldopentose (−9.1% bias), and fructose, a ketohexose (8.8% bias).
Interfering Substances and the Accu-Chek SmartGuide Continuous Glucose Monitor
Design details of the newly launched Accu-Chek SmartGuide system have not been publicly disclosed. As indicated in Table 1, the device is labeled with respect to the usage of ascorbic acid, methyldopa, and gentisic acid. Furthermore, a randomized clinical trial study design using the Accu-Chek SmartGuide CGM has been recently posted by Roche Diagnostics on the clinicaltrials.gov website lists exclusion criteria for the ingestion ≥300 mg quantities of hydroxyurea, levodopa, methyldopa, ascorbic acid, or acetylsalicylic acid, which has not stopped at least 30 days prior to screening. 65 Since methyldopa and gentisic acid have also been associated with influencing CGM readings in other CGM designs, these substances will be further reviewed in Part 3.
Summary
The leading manufacturers have made modifications to their device designs by introduction of permselective membranes to reduce the effect of interferences from certain substances, most notably acetaminophen and ascorbic acid. Such membranes will also act to reduce the flux of other potentially interfering compounds with similar chemical or physical properties. However, such membranes must have exacting requirements beyond reducing the flux of interfering compounds to the sensing element, including promoting adequate flux of glucose to the glucose-recognition agent. Additional membrane layers will, at some level, function as inhibitors to the diffusion of glucose from the surrounding ISF and thus can reduce device sensitivity. Furthermore, their formation must be precisely controlled to minimize sensor-to-sensor variation.
The chemical substances identified as possible interferents within currently or previously widely available CGM systems, as recorded in manufacturers’ labeling, as reviewed in this review, are summarized in Table 2. This table necessarily presents an unbalanced view, in that the CGM interference profiles of the newer systems have not been scrutinized to the same extent as the more established systems. Furthermore, interpretation of published literature is challenging in that little is known regarding the levels of potentially interfering substances in ISF and the fact that no clear guidelines exist regarding recommended test concentrations of potentially interfering substances in ISF. This, in turn, has led to individual investigators conducting CGM interference studies at non-standardized interferent concentrations.
Chemical Substances That Have Been Identified as Interferents Within Currently or Previously Commercially Available CGM Systems.
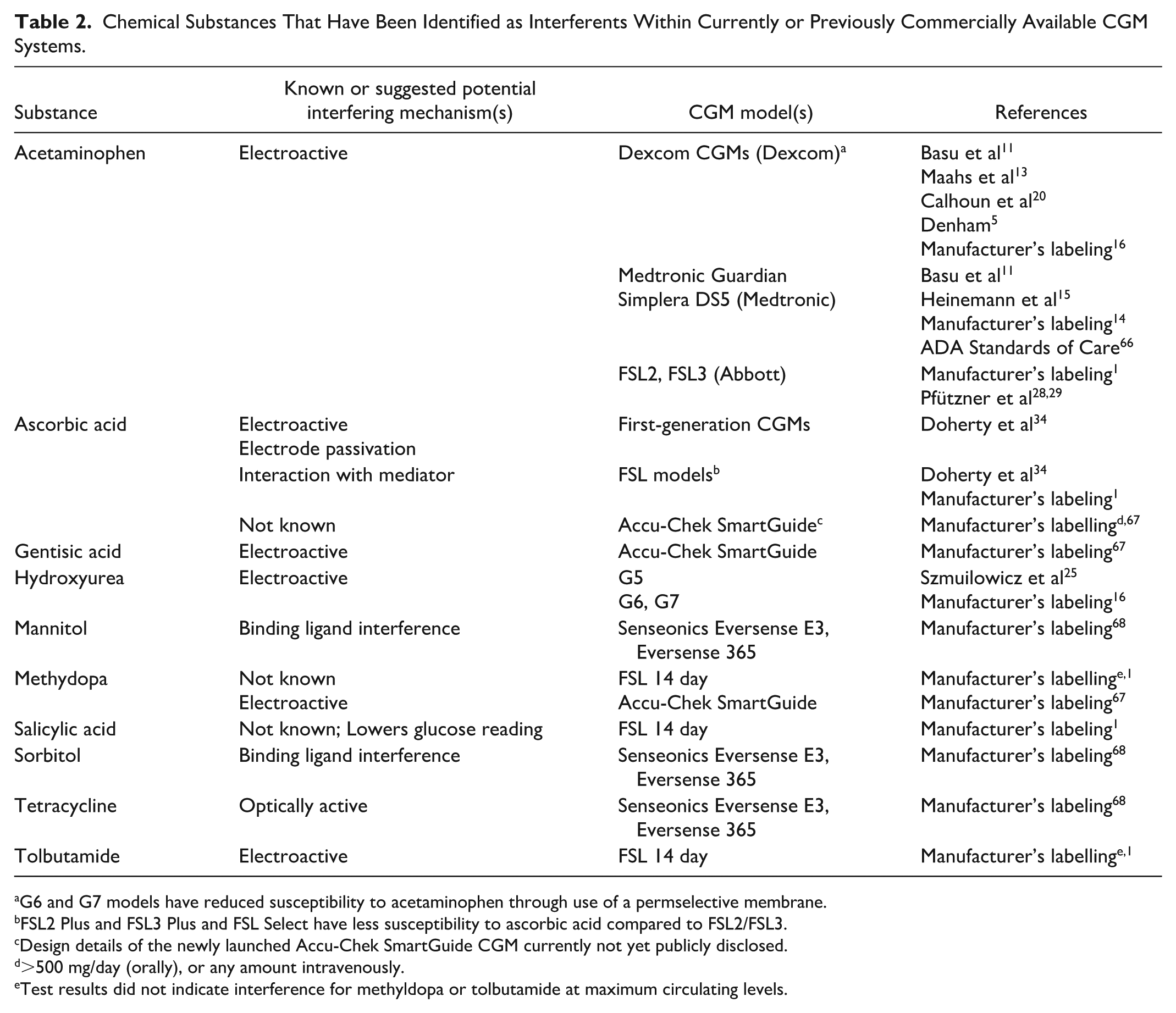
G6 and G7 models have reduced susceptibility to acetaminophen through use of a permselective membrane.
FSL2 Plus and FSL3 Plus and FSL Select have less susceptibility to ascorbic acid compared to FSL2/FSL3.
Design details of the newly launched Accu-Chek SmartGuide CGM currently not yet publicly disclosed.
>500 mg/day (orally), or any amount intravenously.
Test results did not indicate interference for methyldopa or tolbutamide at maximum circulating levels.
The interference behaviors of a wider range of potentially interfering substances on CGM systems will be reviewed in Part 3 of this series. Further detail regarding gentisic acid, salicylic acid, methyldopa, and tolbutamide interference on CGM models beyond those listed in Table 2 is also discussed.
Footnotes
Acknowledgements
The author is indebted to Dr Elizabeth Holt MD, FACE, Head of Global Clinical, Medical and Safety, Dr Mike Grady, Director of Clinical Affairs and Mr Brian Heald, Senior Vice President, Product, for their invaluable suggestions and in-depth review of this manuscript.
Abbreviations
ADA, American Diabetes Association; Ag/AgCl, silver/silver chloride; AID, automatic insulin dosing; BGM, blood glucose monitor; CGM, continuous glucose monitor; FDA, Food and Drug Administration; FSL, FreeStyle Libre; G6, Dexcom G6 CGM; G7, Dexcom G7 CGM; GOx, glucose oxidase; ISF, interstitial fluid; MARD, mean absolute relative difference.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJS is an employee of LifeScan Scotland Ltd.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJS is a salaried employee of LifeScan Scotland Ltd.
